Abstract

To the Editor,
Determination of the faecal pancreatic elastase-1 (PE1) is the first-line laboratory investigation in cases of suspected malabsorption due to exocrine pancreatic insufficiency (EPI), its use being recommended by the British Society of Gastroenterology guidelines. 1 Therefore, we read with great interest the report by Handley et al., 2 in which the authors have verified the performance of the Bühlmann immunoturbidimetric assay for PE1 measurements (fPELA turbo, Bühlmann Laboratories AG, Schoenenbuch, Switzerland) when applied on the Optilite analyser (The Binding Site, Birmingham, UK). The authors concluded that fPELA assay provides good performance, thus representing a valuable alternative for rapid analysis of PE1. To find a valid replacement to the manual ELISA method, that is, the ScheBo PE1 ELISA stool test (ScheBo Biotech AG, Giessen, Germany), routinely used in our laboratory, we recently evaluated another immunoturbidimetric assay option (Elaprest Turbo, Eurospital Diagnostics, Trieste, Italy) that has become commercially available for application on the Optilite analyser. Using excess stool submitted to our laboratory for routine analysis of PE1, we compared measurements of 27 de-identified specimens by Elaprest on Optilite and ELISA performed on the EVOLIS Twin Plus platform (Bio-Rad Laboratories, Hercules, USA). Stool samples (watery samples excluded) were frozen at −20°C, then thawed, extracted, and assayed within 2 weeks from collection. The preanalytical processing, that is, extraction, of stool samples was performed using Master Quick Prep and EasyCal Turbo collection devices for ScheBo and Elaprest Turbo, respectively. Pancreatic elastase-1 concentrations <200 μg/g stool were suggestive of EPI. 3
Assay imprecision data were obtained using control material containing human PE1 in buffer solution. Between-day CV was <5% for ELISA and <9% for Elaprest on Optilite at a PE1 concentration around the decision threshold. Figure 1 shows the diagnostic agreement between the two assays. Pancreatic elastase-1 concentrations were decreased in four samples by both assays, while three samples showed discordant results, two positives (121 and 127 μg/g) for Elaprest and negatives (>500 μg/g) for ScheBo, and one negative for Elaprest (467 μg/g) and positive (80 μg/g) for ScheBo. Being the stool samples de-identified, we were however unable to ascertain a possible explanation to account for the observed discrepancies. The Cohen kappa coefficient, estimated to evaluate the diagnostic concordance of the two assays, was 0.659, indicating substantial agreement among results by the two methods, despite using different stool sample extraction devices. Result agreement between Elaprest Turbo immunoturbidimetric assay performed on the Optilite platform and ScheBo ELISA on the 27 stool specimens evaluated in the study. Dashed lines indicate the adopted cutoff. Squared symbols depict discordant results. Note that the measuring range is up to 500 μg/g stool for ScheBo ELISA and 1200 μg/g stool for Elaprest Turbo assay.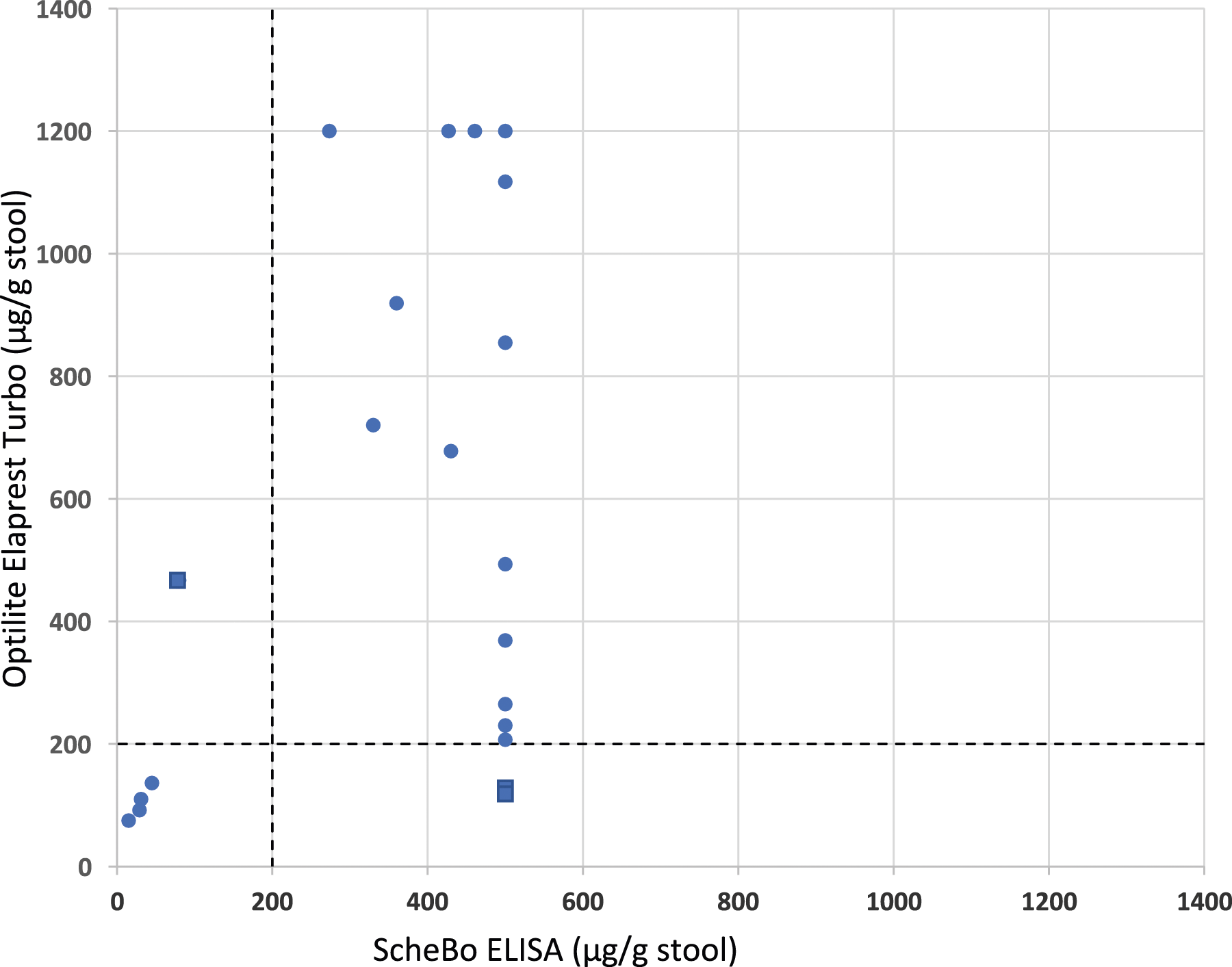
It should be noted that, while fPELA assay uses polyclonal antibodies, Elaprest Turbo is based on a mix of monoclonal antibodies for detecting human PE1. Previous data showed that monoclonal antibody-based assay (ScheBo) is not affected by pancreatic enzyme replacement therapy (PERT), while a polyclonal one (ELISA, Bioserv Diagnostics, Rostock, Germany) is therefore suggesting for the latter less specificity for human PE1 and a significant cross-reactivity against porcine elastase contained in PERT used for EPI management.4,5 Unpublished data obtained by Bühlmann indicate that fPELA is not affected by PERT when a normal recommended dose is taken. However, higher daily doses of porcine enzymes present in different PERT products may give false negative PE1 results.
In summary, when compared with the widely used but cumbersome ScheBo ELISA method, the Elaprest Turbo immunoturbidimetric assay may provide similar performance and a good characterization of the investigated EPI, with significant advantages such as the full automation with improved workflow management, reduced turnaround time, and a wider linearity range, making PE1 determination rapid, effective, and suitable for daily report.
Footnotes
Acknowledgements
We thank The Binding Site for the gift of Elaprest Turbo reagents used in the study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MP.
Contributorship
AC did the study and wrote the first draft of the article. II and MP supervised the study and reviewed the article. All authors approved the final version of the article.
