Abstract
Renal stone disease is a worldwide problem which carries significant morbidity. It frequently requires specialist urology intervention. Patients with recurrent disease and those at high risk require specialist investigations and review. Certain cases benefit from medical and surgical intervention. In this review, we discuss the pathophysiology, risk assessment, specialist investigations and various interventions, their rationale and evidence base. This review aims to provide an update of the previous publication in 2001 in this journal on this topic.
Introduction
This review aims to provide an update of the previous publication in 2001 in this journal on this topic. 1 Renal stone disease (also referred as nephrolithiasis or urolithiasis) was already described more than 2000 years ago by the ancient Egyptians. 2 It remains a disorder which carries significant morbidity with associated health-care cost. Three decades ago, more than $2 billion was spent in the United States on the treatment of renal stones which was mostly due to the cost of surgical intervention. 3 Despite this condition being known for millennia, the medical progress in both preventing recurrent stone formation and understanding the pathophysiology has been limited. Data on the pathophysiology and treatment considerations for renal stone disease have been acquired via basic science experiments, population-based epidemiology, human metabolic and clinical studies. 4
Epidemiology
The yearly incidence of nephrolithiasis is estimated to be about 0.5% in North America and Europe.4,5 In the United States, the prevalence has risen from 3% to 5% in just over two decades from the mid-1970s to the mid-1990s. The lifetime risk is about 10–15% in the developed world, with higher rates between 20 and 25% described in the Middle East. According to two studies6,7 conducted in the UK in the last 25 years, risk factors for stones differ between men and women.6,7 The lifetime risk for a white man is 12–15% and for white women it is 5–6% with lifetime recurrence rates of up to 50%. 8 Similar data were presented in a study conducted in the United States, 9 which estimated the prevalence of stone formation to be around 10% in men and 7% in women.6,9 The interval for recurrences is reported as approximately 10% within one year, 35% in five years and 50% by 10 years.1,8 There are also significant ethnic variations in incidence, with urbanized black South Africans having a reported incidence of <1%. 8 Seasonal variation was described in a study based on the analysis of 43,545 stones.10,11 This showed that more stones were submitted for analysis in the summer than other times of the year.
Pathogenesis
The basic underlying processes involved in stone formation are explained by a phase change from solubility to supersaturation where the dissolved salts condense into solids.2,13
This is best explained by an example of a beaker of water with calcium oxalate crystals in it. The solubility of calcium oxalate is the concentration at which the calcium oxalate salt in the water is just high enough to prevent the crystals from dissolving and not high enough to allow them to grow. The concentration above it is called supersaturation and below it under-saturation. 14 Higher calcium and oxalate excretion and lower urine volume will subsequently increase supersaturation of calcium oxalate. When calcium oxalate supersaturation is higher than its solubility, the dissolved calcium oxalate can form nuclei of its solid phase (this is also known as nucleation).14,15 Nuclei usually form on an existing surface. Possible surfaces where this can occur in the kidney include epithelial linings, cell debris, urinary casts and other crystals. Hence, stone formation requires the formation of a nidus in the urinary tract. When this nidus gets trapped, then a large crystal structure is formed on it which can give rise to clinical symptoms.8,10
The main determinants of calcium oxalate supersaturation are therefore urine volume and urine calcium and oxalate concentration. For calcium phosphate supersaturation urine volume, urine calcium concentration and urine pH are the determining factors, whereas urinary pH is the main determinant of uric acid supersaturation.
Urine citrate reduces supersaturation by binding calcium and inhibits nucleation and growth of calcium crystals.2,5 Phytate (myo-inositol hexakisphosphate) is a natural compound formed during maturation of plant seeds and grains and it is a common constituent of plant-derived foods. 16 Phytate acts as an inhibitor of calcium crystallization both in the intrapapillary tissue and in the urine. In an animal model, calcification in renal tissue was induced in hypercalcaemic rats that were fed a purified phytate-free diet compared with those receiving a diet which included phytates. 16 Furthermore, individuals prone to calcium oxalate stone formation have lower urinary phytate excretion compared with non-stone-forming controls. Other inhibitors of calcium-based stone formation which have been described include osteopontin, prothrombin F1 fragment, calgranulin, Tamm-Horsfall glycoprotein, albumin, RNA and DNA fragments and glycosaminoglycans.2,17–20 All these substances have in common long stretches of polyanion chains that can bond with surface calcium atoms and prevent crystal growth.
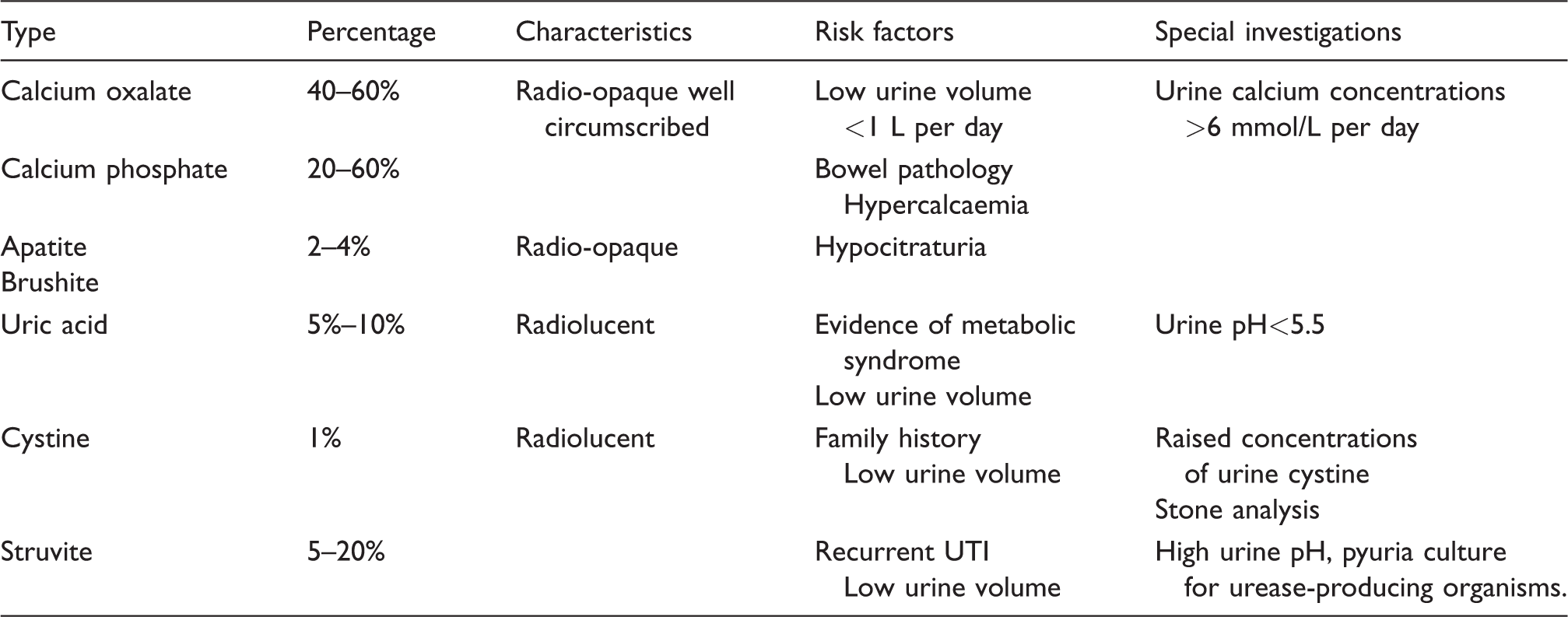
Classification
Calcium oxalate stones
Karl W Scheele (1742–1786) discovered oxalate and uric acid stones in the second half of 18th century. 21 Calcium oxalate stones (CaOx) stones can present in the monohydrate form (also referred to as whewellite) or dihydrate form (also referred to as weddellite). This distinction is important, as the monohydrate stones are associated with underlying hyperoxaluric conditions and the dihydrate stones with hypercalciuric conditions. 22 The vast majority of patients with CaOx stones have no systemic disease and as such can be classified as idiopathic CaOx stone formers. About eight decades ago, Randall 23 described plaque-like lesions in the renal papillae which were invariably present in patients with CaOx. Now called Randall’s plaques, these lesions were believed to be the nidus upon which CaOx arise and grow. 4 Microscopically, these plaques seem to arise from the basement membrane of the thin limbs of the loops of Henle. These can expand through the interstitium encasing the renal tubules and vasrecta and eventually protrude into the uroepithelium in the renal papillae. In patients with enteric hyperoxaluria, the lesions originate from the tubular lumen in the collecting duct.
Calcium phosphate stones
In a majority of kidney stones, CaOx is the main constituent, and calcium phosphate stones (CaP) is present in amounts generally ranging from 1 to 10%. 2 When CaP becomes the main constituent (>50%) of stones, the stones are called CaP stones. CaP is present in urinary stones as either apatite (the principal constituent of bones and teeth) or brushite (calcium mono-hydrogen phosphate). 2 Brushite stones are known to grow rapidly with a correspondingly very high recurrence rate.
Uric acid stones
The history of uric acid stones goes back to late 18th century, when determination of stone composition and urinary constituents became possible. This led to the finding of uric acid as a common stone component by Scheele.21,24 Uric acid stones form when the urine pH is abnormally low. Hyperuricosuria is the other main cause of uric acid stones, although this plays a less important role when compared with urine pH. The total urine urate concentration of healthy individuals is approximately 500 mg/day (∼3 mmol/day). 2 At a pH of 5.35, only 180 mg/L (∼1 mmol/L) of the total urate species can be dissolved. Low urine pH is due in part to low ammonia excretion2,25 and any conditions resulting in excessive acid load or alkali loss such as chronic diarrhoea which can lead to lower urine pH.
The combination of a low urinary pH and uric acid stones is common in patients with gout, diabetes mellitus, metabolic syndrome and a high protein diet. The pathophysiology is not fully elucidated. Insulin resistance may reduce renal ammonia excretion.2,26 Urinary pH falls with increasing body weight2,27 which correlates with insulin resistance. Several studies have described the association of obesity, diabetes mellitus, hypertension and metabolic syndrome with kidney stone disease. One such study demonstrated that an acidic urine is a feature of the metabolic syndrome and this was associated with the degree of insulin resistance.28,29 Another study of about a 1000 renal stone formers reported that the most common presenting metabolic abnormalities among obese patients were gouty diathesis (54%), hypocitraturia (54%) and hyperuricosuria (43%) which presented at concentrations that were all significantly higher than those of the non-obese stone formers. Stone analysis showed that two-thirds of these patients had uric acid calculi. 28 Compared with ∼5% in the general stone-forming population, 30–40% of the stones formed in patients with diabetes are composed of uric acid. 4
Cystine stones
Cystinuria is characterized by renal and intestinal dibasic amino acid transport defects affecting cystine, ornithine, arginine and lysine. Cystine is the least soluble of these and therefore the most likely to precipitate.8,30 The inheritance pattern of cystinuria is complex. Some patients show classic autosomal recessive inheritance, whereas the urinary excretion of cystine and dibasic amino acids can vary considerably among heterozygotes which may result in nephrolithiasis. 31 The disease can also be transmitted in an autosomal-dominant pattern with incomplete penetrance. 32
The first cystine stone was identified in 1810 by Wollaston, who called it cystic oxide.8,33 The diagnosis of cystine stones relies on a high index of suspicion, since urinary cystine is often not routinely measured at the first renal stone presentation. Factors that should raise suspicion are young age of presentation, mildly radio-opaque stones, family history and intraoperatively sulphate odour from stone fragmentation. A 24-h urine test or stone analysis is necessary to make the diagnosis.
A homozygous cystinuric patient excretes between 600 and 1400 mg (2.5–6 mmol/day) of cystine per day. At a pH of 7.0, the solubility of cystine is 275 mg/L (1.14 mmol/L). Simple calculations indicate that a urine output of 5 L per day would be adequate to allow cystine clearance in solution for most homozygous cystinuria patients and indeed, hydration alone can prevent stone recurrence in up to a third of patients. 34 It is important that fluids are taken throughout the 24-h period and it has been recommended to also drink water overnight. 34
Combination stones
Magnesium ammonium phosphate stones (also known as struvite) occur when there is recurrent infection with urease-positive organisms such as
Drug containing renal stones
Several drugs are implicated in producing renal stones. The mechanisms include a reduction in urine citrate concentrations, urinary supersaturation of stone-forming solutes and primary drug containing stones.
Magnesium-trisilicate is used over the counter for gastrooesophageal reflux disease. Excessive consumption can produce silicate stones. Antibiotics like ciprofloxacin and sulphonamides produce renal stones by forming crystalline aggregates of these drugs. Ciprofloxacin and sulphonamide stones are radiolucent, but triamterene is radiopaque. Antiretrovirals such as the protease inhibitor indinavir leads to the formation of pure indinavir stones which are not detectable with standard radiography or computerized tomography. 38 On occasion, these may contain a calcium component. Ceftriaxone is identified in urinary or biliary tract as a calcium salt. 22
Primary conditions
Stone formers are more likely to have primary urinary metabolic abnormalities than those who do not form stones. 16 The pathogenesis of the primary conditions can be understood by metabolic anomalies that favour the formation of the stones due to an excess in promotors of stone formation such as an excess of calcium, uric acid, oxalate and cystine occurring in the urine or due to the reduction in inhibitors of stone formation such as a reduction in citrate and magnesium. 16 Furthermore, the formation of urinary stones is influenced by the urinary pH with alkaline pH favouring the crystallization of calcium and oxalate-containing stones and acidic pH promoting uric acid or cystine stones.
Primary hyperoxalurias
The primary hyperoxalurias are a group of autosomal recessive disorders involving the overproduction of oxalate. Although the initial recognition of the disease is attributed to Lepoutre, who reported it in 1925, the description of the underlying biochemical abnormalities occurred many years later with three types being described namely hyperoxaluria types 1, 2 and 3, each caused by a different enzyme deficiency. 40
The primary defect of inherited hyperoxaluria is the overproduction of oxalate, primarily by the liver, which results in increased excretion by the kidney. The earliest symptoms among those affected are urolithiasis and nephrocalcinosis which lead to progressive renal involvement and chronic kidney disease. 40 Renal damage is ultimately caused by a combination of tubular toxicity from oxalate, nephrocalcinosis (with both intratubular and interstitial deposits of calcium oxalate) and renal obstruction by stones, often with superimposed infection. The second phase of damage that is the result of primary hyperoxaluria occurs when the glomerular filtration rate drops to 30 to 45 ml/min, and the kidney is unable to effectively excrete the oxalate load it receives. At this point, plasma concentrations of oxalate rise and exceed saturation,40,41 and oxalate is subsequently deposited in all tissues (systemic oxalosis), particularly in the skeleton.
Primary hyperoxaluria type 1 is caused by a deficiency of the liver-specific peroxisomal enzyme alanine-glyoxylate aminotransferase that catalyses the transamination of glyoxylate to glycine. 40 This is the most devastating subtype, particularly when it occurs in infancy, but patients who have the Gly170Arg or Phe152Ile mutation have a better overall outcome than other patients with type 1 disease, partly because of their sensitivity to pyridoxine.40,86
Primary hyperoxaluria type 2 is caused by a lack of glyoxylate reductase–hydroxypyruvate reductase which catalyses the reduction of glyoxylate to glycolate and hydroxypyruvate to D-glycerate. It was previously believed that patients with primary hyperoxaluria type 2 generally have a less severe course; however, this condition is characterized by nephrolithiasis (deposition of calcium oxalate in the renal pelvis/urinary tract), nephrocalcinosis (deposition of calcium oxalate in the renal parenchyma) and end-stage renal disease (ESRD). The onset of symptoms is usually in childhood.107
Primary hyperoxaluria type 3 results due to the deficiency of the 4-hydroxy-2-oxoglutarate aldolase enzyme. Primary hyperoxaluria type 3 has the least severe course and may be silent or limited to stone formation, sometimes even improving over time and nephrocalcinosis and ESRD is uncommon. 40
Disorders of purine metabolism
Xanthinuria is a rare autosomal recessive disorder characterized by the excretion of large amounts of xanthine in the urine and a tendency to form xanthine stones. Uric acid is very low in both serum and urine. Two clinically similar but distinct forms of xanthinuria are described. Type I xanthinuria is characterised by an isolated deficiency of xanthine dehydrogenase, and in type II, a dual deficiency of xanthine dehydrogenase and aldehyde oxidase. A wide range of ages presenting with disease has been described ranging from infancy and childhood to incidental findings in older individuals found to have low serum urate concentrations. 87
Dihydroadenine urolithiasis is caused by an autosomal recessively inherited mutation in the adenine phosphoribosyl transferase (APRT) gene leading to APRT deficiency and subsequent excess formation and urinary excretion of 2,8 dihydroadenine (DHA). DHA precipitates and crystallizes as radiolucent stones and have been erroneously treated as urate stones. It can be diagnosed by stone analysis and measurement of APRT activity of red blood cells. As this is a rare under-recognized condition, its diagnosis is often delayed. 42
Lesch-Nyhan syndrome is an X-linked recessive disorder of purine metabolism associated with a virtually complete deficiency of the enzyme hypoxanthine-guanine phosphoribosyl-transferase. The disease is characterized by hyperuricaemia, self-mutilation, choreoathetosis, spasticity and mental retardation. The abnormalities of purine metabolism are present at birth and may lead to uric acid crystalluria and stone formation early in life. 43
Hypercalciuric stone-forming disorders
These rare disorders illustrate the link between genetic variation, urinary calcium excretion and calcium-containing stones. Two reviews44,45 discuss this at length and we provide a brief overview.
Dents disease complex: This X-linked hypercalciuric stone-forming disorder is due to mutations in the gene that codes for the voltage-gated endosomal chloride channel 5.2,46,47 Bartter syndromes: There are five major genetic variants associated with Bartter Syndromes. Cortical and medullary renal calcifications are found in patients with Bartter syndrome types I and II which arise from transport defects that reduce thick ascending limb sodium chloride and calcium reabsorption, leading to hypercalciuria.2,48 In Type III, defects in a thick ascending limb basolateral membrane chloride channel produce only variable hypercalciuria, and crystal deposits are uncommon.
2
Defects in a chloride channel subunit known as Barttin leads to Bartter syndrome type IV with associated deafness, but renal stones disease is not a feature.
2
Familial hypomagnesaemia with hypercalciuria and nephrocalcinosis: Familial hypomagnesaemia with hypercalciuria and nephrocalcinosis (FHHNC) is a complex renal tubular disorder characterized by hypomagnesaemia, hypercalciuria, advanced nephrocalcinosis and progressive renal failure. The mode of inheritance is autosomal recessive.
49
It is caused by the mutations in the gene coding for the renal tight junction protein claudin 16 (CLDN16) gene (also known as ‘paracellin-1’ gene).
50
CLDN16 is exclusively expressed in the thick ascending part of Henle's loop, where it is believed to form paracellular channels that allow reabsorption of Ca2+ and Mg2+ driven by an electrochemical gradient.
51
Consequently, patients with FHHNC experience severe renal Ca2+and Mg2+wasting.
50
The other gene implicated in FHHNC is claudin 19 (CLDN19).
52
CLDN19 is expressed in high concentrations in both the kidney and the eye. Both proteins CLDN16 and CLDN19 are required for renal reabsorption of magnesium in the thick ascending limb of Henle where approximately 50–60% of the filtered magnesium is reabsorbed via a paracellular pathway. CLDN19 mutations are associated with a similar phenotype to that due to CLDN16 mutations except that the patients manifest severe ocular pathology.
52
Distal renal tubular acidosis: Distal renal tubular acidosis (RTA) is associated with renal stone disease. It is caused by the inability of alpha-intercalated cells to acidify urine. The inherited form has three variants: autosomal dominant and autosomal recessive with or without deafness. Acquired forms are more common in clinical settings. Primary distal RTA results in metabolic acidosis with hypokalaemia, hypercalciuria and hypocitraturia. Low urine citrate results from upregulated citrate reabsorption in the proximal tubule to form bicarbonate. Hypercalciuria can be due to multiple factors including increased calcium resorption from the bone in an attempt to buffer systemic acidosis, downregulation of renal calcium transport proteins due to acidosis and increased distal sodium delivery. All of this along with high urine pH leads to hypercalciuria and nephrocalcinosis.
53
Idiopathic hypercalciuria
Idiopathic hypercalciuria (IH) is the result of the interplay between genetic background and environment. About half of patients with IH have a family history of kidney stones. 4 The precise gene or genes which contribute to IH are still unknown, but this is likely polygenic with contributions from several genes which differ within individuals. 54 The diagnosis of IH requires the exclusion of hypercalcaemia and the related underlying conditions such as hyperparathyroidism, hyperthyroidism, malignant neoplasm and sarcoidosis. 2 Subjects with IH have a generalized increase in calcium turnover, which includes increased gut calcium absorption, decreased renal calcium reabsorption and a tendency to lose calcium from bone. 54 High intestinal calcium absorption can raise the load of filtered calcium presented to the renal tubules. The reabsorption of filtered calcium by the renal tubules may also be reduced. Either mechanism can increase urinary calcium concentration, which can be further be increased by glucose or sucrose loads, high sodium intake2,55 and a high-protein diet.2,56 A possible mechanism that could explain the abnormal renal tubule conservation of calcium, as well as increased gut absorption of calcium, would be an altered tissue response to vitamin D. 54 A high tissue response to vitamin D can produce all the manifestations of IH namely: increased gut calcium absorption, decreased renal reabsorption and increased mobilization of calcium from bone. To further study the pathophysiology of IH, a rat model of hypercalciuria has been generated by breeding rats for increased urine calcium excretion. These have been called the genetic hypercalciuric stone-forming (GHS) rats.54,57 Like humans with IH, the GHS rats have increased gut absorption of calcium, decreased renal calcium reabsorption and a tendency to bone demineralization; when placed on low-calcium diet, they excrete more calcium than is absorbed, indicating a loss of bone mineral. Significantly higher concentrations of vitamin D receptors (VDR) have been found in bone, kidney and intestine, compared with normal rats, which would explain their increased sensitivity to their normal concentrations of 1,25 vitamin D.
Secondary and associated conditions
Hypercalcaemia
Conditions such as hyperparathyroidism and sarcoidosis which are associated with chronic hypercalcaemia can lead to renal stone disease and these need to be excluded. 58
Inflammatory intestinal disease
Chronic diarrheal illnesses such as ulcerative colitis and Crohn’s disease are associated with enteric hyperoxaluria which results in an increased risk of developing renal stones.8,59 Renal stones develop in 3–9% of patients with inflammatory bowel disease which is at least twice as high when compared with non-diseased cohorts. 59 Excess oxalate is absorbed through the inflamed bowel wall. 8 However, patients are also at increased risk of uric acid stones as chronic diarrhoea lowers urinary pH which is a significant risk factor for uric acid stones.2,60 These stones are particularly common in patients after colectomy. 8
An additional problem is that of urine supersaturation. Many patients with these conditions restrict their fluid intake to control diarrhoea and consequently have lower urine volumes. 59 Post-ileostomy surgery results in acidic urine due to the loss of alkaline ileostomy fluid and an increased risk of uric acid stones. 8
Coeliac disease is also associated with an increased risk of renal stone disease. The underlying mechanism is likely to be hyperoxaluria. Untreated patients with coeliac disease are at a higher risk compared with patients on a gluten-free diet. One study reported 120% higher oxalate and 43% lower calcium excretion in patients with untreated coeliac disease compared with healthy individuals. These abnormalities normalized when patients were treated on a gluten-free diet. 61 Gastric by-pass surgery is associated with calcium oxalate renal stones disease. The primary pathophysiological process manifests as hyperoxaluria. This is understood to be due to a reduction in fat absorption which leads to a reduction of intestinal calcium which is saponiefied by intestinal fatty acids. There is thus less intestinal calcium available to bind the intestinal oxalate which instead gets absorbed and hyperoxaluria ensues. 62
Drugs
As discussed above, under the ‘classification of renal stones’ section, primary drug containing stones can be formed by crystalline aggregation. Drugs can also increase the risk of stone formation through secondary effects, such as reducing urine citrate concentrations, changing urinary pH or enhancing supersaturation of solutes. 63 Loop diuretics like frusemide and bumetanide inhibit calcium and sodium reabsorption in the ascending limb of loop of Henle, and together with their diuretic effect this leads to a hypercalciuric state. Acetazolamide, a carbonic anhydrase inhibitor, blocks the reabsorption of sodium bicarbonate, leading to an alkaline urine a decrease in citrate and an increased risk of calcium nephrolithiasis. 38 The antiepileptic medication, Topiramate, inhibits carbonic anhydrase, leading to a mild metabolic acidosis. The reduction of bicarbonate reabsorption leads to an increase in urine pH, thereby increasing the risk of calcium oxalate and phosphate stone formation.38,39 Another antiepileptic agent, Zonisamide, is a sulphonamide which increases the risk of renal stones through its weak carbonic anhydrase activity. 38
Kidney malformations
Anatomical abnormalities associated with renal stone disease include: medullary sponge kidney (tubular ectasia), uretropelvic junction obstruction, calyceal diverticulum, calyceal cyst, ureteral stricture, vesicoureterorenal reflux, ureterocele and horseshoe kidney. These conditions lead to impaired drainage and urinary stasis as well as an increased incidence of infection. Horseshoe kidneys are a complex anatomic variant of fused kidneys, with ∼20% of patients suffering from renal stones. 64
Dysglycaemic conditions
Several studies have described the association of obesity, insulin resistance, metabolic syndrome, diabetes mellitus and the propensity to form uric acid stones. The pathophysiology is incompletely understood, but the main mechanism includes a reduction in urine pH, lower citrate concentrations and hyperuricosuria.2,26
Management of renal stone disease
There are no clear guidelines which recommend whom to investigate and what specialist investigations to perform. This is generally guided by the history (e.g. recurrent stones), stone burden (e.g. bilateral renal stones), age of onset (younger patients require investigation), stone factors (e.g. cysteine stones), renal factors (such as patients with single kidney) and relevant co-morbidities (such as hyperparathyroidism or metabolic bone disease). Once a specific diagnosis is made, targeted therapy can be considered. The investigations to enable diagnosis included analysis of urine, blood and radiological imaging.
Special investigations
Urinalysis
Urinalysis should include dipsticks to assess urine pH and indicators of infection. Microscopy and urine culture should be obtained if urinary tract infection is suspected. 65 Additional metabolic testing must be done in high risk and recurrent stone formers. Metabolic testing includes one or two 24-h urine collections obtained, while on the patient’s usual diet and analysed for total volume, pH, calcium, oxalate, uric acid, citrate, sodium, potassium and creatinine. These investigations have been recommended upon the first presentation 65 of renal stones, but this may be less practical and should also be guided by the availability of services and risk of future stone disease.
Various schedules for urine collection have been proposed. The most commonly used is collection over 24 h. Alternative collection regimens which include 16 h and/or 8 h have also been described.8,66 Although these are more practical from a patient perspective, test interpretation is likely more complex.
Normal values of relevant analytes in urine.106

Blood investigations
The recommended blood investigations include serum sodium, potassium, bicarbonate, calcium, creatinine and urate. 65 Any abnormalities in these investigations will guide further investigations such as parathyroid hormone, magnesium and phosphate concentrations in the examination of an abnormal calcium concentration.
Imaging
Abdominal computed tomography (CT) has become the most common initial imaging test for suspected nephrolithiasis because of its high sensitivity for the diagnosis of renal stone disease.70,71 However, CT entails exposure to ionizing radiation with long-term cancer risk 72 and it is associated with a high rate of incidental findings70,72,73 that can lead to inappropriate follow-up referral and treatment. All these contribute to the growing costs associated with renal stone disease. Although ultrasonography was less sensitive than CT for the diagnosis of nephrolithiasis, using ultrasonography as the initial test in patients with suspected nephrolithiasis (and using other imaging as needed) resulted in avoiding CT in most patients, lower cumulative radiation exposure and no significant differences in the risk of subsequent serious adverse events, pain scores, return emergency department visits or hospitalizations. 70 Either of these investigations can be used for surveillance depending on initial findings and cumulative radiation exposure. CT has the additional advantage of assessing the stone’s radiodensity (reported in Hounsfield units) which helps to guide surgical management. Abdominal X-rays are also used for surveillance, although this investigation is used less often due to the risks of radiation exposure and the inability to monitor radiolucent stones such as uric acid stones.
Treatment
Fluid intake
Increasing urine volume can lower the supersaturation and therefore the risk of renal stone disease. This forms the cornerstone of general recommendations. Randomized controlled trials have demonstrated that increase fluid intake reduces the risk of stone formation and a general goal is the excretion of at least 2.5L of urine daily.65–74 The practical recommendation is to have colourless and thus sufficiently dilute urine. 8 Studies investigating the adherence to an increased fluid intake show that this is often poor. This is exacerbated by heavy exercise, hot weather and long distance travel.8,75
Diet
There is compelling evidence that certain dietary measures limit the risk for stone formation.74,76 The most important dietary factors which can reduce the risk of renal stone formation include: a reduction in animal protein and sodium consumption and a normal calcium intake. Historically, patients with calcium-containing stones were advised to reduce calcium intake; however, such recommendations lacked a robust evidence base. Although dietary calcium is the main contributor to high urinary calcium concentrations, a reduction in calcium intake has not been proven to be effective to prevent renal stone disease in patients with IH.2,76 An epidemiological study of men without a history of nephrolithiasis reported that those with a high intake of calcium (>26.2 mmoL per day) had a 34% lower risk of stone formation than in those with a low calcium intake (<15.1 mmoL per day). 77 In a randomized controlled trial which included 120 men (60 in each arm) with recurrent CaOx and hypercalciuria, a restricted intake of animal protein and salt, combined with a normal calcium intake, provided greater protection (12 recurrent events) than the historically suggested low-calcium diet where there were 23 recurrent events. 76 Furthermore, a low calcium may also predispose to a reduced bone mineral content and this strategy should not be advocated.2,20
Dietary intake of animal protein is associated with the risk of stone formation, whereas potassium intake is inversely related to risk. Animal protein intake increases the excretion of uric acid, 3 calcium 78 and lowers urinary citrate excretion 78 and urinary pH. 79 All these factors increase the risk of forming calcium-based stones. 3 This is supported by a randomized controlled trial in recurrent calcium oxalate stone formers which demonstrated a reduced recurrence rate in patients on high calcium, low sodium and low animal protein diet.65,80 There is a clear association between a high salt intake and nephrolithiasis. High salt intake correlates with other risk factors of renal stone disease, most notably hypercalciuria and hypocitraturia. Dietary salt restriction has been shown to reduce calcium excretion in several studies. 81 However, this is not clearly demonstrated in all individuals, and as in the field of hypertension, this may be explained by the concepts of salt sensitivity and salt resistance. 81
There is little proof in recommending restrictions on oxalate intake as only 10–15% of urine oxalate is derived from the diet. 8 Spinach and rhubarb contain large quantities of bioavailable oxalate.8,82 Nuts (almonds, hazelnuts, peanuts, pecans and walnuts), blackcurrants, strawberries, beetroot, parsley, chives, cocoa, wheat germ, brown rice, cola, instant tea, and chocolate contain moderate amounts.8,59,88 It has been shown that a high dietary oxalate intake can be offset for by a high calcium intake. 83
Ascorbic acid (vitamin C) can be metabolized to oxalate. In one prospective study which included 22,000 men who consumed greater than 1 g of vitamin C had a greater than 40% risk of stone formation.77,84
Although hyperuricosuria results from high dietary intake of beef, poultry and fish and can be abolished by dietary changes, no clinical trials have documented the effectiveness of this approach. 2
Phytates which are a common constituent of plant-derived foods can inhibit calcium crystallization both in the intrapapillary tissue and in the urine. 16 No prospective studies specifically related to phytate consumption and renal stone disease has been conducted.
The trial data on dietary intervention and pharmacotherapy are limited by short-term studies in relatively small cohorts with very few comparative effectiveness studies. Dietary intervention studies are notoriously difficult to conduct as this often relies on self-reporting, the lack of biomarkers to monitor adherence and the presence of confounders in epidemiological studies and the difficulties related to blinding. Surrogate markers such as urine calcium excretion are often used as primary endpoints as opposed to demonstrating an actual reduction in recurrent events.
Pharmacotherapy
Hypercalciuria
Hypercalciuria is generally regarded as a 24-h calcium excretion of >7.5 mmoL/day in men and > 6.2 mmoL/day in women. 1 A number of randomized controlled trials have shown that thiazide diuretics reduce the formation of calcium stones. 65 The drugs which have been investigated include hydrochlorothiazide, chlorthalidone, bendroflumethiazide and indapamide. These medications all reduce hypercalciuria by increasing the fractional reabsorption of calcium in the distal nephron and decreasing intestinal calcium absorption.8,88
Hyperoxaluria
Oxalate absorption can be blocked by magnesium carbonate 1 or cholestyramine. 88 The recommended dose of magnesium is 650–1300 mg magnesium hydroxide/day; however, no prospective randomized trials have reported a reduction in renal stone disease. 8
In cases of intestinal hyperoxaluria, cholestyramine can be used to reduce intestinal absorption of oxalate. Cholestyramine is an anionic exchange resin that binds bile acids which prevents their absorption. 88 The increase in faecal loss of bile acids results in greater oxidation of cholesterol into bile acids (leading to a reduction in serum cholesterol) and a decrease in oxalate absorption (leading to a reduction in serum and urinary oxalate). 8 Pyridoxine (Vitamin B6) is another option for the treatment of hyperoxaluria. 8 Pyridoxine is the co-factor in the alanine–glyoxalate–transaminase pathway, which converts glyoxalate to glycine, and may reduce oxalate production by enzyme induction. 85 Pyridoxine supplementation is helpful in primary hyperoxaluria type 1 but not in other forms of primary hyperoxaluria.40,85 The ultimate treatment of primary hyperoxaluria may require transplantation of the kidney alone or combined liver and kidney transplantation.
Hyperuricosuria
Hyperuricosuria is diagnosed when the uric acid excretion exceeds 4800 mmoL/day in men or 4400 mmol/day in women, or the uric acid:creatinine ratio >530 mmol/mmoL. 89 As uric acid stones are classically formed in acidic urine, the cornerstone of therapy is urinary alkalization. The urine can be alkalinized with potassium citrate aiming to achieve a urine pH of 6.5–7. 2 Alkalization can completely dissolve uric acid stones, thereby avoiding any surgical intervention. Allopurinol, which is a xanthine oxidase inhibitor that prevents the conversion of hypoxanthine to xanthine and ultimately uric acid, is used for the treatment of gout. It can be used as an adjunct to alkalization. 65
Febuxostat is a newer xanthine-oxidase inhibitor which demonstrated superior reductions in serum uric acid concentrations and urinary uric acid secretion compared with allopurinol.90–92 Hyperuricosuria may also promote calcium oxalate nephrolithiasis by heterogeneous nucleation or by adsorption of macromolecular inhibitors. It may therefore be beneficial to treat hyperuricosuria to prevent calcium stone formation. 8 A randomized controlled trial comparing febuxostat to allopurinol or placebo in patients with a history of hyperuricosuria and CaOx demonstrated that febuxostat led to a significantly greater reduction in 24-h urinary uric acid excretion than either allopurinol or placebo after six-month treatment period. There was no change in stone burden at the end of treatment period. 93
Hypocitraturia
Citrate plays a role in inhibiting stone formation as it reduces the supersaturation of calcium oxalate by binding to calcium. Hypocitraturia is found in up to 20% of stone formers and may be idiopathic or secondary to intestinal, renal, dietary or pharmacological causes.8,94
Alkalinizing salts such sodium citrate, potassium citrate, potassium–magnesium citrate, sodium bicarbonate and potassium bicarbonate can be used to increase urinary citrate concentrations. Only potassium citrate and potassium–magnesium–citrate have been tested in randomized trials.8,94,95 In one double-blinded randomized controlled trial which included 57 patients, potassium citrate was compared with placebo over a three-year period. This demonstrated a significant reduction in recurrence of renal stone disease in the potassium citrate arm. 95
Hypomagnesuria
Magnesium is another inhibitor of renal stone formation. Its urine concentration depends on diet. In one study conducted in 1982, 55 patients with recurrent renal calcium stone disease without any signs of Mg deficiency received magnesium supplementation. Results showed a 10-fold reduction in stone recurrence compared with the control arm. 96 Supplementation with magnesium salts may lead to diarrhoea, intestinal colic and loose bowel action.
Cystinuria
The cornerstone of cystine stone prevention is hydration and urinary alkalization.34,97 The most important cystine stone-prevention technique is hyper diuresis which decreases urinary cysteine concentration. The goals of hydration therapy are urine volumes in excess of 3 L/d. This goal may require ingesting 4–4.5 L of water per day to allow for insensible loss. Alkalizing with potassium citrate is recommended and the addition of citrus juices may provide additional benefit. 34 Chelating agents may be used in addition to alkalization and optimizing fluid intake. D-Penicillamine, is a first-generation chelating agent which produces cysteine–penicillamine heterodimers that are 50 times more soluble than cysteine–cysteine (cystine) homodimers. 8 D-Penicillamine was associated with a wide variety of side-effects, and long-term treatment may result in vitamin B6 deficiency. 8 A retrospective study of D-Penicillamine showed decreased stone event and dissolution of stone. 98 Alpha-mercaptopropionylglycine (Tiopronin) is a second-generation chelating agent that has a similar mode of action to penicillamine with fewer side-effects and its benefit was demonstrated in a clinical trial which included 66 patients with cystinuria.8,99,100
The angiotensin-converting enzyme inhibitor captopril forms a thiol–cystine mixed disulphide which is 200 times more soluble than cysteine. At a dose of 75–100 mg, captopril can be used in those who do not respond to other treatment. 34 It seems less efficacious when compared with D-Penicillamine and alpha mercaptopropionylglycine.101,102
Bucillamine is a third-generation chelating agent which is theoretically superior to penicillamine, but experience and data on this medication are limited.8,102
Recurrent urine tract infection
It is possible to try to prevent recurrent infection by chronic antibiotic treatment, but this may only slow the progression of the disease rather than be curative.8,35 Other considerations include antibiotic resistance and antibiotic-induced reduction in intestinal bacteria such as
Surgical intervention
The surgical management of renal stone disease can be categorized into non-invasive and invasive interventions.
Non-invasive intervention
The most common non-invasive modality is extracorporeal shock wave lithotripsy (ESWL). This was introduced in the early 1980s and remains the first-line treatment in the management of renal stones as the majority of stones can be treated with this method. 77 Indications for ESWL revolve around the stone size and position. It is usually the first-line approach in patients with stone burden less than 10 mm and in children. In renal stone burden between 10 and 20 mm, it is considered if other approaches are deemed to have a higher complication risk. 77 Stone composition is an important determinant. Very hard stones (>1000 Hounsfield units) such as brushite (calcium mono-hydrogen phosphate) and whewellite (calcium oxalate monohydrate) respond poorly to ESWL.12,68
Invasive interventions
Retrograde surgery
This includes ureteroscopy for ureteric stones and flexible ureterorenoscopy also known as retrograde intrarenal surgery. Flexible ureterorenoscopy is suitable for stone sizes between 0.5 and 1.5 cm and suitable for all types of stone compositions apart from the lower pole stones which are hard to access. The mainstay of treatment is a holmium-YAG (yetrium-Aluminium-Garnet) laser. It delivers a pulsatile energy that allows lithotripsy to be performed. 77
Percutaneous nephrolithotomy
It offers a reduced morbidity rate and a lower length of stay and cost compared with open pyelolithotomy. It involves the removal of the renal stone as a whole or in fragments through a specifically created tract between the skin surface and the collecting system of the kidney. 77
Future developments
For such a common condition, there is surprisingly little basic and clinical research on renal stone disease. The increased knowledge base of causal genetic conditions as well as the role which the intestinal microbiome may play in this condition provides promise for future research and our understanding of the pathophysiology. 103 A hand full of clinical trials have recently reported or are underway. ALLN-177 is an oral formulation of a recombinant oxalate-specific microbial enzyme, oxalate decarboxylase intended to treat secondary hyperoxaluria by degrading dietary oxalate in the gastrointestinal tract, thereby reducing its absorption and subsequent excretion in the urine. A double-blind placebo controlled randomized cross-over study with ALLN-177 demonstrated a significant reduction in urinary oxalate excretion and this may present a new approach for managing secondary hyperoxaluria and its complications. 104 The Nephrolithiasis Prevention by Lemon Juice trial is investigating the effect of lemon juice in reducing the risk of new stone formation in patients with recurrent idiopathic calcium oxalate nephrolithiasis. This trial is currently underway. 105
One comparative effectiveness study is currently underway which is investigating the cysteine-binding thiol drugs tiopronin and D-Penicillamine. 106
Conclusion
Taken that this a relatively common condition, there is surprisingly little basic science and clinical research undertaken. The trial data on dietary intervention and pharmacotherapy are limited by short-term studies in relatively small cohorts with very few comparative effectiveness studies. Dietary intervention studies are notoriously difficult to conduct as this often relies on self-reporting, the lack of biomarkers to monitor adherence and the presence of confounders and the difficulties related to blinding. Surrogate markers such as urine calcium excretion are often used as primary endpoints as opposed to demonstrating an actual reduction in recurrent events.
Renal stone disease is associated with a diverse range of pathological conditions. The causal relationship between dietary and other factors as well as the influence of genetic susceptibility is still poorly understood. There remains a need for and an improved understanding of the pathophysiology, the genetic basis, as well as more robust evidence base to guide the management and investigations of this condition.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
AV.
Contributorship
All authors contributed equally to the manuscript.