Abstract
Lymphoplasmacytic lymphoma (LPL) is a neoplasm of small B lymphocytes, plasmacytoid lymphocytes and plasma cells usually involving the bone marrow (BM). A subset of LPL which is associated with IgM monoclonal gammopathy is called Waldenstrom’s macroglobulinaemia (WM), and usually requires therapeutic intervention when a patient becomes symptomatic (Bone Marrow failure characterised by cytopenia or hyperviscosity syndrome). Here, we report the case of an 80-year-old female with clinically unsuspected WM who initially presented to the Emergency Department (ED) with nausea and vomiting. The patients’ gastrointestinal symptoms subsequently settled and was awaiting discharge. Non-specific, borderline size significant lymph nodes on CT chest was the only substantial past medical history. The diagnosis of WM was made after the Biochemistry Biomedical Scientist (BMS) detected the presence of a Type I monoclonal cryoglobulin. A potential cryoprecipitate was suspected when repeated ‘clotting’ error flags occurred during routine laboratory analyses; the sample aspiration difficulties being attributed to the viscous nature of the sample. The investigation of inaccessible low volume lymphadenopathy in the elderly should include serum protein electrophoresis and immunoglobulins as this may have established an earlier diagnosis in this case. The application of good scientific principles informed the laboratory investigation and resulted in the identification of a large IgM monoclonal cryoglobulin that prompted further appropriate investigations resulting in the diagnosis of WM. This case also highlights the importance of good communication between the laboratory and clinical staff.
Introduction
B cell malignancies are a broad and complex group of cancers that arise at various developmental stages of the B lymphocyte. The median age of LPL or WM at diagnosis is 74 years with a male predominance. 5 LPL is a neoplasm of small B lymphocytes, plasmacytoid lymphocytes and plasma cells usually involving the BM. WM belongs to a subset of LPL’s which is associated with IgM monoclomal gammopathy, ninety percent of cases demonstrating the MYD88 L265P mutation which can help with diagnostic certainty.2,5Approximately five percent of patients secrete an IgG or IgA M-protein, the vast majority of these being the IgG isotype. 6 The light chain only and non-secretory variants have also been described. 4
Clinical features are dependent on lymphocyte infiltration within organs (lymph nodes & spleen) and/or an IgM M-protein. 8 These include evidence of BM failure (anaemia, thrombocytopenia), hepatosplenomegaly, lymphadenopathy and hyperviscosity syndrome associated with a high level of IgM M-protein.
Cryoglobulins are proteins found in the blood that precipitate at temperatures below 37°C and re-dissolve on rewarming. They are, according to Brouet, classified into three types: type I, type II, and type III. 1 Type I is seen exclusively in clonal haematological disease whereas types II and III are found in many autoimmune, inflammatory and chronic infectious diseases. 3 This classification considers correlations between the protein immunochemistry findings and the clinical features as well as any underlying disease.
Case report
Here, we report a case of an 80-year-old woman who presented to the ED with nausea and vomiting. She had routine bloods sent to the laboratory and was diagnosed with a viral trigger. The only significant past medical history was non-specific, non-progressive lymph nodes on a previous CT chest.
The on-call BMS was unable to obtain analytical results on the initial and repeat blood tests because of ‘clotting’ flags (aspiration errors) on the Roche Cobas analyser. Analytical difficulties were also encountered when performing the full blood count (FBC). Agglutination, lipaemia and viscosity error flags were recorded in the context of an elevated MCHC and resulted in a haemoglobin being incorrectly reported. During the morning handover, the BMS noted that the samples stored overnight at 4°C appeared to be gelatinous in nature and consequently the presence of a cryoprecipitate was suspected (Figure 1(a)). The Duty Biochemist was notified, and the patient history reviewed on Northern Ireland Electronic Care Record (NIECR). ED was informed and the patient consented for cryoprotein investigation. Positive cryoglobulin and confirmation of an IgM monoclonal gammopathy. (a) Patient sample at 4°C: hyperviscous sample confirmed the presence of a cryogel (b) Capillary electrophoresis of the sample taken at 37°C showing a large M-protein in the gamma region measuring 27 g/L (Helena V8 Nexus). (c) Immunofixation confirmed the isotype of the M-protein, IgM Kappa (Helena SAS-4). (d) The 4°C sample, post centrifugation, after 48 hrs incubation at 4°C demonstrating a cryogel which was 50% of the 4 mL serum sample. (e) Capillary electrophoresis of the cryoprecipitate resolubilised to original volume (4 mL) using PBS. (f) Immunofixation of the cryoprecipitate resolubilised showing IgM Kappa with polyclonal IgG.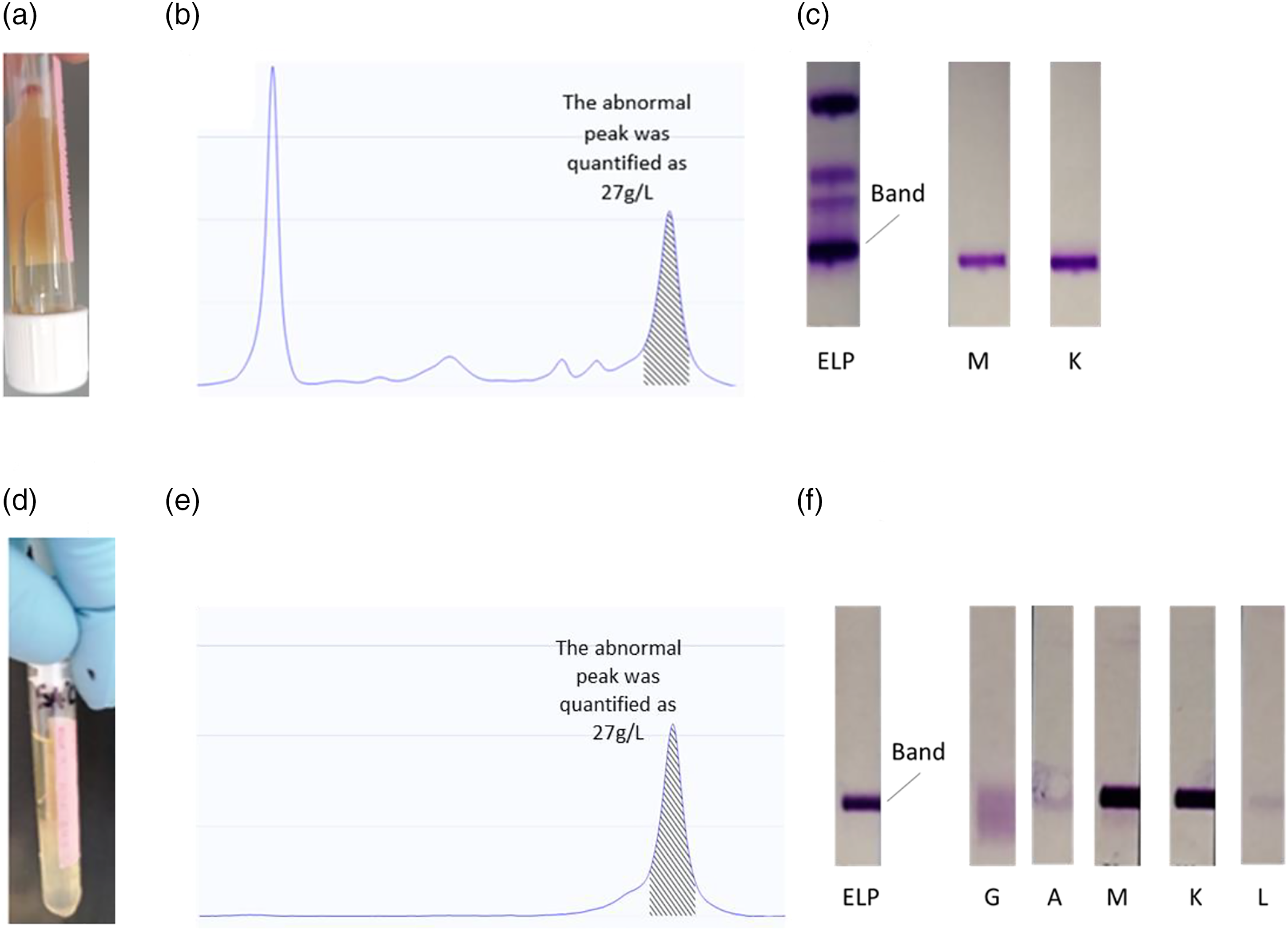
The samples were centrifuged at 37°C (10 mins; 2500 rpm.) and the serum separated as quickly as possible to avoid loss of protein within the clot. Two 4 ml aliquots were stressed at 4°C and a third aliquot maintained at 37°C. After aliquoting, the sample fluidity decreased very quickly which suggested the presence of a cryoprotein with high thermal insolubility. The 37°C sample was placed in the portable incubator (Grant 95) which had been pre-set to 40°C. This was to facilitate the analysis of the sample in the liquid state, thus preventing precipitation below 37°C and was performed when no other samples were on board the Roche 8000 analyser.
The serum maintained at 4°C instantly formed a cryogel and within 48 hrs was approximately 90% of the total serum volume. The sample was then centrifuged at 0°C for five minutes at 3000 rpm, which reduced the cryogel to approximately 50% of the total sample volume.
Immunochemical analysis (Roche 8000) of the S37, supernatant and resolubilised cryoprecipitate.
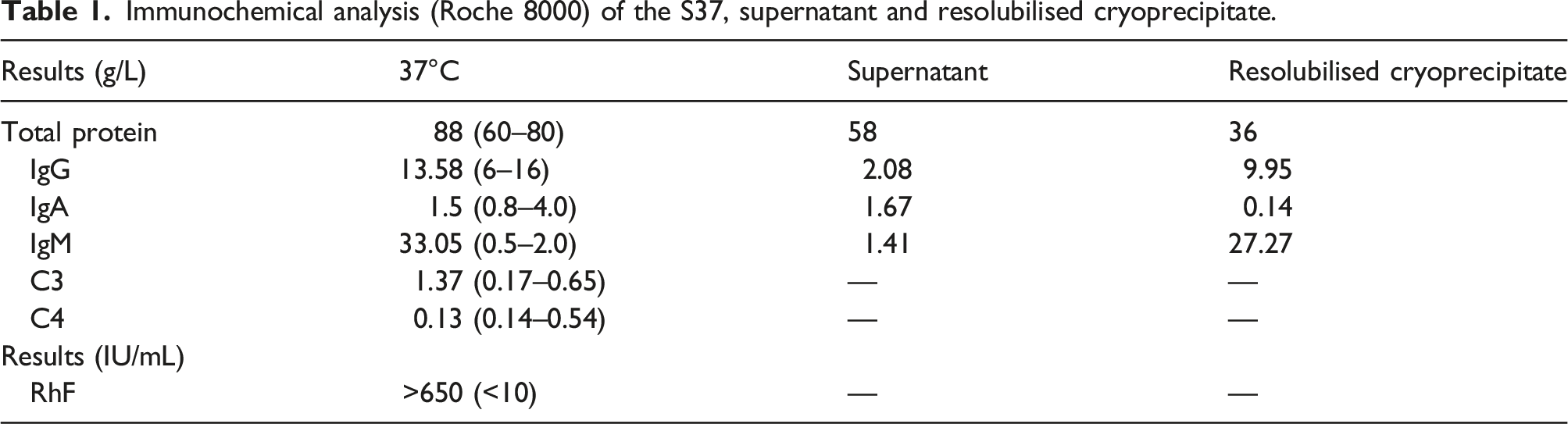
The isolated cryoprotein was subsequently characterised as a Type I cryoglobulin (IgM Kappa) of 27 g/L. A FBC sample incubated and analysed at 37°C indicated anaemia (Hb 111 g/L) and a negative direct Coombs test helped exclude the presence of a cold agglutinin disease. The serum viscosity was markedly elevated at >5.0 (1.5–1.72 mPa.s). Previous biochemical and haematological bloods showed no abnormality that would have indicated a diagnosis of LPL.
Discussion
This unusual and challenging laboratory presentation demonstrates the importance of a thorough pre-analytical and analytical approach, when repeated aspiration errors, suggested by ‘clotting’ flags, occur during routine laboratory analyses.
The subsequent laboratory workup revealed the presence of a large cryoprecipitate which was characterised as a Type I (IgM Kappa) cryoglobulin (Figure 1(c), (f)). Type I cryoglobulinemia is associated mostly with LPL/WM followed by other IgM disorders such as IgM Monoclonal Gammopathy of Undetermined Significance (MGUS), marginal zone lymphoma (MZL), small lymphocytic lymphoma (SLL), or in rare cases IgM myeloma. 7 IgG MGUS and IgG myeloma are also common, with IgA myeloma and free light chain to a lesser degree. 7
Serum Protein Electrophoresis (SPEP) is routinely performed to exclude a myelomatous malignancy. The assay is carried out in conjunction with immunoglobulins and serum free light chains. The majority of Plasma Cell Disorders (PCD) are of the IgG and IgA isotypes. Clinically, a patient presenting with Plasma Cell Myeloma (PCM) may have related organ or tissue impairment (ROTI). This includes hypercalcaemia, renal impairment, anaemia, or lytic bone lesions (CRAB).
When there is suspicion of lymphoma the accompanying symptoms include weight loss, night sweats, fever, and lymphadenopathy. 9 A biopsy of affected tissue is performed to confirm the diagnosis. This case highlights the importance of SPEP check at the initial investigation of lymphadenopathy. Serum viscosity analysis should be requested if the patient presents with headache, epistaxis, or visual disturbance.
As the patient was a new admission to ED, their biochemistry and haematology bloods were collected in lithium heparin and Ethylenediamin tetra-acetic acid (EDTA) sample tubes respectively. However, identifying a cryoprecipitate in lithium heparin tubes can be problematic due to the possible presence of heparin perceptible fraction. It is therefore imperative that cryoprotein investigations are performed in plain Serum Separator Tube (SST) and EDTA sample tubes.
The initial haematology FBC suggested the potential presence of a cold agglutinin, whereas the biochemistry error flags were possibly indicative of a cryoprecipitate. This prompted the biochemistry BMS to inspect the patients’ primary samples stored at 4°C. The samples appeared gelatinous in nature, which is the characteristic appearance of a cryogel. Observation of samples that cause analytical ‘clotting’ flags errors is an important step in the analytical process.
An IgM M-protein was discovered (Figure 1(b)) on her 37°C sample with no other abnormalities noted in her biochemistry bloods at that time. Although the sample viscosity was very elevated, the patient was essentially asymptomatic. IgM gammopathies tend to form aggregates at high concentrations in-vitro, creating a sticky/viscous sample. Figure 1e showed a cryogel with no M-protein detected in her supernatant indicating that the M-protein was completely cryoprecipitable. Significant cytopenias were discovered when her FBC was collected at 37°C.
A BM aspirate was performed which demonstrated the presence of 11% lymphoplasmacytic cells. Flow cytometry analysis identified 11% kappa restricted clonal lymphoplasmacytic cells (CD138−, CD38+, CD19− & CD45+). Molecular analysis failed for MYD88 L265P mutation. The patient was commenced on first line treatment (Dexamethasone, Cyclophosphamide and Rituximab) (DRC).
Conclusion
This case of WM presenting with a type I cryoglobulin is unusual because the patient lacked the common clinical features of LPL or WM, except for borderline lymphadenopathy. It also highlights the need for initiating protein immunochemistry investigations, especially in the elderly, in the setting of small volume lymphadenopathy and when routine biochemistry and haematology results are all normal.
There were also no signs or symptoms suggestive of cryoglobulinemia or hyperviscosity even though the plasma viscosity was very elevated. The unexpected malignancy was diagnosed because the BMS utilised his knowledge and training, and by the application of good scientific principles, initiated the appropriate investigations in a timely manner. If reflex testing had not been undertaken and concerns raised regarding the possible presence of a cryoglobulin, the patient would have been discharged back into the community. Unfortunately, the patient would have presented subsequently, with a higher M-protein level, and increased risk of morbidity and mortality.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not required as a clinical concern was raised with regards to underestimating M-proteins paraproteins in patients with undiagnosed cryoglobulinemia.
Guarantor
PL.
Contributorship
PL discovered this patient to have cryoglobulinemia and conducted the laboratory work, data analysis and reporting of results. SG was duty biochemist who contacted ED and haematology consultant with the patient results. AN admitted the patient under his care for subsequent treatment and monitoring. PL wrote the first draft of the manuscript. WID assisted in our hospital validation of cryoprotein investigations. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
