Abstract
Background
Small dense low-density lipoprotein a smaller and denser subfraction among whole low density lipoprotein particles is known to be highly atherogenic. The reference interval (RI) is not strictly defined for serum concentration of sdLDL-cholesterol (sdLDL-C) in Japan. The purpose of this study is to set the reference interval for sdLDL-C in healthy subjects.
Methods
The population of this cross-sectional study was consisted of 40,862 individuals who had annual health checkups, and healthy subjects were extracted based on exclusion criteria such as medical history, social history, and blood sampling test results. Their serum sdLDL-C values were statistically analyzed and the RIs were set in men, premenopausal women, and postmenopausal women separately.
Results
The mean values of serum sdLDL-C in healthy subjects were 23.9 mg/dL in men, 20.0 mg/dL in premenopausal women and 23.7 mg/dL in postmenopausal women, and the RIs were 12.6–45.3 mg/dL in men, 11.4–35.1 mg/dL in premenopausal women and 14.6–38.6 mg/dL in postmenopausal women. Serum sdLDL-C values were significantly higher in men than in women. Besides, sdLDL-C values were significantly higher in postmenopausal women than in premenopausal women. In both genders, sdLDL-C values tended to increase with age.
Conclusion
These results suggest that the RIs for sdLDL-C are recommended as follows: 13–45 mg/dL in men, 11–35 mg/dL in premenopausal women, and 15–39 mg/dL in postmenopausal women, respectively. Aside from these RIs, it is also necessary to define clinical cutoff values graded according to individual risk levels for atherosclerotic cardiovascular diseases.
Introduction
Atherosclerotic cardiovascular diseases (ASCVDs) including coronary heart disease (CHD) and cerebrovascular disease are one of leading causes of death worldwide, and the prevention and management of ASCVDs become increasingly important. One of risk factors for ASCVDs, especially for CHD is dyslipidemia. An increase in low density lipoprotein (LDL) cholesterol (LDL-C) is recognized as an important risk factor. Even though a lot of efforts have been made for decreasing LDL-C levels, a part of patients with dyslipidemia come to the development of CHD. In a certain number of these patients, their serum LDL-C levels are controlled under the target levels. 1 LDL is classified into some subfractions according to its size and density, and the smaller and denser one is called small dense LDL (sdLDL). A sdLDL particle does not easily bind to LDL receptors, and consequently it stays in arterial walls for a long time. In addition, it is easily denatured into oxidized LDL due to its scarce antioxidants, leading to the formation of atherosclerosis.2-6 It has been reported in Suita study and Hisayama study that high sdLDL-cholesterol (sdLDL-C) levels are significantly associated with the onset of CHD, and sdLDL-C is considered to be an important predictive marker for CHD in Japanese population.7-9 A homogenous assay for the measurement of sdLDL-C has been developed, and moreover the measurement has become convenient by an automatic analyzer application.2-4 This assay kit has already received the certification by Food and Drug Administration (FDA) as an in vitro diagnostic test reagent in the United States. However, while the importance of sdLDL-C evaluation is recognized through Japanese clinical trials, it has not yet been approved as an in vitro diagnostic reagent in Japan. In addition, no consensus has been made yet for standard values or reference intervals (RIs) for serum sdLDL-C concentrations.
In this study, we aimed to examine the RIs for serum sdLDL-C concentration in healthy Japanese subjects with reference to the collaborative derivation of RIs for major clinical laboratory tests by the Japanese Committee for Clinical Laboratory Standard (JCCLS). 10
Methods
Subjects
The present cross-sectional study population consisted of 40,862 individuals over 20 years old who received medical examinations at the health evaluation centers in Osaka, Japan, between April of 2016 and March of 2019. All subjects agreed to participate in this study and the study protocol had previously been approved by the Medical Research Ethics Committee of Tokyo Medical and Dental University, Tokyo, Japan (M2000-2209). In subjects who visited more than once during the study period, data at the first visit was used for this study.
Measurements
Past medical history and social history of each subject were investigated by self-reporting questionnaire. Blood samples were collected from all subjects after an overnight fast. SdLDL-C was measured automatically using a homogeneous assay kit (Denka Co., Ltd, Japan) on TBA-FX8 instruments (Canon Medical Systems, Japan), as described previously. 2 Serum total protein (TP), albumin (Alb), uric acid (UA), aspartate transaminase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH), γ-glutamyl-transferase (GGT), creatine kinase (CK), C-reaction protein (CRP), total cholesterol (TC), triglycerides (TG), direct LDL-C and high-density lipoprotein cholesterol (HDL-C) were also measured by an automated standardized enzymatic analysis. Plasma glucose was measured using the hexokinase G-6-PDH methods (FUJIFILM Wako Pure Chemical CO., Japan), and HbA1c was measured using high performance liquid chromatography on HL-723 G9 instruments (Tosoh, Japan). White blood cell count (WBC), hemoglobin (Hb) and platelet (Plt) were measured on DxH800 (BECKMAN COULTE R, USA). All assays had within and between run coefficients of variation< 5.0%. Non-HDL-C was calculated by subtracting HDL-C from TC. Globulin (Glb) was calculated by subtracting albumin from total protein.
Exclusion criteria for healthy subjects
We have set 10 exclusion criteria on the following data of medical and social history and laboratory tests, and subjects who did not meet any criteria were defined as reference individuals, namely “healthy subjects” in the present study. 1. Body Mass Index (BMI) ≥ 28 kg/m2 This criterion was set based on the exclusion criteria of the collaborative derivation of RIs for major clinical laboratory tests by JCCLS.
10
. 2. Average daily drinking (ethanol equivalent) ≥ 25 g/day 3. Current smoker 4. Under treatment for any disease 5. Past histories of hepatitis B or C 6. Dyslipidemia (LDL-C ≥ 140 mg/dL or/and triglycerides ≥ 150 mg/dL or/and high-density lipoprotein cholesterol (HDL-C) < 40 mg/dL or/and non-HDL-C ≥ 170 mg/dL) 7. Diabetes (Fast plasma glucose ≥ 126 mg/dL and/or HbA1c ≥ 6.5%) 8. Hypertension (systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg) 9. Based on the latent abnormal values exclusion (LAVE) method
10
on the collaborative derivation of RIs, those subjects having two or more outside the respective RIs in the following items; Alb (4.1–5.1 g/dL), Glb (2.2–3.4 g/dL), UA (male (M) 3.7–7.8 mg/dL, female (F) 2.6–5.5 mg/dL), AST (13–30 U/L), ALT (M 10–42 U/L, F 7–23 U/L), LDH (124–222 U/L), GGT (M 13–64 U/L, F 9–32 U/L), CK (M 59–248 U/L, F 41–153 U/L), CRP (0–0.14 mg/dL) 10. Those subjects having one or more outside the respective RIs in the following items: WBC (3300–8600 /μL), Hb (M 13.7–16.8 g/dL, F 11.6–14.8 g/dL), Plt (158–348 103/μL)
Subjects who were pregnant, within 1 year after delivery, within two weeks after admission for surgery or acute illness, or HIV carrier should be excluded, but could not be determined from the date obtained in this study. However, it is considered that they can be almost excluded by the above criteria.
Subjects with metabolic syndrome or with history of myocardial infarction or angina
To examine the values of sdLDL-C in subjects at high risk for atherosclerosis, we selected subjects with metabolic syndrome and subjects who were undergoing treatment or have histories of myocardial infarction (MI) or angina. Metabolic syndrome was diagnosed by meeting the following conditions: being two or more positive about the criteria for lipids (TG ≥150 mg/dL and/or HDL-C ≤ 40 mg/dL), blood pressure (sBP ≥ 130mmHg and/or dBP ≥ 85mmHg) and fast plasma glucose ≥ 110 mg/dL are met in addition to the criteria for waist circumstance (≥ 85 cm for men, ≥ 90 for women) according to the definition of the metabolic syndrome for the Japanese announced by the Japanese Society of Internal Medicine. 11
Statistical Analysis
Since the lipid profile varies depending on differences in gender and age, sdLDL-C was examined by gender and age groups.3,7 The average age of menopause for Japanese women was reported to be 52.1 years, and consequently female subjects were divided into two groups with 55 years as the age of menopause. 12
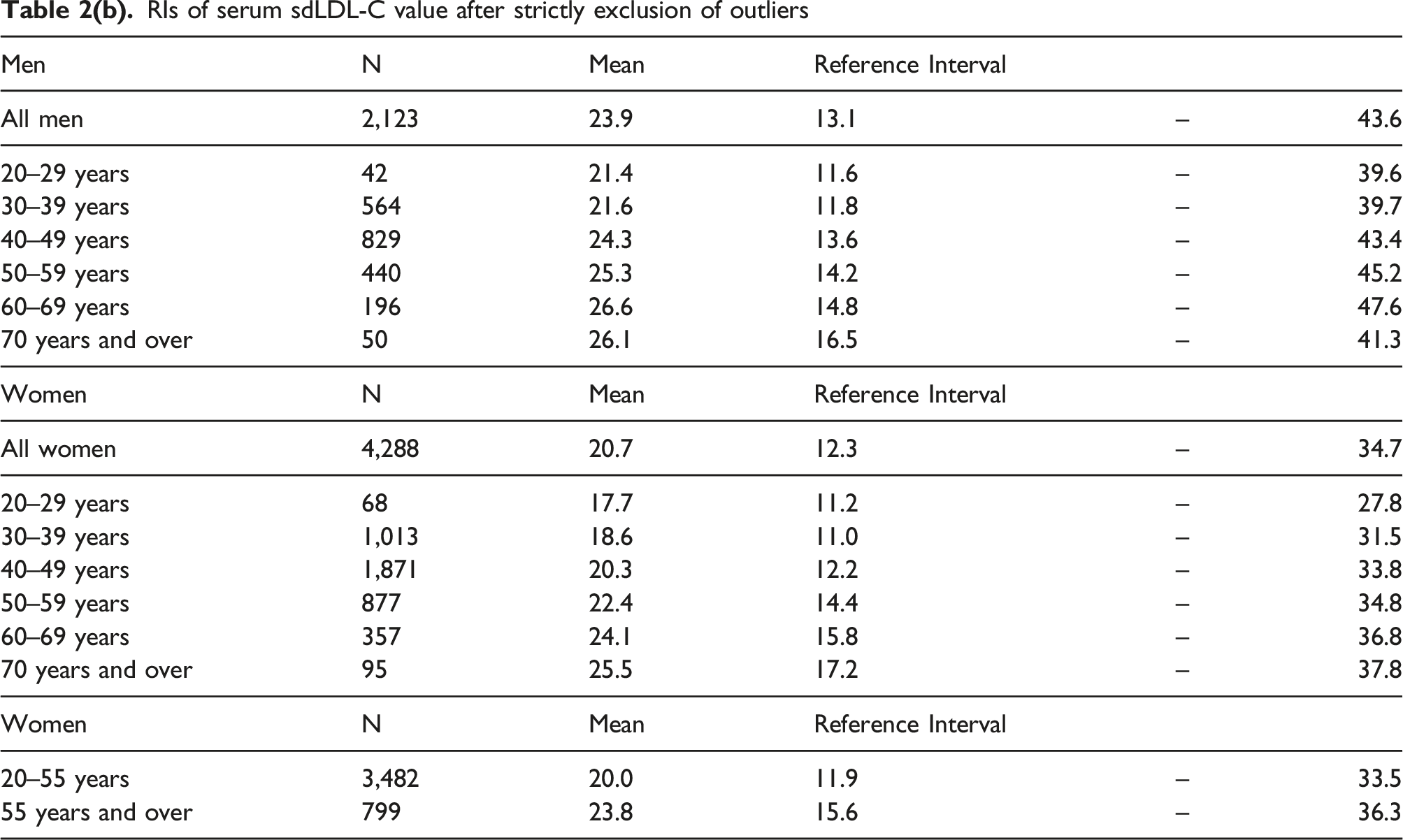
After normalizing data of sdLDL-C levels and sdLDL-C/LDL-C ratios through the logarithmic conversion, the mean and standard deviation (SD) were calculated, and the RIs (mean ±1.96 SD) were determined in accordance with the method recommended by Japan Society of Clinical Chemistry. 10 The RIs were expressed after restoring logarithmic data to real numbers. In addition, to strictly exclude outliers, samples exhibiting measured values outside the mean ±2.58 SD were excluded, and the new mean and SD were calculated. This process was repeated until exception data were not found any longer. 13 In addition, 2.5–97.5 percentiles of sdLDL-C and sdLDL-C/LDL-C as the reference intervals by a non-parametric analysis were calculated.
To examine whether the reference range of sdLDL-C should be set by gender and age, we calculated SD ratio (SDR), corresponding to the SD due to between-subgroup variations divided by SD representing the RI (corresponding to the between-individual SD).10,14 SDR ≥ 0.3 was regarded as indicating apparent between-subgroup differences. 10
Comparisons between the two groups, such as gender differences and pre- and post-menopausal differences in women, were tested using unpaired t-test. And, the Jonckheere-Terpstra test was performed for age-specific trends. The data of the characteristics of three subgroups, healthy subjects, subjects with histories of MI or angina and subjects with metabolic syndrome, were tested by the Kruskal–Wallis test and the p values were calculated with Bonferoni’s correction. All statistical analyses were performed using SPSS software, version 23 and 28 (IBM, Armonk, NY). A p value of <0.05 was considered statistically significant.
Results
(a) Baseline characteristics of the study population in men.
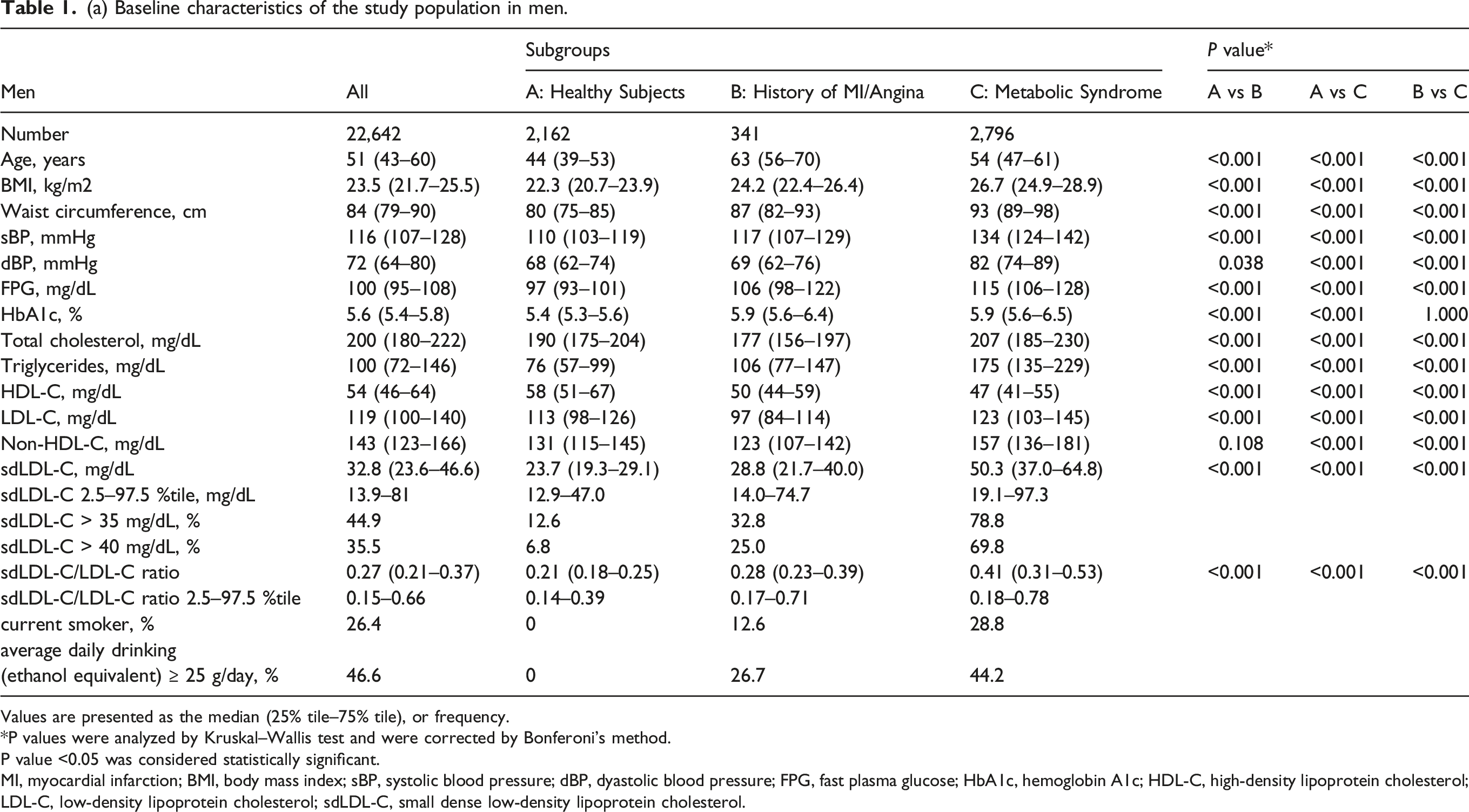
Values are presented as the median (25% tile–75% tile), or frequency.
*P values were analyzed by Kruskal–Wallis test and were corrected by Bonferoni's method.
P value <0.05 was considered statistically significant.
MI, myocardial infarction; BMI, body mass index; sBP, systolic blood pressure; dBP, dyastolic blood pressure; FPG, fast plasma glucose; HbA1c, hemoglobin A1c; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; sdLDL-C, small dense low-density lipoprotein cholesterol.
(b) Baseline characteristics of the study population in women.

Values are presented as the median (25% tile–75% tile), or frequency.
*P values were analyzed by Kruskal–Wallis test and were corrected by Bonferoni’s method.
P value <0.05 was considered statistically significant.
MI, myocardial infarction; BMI, body mass index; sBP, systolic blood pressure; dBP, dyastolic blood pressure; FPG, fast plasma glucose; HbA1c, hemoglobin A1c; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; sdLDL-C, small dense low-density lipoprotein cholesterol
Reference Intervals for serum sdLDL-C value (a) RIs of serum sdLDL-C value.
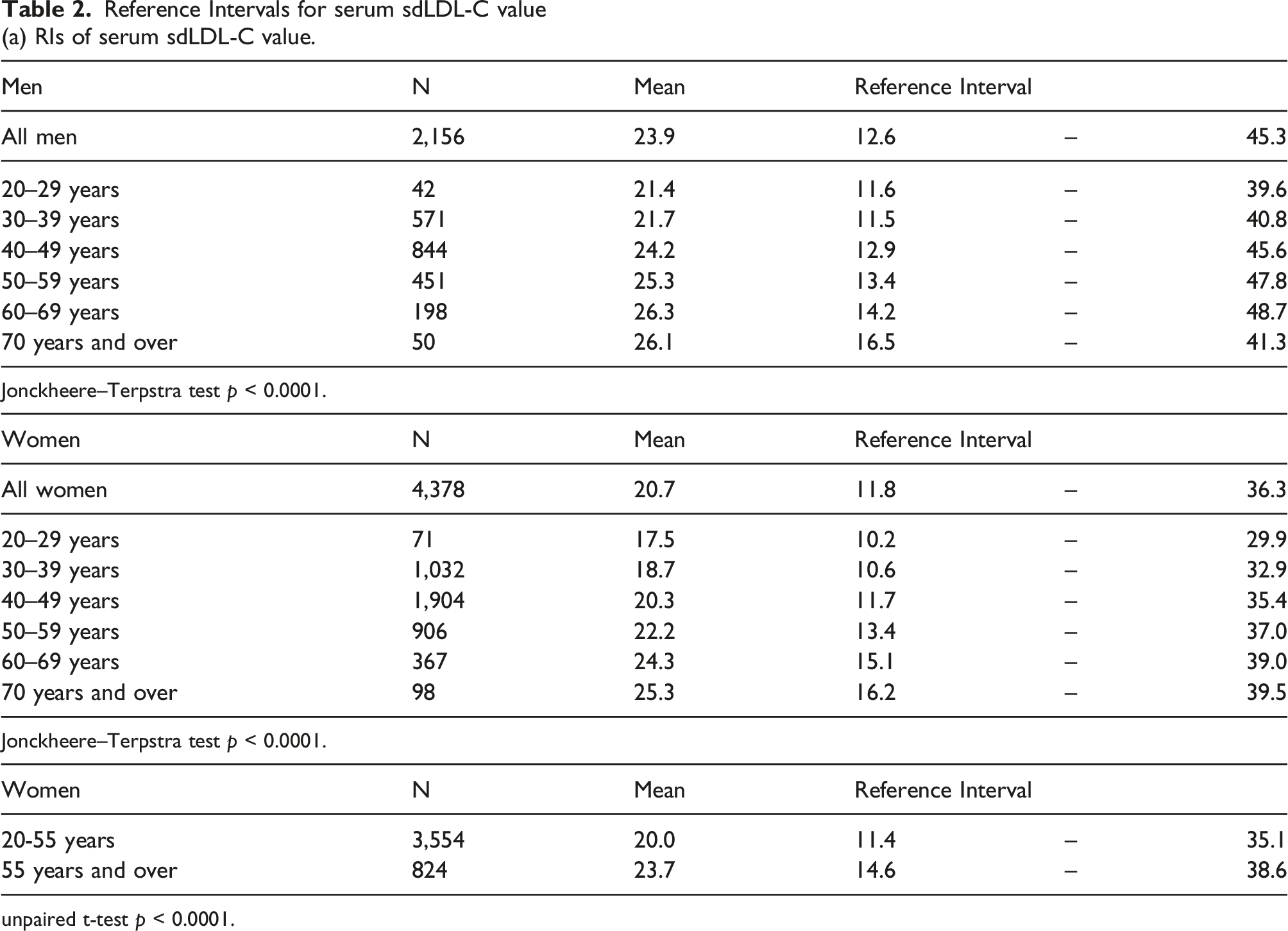
unpaired t-test p < 0.0001.
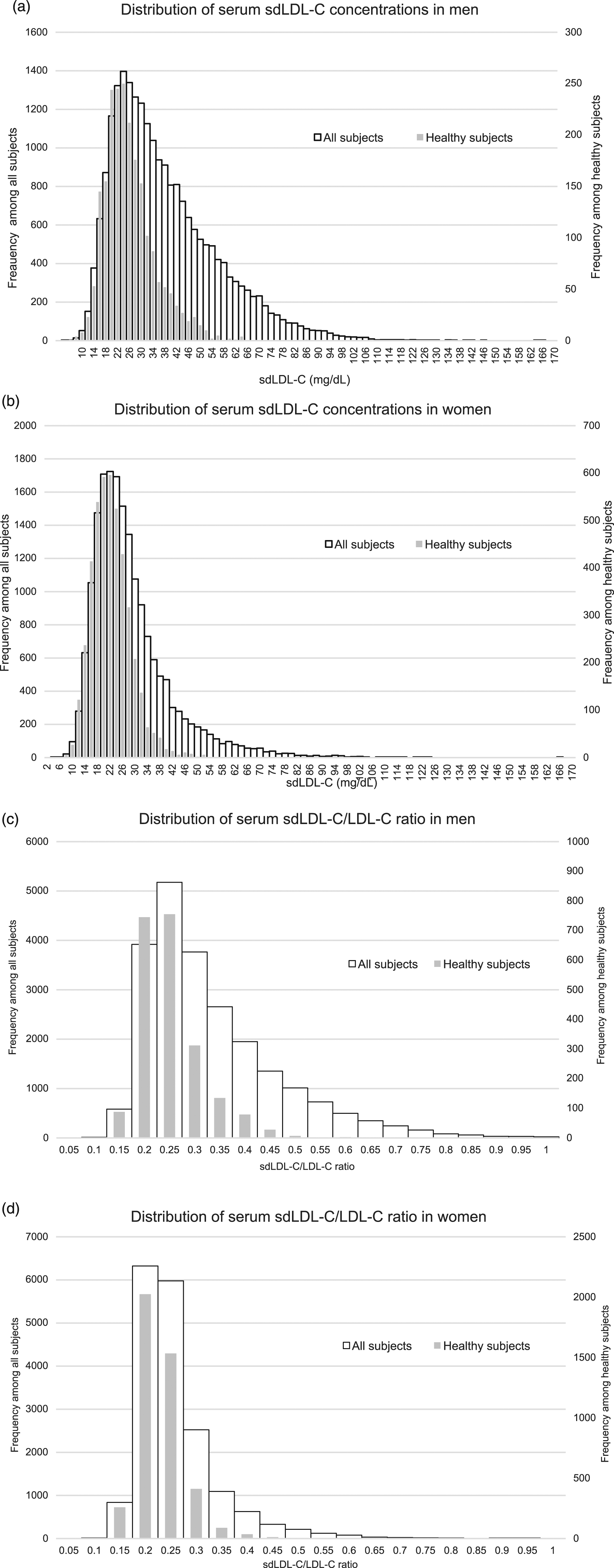
Distribution of serum sdLDL-C and sdLDL-C/LDL-C ratio concentrations. Panels (a) and (c) are shown for distributions in men, and (b) and (d) are in women, respectively. The gray bar part means the distributions of healthy subjects, and the white bar part means the distributions of all subjects.
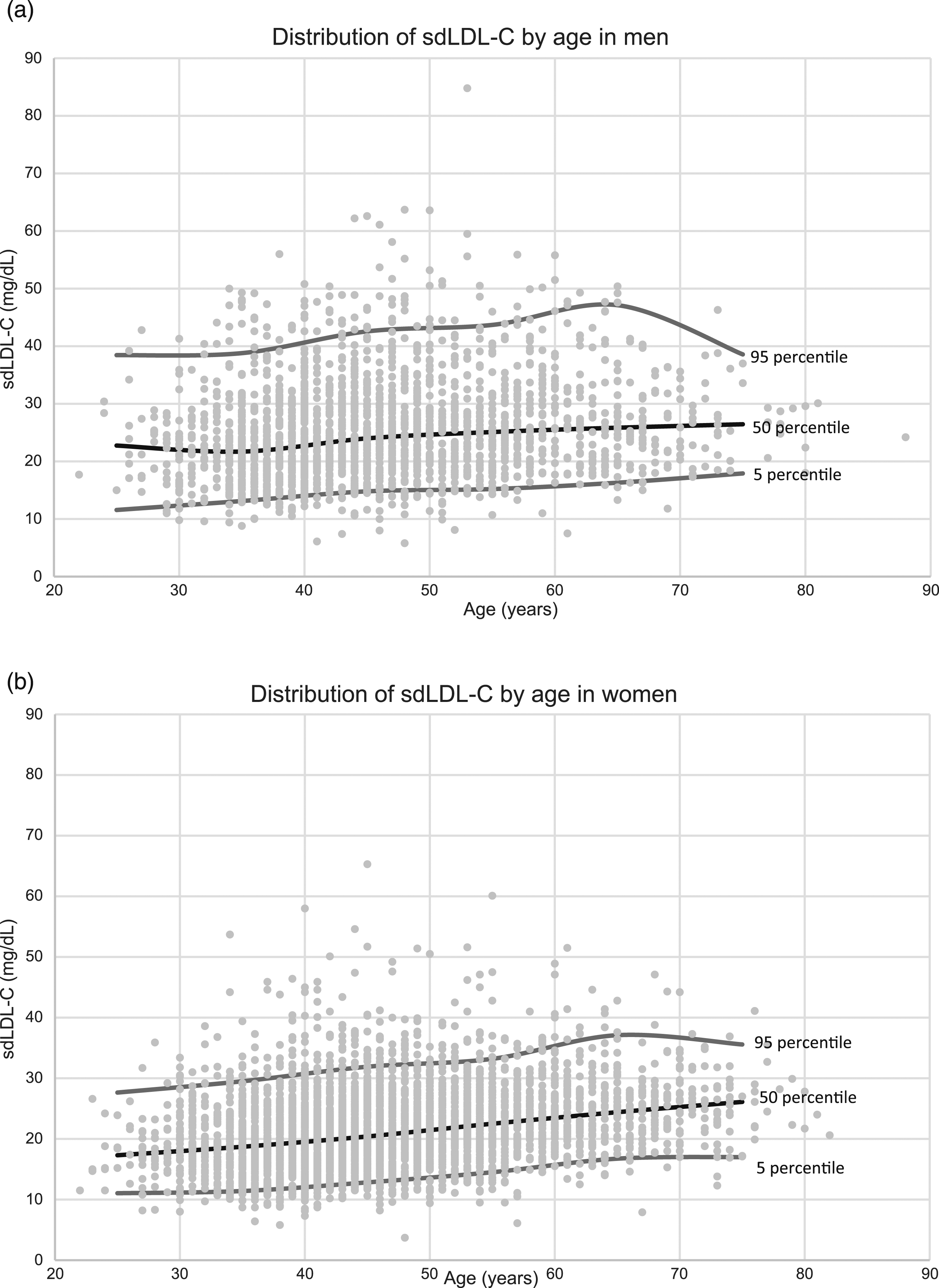
Serum sdLDL-C concentrations by age in healthy subjects. Panels (a) and (b) are shown for serum sdLDL-C concentrations in men and women, respectively.
RIs of serum sdLDL-C value after strictly exclusion of outliers
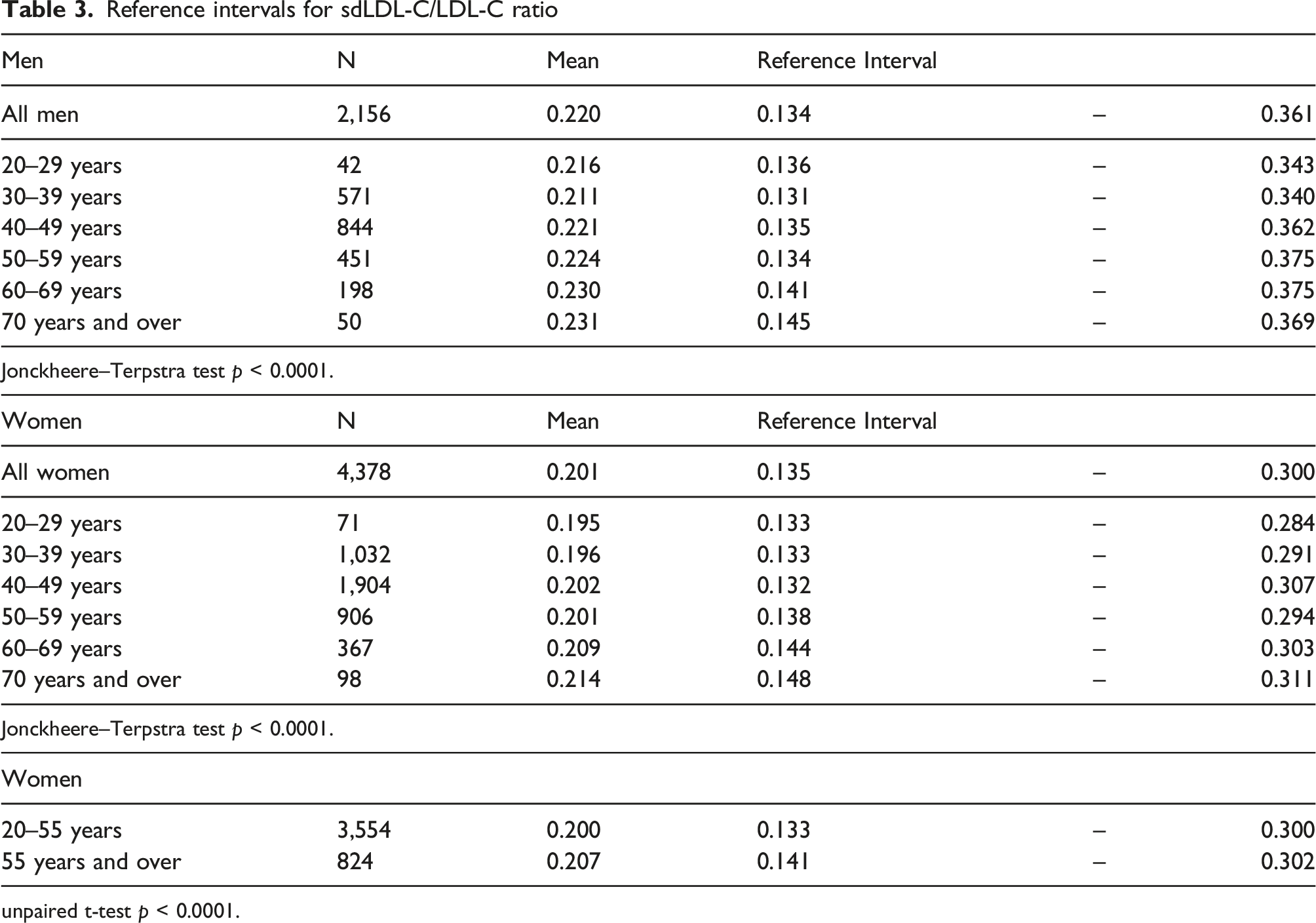
Reference intervals for sdLDL-C/LDL-C ratio
unpaired t-test p < 0.0001.
Regarding menopausal status, the mean value of sdLDL-C was significantly higher in postmenopausal women (55 years old and over, 23.7 mg/dL) than in premenopausal women (under 55 years old, 20.0 mg/dL), as shown in Table 3. The RI was 11.4–35.1 mg/dL for premenopausal women and 14.6–38.6 mg/dL for postmenopausal women. The SDR before and after menopause was 0.43. The sdLDL-C/LDL-C ratio was also significantly higher in postmenopausal women (0.207) than in premenopausal women (0.200), as shown in Table 3.
Subjects who met the criteria for metabolic syndrome and had histories of MI or angina were older than healthy subjects. Both sdLDL-C and sdLDL-C/LDL-C ratio were significantly higher in subjects with histories of MI or angina and subjects with metabolic syndrome than in healthy subjects, as were other cardiovascular disease risk factors (Table 1).
Discussion
In this study, we examined the distribution of serum sdLDL-C values among healthy subjects in Japan. The mean value of sdLDL-C was 23.9 mg/dL for men and 20.7 mg/dL for women, which was significantly higher in men than in women, and the serum sdLDL-C value increased with age in both genders, even in healthy subjects. These results were similar to the results of previous studies in Japanese.15,16 On the other hand, in the Korean study, the median value of sdLDL-C in the healthy population was 33.8 mg/dL in men and 24.6 mg/dL in women, which was higher than the results of the present study. 17 In the Korean study, the definition of healthy population may be less strict than in the present study, which might affect the results.
Exclusion criteria for the selection of healthy subjects in this study were set according to the JCCLS method. 10 In the exclusion criteria of JCCLS, the cutoff for obesity was set at BMI 28. Although this is a loose criterion for obesity, JCCLS reasoned that the cutoff at BMI 25 would exclude more than half of middle-aged men and that other exclusion criteria, such as medication history, would exclude subjects with abnormalities. In our study, when subjects with BMI ≥ 25 kg/m2 were excluded, approximately 300 men and 150 women were additionally excluded, but the mean and RI of serum sdLDL-C value remained the same as when BMI 28 kg/m2 was used as the cutoff. This may be due to the strict exclusion of subjects with abnormal values by criteria other than BMI.
Since SDR for gender was 0.320, it is considered appropriate to set RI for each gender, separately. SDRs by age group were 0.26 for men and 0.51 for women. Moreover, SDR was 0.43 between premenopausal and postmenopausal women. Based on these results, it is not meaningful for men to set RI by age group, while for women, it is necessary to stratify RI by age group or menopausal status. Therefore, in this study the subjects were divided into three groups: men, premenopausal women and postmenopausal women, and set the RIs separately. Overall, our results indicate that the RIs of sdLDL-C in healthy subjects are recommended as follows: 13–45 mg/dL for men, 11–35 mg/dL for premenopausal women and 15–39 mg/dL for postmenopausal women.
The mean value of serum sdLDL-C concentration in healthy subjects did not differ between before and after excluding outliers, and the RI became slightly narrower after excluding outliers by 2 mg/dL. Since the individuals with abnormalities affecting sdLDL-C levels were excluded according to the criteria, only small number of subjects showed outliers.18-20 As a result, the effect of exclusion of outliers was quite small. Moreover, the 2.5–97.5th percentiles of sdLDL-C or sdLDLC/LDLC ratio obtained in non-parametric analysis were almost the same as the results of analysis using normalizing data through the logarithmic conversion.
The sdLDL-C/LDL-C ratio is another index that correlates with ASCVD. 3 In the current study, the sdLDL-C/LDL-C ratio was significantly higher in men than in women and higher in high-age groups. Women have a lower risk of developing MI and cardiovascular death than men, but such gender difference decrease with age. 21 The results of this study revealed that being male and aging affect not only the quantitative increase in sdLDL-C but also the qualitative change in LDL.
In the Hisayama study, a prospective analysis for 8 years, subjects with high sdLDL-C (fourth quartile, sdLDL-C ≥ 43.7 mg/dL) compared with subjects with low sdLDL-C (first quartile, sdLDL-C < 24.4 mg/dL) has been reported to increase the CHD risk 5.41 times. 9 Moreover, it has been reported that the CHD risk is 2.09 times higher in those whose serum sdLDL-C levels are over 35 mg/dL than in others. 8 The upper limits of RIs of serum sdLDL-C levels that we set in this study in men and postmenopausal women were higher than 35 mg/dL. Therefore, the individuals whose serum sdLDL-C levels are over 35 mg/dL have a higher risk for ASCVD, even their sdLDL-C levels are within the RIs. In this study, the values of sdLDL-C in subjects with metabolic syndrome were significantly higher than those in healthy subjects. It showed similar trends of previous reports in Japanese and Korean subjects.15-17 In subjects with histories of MI or angina, the values of sdLDL-C were higher than those in healthy subjects, but lower than those in subjects with metabolic syndrome. The serum values of LDL-C in subjects with history of MI or angina were lower than those in healthy subjects. This may be attributed to presumable conditions that subjects with histories of MI or angina have been already receiving therapeutic interventions such as statins. According to the self-reports, about half of the subjects with histories of MI or angina were not on medication for dyslipidemia. The median of serum LDL-C and sdLDL-C were 103 mg/dL and 29.6 mg/dL in men without medication for dyslipidemia, and 93 mg/dL and 28.0 mg/dL in men with medication for dyslipidemia, and serum LDL-C values were lower in subjects with MI or angina regardless of the medication for dyslipidemia than in healthy subjects, and the sdLDL/LDL-C ratio was 0.28. Because of self-reported medication histories, there was a possible misidentification or a lack of information that the study subjects did not recognize statins as a lipid-lowering treatment, or that they were using drugs other than statins. In men, 45% of the total population, 13% of healthy subjects and 79% of subjects with metabolic syndrome, and in women, 20% of the total population, 3.1% of healthy subjects, and 70% of subjects with metabolic syndrome, had sdLDL-C levels above 35 mg/dL, a cutoff value proposed in the Hisayama study.8,9 The sensitivity was high in subjects with metabolic syndrome who are at high risk of cardiovascular disease. However, the sdLDL-C levels were higher than the cutoff value in about half of all male subjects so that the cutoff value may lead to overdiagnosis (Table 1). Since our analysis was not based on any cohort or clinical study on the prevention of ASCVD, the cutoff values of serum sdLDL-C level regarding risk assessment and/or prevention for ASCVD needs to be considered separately in the future.
The Japan Atherosclerosis Society guidelines for prevention of atherosclerosis 21 set diagnostic criteria for dyslipidemia, and the Japan Society of Ningen Dock (extended health check-ups) also sets the criteria for health screening.22,23 As with other lipids, it is necessary to set standard values, RIs and diagnostic values for serum sdLDL-C concentration. Meanwhile, the current study provided these values for the first time in Japan.
In this study, we examined RIs of the sdLDL-C value using the data of over 6000 healthy Japanese subjects selected by strict criteria, which is the strength of our study. On the other hand, our study has some limitations. Our study was cross-sectional in nature. Another shortcoming was that our data such as past medical history, smoking habits and drinking habits were collected based on self-reporting questionnaires, and therefore, subjects may have underreported these habits. Moreover, since the data were collected in only one regions of Japan, the results might differ in other regions of Japan.
Conclusion
Overall, these results indicate that the RIs for sdLDL-C are recommended as follows: 13–45 mg/dL in men, 11–35 mg/dL in premenopausal women and 15–39 mg/dL in postmenopausal women. Beyond these RIs, it is also necessary to define clinical cutoff values graded according to individual risk levels for ASCVD.
Supplemental Material
sj-xlsx-1-acb-10.1177_00045632211066334 – Supplemental Material for Reference interval for serum concentration of small dense low-density lipoprotein cholesterol in the healthy Japanese population
Supplemental Material, sj-xlsx-1-acb-10.1177_00045632211066334 for Reference interval for serum concentration of small dense low-density lipoprotein cholesterol in the healthy Japanese population by Marie Nakamura, Yasushi Yamamoto, Wataru Imaoka, Toshio Kuroshima, Ryoko Toragai, Yasuki Ito, Hiroshi Yoshida and Masumi Ai in Annals of Clinical Biochemistry
Supplemental Material
sj-xlsx-2-acb-10.1177_00045632211066334 – Supplemental Material for Reference interval for serum concentration of small dense low-density lipoprotein cholesterol in the healthy Japanese population
Supplemental Material, sj-xlsx-2-acb-10.1177_00045632211066334 for Reference interval for serum concentration of small dense low-density lipoprotein cholesterol in the healthy Japanese population by Marie Nakamura, Yasushi Yamamoto, Wataru Imaoka, Toshio Kuroshima, Ryoko Toragai, Yasuki Ito, Hiroshi Yoshida and Masumi Ai in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
The contribution of all subjects in this study is gratefully acknowledged and gratefully appreciated.
Declarations of conflicting interests
YI is an employee of Denka Co., Ltd, MA is supported by a research grant from Denka Co., Ltd, Tokyo, Japan. HY received honoraria for speaking activities from Denka. Other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the university research fund (No. 32,782) and the research grant (No. 2A232) from Denka Co., Ltd, Tokyo, Japan to MA.
Ethical approval
All subjects agreed to participate in this study and the study protocol had previously been approved by the Medical Research Ethics Committee of Tokyo Medical and Dental University, Tokyo, Japan (M2000-2209). This study was performed according to the Declaration of Helsinki.
Guarantor
MA.
Contributorship
MN, YI, HY and MA designed and conducted the study. YY, WI, TK and RT recruited study subjects and collected data. MN and MA analyzed the data. MN, YI, HY and MA discussed and wrote the manuscript. YI, an employee of Denka Co., Ltd, wasn’t involved in the measurement and the analysis of the clinical data. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
