Abstract

Dear Editor,
Retinol (Vitamin A) and α-tocopherol (Vitamin E) are routinely measured in patients predisposed to fat malabsorption, including those with cystic fibrosis, inflammatory bowel disease and short-bowel syndrome. 1 Data from the United Kingdom National External Quality Assurance Scheme indicate that high-performance liquid chromatography is the predominant method employed for retinol and α-tocopherol analysis in UK laboratories, most of which use the Chromsystems assay.
Chromsystems recently updated the instruction manual for the one-step version of their vitamin A and E method, specifying that retinol should not be quantified in samples containing haemoglobin above 0.3 g/L, and α-tocopherol should not be quantified in any ‘haemolytic’ samples. Our aim was to determine the degree to which haemolysis interferes with retinol and α-tocopherol quantitation by the Chromsystems assay, and to investigate a mechanism of interference.
Using the osmotic shock protocol,
2
we first prepared a haemolysate from anonymised surplus blood collected into lithium heparin tubes, and generated six haemolysate standards with incrementally increasing haemoglobin concentrations, measured using the Abbott Alinity haemolysis index. Anonymised redundant serum samples (n = 12) and quality control samples (n = 4) were also collected, with baseline retinol and α-tocopherol concentrations across the linear measuring range. Aliquots of each sample were separately spiked 1:1 with the haemolysate standards. Extraction was performed using the Chromsystems pre-mixed solution, chromatography using the Chromsystems vitamin A/E column and mobile phase, and ultraviolet detection on the Thermo Fisher UltiMate 3000 HPLC system. Retinol and α-tocopherol were analysed simultaneously and in duplicate in each aliquot. Percentage difference in numerical results from baseline values of the corresponding neat samples was determined, with ≤ 15% deviation considered acceptable (Figure 1). The effect of increasing haemolysis on retinol and α-tocopherol quantitation using the Chromsystems one-step HPLC assay. Each data point represents the percentage change in retinol and α-tocopherol numerical results in an individual specimen spiked with haemolysate compared to the baseline values of the corresponding neat sample, and after adjusting for the x2 dilution factor. Horizontal dotted lines indicate +/−15% acceptable deviation limits.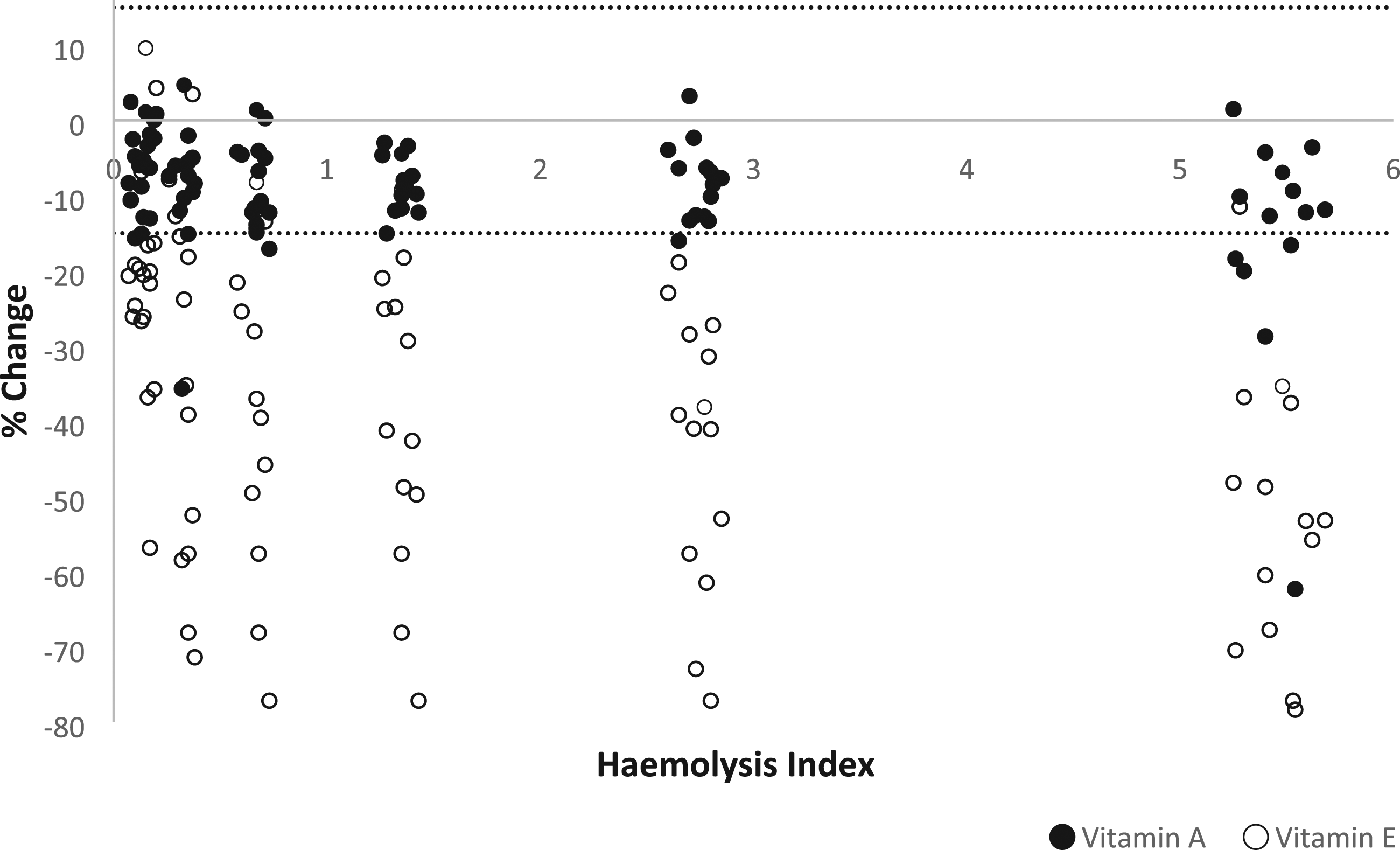
Retinol results were within 15% of baseline results obtained for the corresponding neat samples, except for several specimens with the highest degree of haemolysis. By contrast, α-tocopherol results for the majority of spiked specimens were substantially lower than baseline values, regardless of haemolysis level. Notably, even in the 16 specimens spiked to achieve the lowest concentration of haemoglobin between 0.1 and 0.2 g/L, measured α-tocopherol concentrations were more than 15% lower than the respective baseline values in 13 specimens, with a 56% decrease being the greatest deviation. This finding suggests that α-tocopherol is susceptible to negative interference even at low levels of haemolysis commonly seen in clinical practice.
To investigate the mechanism of interference, we subsequently spiked specimens with increasing concentrations of purified human haemoglobin as opposed to haemolysate. Retinol results were similarly unaffected, whereas negative interference of a similar magnitude was observed for α-tocopherol. These findings suggest haemoglobin itself is the primary cause of interference and not another mechanism such as degradation by proteases released during haemolysis. However, spectrophotometric interference is unlikely to be the mechanism of interference given the negative interference and the lack of overlap between haemoglobin’s absorbance spectrum 3 and α-tocopherol’s peak absorbance measurement at 295 nm.
In conclusion, while our findings demonstrate that haemolysis has a minimal effect on retinol quantitation by the Chromsystems assay, even low levels of haemolysis have the potential to significantly decrease α-tocopherol results. Such decreases in α-tocopherol could misclassify vitamin E–replete patients as deficient, and levels above reference ranges could be falsely reported within reference ranges. Accordingly, laboratories offering this assay should consider measuring the haemolysis index on all requests, and take into account the possibility of artefactually decreased levels when reporting results in affected specimens.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
Catherine Collingwood.
Contributorship
CC conceived the study. CH performed the practical work, data analysis, and wrote the first draft of the manuscript. All authors reviewed, edited and approved the final manuscript.
