Abstract
Background
Periostin, a matricellular protein that modulates cell functions having various pathophysiological roles, has the potential to be a useful biomarker for various diseases. We recently found that periostin forms a complex with IgA in human serum, which may affect the periostin measurement.
Methods
We investigated (1) whether the formation of the periostin-IgA complex affects the original periostin ELISA system, decreasing the values of serum periostin? (2) bow each domain of periostin affects periostin measurement by the original periostin ELISA system? (3) whether we can establish a novel ELISA system that is not affected by formation of the IgA complex?
Results
The periostin value at the reducing condition was significantly higher than that of the non-reducing condition, demonstrating that formation of the IgA complex affects periostin measurement. The monoclonal antibodies (mAbs) for periostin recognizing the EMI and R1 domains immunoprecipitated serum periostin in the reducing condition more than in the non-reducing condition, whereas the mAbs recognizing the R2 or R3 domain immunoprecipitated comparable amounts of serum periostin in the reducing and non-reducing conditions, suggesting the EMI and R1 domains contribute to formation of the complex with IgA. Using SS16A recognizing the R3 domain combined with SS17B recognizing the R4 domain, we established an ELISA system that was able to measure periostin independently of the IgA complex.
Conclusions
We have established a novel ELISA system that measures periostin independently of the IgA complex. This system is promising in identifying periostin as a biomarker for various diseases.
Introduction
Periostin is a matricellular protein that modulates cell functions by binding to several integrins, playing important roles in development, maturation, and remodeling of bones, cutaneous, and connective tissues as well as cardiovascular and respiratory systems. 1
Moreover, periostin is important for oncogenesis in that it interacts with the microenvironment of tumors. 2 Periostin has the EMI domain in the N terminus, four fasciclin domains in the middle, and an alternative splicing domain in the C terminus.1,3 Various factors are known to induce expression of periostin. These include interleukin (IL)-4/IL-13, transforming growth factor β, homologous to lymphotoxin, exhibits inducible expression and competes with HSV glycoprotein D for binding to HVEM, a receptor expressed on T lymphocytes, connective tissue growth factor 2, bone morphogenetic protein 2, and mechanical stretch. 4 Based on this knowledge, we have shown that periostin is highly expressed in the lesions of various allergic diseases—asthma, 5 atopic dermatitis, 6 chronic rhinosinusitis with nasal polyp, 7 and allergic conjunctivitis 8 —and in fibrotic diseases such as scleroderma 9 and idiopathic pulmonary fibrosis (IPF). 10 Thus, the pathophysiological roles of periostin have been extensively studied.
Periostin has the characteristic of easily moving from the produced sites to various body fluids—blood, 11 sputum, 12 exhaled breath condensate, 13 tear, 8 and urine. 14 Periostin therefore can potentially be used as a biomarker for various diseases in which it is highly expressed. Based on this notion, several ELISA systems for periostin have now become available. 15 However, since the properties of periostin differ depending on the pathological conditions or on the excreted sites, we need to establish corresponding systems to detect periostin. One example is the detection of monomeric periostin in the serum of IPF patients. 16 Serum periostin mostly exists in the oligomeric form, whereas only small amounts occur in the monomeric form. We found that the monomeric form of periostin is significantly upregulated in IPF patients, correlated with decline of lung functions. Based on this finding, we established an ELISA system to detect only the monomeric form, but not the oligomeric form, for application of serum periostin to IPF. Another example is detection of periostin in the sputum of asthma patients (Ono, unpublished data). Sputum periostin, known to be upregulated in asthma patients,12,17 mostly exists in the cleaved form, probably due to proteases expressed in the airways. Since the usual ELISA systems for periostin cannot detect the cleaved form (Ono, unpublished data), we have recently established a new ELISA system to do so.
We recently found that periostin forms a complex with IgA in human serum via intermolecular disulfide bonds. 18 The periostin-IgA complex is functional, sustaining the ability to bind to a periostin receptor, αVβ3 integrin, on cell surfaces. Moreover, we found that the affinities of various monoclonal antibodies (mAbs) to periostin purified from human serum are decreased compared to those with recombinant protein. Surface plasmon resonance sensor analysis suggested that formation of the periostin-IgA complex would interfere with the binding of the periostin mAbs to periostin. Therefore, in this study, we addressed the following questions: (1) Does the formation of the periostin-IgA complex affect the original periostin ELISA system, decreasing the values of serum periostin? (2) If yes, how does each domain of periostin affect periostin measurement by the original periostin ELISA system? (3) Can we establish a novel ELISA system that is not affected by the formation of the IgA complex?
Methods
Periostin protein and anti-periostin mAbs
We prepared recombinant human periostin protein using Drosophila S2 cells, as previously described. 5 We then purified monomeric periostin using the affinity purification method, as previously reported. 16 We generated 11 kinds of mAbs against periostin (SS16A, SS17B, SS18A, SS19A, SS19B, SS19C, SS19D, SS20A, SS21A, SS23A, and SS27D) by immunizing rats or mice with recombinant human periostin, as previously described. 16
ELISAs for periostin
We used two kinds of periostin ELISA systems: (1) the original ELISA system recognizing the total periostin composed of SS18A and SS17B (Shino-Test Corporation, Tokyo, Japan) 11 and (2) a novel ELISA system recognizing the total periostin composed of SS16A and SS17B established in this study. After we incubated 2 μg/mL of SS16A mAb overnight at room temperature on ELISA plates (Immuno Clear Standard Modules; Thermo Fisher Scientific, Waltham, MA, USA), we added 200 μL of the blocking buffer (0.5% casein, 50 mM Tris, 100 mM sodium chloride, pH 8.0) followed by incubation overnight at 4°C. We then washed the plates three times with 450 μL of the washing buffer (0.05% Tween20 in PBS). We added 500-fold or 1000-fold diluted serum samples to the wells followed by incubation for 18 h at room temperature and washed the plates five times. We added 50 ng/mL of peroxidase-conjugated SS17B mAb followed by incubation for 90 min at room temperature. After we washed the plates five times, we added the reaction solution (0.8 mM 3,3,5,5–Tetramethylbenzidine, 2.5 mM H2O2) followed by incubation for 10 min at room temperature. The reaction was stopped by adding the stop solution (0.7 N H2SO4). We used the recombinant periostin that we produced for the standard diluted by the solution same as for the samples (0.5% casein, 50 mM Tris, 100 mM sodium chloride, 0.9% sodium azide). The values were calculated based on the absorbance at 450 nm measured by a microplate reader (Tecan, Mannedorf, Switzerland). In some experiments, we added dithiothreitol (DTT) to set the final concentration of 1 mM and confirmed that this DTT concentration could dissociate the complex of periostin and IgA and we calculated the periostin values using the calibration curve in the presence of 1 mM DTT. We could exclude the possibility that the periostin values in the reducing condition are mainly attributed to the fragmented IgA, because we detected only slight signal in the ELISA system, when we measured IgA in the reducing condition (data not shown). Two commercially available ELISA kits were as supplied by R&D Systems (Minneapolis, MN, USA) and Biomedica (Vienna, Austria).
Serum samples
We purchased pooled human serum and 50 serum samples obtained from individual subjects from Cosmo Bio Co., Ltd., (Tokyo, Japan) and VERITAS Corporation (Tokyo, Japan), respectively.
Immunoprecipitation and Western blotting
We carried out immunoprecipitation and Western blotting as previously described using the indicated Abs for immunoprecipitation and SS19C for immunoblotting, respectively.16,18 We confirmed that the intensities of the bands that correspond to periostin showed a linearity with the amounts of periostin (Supplemental Figure 1). In the reducing condition, we added DTT to set the final concentration of 4 mM. We calculated periostin concentrations of immunoprecipitated samples by comparing the signals with those of recombinant periostin standards using Image J software.
Subject IPF patients
We used serum samples from 56 IPF patients and 137 healthy controls collected by a previously published study, combining the CoDD-PF study and the Kurume study. 16 The details of the subject IPF patients and diagnosis of IPF were described in the previous report. 16 This study was approved by the ethics committee of Kurume University School of Medicine (#11216 issued on 29/2/2012).
Statistics
Differences were examined by the Student t-test. Results are shown as mean ± SD. The correlation between different methods was arrived at using nonparametric Spearman rank correlations. A linear relationship between two analytes was evaluated with a linear regression model. Differences were considered statistically significant if p < .05. All statistical analysis was performed by SPSS software version 26.
Results
Effect of formation of the IgA complex on periostin measurement by the original ELISA method
We recently found that periostin forms a complex with IgA in human serum via intermolecular disulfide bonds.
18
We first examined whether formation of the complex with IgA affects periostin measurement, using the original ELISA method. When we added 1 mM DTT in serum, periostin forming the complex with IgA migrated at around 240 kDa and was dissociated into the monomeric periostin migrated at 79 kDa, as we previously demonstrated. (Figure 1(a)).
18
When we measured the periostin concentration of pooled serum by the original ELISA method, those of the non-reducing and reducing conditions were 52 ng/mL and 783 ng/mL, respectively, which means that the value at the reducing condition is 15 fold higher than that of the non-reducing condition (Figure 1(b)). Such an increase of the periostin values was also observed with two kinds of ELISA kits commercially available (Supplemental Figure 2). These results demonstrate that formation of the complex with IgA affects periostin measurement by the original ELISA method. Effect of formation of the IgA complex on periostin as measured by the original ELISA method. (a) Western blotting of the periostin-IgA complex and the dissociated periostin. Periostin or IgA was immunoprecipitated by SS19A from pooled serum in either the non-reducing or reducing conditions followed by blotting with SS19C or anti-IgA Ab. (b) Perisotin values in pooled serum estimated by the original ELISA system in the non-reducing and reducing conditions. Data are shown as mean ± SD (n = 3) ***p < .001 (c) Western blotting of periostin. Periostin was immunoprecipitated from either recombinant protein or pooled serum by SS18A or SS17B in the non-reducing and reducing conditions followed by blotting with SS19C. The correlations of periostin amounts in serum from 20 individuals, estimated by Western blotting with SS18A (open circle) or SS17B (closed triangle) in the non-reducing or reducing condition, are depicted. IP denotes immunoprecipitation.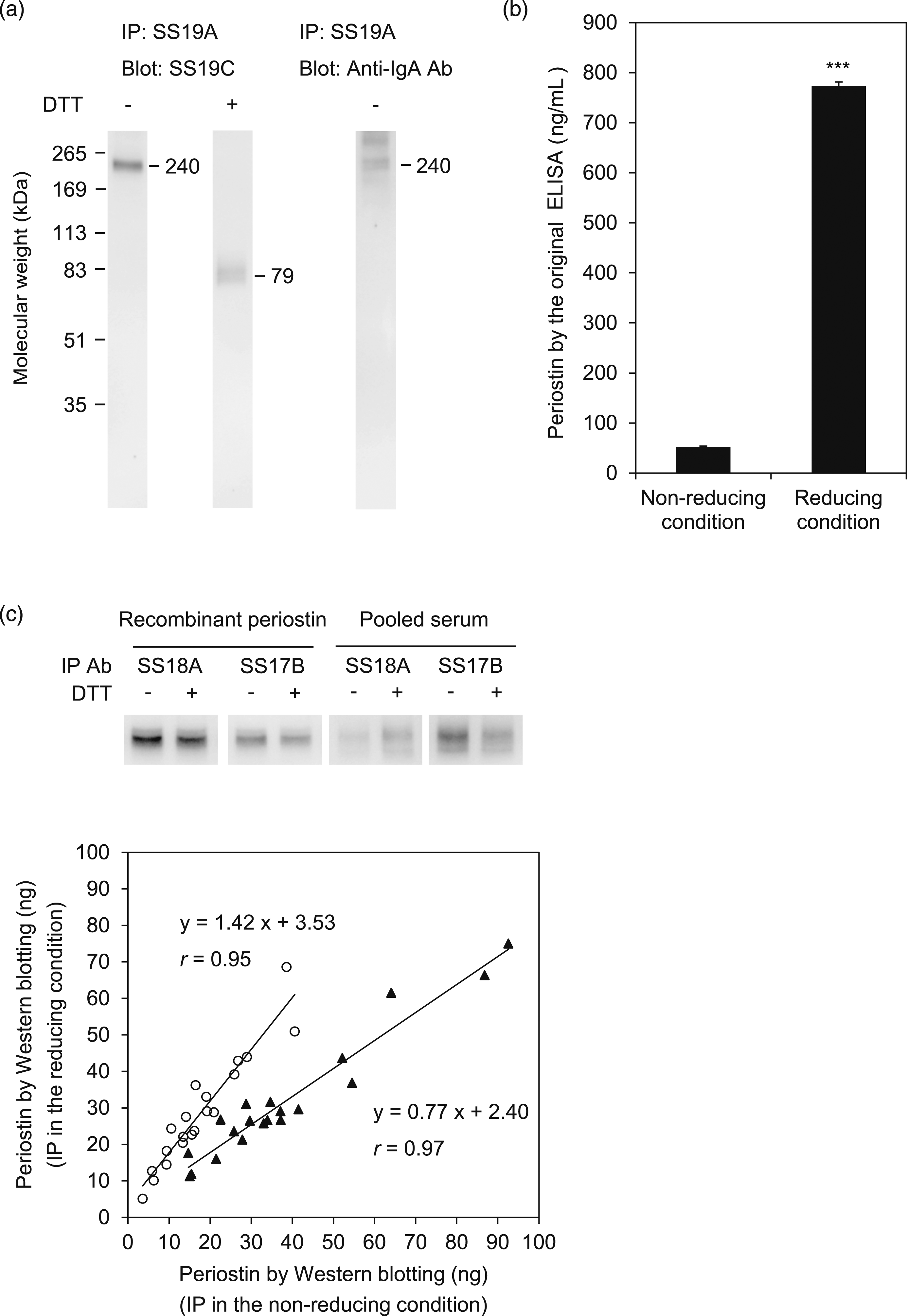
We next addressed which primary Ab (SS18A) or secondary Ab (SS17B) comprising the original ELISA method causes the difference in periostin measurement of the non-reducing and reducing conditions. SS18A immunoprecipitated comparable amounts of periostin from recombinant periostin that did not form a complex with IgA in both the non-reducing and reducing conditions, whereas SS17B showed an only slightly weaker ability to immunoprecipitate periostin in the reducing than in the non-reducing condition (Figure 1(c)). When we immunoprecipitated periostin from pooled serum, SS18A was more effective in the reducing than in the non-reducing condition, whereas SS17B was the opposite. These results demonstrate that the increase of periostin values measured by the original ELISA method in the reducing compared to the non-reducing condition is attributable to SS18A, not to SS17B.
Identification of the periostin domains that affect periostin measurement by the original ELISA method
We next examined how each domain of periostin affects periostin measurement using SS18A. We prepared 10 kinds of anti-periostin mAbs that recognize the EMI, R1, R2, R3, R4, and C-terminal domains (Figure 2(a)), examining their abilities to immunoprecipitate periostin from either recombinant periostin or pooled serum in the non-reducing and reducing conditions. Both SS20A and SS18A recognizing the EMI and R1 domains, respectively, immunoprecipitated more periostin in the reducing condition of pooled serum, but not of recombinant periostin, than in the non-reducing condition (Figure 2(b)–(d)). SS21A and SS19B in addition to SS17B showed weaker abilities to immunoprecipitate periostin in the reducing than in the non-reducing condition. The four mAbs—SS19C, SS23A, SS16A, and SS27D—recognizing the R2 or R3 domain immunoprecipitated comparable amounts of periostin from pooled serum in the reducing and non-reducing conditions. These results demonstrate that both the EMI and R1 domains rich in cysteine residues contribute to formation of the complex with IgA, affecting the abilities of the mAbs recognizing these domains to bind to periostin. Identification of the periostin domains that affect periostin measurement by the original ELISA method. (a) The schematic model of the periostin structure and the epitopes of 10 kinds of anti-periostin mAbs. (b) Western blotting of periostin. Periostin was immunoprecipitated from either recombinant protein or pooled serum by the indicated mAbs in the non-reducing and reducing conditions, followed by blotting with SS19C. (c, d) The amounts of periostin estimated by Western blotting. Periostin was immunoprecipitated from recombinant protein (c) or pooled serum (d) by the indicated anti-periostin mAbs in either the non-reducing (open bar) or reducing (closed bar) condition, followed by blotting with SS19C. Data are expressed as mean ± SD (n = 3)
**
p < .01, *p < .05.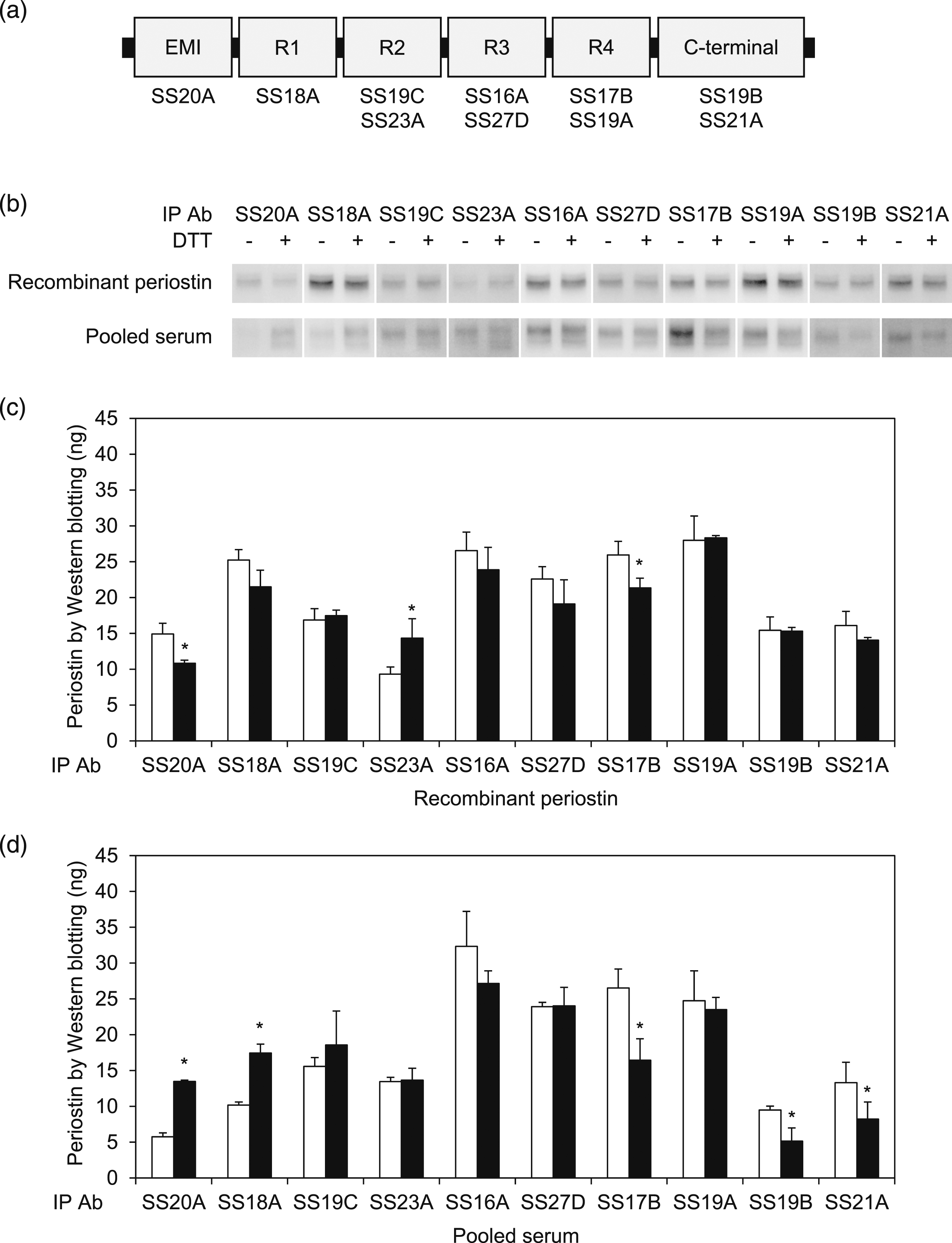
Establishment of a novel ELISA system that can measure periostin independently of the IgA complex
In order to establish an ELISA system that could measure periostin independently of the IgA complex, we chose as the primary mAb SS16A recognizing the R3 domain that does not change the ability to immunoprecipitate periostin in the reduction condition. We then established a novel ELISA system combining SS17B as the secondary mAb. We validated the accuracy of the ELISA system in the presence or absence of formation of the IgA complex by comparing the values in the reducing and non-reducing conditions for the original and novel ELISA systems. The periostin concentrations measured by the original ELISA system provided values approximately tenfold higher in the reducing than the non-reducing condition (r = 0.76, y = 8.80x + 209.41), whereas the novel ELISA system showed almost the same values (r = 0.92, y = 1.03x + 184.46) in the non-reducing and reducing conditions (Figure 3(a)). Estimation of periostin concentrations by immunoprecipitation also showed a good correlation with what had been estimated by the novel ELISA system (Figure 3(b)). These results demonstrate that the novel ELISA system using SS16A instead of SS18A as the primary mAb can measure periostin independently of the IgA complex. Establishment of a novel ELISA system that can measure periostin independently of the IgA complex. (a) The comparison of periostin amounts in serum samples from 50 individuals, as estimated by the original ELISA system (open circle) or the novel ELISA system (closed triangle) in the non-reducing or the reducing condition. (b) The correlations of periostin amounts in serum samples from 20 individuals as estimated by Western blotting and ELISA. Western blotting was performed by SS17B for immunoprecipitation and by SS19C for blotting in the non-reducing condition. ELISA was performed by the original ELISA (open circle) or the novel ELISA (closed triangle) system in the non-reducing condition.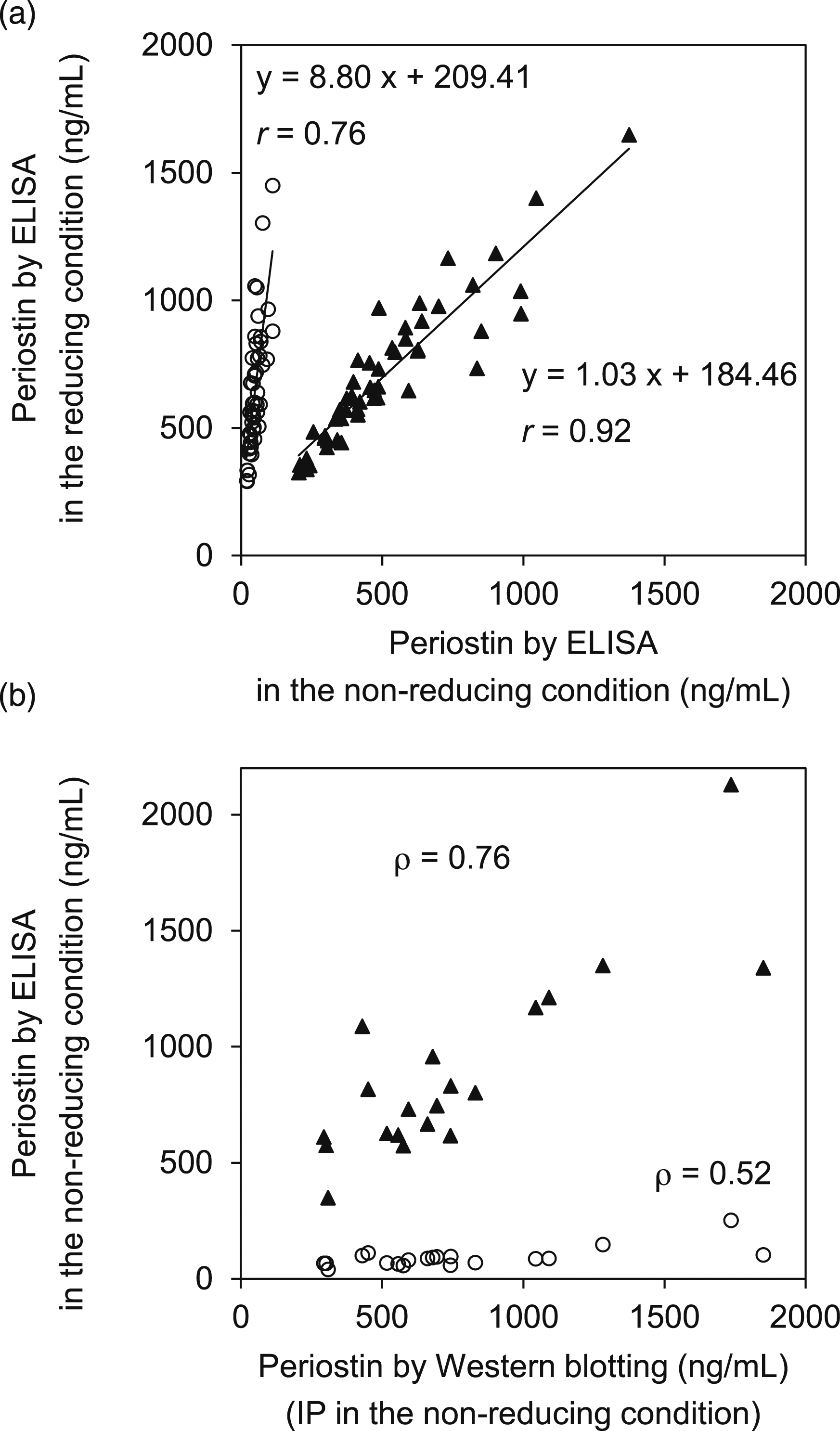
Validation of the novel ELISA system for periostin
We validated the assay performance of the novel ELISA system for periostin according to the Clinical and Laboratory Standards Institute guidelines. The novel ELISA system showed good representative calibration curve (Figure 4(a)). We always calculated periostin values by appropriately diluting samples to let the detected values less than 4 ng/mL. Presence of 1 mM DTT did not affect the calibration curve, whereas the original ELISA system showed lower values in the presence of 1 mM DTT (Supplemental Figure 3). The lower limits of blank, of detection, and of quantification (LOQ) of the novel ELISA system were 2.0, 4.0 (data not shown), and 18 pg/mL, respectively (Figure 4(b)). The intra- and inter-assay CV values ranged from 1.8% to 2.2% (n = 15) and from 1.9% to 2.3% (n = 15), respectively (Table S1). Recoveries of periostin ranged from 99% to 102% (Figure 4(c)). Serially diluted analyses showed close linearity in each sample (r > 0.997, data not shown). There was no substantial interference from hemoglobin (<500 mg/dL), bilirubin (<50 mg/dL), IgA (<5 mg/mL), βIG-H3 (<500 ng/mL), ascorbic acid (<20 mg/dL), chyle (<3000 formazin turbidity unit), or DTT (<0.1 mM), indicating that the assay was resistant to interference from a wide range of biological constituents (Figure 4(c)). These results guarantee the quality of the novel ELISA system for periostin established in this study. Validation of the novel periostin ELISA system. (a) The calibration curve of periostin values estimated by the novel ELISA system. The calibration was performed by the cubic regression model without (solid line) or with (dashed line) 1 mM DTT. Data are expressed as mean ± SD (n = 3). (b) The lower limit of quantification of the novel ELISA system. Eight serum samples were measured in triplicate for 3 days. The bias was calculated as the measured mean minus the reference value. The total error was calculated by adding two standard deviations to absolute value of the bias. Total error % was the total error divided by the reference value of the sample. The LOQ value was estimated by the power function model parameter, calculated with total error % (x-axis) and the reference value (y-axis) (total error ≤20%). (c) The spike and recovery test assessed by measuring three serum samples added to 39 (white bar), 76 (gray bar) or 159 ng/mL (black bar) of recombinant periostin. Each sample was measured in triplicate. The expected values were calculated as the periostin levels of base serum plus the added periostin amount. The recovery rate was calculated from the observed values divided by expected values. Periostin levels of base serums were indicated as follows: Serum 1: 565 ng/mL, Serum 2: 189 ng/mL, and Serum 3: 717 ng/mL. Data are expressed as mean ± SD (n = 3). (d) Interference tests for the novel ELISA system. Pooled serums with or without these interferents were measured by the novel ELISA system. The evaluated concentrations are indicated in square brackets. Mean values represent the average of three replicates. Recovery indicates the ratio of the periostin levels in serum with and without interferent.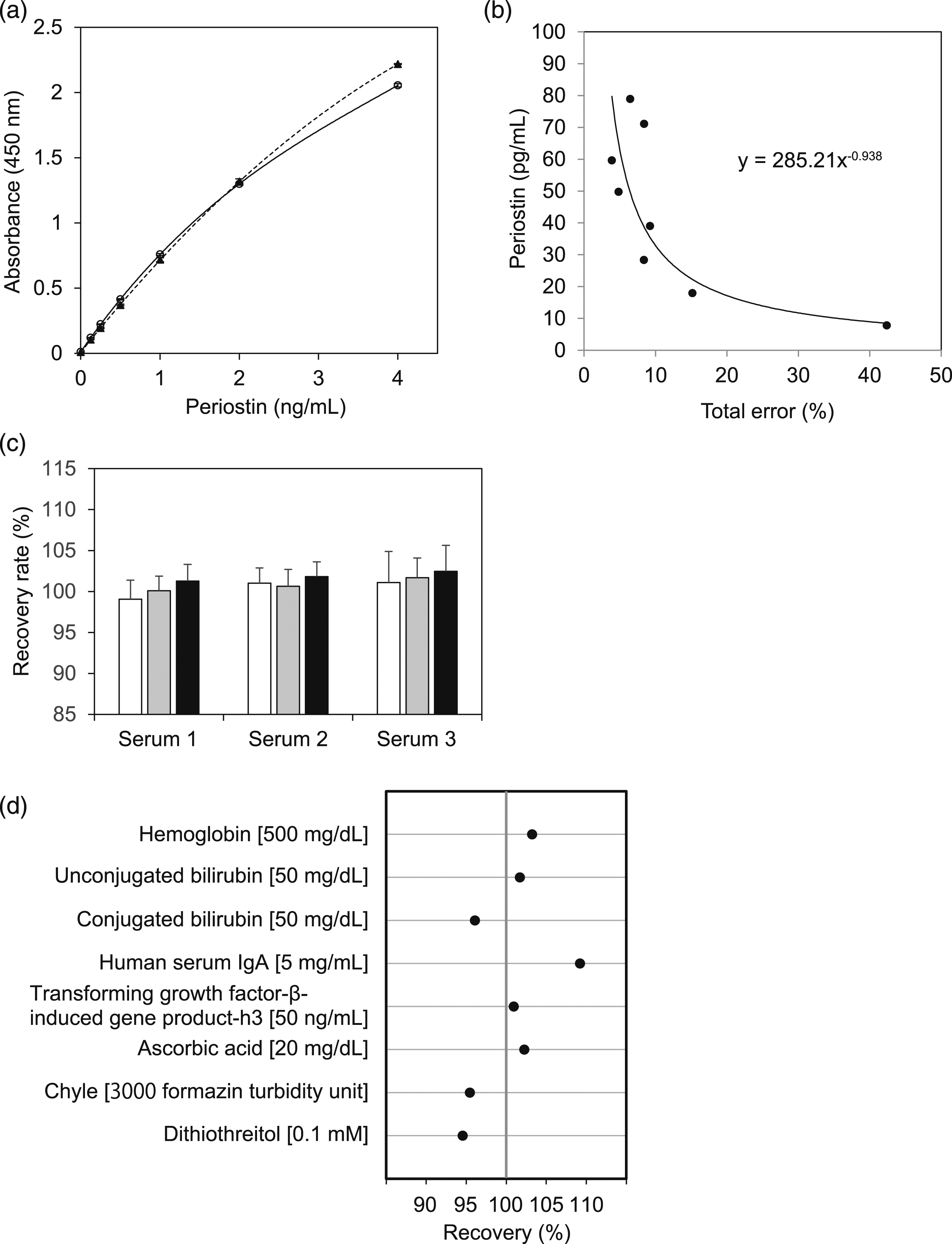
Comparison of the serum periostin values estimated by the original and novel ELISA systems for periostin
We finally compared serum periostin values estimated by two kinds of ELISA systems for periostin—the original ELISA system composed of SS18A and SS17B detecting the periostin-IgA complex and the monomer and the novel ELISA system established in this study, composed of SS16A and SS17B detecting the whole periostin independently of the formation of the periostin-IgA complex. For this analysis, we used 56 IPF patients as an example of a pathological condition in which serum periostin levels are upregulated and 137 healthy controls. The serum periostin values estimated by the novel ELISA system showed good correlations with those estimated by the original ELISA system by both the non-reducing condition (Figure 5(a)) and the reducing condition (Figure 5(b)) in both healthy controls and IPF patients (p < .001). However, the values in the reducing condition were completely concordant in both healthy controls (r = 0.99) and IPF patients (r = 1.00), whereas those of the non-reducing condition were less concordant in either healthy controls (r = 0.68) or IPF patients (r = 0.57). Moreover, the periostin values by the novel ELISA system were very accordant in both IPF patients (r = 0.94) and healthy controls (r = 0.94). Taken together, we conclude that the novel ELISA system measures accurately whole periostin even in non-reducing condition. Comparison of the serum periostin values estimated by two ELISA systems for periostin.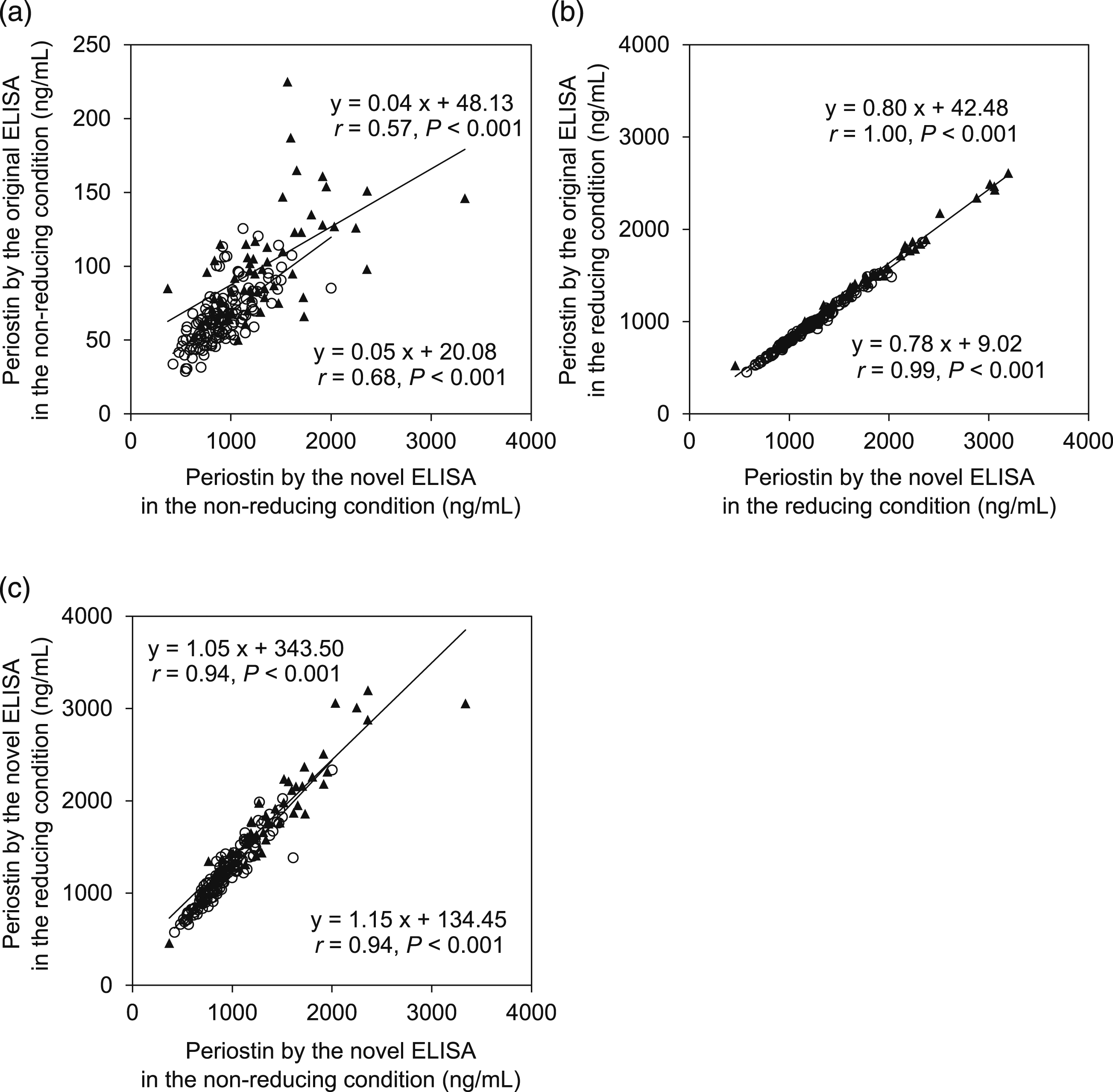
Discussion
In this study, we established a novel ELISA system that could measure periostin independently of the IgA complex. Thus far, we have established four kinds of ELISA system for periostin: (1) the original periostin ELISA system composed of SS18A and SS17B that has been widely used for various diseases.
11
(2) The ELISA system composed of SS20A and SS19D detecting specifically the monomeric form of periostin.
16
(3) The ELISA system composed of SS18A and SS19C detecting the cleaved form of periostin in sputum (Ono, unpublished data). (4) The ELISA system established in this study. Although both the original ELISA system and the ELISA system established in this study can detect all full-length periostin, the former, but not the latter, is affected by the formation of the complex with IgA in serum (Figure 6). We do not know at this moment how periostin forms a complex with IgA in serum and what the proportion is of the complex and periostin alone in serum. Therefore, the periostin values estimated by the novel ELISA system established in this study rather than by the original ELISA system would accurately reflect periostin existing in serum. The basal level of serum periostin is likely to be mainly regulated by periostin derived from bones, because the serum periostin levels parallel the activity of bone metabolism in children.
19
Then periostin that is excessively produced in various pathological conditions can be detected as increased periostin in serum. This is because periostin characteristically moves easily from the produced sites to vessels and because its basal levels in serum are relatively low compared to those of other extracellular matrix proteins.
3
The increase of periostin values estimated by the novel ELISA system established in this study would accurately reflect production of periostin in various pathological conditions as well. Schematic model of three ELISA systems for periostin. The schematic model of the original and novel ELISA systems for periostin in the non-reducing condition (a) and in the reducing condition (b).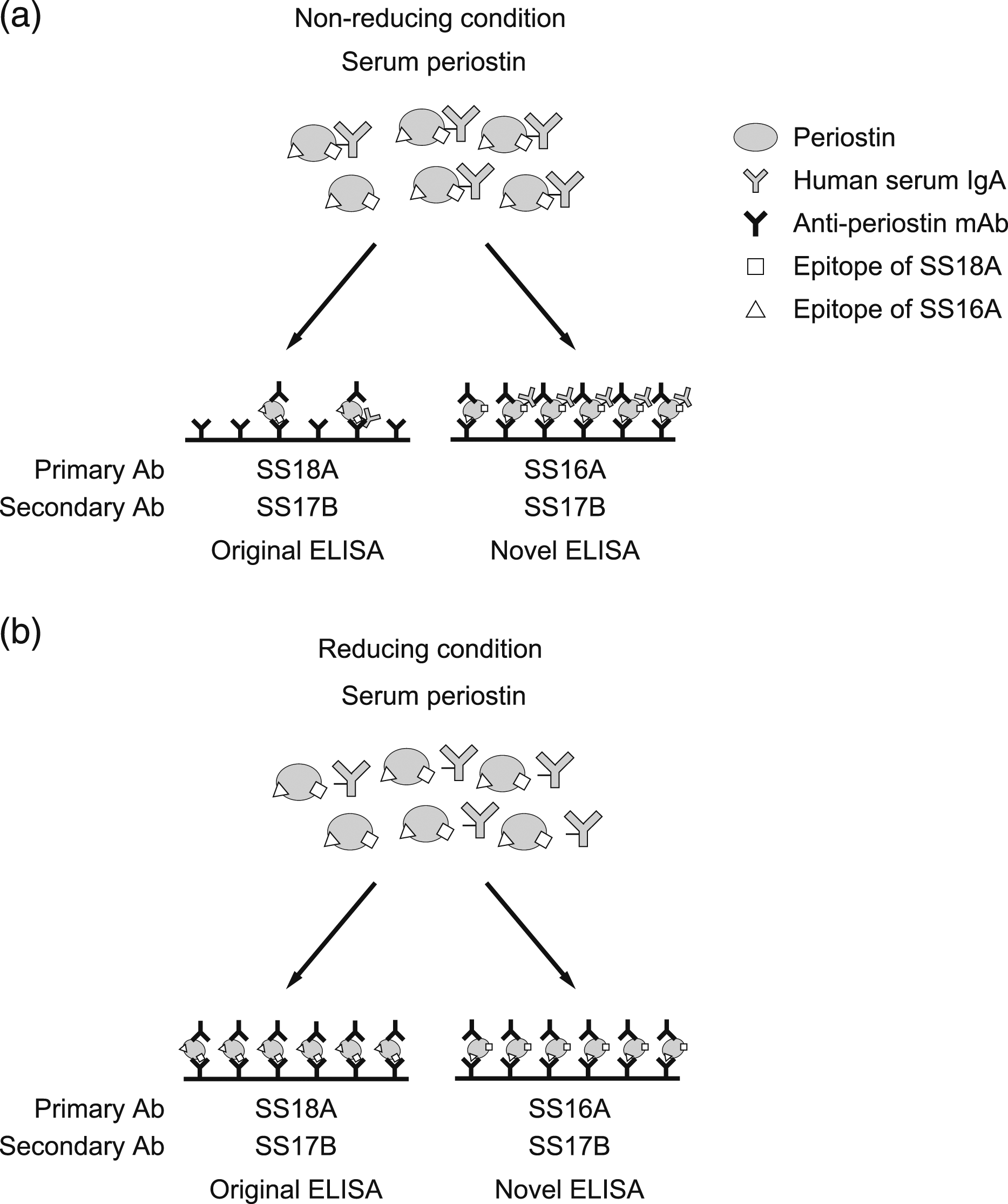
We have previously demonstrated that periostin forms a complex with IgA in serum via (a) disulfide bond(s). 18 Structural analysis of periostin showed that 10 cysteine residues located at the EMI domain or at the fasciclin domains contribute to formation of intramolecular disulfide bonds. The analysis also showed that the cysteine residue at 60 (Cys60) causes cysteinylation, suggesting that Cys60 would be involved in formation of intermolecular disulfide bonds with other proteins. 20 This indicates that periostin would form a complex with IgA via Cys60. This is a reasonable assumption; our present result shows that both the EMI and R1 domains contribute to the complex formation (Figure 2). Structural analysis also demonstrated that Cys60 does not affect formation of the homo-dimeric periostin via hydrogen bonds. 20 This corresponds to our previous result that the periostin-IgA complex is still functional, sustaining the ability to bind to αVβ3 integrin on cell surfaces. 18 Taking these results together, it is reasonable that the formation of the complex with IgA does not interfere with the binding to αVβ3 integrin via the binding sites assumed to be located at least at the R1 and R2 domains.21,22 Our present study supports the notion that the formation of the complex with IgA does not prevent the access of SS16A and SS17B to their epitopes, the R3 and the R4 domains, respectively (Figures 2 and 3).
A number of studies have been performed aiming to elucidate the potential of periostin as a biomarker in various pathological conditions. These studies have shown the usefulness of this biomarker for allergic diseases, 3 fibrosis in the lung, 23 kidney, 24 and skin, 25 and tumorigenesis. 26 In particular, a great deal of attention has been paid to periostin’s potential in the management of asthma—to diagnose type 2 inflammation-dominant asthma,11,27 to predict the effect of biologics targeting type 2 mediators,28–30 and to predict resistance to inhaled corticosteroids.11,31,32 Thus far, we do not know the advantage of the novel ELISA system established in this study over the original system. However, the periostin values estimated by the novel system would accurately reflect the pathological production without the formation of the IgA complex. It is certain that although the periostin values estimated by either the original or the novel system are well correlated, these two values are more concordant in the reducing condition rather than in the non-reducing condition, as shown in Figure 5(a) and (b), suggesting the possibility that our novel system may have a unique utility for clinical applications, superior to the original system. Further studies aiming at this point are needed in the future.
Supplemental Material
Supplemental Material - Establishment of a novel ELISA system for measuring periostin independently of formation of the IgA complex
Supplemental Material for Establishment of a novel ELISA system for measuring periostin independently of formation of the IgA complex by Masayuki Takai, Junya Ono, Masaki Okamoto, Kiminori Fujimoto, Ayami Kamei, Satoshi Nunomura, Yasuhiro Nanri, Shoichiro Ohta, Tomoaki Hoshino, Arata Azuma, and Kenji Izuhara in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We thank Dr. Dovie R. Wylie for the critical review of this manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MT, JO, and AK are employees of Shino-Test Corporation. KI received research grants from Shino-Test Corporation. The other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Grant-in-Aid for Scientific Research (JP21K08476); Japan Society for the Promotion of Science to Dr. Kenji Izuhara. No additional external funding was received for this study.
Ethical approval
This study was approved by the ethics committee of Kurume University School of Medicine (#11216 issued on 29/2/2012).
Guarantor
KI.
Contributorship
MT, JO, and AK made the kit and analyzed the samples. MT, JO, MO, KF, AA, and KI designed the work. MT and KI wrote the paper. All authors contributed to drafting the article and approved the published version of the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
