Abstract
Background
During normal pregnancy, the glomerular filtration rate (GFR) increases dramatically. Failure to obtain this physiological increase is an important risk factor for morbidity and mortality for both mother and child. The estimated GFR (eGFR) using serum creatinine levels is unsuitable for accurate measurement of renal function during pregnancy. Therefore, new biomarkers have been proposed. Elevated levels of Cystatin C (CysC) and Neutrophil Gelatinase-Associated Lipocalin (NGAL) are associated with renal failure and preeclampsia (PE). In this study, we determined reference intervals for CysC and NGAL during pregnancy.
Methods
Healthy pregnant women were recruited and blood samples were collected at 9–13 weeks (T1), 27–29 weeks (T2), and 36–39 weeks (T3) of gestation and at 4–13 weeks postpartum (PP). The samples from women with uncomplicated pregnancy were analyzed to determine median values and upper reference limits (URLs, 97.5 percentiles) of creatinine, CysC, and NGAL.
Results
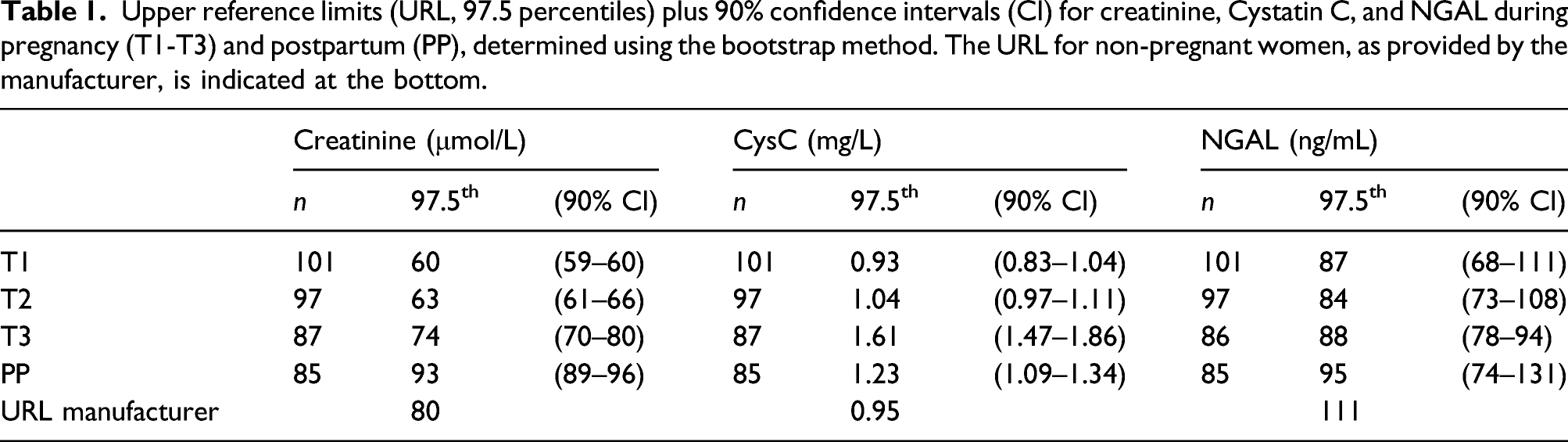
A total of 175 women were included. Longitudinal changes and median values of creatinine, CysC, and NGAL were determined using only complete data sets (n=59). URLs were determined using all available data. The URL at T1, T2, T3, and PP were 60, 63, 74, 93 μmol/L for creatinine; 0.93, 1.04, 1.61, 1.23 mg/L for CysC; and 87, 84, 88, 95 ng/mL for NGAL.
Conclusions
CysC concentrations are highly dynamic and increase during pregnancy. NGAL concentrations are less dynamic, but well below the URL specified by the manufacturer for non-pregnant women. It is therefore recommended to use trimester-specific reference values for both CysC and NGAL.
Introduction
During normal pregnancy, glomerular filtration increases up to 50% due to an increased plasma flow and dynamic changes in filtration fraction. 1 The estimated glomerular filtration rate (eGFR), determined using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation based on serum creatinine, is only suitable for use in stable situations. During pregnancy, creatinine levels are decreased in the first two trimesters and start to rise again in the third trimester, resulting in slightly elevated creatinine levels shortly after delivery. 2 Serum creatinine and eGFR are therefore not suitable for accurate determination of renal function in pregnant women. 3
The kidneys play a central role in hypertensive disorders of pregnancy, which includes pre-eclampsia (PE) and HELLP syndrome (hemolysis, elevated liver enzymes and low platelets). 4 The prevalence of chronic kidney disease (CKD) in pregnancy and PE are both estimated at ±3%, and are associated with a significant increase in adverse pregnancy outcomes for both mother and child. 4 Failure to manage the physiologically expected rise in GFR during early pregnancy is associated with spontaneous abortions. 5 The ability to determine a reliable eGFR is thus critical in the care of pregnant women.
Several new biomarkers have been proposed to determine renal injury more reliably during pregnancy. Cystatin C (CysC) is a 13-kDa cysteine protease inhibitor which is constantly produced by all nucleated cells. It is not actively secreted by the renal tubules and filtered freely through the glomerulus, after which it is degraded by proximal tubular cells, making it a suitable candidate biomarker for renal function. 6 It has been shown that serum CysC levels rise significantly during the third trimester of uncomplicated pregnancies due to placental expression, and decrease to normal levels postpartum.7,8 In women with PE, placental expression of CysC is elevated in comparison to women with uncomplicated pregnancy. 9 A recent meta-analysis shows that maternal serum CysC levels during the third trimester could serve as a useful diagnostic marker for PE, with a sensitivity of 85%, a specificity of 84%, and pooled positive and negative likelihood ratios of 5.36 and 0.18, respectively. 10 Moreover, several studies showed that elevated CysC levels during early pregnancy are associated with development of PE,11–13 indicating that it could serve as a predictive marker for PE. 14 Furthermore, CysC elevation in the second trimester is associated with gestational diabetes mellitus (GDM). 15
Another promising biomarker to determine renal injury during pregnancy is Neutrophil Gelatinase-Associated Lipocalin (NGAL), a 25-kDa glycoprotein expressed and secreted by immune cells, hepatocytes and renal tubular cells. 16 Several studies showed that NGAL levels were significantly increased in women with PE.17–19 Similar to CysC, NGAL levels are elevated during early pregnancy in women who develop PE, making it a potential predictive marker.20,21 One recent study showed that the combination of CysC and NGAL levels at the end of the first trimester was the best predictor for development of PE, with an area under the receiver-operating curve (ROC) of 0.877. 22
To use CysC and NGAL as predictive or diagnostic markers for kidney disease during pregnancy, reference values for women with uncomplicated pregnancy have to be available. Those have not been established yet. Therefore, the aim of this study is to determine trimester-specific reference values for CysC and NGAL in women with uncomplicated pregnancy.
Materials and methods
Design
175 women with uncomplicated pregnancy (98% Caucasian) were recruited via primary midwife practices in The Netherlands, as described before. 23 Exclusion criteria were pre-existing renal disease, cancer, endocrine disease, twin pregnancy, thyroid medication and hormonal stimulation related to the pregnancy. Women who developed complications during the current pregnancy (hyperemesis gravidarum, gestational diabetes, PE, HELLP) and women who delivered prematurely or of a dysmature child were excluded prior to the data analysis. None of the women used medication for hypertension or cardiovascular disease. The Medical Research Ethics Committee (MREC) Brabant and the local MREC at Amphia Hospital exempted this study from the Medical Research Involving Human Subjects Act (WMO) since it did not meet the criteria for medical-scientific research and hence approved this study. Written informed consent was obtained from all subjects.
Non-fasting blood samples were collected in EDTA tubes at 9–13 weeks (T1), 27–29 weeks (T2), and 36–39 weeks (T3) of gestation, as well as 4–13 weeks postpartum (PP). Plasma aliquots were frozen and stored at −80°C. Aliquots underwent only one freeze-thaw cycle before the measurements.
Methods
Creatinine, CysC, and NGAL were measured using CE-marked assays on a Cobas c501 (Roche Diagnostics, Mannheim, Germany). For creatinine, an enzymatic method was used which is standardized against ID/MS (Roche Diagnostics). Particle-enhanced immuno-turbidimetric methods were used for CysC (Roche Diagnostics, reference number 06729371) and NGAL (BioPorto Diagnostics, Gentrofte, Denmark, reference number ST001CA). The 95% reference interval (RI) for women provided by the manufacturer and used in clinical practice at the time of the study was 45–80 μmol/L for creatinine. Provided RI for women were 0.61–0.95 mg/L for CysC and <111 ng/mL for NGAL.
Statistical analysis
Creatinine, CysC and NGAL concentrations were not normally distributed. Therefore, the upper reference limits (97.5 percentiles) were estimated using the bootstrap method described by Solberg, 24 creating 500xn data points for each time-point. Outliers were identified using the D/R ratio, with D the absolute difference between the extreme value and the next largest value and R the range of all observations. If D/R was >0.33, the data point was considered to be an outlier and excluded from analysis. 25
Longitudinal changes of the markers were assessed with the Friedman non-parametric test for paired observations, using only the data from the women with a complete data set. p < 0.05 was considered statistically significant. Changes between two individual time-points were assessed with the Wilcoxon signed rank test for two paired observations. p < 0.01 was considered statistically significant, considering multiple testing. Microsoft Excel and Analyse-it (Analyse-it Software, Ltd, Leeds, UK) were used for statistical analysis.
Results
Women who miscarried (n = 4), developed significant disease during pregnancy [hyperemesis gravidarum (n = 1), pregnancy-induced hypertension, PE or HELLP (n = 17), diabetes (n = 4) or other disease (n = 2)], delivered prematurely (n = 2) or had a small for gestational age neonate (n = 4) were excluded from analyses, while nine women never showed up for blood collection, as described before. 23 Fifty-nine women had a complete data set (T1-T2-T3-PP) and were used for longitudinal analysis of creatinine, CysC and NGAL. The mean (±SD) age at T1 was 30 (±4) years and the mean (±SD) gestational period was 40 (±1) weeks. For each of the three markers, one outlier was removed at T1 (all different subjects). For NGAL, one additional outlier was removed at T3.
Longitudinal changes
Longitudinal changes in creatinine, CysC and NGAL concentrations are presented as median values and interquartile ranges per time point (see Figure 1). Median creatinine values are low-normal compared to the non-pregnant reference range for women during the first part of pregnancy (T1, T2) and rise towards high-normal values postpartum (PP). Median CysC and NGAL increase during pregnancy (T1-T3), and decrease again after delivery (PP). Median CysC levels are higher than the non-pregnant reference range for women during the third trimester (T3), while median NGAL levels stay well below the upper reference limit (URL) of the manufacturer at all times (see Figure 1). Median values (black lines) and interquartile range (dotted gray lines) for creatinine (left), Cystatin C (middle), and NGAL (right), determined in the longitudinal subgroup (n=59). Reference values for non-pregnant women defined by the manufacturers are depicted with gray intermittent lines. Significantly elevated creatinine, cystatin C, and NGAL levels compared to other time-points are indicated with asterisks above the graphs.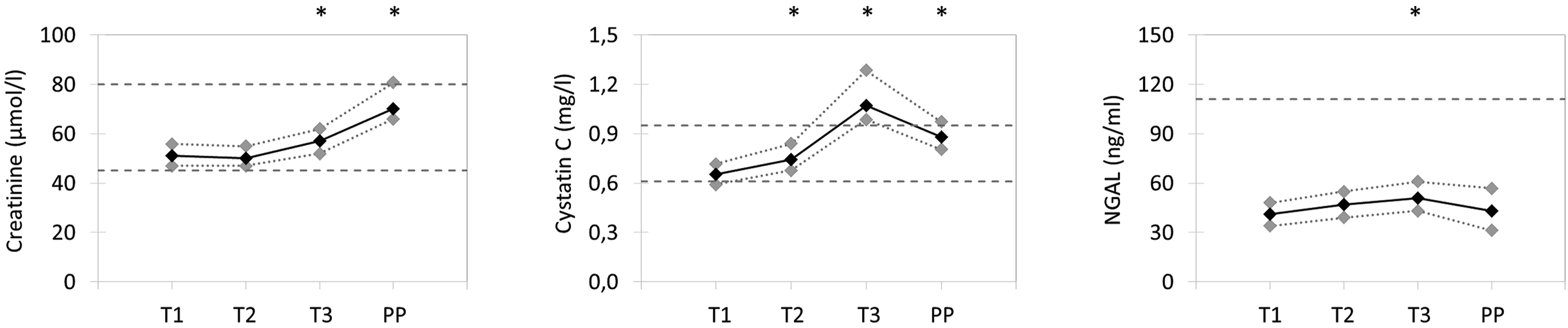
The Friedman test indicated that the overall longitudinal changes for all three markers were statistically significant (p<0.001). For creatinine, the Wilcoxon test showed that there was no significant difference between T1 and T2 (p = 0.02), but all other combinations of time-points showed a statistically significant difference (p < 0.001). CysC concentrations were statistically significantly different between all time-points (p < 0.001). NGAL levels at T1, T2, and PP were not statistically significantly different (p ≥ 0.19), but NGAL levels at T3 were significantly higher than all other time-points (p < 0.001).
Cross-sectional analysis
Upper reference limits (URL, 97.5 percentiles) plus 90% confidence intervals (CI) for creatinine, Cystatin C, and NGAL during pregnancy (T1-T3) and postpartum (PP), determined using the bootstrap method. The URL for non-pregnant women, as provided by the manufacturer, is indicated at the bottom.
Discussion
In this study, trimester-specific reference values for CysC and NGAL have been determined in women with uncomplicated pregnancy. The longitudinal analysis showed increasing levels of CysC during pregnancy, which decreased again postpartum. NGAL levels remained almost constant during pregnancy. Creatinine levels were low-normal in the first part of pregnancy, increased in the third trimester and were high-normal postpartum, in comparison to the reference interval for non-pregnant women. As creatinine, CysC, and NGAL have different concentration profiles during pregnancy, CysC and NGAL could provide different information than creatinine, and thus be of additional value to predict or diagnose kidney (related) disease.
Cystatin C
CysC is generally considered to be a better indicator of renal function than creatinine, since it is independent of age, sex and lean muscle mass and correlates better with mortality. 27 Several studies showed that calculation of the eGFR including CysC performed better than creatinine-based eGFR to determine kidney function in different populations.28–30 However, it is likely that CysC is unsuitable to calculate the eGFR in pregnant women, since the placenta produces (extra) CysC. 10 Indeed, several studies showed that CysC did not correlate with creatinine-based (e)GFR in pregnancy7,8 and CysC levels increase significantly during the third trimester in healthy pregnant women. 10 Nevertheless, CysC levels proved to be a good predictor of PE. Several studies showed that women with PE have higher CysC levels than healthy pregnant women,9–14 showing that it could serve as a diagnostic and predictive marker for PE.
With our method, a median value of 1.07 mg/L was found, which is concordant to the values found in a recent meta-analysis (0.55–1.22 mg/L). 10 However, in order to use CysC as a marker for PE, upper reference limits are needed and not median values. For our assay, we determined an upper reference limit of 1.61 mg/L for the third trimester.
Another limitation of the meta-analysis 10 is that they only analyzed data for the third trimester and did not provide longitudinal data. In our study, we also found significantly increased CysC levels during the second trimester and postpartum in comparison to the first trimester. The upper reference limits for women with uncomplicated pregnancy of T2, T3, and PP were higher than the manufacturer’s URL of 0.95 mg/L for non-pregnant women. The manufacturer’s URL for non-pregnant women will thus lead to many false-positive results if used to detect PE during pregnancy. Therefore, we advise the use of trimester-specific reference values for CysC.
Neutrophil gelatinase-associated lipocalin
In our population of women with uncomplicated pregnancies, the NGAL levels were relatively constant. NGAL is not a marker for kidney function such as creatinine, but for kidney injury, and is not expressed by the placenta. NGAL could therefore be a very useful marker to detect kidney injury in pregnant women, independent of kidney function. It was shown by multiple studies that NGAL levels are indeed elevated in pregnant women diagnosed with PE, 18 and in women who would develop PE later in pregnancy.18,21
A limitation of these studies is that they only used median values. As mentioned before, in order to use NGAL as a marker for PE, upper reference limits are needed and not median values. In our study, the URLs ranged from 84-88 ng/mL during pregnancy to 95 ng/mL postpartum. Those values are well below the manufacturer’s upper reference limit for women in EDTA samples (111 ng/mL). This might be explained by differences in population. Another possible explanation is that NGAL levels are lower during pregnancy due to the rise in plasma volume (hemodilution).
To our knowledge, the BioPorto assay for evaluation of NGAL levels has not been studied yet in pregnant women. Several CE-approved NGAL assays use the same anti-NGAL antibody and reagents. However, different analytical platforms are used. It is therefore important to validate each assay before use. For the Bioporto assay on the Cobas c501, we found that the manufacturer’s URL is too high for pregnant women, possibly leading to false-negative results. Therefore, we recommend using trimester-specific reference values for NGAL.
Limitations of this study are the low number of participants, which was mathematically overcome using the bootstrap method to determine upper reference values. Most of the women included were Caucasian (98%), so we were unable to determine possible ethnical differences in CysC and NGAL levels. The CysC and NGAL assays are not yet standardized, so the reference values we proposed will need to be verified in other labs.
In conclusion, CysC and NGAL show distinct patterns during pregnancy and add valuable extra information compared to creatinine, which is a suboptimal biomarker for kidney function during pregnancy. For clinical use in prediction and diagnosis of PE, suitable reference intervals are pivotal. We recommend to use trimester-specific reference intervals for CysC and NGAL in pregnant women.
Footnotes
Acknowledgments
We would like to thank Donna van den Herik for analyzing the samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Medical Research Ethics Committee (MREC) Brabant and the local MREC at Amphia Hospital exempted this study from the Medical Research Involving Human Subjects Act (WMO) since it did not meet the criteria for medical-scientific research and hence approved this study.
Guarantor
AJ.
Contributorship
AJ, TE and MG conceived the study, developed the protocol and gained ethical approval. AJ was involved in participant recruitment. ES and AJ searched literature. ML was responsible for analysis of the samples. ES performed the data analysis and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
