Abstract
Background
The Royal College of Pathologists of Australasia Quality Assurance Programs runs a Quality Assurance Program for the assessment of synovial fluid crystals. It provides aliquots of synovial fluid to various laboratories. The quality of specimens can deteriorate prior to being examined. We aimed to assess whether the addition of dimethyl sulfoxide (DMSO) to synovial fluid specimens helps maintain cellular morphology.
Methods
Synovial fluid specimens were obtained from 15 patients. Each specimen was aliquoted into 24 samples, with half having DMSO added at a concentration of 10%. For each specimen, six samples containing DMSO and six samples not containing DMSO were stored at−80°C and room temperature. Samples from each group were examined at 1, 2, 3, 6, 7 and 8 weeks.
Results
Conclusions
DMSO contributes to the maintenance of cellular morphology in synovial fluid when stored in frozen conditions.
Introduction
The accurate diagnosis of crystal-induced arthritis such as gout and calcium pyrophosphate arthritis is helped by the detection of the relevant crystals in synovial fluid. 1 There is room for improvement in the accurate identification of crystals. 2 Given that the sensitivity of detecting monosodium urate monohydrate crystals and calcium pyrophosphate dihydrate (CPPD) crystals in synovial fluid is approximately 70%, quality control is of great importance. 3
The Royal College of Pathologists of Australasia Quality Assurance Programs (RCPAQAP) runs a Quality Assurance Program (QAP) for assessment of synovial fluid, which helps to ensure the accurate identification of crystals. All laboratories in Australia which are involved in the examination of synovial fluid must partake in the QAP. Overall, approximately 130 laboratories partake in the QAP twice per year, most of which are located in Australia and New Zealand. Other countries include Singapore, the United Kingdom, France, the Netherlands and South Africa. During the 1 to 7 days between dispatch and receipt at the receiving laboratory, the quality of the synovial fluid specimens deteriorates. Feedback from participating laboratories was that synovial fluid specimens did not always closely resemble fresh synovial fluid specimens obtained in clinical practice because of degradation of the cells and increased cellular debris.
For the purposes of the QAP, synovial fluid specimens are collected into plain tubes (non-EDTA) and stored at −80°C as soon as possible. To be used in the QAP, two independent assessors must reach the same conclusion as to which crystals are present. Once deemed appropriate, the synovial fluid specimens are thawed and aliquoted into micro tubes (volume 25 μL–50 μL) before being distributed to laboratories for examination. Evidence-based methods to ensure the optimal preservation of synovial fluid pending final examination are scarce. 4 Previous studies assessing synovial fluid at various time points have done so using a variety of storage materials and temperatures. A study focusing on CPPD crystals showed that freezing synovial fluid is beneficial for maintaining CPPD crystal counts, as well as reducing the likelihood of cell clumping, which can impact upon the number of crystals seen. 5 A recent study looking at monosodium urate monohydrate crystals and CPPD crystals showed that refrigerating synovial fluid in heparin-containing tubes was favourable with respect to facilitating the delayed identification of crystals on microscopy. 6
Cryopreservation is used to maintain various biological constructs for prolonged periods of time. 7 The most commonly used agent for the cryopreservation of cells is dimethyl sulfoxide (DMSO), which is generally added at concentrations of 5–10%. 7 Cryoprotectants such as DMSO are effective at minimising cell damage in frozen biological systems, as biochemical reactions can occur down to−80°C. 7
In this study, we aimed to assess whether the addition of DMSO to synovial fluid specimens would contribute to the maintenance of cellular morphology, which influences the likelihood of correctly identifying the presence and type, or absence, of crystals.
Materials and methods
Ethics approval to undertake this study was granted by the Sydney Local Health District. All patients provided written informed consent.
Inclusion criteria
All patients were at least 18 years of age. All patients who required a synovial fluid joint aspiration for diagnostic or management purposes were considered for inclusion in the study, irrespective of their underlying condition. All synovial fluid joint aspirations were undertaken between June 2018 and January 2019 at either Royal Prince Alfred Hospital or the Private Practice of Assessor 1.
Processing and photographing of synovial fluid specimens
Following the collection of each synovial fluid specimen, a baseline examination was performed via microscopy using a Zeiss Axio Lab.A1 polarisation microscope with rotatable stage and EC Plan Neofluar 20x and 40x objectives. Photography was performed using a Jenoptik Gryphax NAOS Back Illuminated CMOS 1″ camera, allowing capture of an electronic image. Each synovial fluid specimen was then aliquoted into 24 samples using 0.5 mL polypropylene micro tubes and accompanying polypropylene screw caps, with half having DMSO added at a concentration of 10%. For each synovial fluid specimen, six of the samples containing DMSO and six of the samples not containing DMSO were stored at−80°C and room temperature. Samples from each group (i.e. Group 1 = DMSO at−80°C [DMSO−80°C], Group 2 = DMSO at room temperature [DMSOrtemp], Group 3 = no DMSO at−80°C [noDMSO-80°C] and Group 4 = no DMSO at room temperature [noDMSOrtemp]) were examined via microscopy and electronic images captured at sequential time points (i.e. at weeks 1, 2, 3, 6, 7 and 8). For each sample, one drop of synovial fluid was placed onto a glass slide and a cover slip was applied. A 40x objective was used and the microscope was linked to a computer and camera. The images were stored electronically. See Figure 1. Example of synovial fluid specimen at baseline examination and 3 weeks. These samples were derived from the same synovial fluid specimen; a) baseline examination, b) DMSO at −80°C at 3 weeks and c) no DMSO at −80°C at 3 weeks.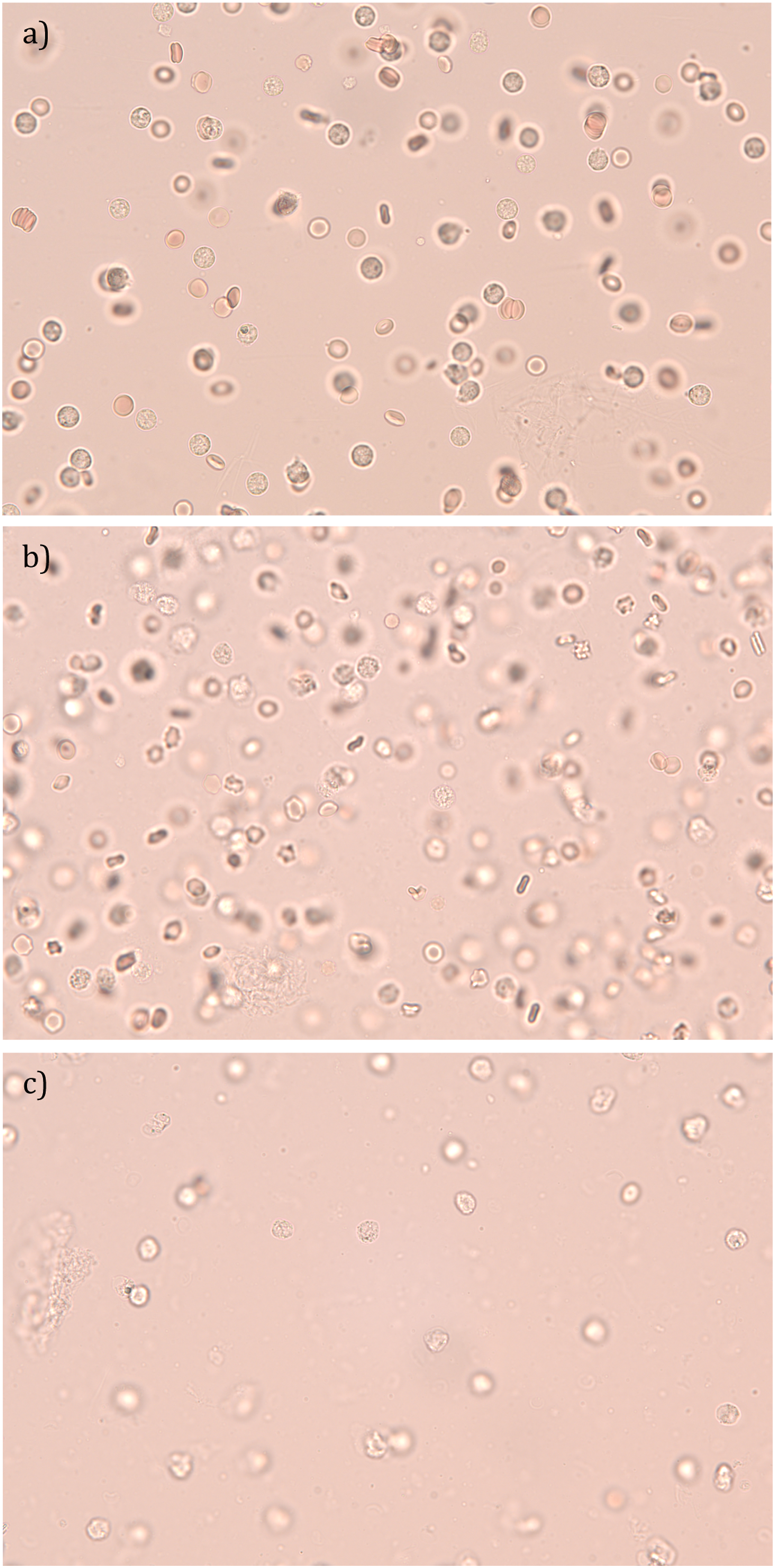
Assessment of synovial fluid specimens
Comparative analysis
For each synovial fluid specimen, the final remaining aliquoted samples containing DMSO and not containing DMSO which were stored at−80°C (generally for 8 weeks following collection) were directly compared to each other, to determine if one image looked “better”, or both images looked the “same”. Both Assessor 1 and Assessor 2 were blinded. The images were arranged in a random manner with the assistance of www.randomizer.org, which was used to generate random sequences of numbers.
Quantitative analysis
A system for grading cellular morphology and assessing the presence of artefacts and cellular clumping was developed and applied by two independent assessors. Assessor 1 was blinded, as he was not involved in the processing or photographing of the synovial fluid specimens. Assessor 2 was not blinded, as she was involved in the processing and photographing of the synovial fluid specimens.
A four-point system was applied for the grading of cellular morphology, with “1” being the best and “4” being the worst. Grade 1 = appearance in keeping with a “fresh” sample with intact cellular membranes. Grade 2 = appearance in keeping with partially ruptured cellular membranes. Grade 3 = appearance in keeping with severely ruptured cellular membranes, or invisible cellular membranes but identifiable cells. Grade 4 = appearance in keeping with cellular debris not confined to the shapes of cells.
A “yes” or “no” system was applied for assessing artefacts and cellular clumping.
Data analysis
For the paired nominal data in the Comparative Analysis, the McNemar-Bowker test was used.
For the continuous outcome variables, multiple regression analyses were performed, with the “groups” and “weeks” being the independent variables and the outcome variables being the dependent variables. For the binary outcome variables, logistic regression analyses were performed, with the “groups” and “weeks” being the independent variables and the outcome variables being the dependent variables. The analyses were performed within the framework of Generalised Estimating Equations, which corrects for any bias in the estimates due to having multiple observations per synovial fluid specimen.
All statistical tests were 2-tailed, with the significance level set at 0.05. All statistical analyses were conducted using SPSS Version 24, with the assistance of a statistician (MD’S).
Results
Comparative analysis
Assessor 1
A significant difference was found between samples containing DMSO and not containing DMSO which were stored at −80°C (p = .007), in favour of those containing DMSO.
Assessor 2
A significant difference was found between samples containing DMSO and not containing DMSO which were stored at −80°C (p = .007), in favour of those containing DMSO.
Assessor 1 and Assessor 2 (Combined)
A significant difference was found between samples containing DMSO and not containing DMSO which were stored at −80°C (p = .000), in favour of those containing DMSO.
Quantitative analysis
Assessor 1
Grading of cellular morphology
A significant difference was found according to “groups” (p = .003). DMSO-800C was found to have a lower (better) grade than DMSOrtemp (mean difference =−0.46, p = .000) and noDMSOrtemp (mean difference =−0.42, p = .005). noDMSO-800C was found to have a lower grade than DMSOrtemp (mean difference =−0.35, p = .001) and noDMSOrtemp (mean difference =−0.31, p = .015). No significant difference was found according to “weeks” (p = .308).
Assessing for artefacts
No significant difference was found according to “groups” or “weeks” (p = .465 and p = .294, respectively).
Assessing for cellular clumping
No significant difference was found according to “groups” or “weeks” (p = .374 and p = .610, respectively).
Assessor 2
Grading of cellular morphology
A significant difference was found according to “groups” (p = .000). DMSO−800C was found to have a lower (better) grade than DMSOrtemp (mean difference =−0.21, p = .027), noDMSO−80°C (mean difference =−0.34, p = .000) and noDMSOrtemp (mean difference =−0.27, p = .006). No significant difference was found according to “weeks” (p = .083).
Assessing for artefacts
No significant difference was found according to “groups” or “weeks” (p = .078 and p = .150, respectively).
Assessing for cellular clumping
A significant difference was found according to “groups” and “weeks” (p = .001 and p = .000, respectively). DMSOrtemp was found to have less cellular clumping than noDMSOrtemp (OR = 0.55, p = .011, 95% CI = −1.075 to −0.137). Further, Week 1 was found to have less cellular clumping than Week 6 (OR = 0.46, p = .037, 95% CI = −1.487 to −0.047) and Week 8 (OR = 0.40, p = .024, 95% CI = −1.691 to −0.121). Week 3 was found to have less cellular clumping than Week 6 (OR = 0.59, p = .040, 95% CI = −1.039 to −0.023), Week 7 (OR = 0.47, p = .023, 95% CI = −1.388 to −0.103) and Week 8 (OR = 0.51, p = .000, 95% CI = −1.005 to −0.335).
Assessor 1 and Assessor 2 (Combined)
Grading of cellular morphology
Assessor 1 and Assessor 2 (Combined) Grading of Cellular Morphology - Pairwise Comparisons for “Groups”.
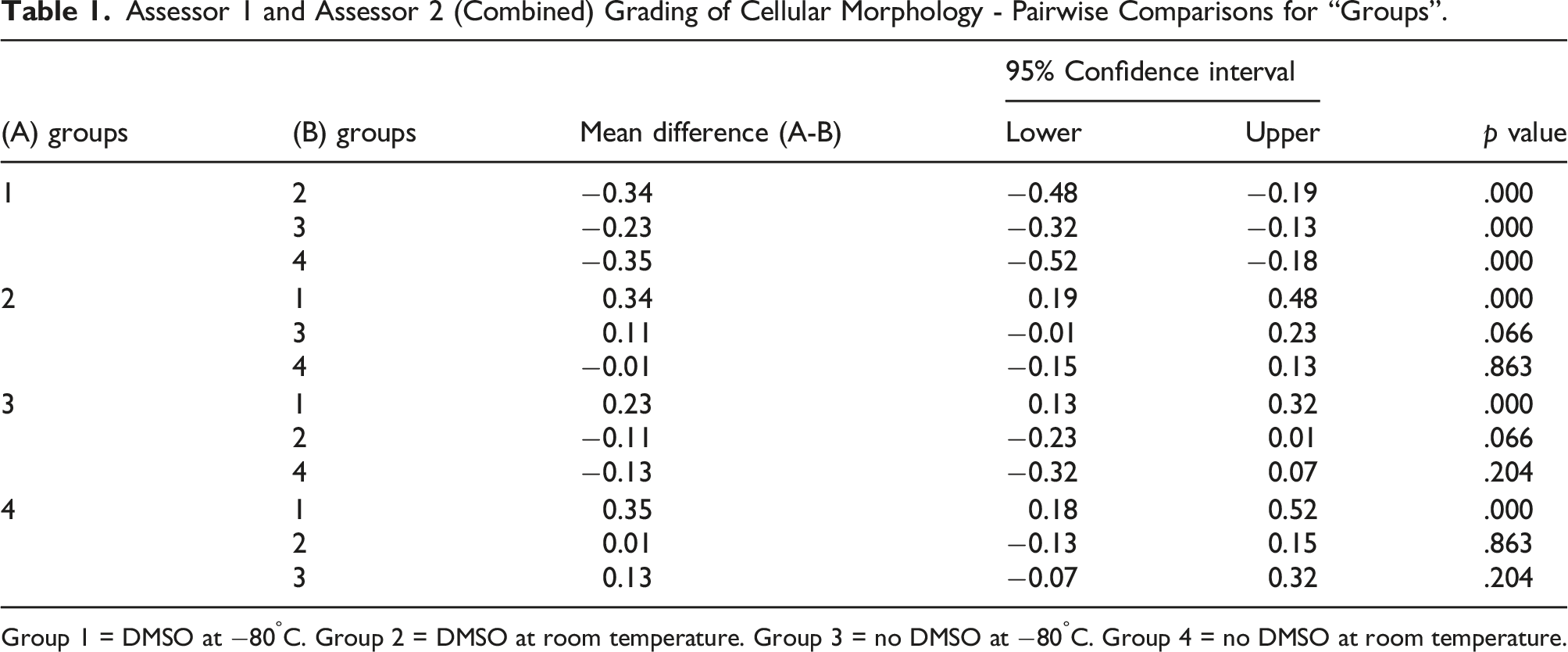
Group 1 = DMSO at −80°C. Group 2 = DMSO at room temperature. Group 3 = no DMSO at −80°C. Group 4 = no DMSO at room temperature.
Assessor 1 and Assessor 2 (Combined) Grading of Cellular Morphology - Pairwise Comparisons for “Weeks”.
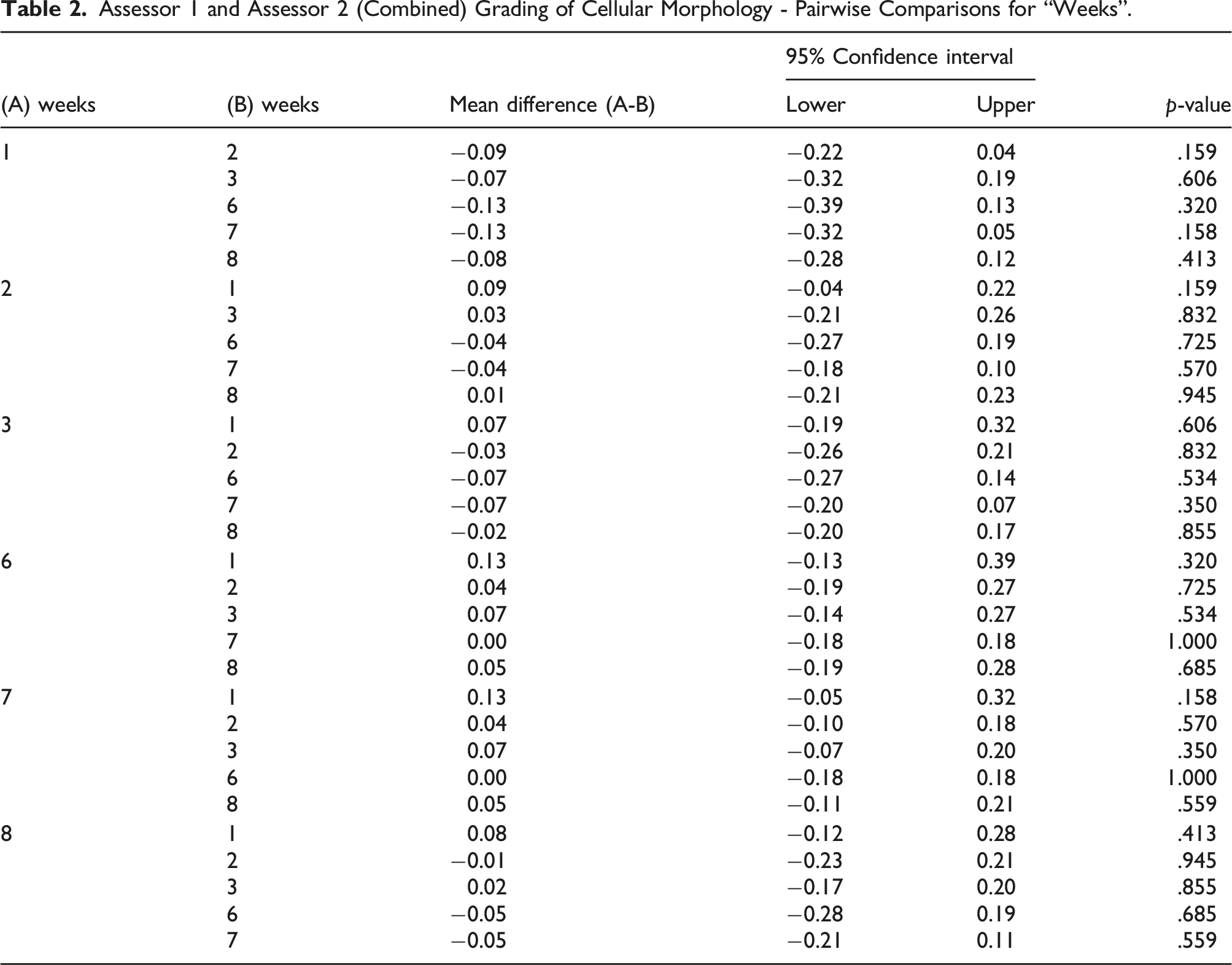
Assessing for artefacts
A significant difference was found according to “weeks” (p = .011). Week 1 (OR = 0.41, p = .001, 95% CI = −1.418 to −0.345), Week 2 (OR = 0.57, p = .008, 95% CI = −0.988 to −0.148), Week 3 (OR = 0.52, p = .012, 95% CI = −1.173 to −0.146) and Week 8 (OR = 0.62, p = .038, 95% CI = −0.934 to −0.026) were found to have less artefacts than Week 7. See Supplemental Table 1 and Supplemental Table 2.
Assessing for cellular clumping
No significant difference was found according to “groups” (p = .291). A significant difference was found according to “weeks” (p = .038). Week 3 was found to have less cellular clumping than Week 7 (OR = 0.52, p = .024, 95% CI = −1.205 to −0.087). See Supplemental Table 3 and Supplemental Table 4.
Level of agreement
The Kappa agreement between Assessor 1 and Assessor 2 for grading of cellular morphology, assessing for artefacts and assessing for cellular clumping was 0.1045, 0.1474 and 0.0523, respectively.
Discussion
Ideally, synovial fluid specimens should be examined as soon as possible following collection, as delays can lead to incorrect findings. Delay allows elements within synovial fluid to deteriorate, and artefacts can develop. 8 Where synovial fluid specimens are to be examined at a later date, the amount of time that can safely elapse without interfering with the accurate detection of crystals is uncertain. 3
In this study, significant differences were found with respect to the grading of cellular morphology according to “groups”. For the combined findings of Assessor 1 and Assessor 2, samples containing DMSO which were stored at −80°C had significantly lower (better) grades than those containing DMSO which were stored at room temperature, those not containing DMSO which were stored at −80°C and those not containing DMSO which were stored at room temperature. For samples containing DMSO which were stored at −80°C and samples not containing DMSO which were stored at −80°C, significant differences were found by Assessor 2 but not by Assessor 1. In the Comparative Analysis, which directly compared the final remaining aliquoted samples which were stored at −80°C, either containing or not containing DMSO, those containing DMSO looked significantly better according to Assessor 1, Assessor 2 and the combined findings of Assessor 1 and Assessor 2. Overall, these findings suggest that the addition of DMSO to synovial fluid specimens which are to be stored in frozen conditions is of benefit for preserving synovial fluid cellular morphology. To the best of our knowledge, this is the first time that DMSO has been shown to be of benefit in preserving synovial fluid. With further research, this could enhance the value of the QAP for the participating laboratories.
No drawback was detected with respect to the development of artefacts and cellular clumping from the addition of DMSO to synovial fluid specimens which are to be stored. Given the pilot nature of this study, there were limitations. The sample size was quite small and there were some missing data, with not all images captured for every possible group and time point for each synovial fluid specimen due to logistical issues. Further, some images were captured slightly behind schedule for their intended time point, again due to logistical issues. At one point, the microscope malfunctioned, and so images had to be captured at a different objective (20x) for part of the study. We do not believe that this setback altered the quality of the images enough to significantly affect the analysis. Finally, the Quantitative Analysis was not double blinded.
In conclusion, the addition of DMSO to synovial fluid specimens contributes to the maintenance of cellular morphology when stored in frozen conditions. This could improve the likelihood of correctly identifying the presence or absence of crystals, with benefit to the ongoing long-term (more than 20 years) RCPAQAP Synovial Fluid QAP and to future research projects reliant upon the examination of synovial fluid.
Supplemental Material
sj-pdf-1-acb-10.1177_00045632221076349 – Supplemental material for The preservation of synovial fluid using dimethyl sulfoxide
Supplemental material, sj-pdf-1-acb-10.1177_00045632221076349 for The preservation of synovial fluid using dimethyl sulfoxide by Katrina Pavic, Vicki McGill, Mario D’Souza and Neil McGill in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethics approval to undertake this study was granted by the Sydney Local Health District.
Guarantor
NM.
Contributorship
NM and VM conceived the study. KP researched the literature. NM and VM were involved in gaining ethical approval. NM and KP were involved in protocol development. NM and KP were involved in patient recruitment. KP conducted the experiment. MDS, NM and KP were involved in data analysis. KP wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
