Abstract
Background
In samples from patients administered rasburicase, ex vivo uricolysis leads to spuriously low uric acid results. The manufacturer’s recommendation of storing the sample in ice-water until analysis, however, does not fully arrest uricolysis. Since uricase activity is affected by pH and metal chelators, we assessed uricolysis inhibition in sodium fluoride-ethylenediaminetetraacetic acid (EDTA)-citrate sample tube (FC Mix tube, Greiner) used primarily for plasma glucose.
Method
A serum pool was spiked with rasburicase and uric acid measured at 15, 45, 90, 150, 240 and 1080 min in a lithium heparin tube in ice-water, plain tube at room temperature (RT), EDTA tube at RT, FC Mix tube in ice-water, FC Mix tube at RT and FC Mix tube at RT prepared by dissolving FC Mix in serum.
Results
The rate of urate decay was lowest in the FC Mix tube independent of temperature, then lithium heparin tube in ice-water, then EDTA tube at RT and highest in the plain tube at RT. Uric acid concentrations in the prepared FC Mix tube at RT and heparin tube in ice-water were, respectively, 98.2% and 93.8% of control values at 90 min, 97.1% and 89.3% of control values at 4 h, and remained higher in the prepared FC Mix tube at all time points.
Conclusion
NaF-EDTA-citrate mixture largely arrested rasburicase mediated ex vivo uricolysis without the need for sample cooling. We propose that sample tubes containing NaF-EDTA-citrate be used for the measurement of uric acid in patients administered rasburicase.
Keywords
Background
Rasburicase, a recombinant urate oxidase produced by Saccharomyces cerevisiae, is used for the prophylaxis and treatment of acute hyperuricaemia in patients at high risk of tumour lysis syndrome. Intravenous rasburicase is administered daily for up to 7 days according to plasma uric acid concentrations. 1
In patients administered rasburicase, uricolysis continues ex vivo following blood sample collection leading to spuriously low uric acid results. The manufacturer, therefore, recommends collection in a pre-chilled heparin tube, immersion in an ice-water bath, immediate centrifugation at 4°C and the separated plasma maintained in an ice-water bath until analysed within 4 h. 2 Sample storage and processing at 4°C, however, does not arrest rasburicase activity leading to falsely low uric acid results and therefore immediate sample analysis has been proposed3,4 but this is impractical in routine clinical and laboratory practice.
Microbial origin uricase is affected by temperature, metal chelators like ethylenediaminetetraacetic acid (EDTA) 5 and pH.5-7 Acidification with perchloric acid achieves near-complete inhibition of uricolysis by rasburicase and its use has been proposed for accurate measurement of uric acid in the presence of rasburicase. 8 This, however, requires bespoke blood collection tubes with added perchloric acid and neutralisation with tripotassium phosphate before analysis, 8 which may not be practical in routine laboratory and clinical practice and sample dilution may increase the limit of quantitation.
The utility of sample tubes containing sodium fluoride (NaF), EDTA and citrate buffer for plasma glucose measurement is well-established9,10 and these tubes are commercially available (Greiner FC Mix, Sarstedt GlucoEXACT). The citrate buffer, immediately lowering pH to below 5.9, inhibits hexokinase and phosphofructokinase activities arresting glycolysis. 10 We, therefore, investigated the effect of NaF-EDTA-citrate in inhibiting rasburicase induced uricolysis. If of proven utility, NaF-EDTA-citrate tubes offer a simple solution to the current labour intensive and impractical recommended method for sample collection, processing and analysis of uric acid in patients administered rasburicase.
Methods
Inhibition of rasburicase induced uricolysis in NaF-EDTA-citrate tube (FC Mix tube, Greiner item 454510) was assessed and compared with other Greiner tube types at room temperature (RT, ∼22°C) and in ice-water. The study, outlined in Figure 1, was designed to evaluate the inhibitory effect on rasburicase activity of (a) EDTA, (b) pH change (citrate buffer) and NaF in addition to EDTA, and (c) cold temperature in addition to EDTA, pH change and NaF. Study outline. RT: room temperature.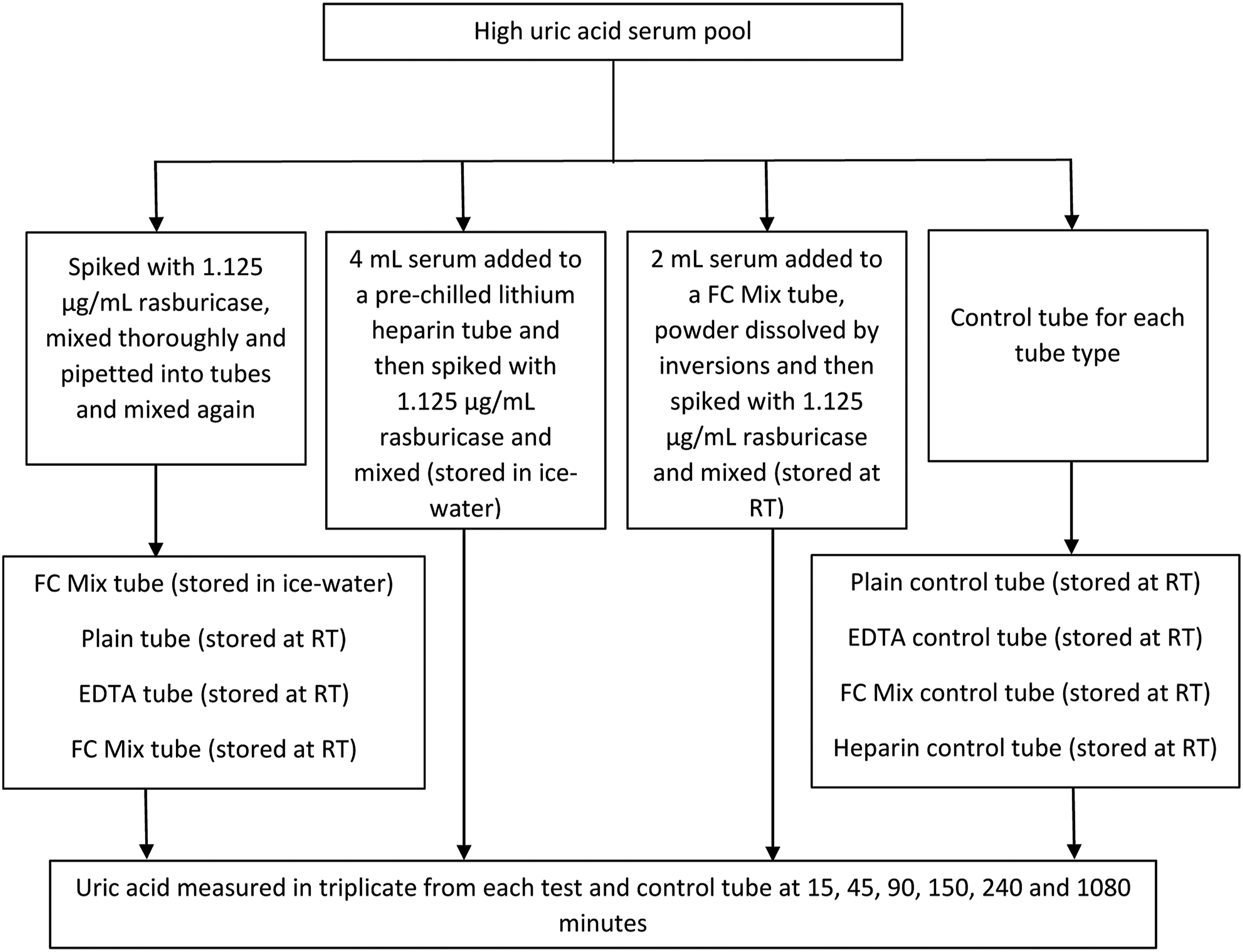
Hyperuricaemic serum samples (uric acid >450 μmol/L) were identified and pooled within 24-h of collection. A part of the pool was spiked with 0.75 μL rasburicase (Fasturtec 1.5 mg/mL, Sanofi) per mL of serum to achieve a rasburicase concentration of 1.125 μg/mL. This is equivalent to a serum rasburicase concentration after distribution of a 7.5 mg vial in a 70 kg person (volume of distribution 75.8–138 mL/kg in adults). 2 Rasburicase was spiked at a lower concentration than the product characteristics document 2 and NICE 1 recommended 200 μg/kg due to widespread use of a lower dose of rasburicase including at our centre.11,12 After thorough mixing, serum was pipetted in one tube each of plain (no additive, Greiner item 454001) and tripotassium-EDTA (Greiner item 454086), and two FC Mix tubes (Greiner item 454510) to the recommended volume and mixed by inversions as per Greiner tube recommendation. Additionally, a pre-chilled (ice-water) lithium heparin tube was spiked with the same concentration of rasburicase. Since the powdered mixture in the FC Mix tube takes considerable time to dissolve, an additional FC Mix tube was also prepared by dissolving the powdered mixture in 2 mL of serum and then spiking with the same concentration of rasburicase. The pre-chilled lithium heparin tube and one FC Mix tube were kept on ice-water between measurements. The plain tube, EDTA tube, one FC Mix tube and the prepared FC Mix tube with pre-dissolved powder remained at RT between measurements.
The remainder of the pool was used to create a control tube for each tube type by adding a volume of saline equivalent to rasburicase volume (Figure 1).
Test and control sample tubes were analysed in triplicate on a single Abbott Architect c16000 at 15, 45, 90, 150, 240 and 1080 min. Inter-assay coefficient of variation (CV) for uric acid was 1.76%, 0.99% and 0.95% at 138, 347 and 484 μmol/L, respectively. Serum pH in the FC Mix tube was measured using Dosatest 100 colour fixed indicator strips (VWR chemicals ref 85410.601).
Data were tabulated in Excel (Microsoft Corp.), Figure 2 prepared in Prism version 9.2.0 (GraphPad software) and statistical analyses were performed in SPSS Statistics for Windows version 26 (IBM Corp.). One-way ANOVA was used to assess the significance of the difference between triplicate paired uric acid results from different tubes and at different times. Tukey’s HSD (honestly significant difference) honestly significant difference was used as the post hoc test. The threshold for statistical significance was 5%. A difference exceeding the reference change value (RCV) of 20.2% for unidirectional change with 95% probability (RCV = 21/2 *1.65 *(CVa2 + CVi2)1/2] was considered clinically relevant.
13
Within-subject biological variation (CVi) of 8.6% for serum urate was used in the RCV calculation.
14
Uric acid results in different tubes at chosen time points. The results are expressed as a percentage of control. Vertical lines at the markers indicate error bars of 95% confidence interval. Only the error bars larger than the markers are visible. RT: tube at room temperature; IW: tube in ice-water; RCV: reference change value.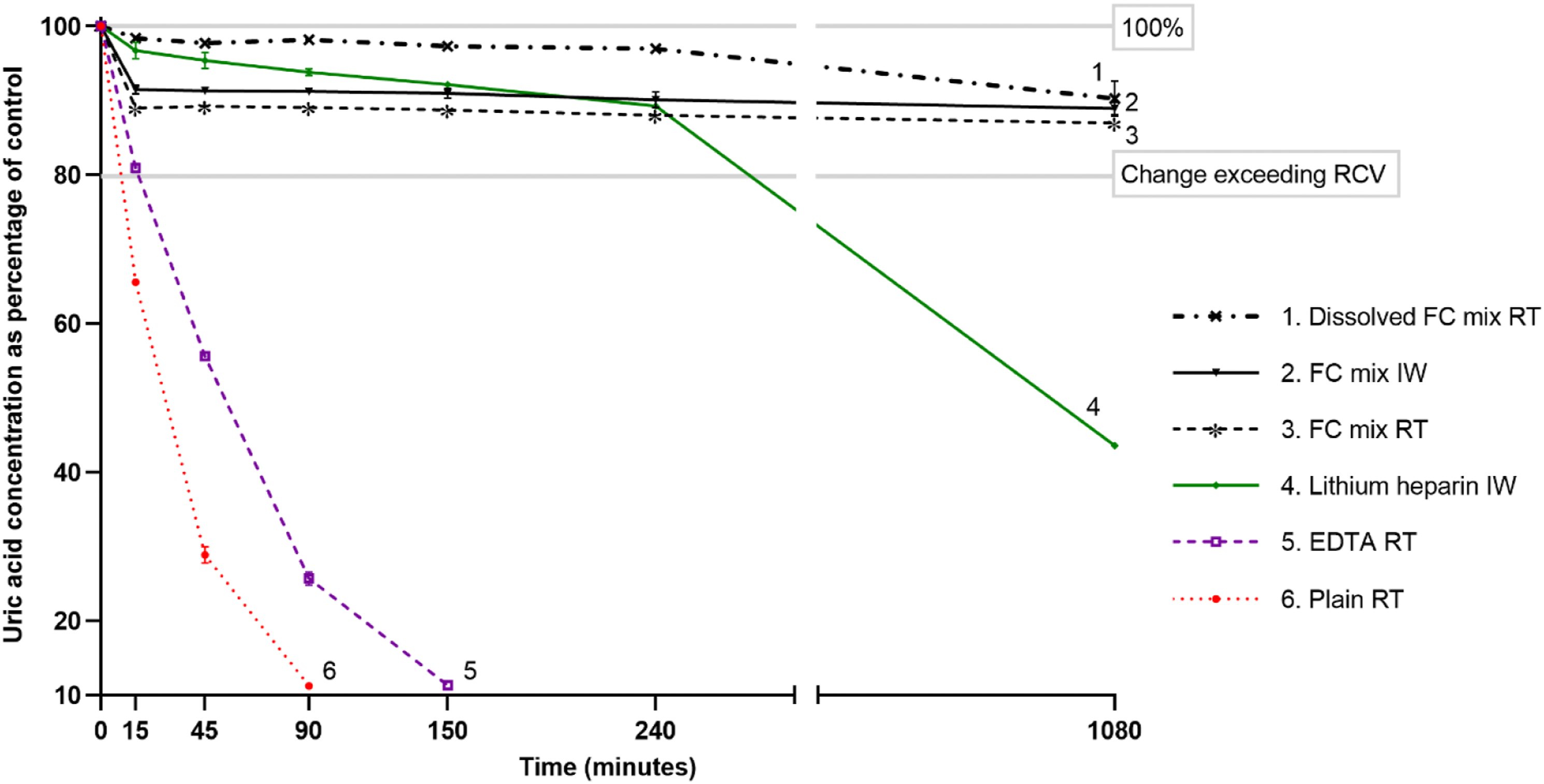
Results
The uric acid concentration in control samples was 532 ± 3.4 μmol/L at baseline and 531.7 ± 2.5 μmol/L at 18 h and there was no difference between plain, EDTA, heparin or FC Mix control tubes (p = 0.236). Serum pH in the FC Mix tube was between 5 and 6.
Uric acid concentration (μmol/L) in tubes over time.
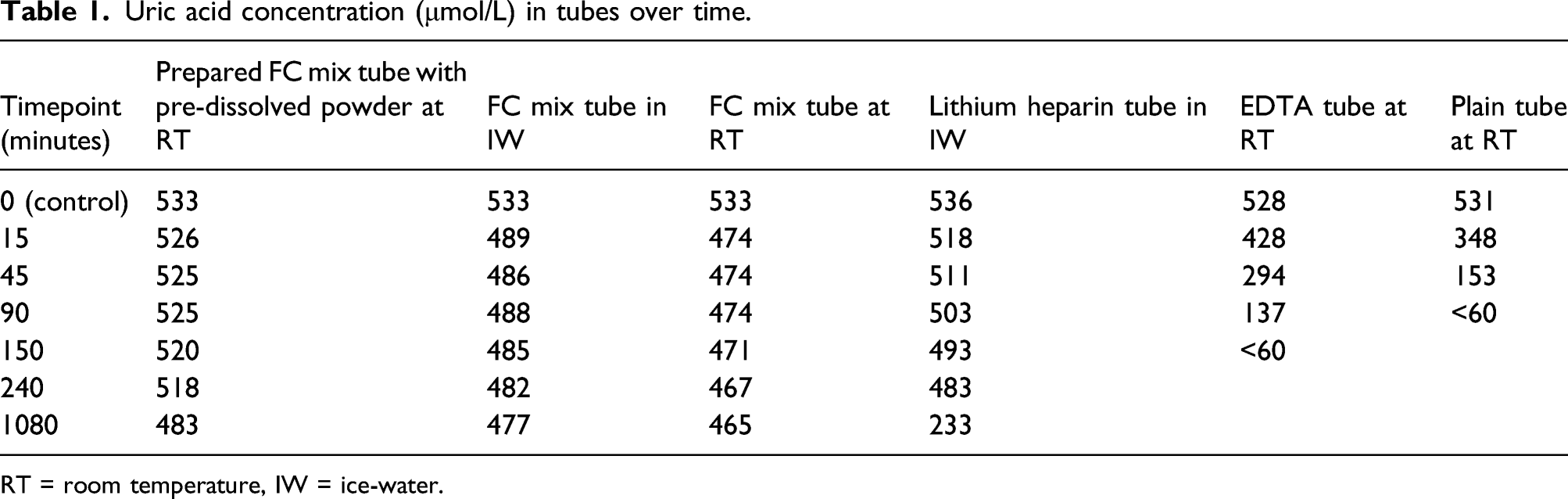
RT = room temperature, IW = ice-water.
Uric acid concentrations in the prepared FC Mix tube at RT were higher than that in the lithium heparin tube in ice-water at 15 min (p = 0.042) and at all time points thereafter (p < 0.001). Uric acid concentrations were below the limit of quantitation (60 μmol/L) in the plain tube at RT and EDTA tube at RT at 90 and 150 min, respectively.
At 18 h, uric acid concentrations in the prepared FC Mix tube at RT, FC Mix tube in ice-water, FC Mix tube at RT and lithium heparin tube in ice-water were lower (p < 0.05) than their respective uric acid concentrations at 15 min. The uric acid concentration did not exceed RCV for 18 h in the three FC Mix tubes (Figure 2).
Compared to controls at 90-min, 4 h and 18 h, the respective uric acid concentrations were 98.2%, 97.1% and 90.0% for the prepared FC Mix tube at RT; 91.2%, 90.1% and 89.0% for FC Mix tube in ice-water; 89.1%, 88.0% and 87.0% for FC Mix tube at RT; and 93.8%, 89.3% and 43.6% for the lithium heparin tube in ice-water (Figure 2).
The rate of uric acid decay between 15-min and 4 h in prepared FC Mix tube at RT, FC Mix tube in ice-water, FC Mix tube at RT and lithium heparin tube in ice-water was, respectively, 2.2 μmol/hr, 2.3 μmol/hr, 1.7 μmol/hr and 9.3 μmol/hr. The rate of uric acid decay between 15- and 45-min in the plain tube at RT and EDTA tube at RT was, respectively, 389 μmol/hr and 266 μmol/hr.
Discussion
The British National Formulary (BNF) recommends rasburicase dosing is based on uric acid concentrations. 1 To prevent ex vivo rasburicase uricolysis, the rasburicase manufacturer recommends collection in a pre-chilled heparin tube, immersion in an ice-water bath, immediate centrifugation at 4°C and the separated plasma maintained in an ice-water bath until analysed within 4 h. 2 The uric acid concentration in the heparin tube in ice-water was 93.8% and 89.3% of control value at 90 min and 4 h, respectively. This, however, is impractical and in our experience and others, uric acid in patients administered rasburicase is analysed in a serum sample tube as part of routine biochemical testing. 15 The uric acid concentration in the plain tube was 28.8% of the control value and below the limit of quantitation at 45 min and 90 min, respectively. Such uric acid results are of limited or no value as they markedly underestimate in vivo uric acid concentrations as exemplified by our data and previous studies.3,15 Our data also confirm that the rasburicase manufacturer recommendation for sample collection and processing does not arrest uricolysis. 3
The uric acid concentration in the prepared FC Mix tube at RT was 98.2% and 97.1% of control at 90 min and 4 h, respectively. NaF-EDTA-citrate achieved near-complete arrest of uricolysis, irrespective of temperature, and the FC Mix tube was superior to other tube types and the manufacturer recommended procedure. Commercially available NaF-EDTA-citrate mix tubes, primarily for plasma glucose measurement, are easy to use without any special sample handling requirements. The arrest of uricolysis by NaF-EDTA-citrate is likely due to lowering of pH which inhibits uricase activity5-7 but may also partly be due NaF 7 and EDTA 5 inhibiting uricolysis. Our data also verify that the contents of the NaF-EDTA-citrate tube do not interfere with the measurement of uric acid.
FC Mix tube at RT had greater uric acid decay compared to the FC Mix tube in ice-water during the first 15 min but thereafter there was no difference in the rate of decay (tubes 2 and 3 in Figure 2). The tube at 4°C was prepared and mixed first after spiking the pool with rasburicase and the difference between the tubes may indicate the continued uric acid decay whilst the FC Mix tube at RT was prepared and mixed rather than an additional effect of temperature. This is substantiated by the third FC Mix tube where NaF-EDTA-citrate powder was dissolved in serum before spiking with rasburicase and had least uric acid decay despite storage at RT (tube 1 in Figure 2). Uric acid results in neither of the three FC Mix tubes changed greater than the RCV (20.2%) up to 18 h and the results in the FC Mix tube with pre-dissolved powder were within 3% of the control till 4 h and within 10% at 18 h.
Although NaF-EDTA-citrate mixture arrests uricolysis, it could haemolyse erythrocytes.9,10 The incidence, however, of haemolysis with NaF-EDTA-citrate powder mixture tubes is low. In a study using NaF-EDTA-citrate powdered mixture tube from Terumo Corp., free haemoglobin was >2.0 g/L in only 1% of tubes. 10 Haemolysis with free haemoglobin up to 5 g/L does not significantly affect uric acid.16,17 Haemolysis in NaF-EDTA-citrate powdered mix tube is believed to be mainly due to the exposure of a small volume of blood to a high concentration of the powdered contents as a result of improper mixing 9 and maybe mitigated by gentle and thorough mixing. Some sample tube manufacturers provide NaF-EDTA-citrate tubes containing a liquid buffer mixture, S-Monovette® GlucoEXACT from Sarstedt, for example, which may be better than tubes containing powdered mixture by achieving earlier inactivation of rasburicase and perhaps less haemolysis due to more rapid mixing; this, however, requires further study. FC Mix sample with pre-dissolved powder in our study represents this scenario. Liquid NaF-EDTA-citrate tubes require a correction factor to compensate for sample dilution and, therefore, require accurate sample tube filling for reproducible results.9,18
In summary, the NaF-EDTA-citrate mixture almost completely arrested rasburicase mediated ex vivo uricolysis without the need for cooling. We suggest the NaF-EDTA-citrate mix tube offers an easily implementable pragmatic solution for the accurate measurement of uric acid in patients administered rasburicase. A further study comparing the NaF-EDTA-citrate tubes with the existing practice of uric acid measurement in patients administered rasburicase is required to assess if the accurate uric acid measurement has any impact on rasburicase dosage decisions.
Footnotes
Acknowledgement
The authors thank Greiner Bio-One for providing FC-mix tubes. Greiner had no input in any aspect of the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
Data are available from the corresponding author on request.
Guarantor
TK.
Contributorship
TK and JF conceived the study, did laboratory experiments, compiled and analysed the data, and wrote the first draft of the manuscript. All the authors contributed to data interpretation, critically reviewed the manuscript and approved the final version.
