Abstract
Background
Therapeutic monitoring of lithium (Li) is important because of its narrow therapeutic range and therapeutic index. Here, the authors present the evaluation of an accurate method for the determination of lithium in serum.
Method
Serum samples were diluted with 0.3% ultrapure nitric acid and were spiked with an internal standard germanium (Ge). The Li/Ge ratio was detected in He mode; we utilized standard addition method to quantify lithium in human serum. The new inductively coupled plasma mass spectrometry (ICP-MS) assay was characterized for linearity, specificity, imprecision, trueness, accuracy, and comparison.
Results
The correlation coefficients (r) of linearity were all > 0.9999. The specificity proved to be good. The total coefficients of variation (CV) were 1.11% and 0.49% for the two serum samples. The mean bias from target values of standard reference materials (SRM 956d) was −0.71% for Level I, −017% for Level II, and 2.20 for Level III. External Quality Assessment Scheme for Reference Laboratories in Laboratory Medicine (RELA) gave satisfied results for the new method. Comparison with the ion-selective electrode routine method got reasonable results.
Conclusion
This high accuracy method is an attractive alternative for lithium measurement and can be used as a candidate reference method to improve quality of serum lithium in China.
Keywords
Introduction
Monitoring serum lithium concentrations is accepted as a necessary feature of lithium therapy because the consequences of lithium toxicity can be quite serious. The therapeutic range of lithium in serum is 0.6–1.2 mmol/L for acute treatment and 0.5–0.8 mmol/L for maintenance treatment; lithium concentrations exceeding 2.0 mmol/L are considered potentially toxic.1,2
Clinical lithium measurements are performed using ion-selective electrode (ISE) 3 and photometric technology. 4 However, in order to improve the measurement quality, a high accuracy and metrologically traceable reference method is required. Up to now, the Joint Committee for Traceability in Laboratory Medicine (JCTLM) database (www.bipm.org/jctlm/) lists different reference measurement methods for lithium.5–8 Even though the reference method for the determination of lithium already exists, establishing additional reference measurement method such as inductively coupled plasma mass spectrometry (ICP-MS) is justified. Lithium is free from mass spectrum interference and is suitable for ICP-MS analysis. Here, a high accuracy method was developed in the calibration laboratory of the Shanghai Center for Clinical Laboratory (SCCL). The calibration laboratory is accredited according to ISO 17025 9 and ISO 15195. 10
Materials and methods
Instruments
A PerkinElmer 300x quadrupole ICP-MS instrument (Waltham, USA) was used in this study. Calibration solution, internal standards, and samples were prepared by a Mettler XP205 analytical balance (Zurich, Switzerland). A K-Lite8 Series electrolyte analyzer (Meizhou, China) was used for method comparison. Ultrapure deionized water was prepared by a Millipore Advantage water purifier (Billerica, USA).
Reagents and samples
Nitric acid (Lot: 1213070, 69% w/w) was obtained from Fisher Scientific (Canada) and prepared as a dilute nitric acid (0.3% w/w) solution with ultrapure deionized water. Standard reference material (SRM) 3129a (Lot: 100714) from the National Institute of Standards and Technology (NIST, Gaithersburg, USA) was used for calibration. Internal standard Germanium (Lot: GSB 04-1728-2004, 13.772 mmol/L) was obtained from the National Institute of Standard Materials (Beijing, China). SRM 956d (frozen serum, stored at −80 C) from NIST was used for accuracy assessment. Clinical serum samples (stored at −80 C) were collected from Shanghai Mental Health Center.
Internal standard solution, calibration solution, and serum preparation for ICP-MS
Before sampling, all plastic tubes and pipette tips were washed using 0.3% nitric acid and ultrapure deionized water. Diluting SRM 3129a with 0.3% nitric acid was prepared for a lithium standard solution (1.180 mmol/L). Diluting germanium with 0.3% nitric acid was prepared for a germanium internal standard solution (0.275 mmol/L).
Blank solution: 0.3% nitric acid.
Samples: accurately weighing 0.5 g of serum and 0.5 g of the internal standard germanium solution into a plastic tube and diluting 100 times with 0.3% nitric acid.
Standard addition solution 1: accurately adding 0.05 g standard solution to 10.0 g of sample. Standard addition solution 2: accurately adding 0.1 g standard solution to 10.0 g of sample. Standard addition solution 3: accurately adding 0.2 g standard solution to 10.0 g of sample.
ICP-MS measurements
The ratio of the ICP-MS signals of lithium and germanium was proportional to the concentration of the element in the material of investigation. For calibration, the calibrators were measured before and after the samples. The mean of the calibrators was used for calculation. Method parameters are as follows:
Scan Mode: Peak Hopping, Dwell Time = 100 ms, Integration = 3000 ms, Analysis Mode: KED, He gas flow rate = 3 ml/min, Rpa = 0, Rpq = 0.25, Sweeps = 40, Readings = 1, Replicates = 3.
Calculations
Lithium concentrations (mmol/L) were calculated according to the standard addition method
11
(see Figure 1). Addition 0 is unknown sample without standard solution, Addition 1 is Standard addition solution 1, Addition 2 is Standard addition solution 2, Addition 3 is Standard addition solution 3, and c is concentration of the unknown sample. Standard addition method.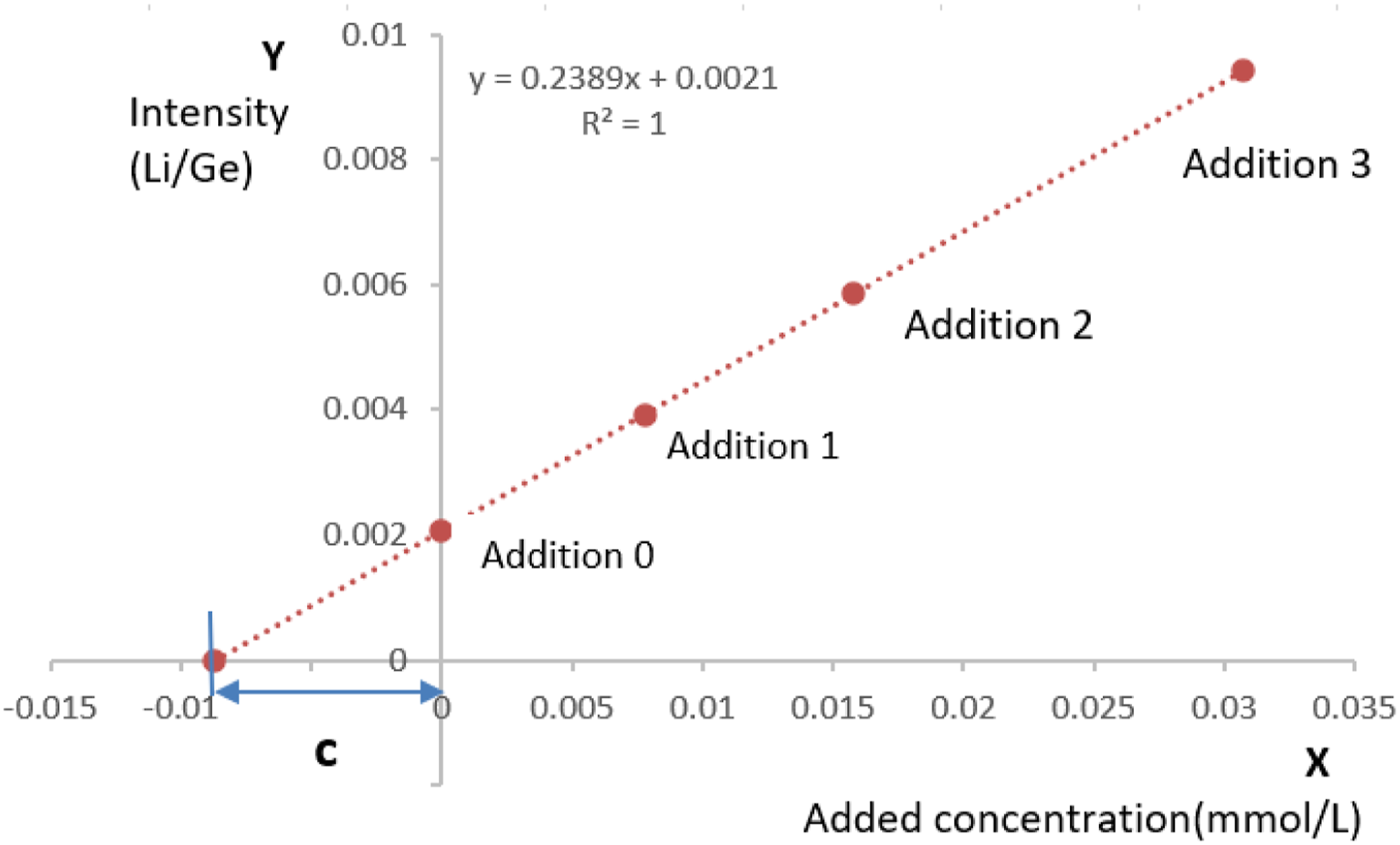
Y = kX + b, Y is the Li/Ge signal ratios of the calibration solutions and X is the added lithium concentration in the calibration solutions, and k is the slope of the calibration curve.
c = I/k, I is the Li/Ge signal ratios of the unknown serum samples.
C = (c × n × D)/(1000 × 6.941), C is the original lithium concentration in the serum samples, n is the dilution factor of the serum samples, D is the density of the serum samples, and 6.941 is the relative atomic weight of lithium.
Measurement uncertainty
The uncertainty of measurement was evaluated according to GUM. 12 The following uncertainty components and type were considered:
u(Rep): the uncertainties of all the daily repeated procedures (type A).
u(p): the uncertainties of Purity of reference material (type B).
u(m): the uncertainties of weighing procedures (type B).
Combined relative standard uncertainty uc
Combined standard uncertainty uc (y) = uc × mean value. The expanded combined measurement uncertainty Ue = k × uc (y) (using the number of the degrees of freedom, the coverage factor k was estimated for a coverage probability 95%, K=2);
Method validation
The linearity study in this work was based on the linearity of the serum lithium matrix calibration curve. The linearity was considered to be satisfactory when the r value was higher than 0.9999.
The specificity of the method was evaluated by measuring the intensity of the lithium signal of the blank with the added internal standard, compared with the intensity of the lithium signal of the serum samples. 13
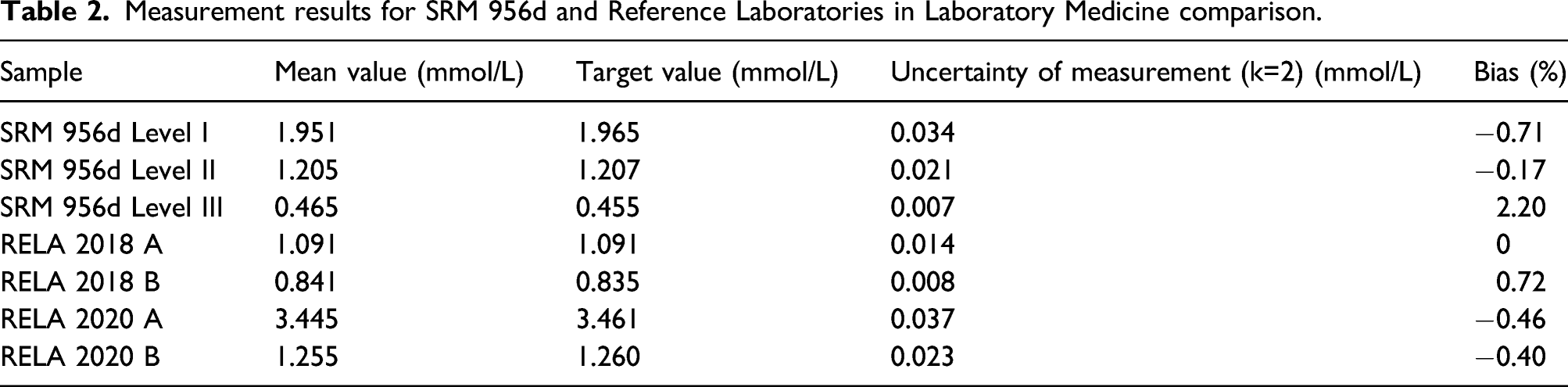
Measuring samples three times a day, repeated for 3 days for imprecision, trueness, and accuracy evaluation. The trueness of the method was estimated using SRM 956d. Moreover, the ICP-MS results were compared to the participant results of External Quality Assessment Scheme for Reference Laboratories in Laboratory Medicine (RELA) intercomparison studies for accuracy evaluation. The relative biases should be < 3.00%. 14
Method comparison
According to the Clinical and Laboratory Standards Institute document EP09-A3, to validate the correlation of the ICP-MS method, 40 clinical serum samples were measured and compared with the results of the ion-selective electrode (ISE) routine method (K-Lite8 Series electrolyte analyzer, indirect method). Passing-Bablok plots and Bland–Altman plots were used to compare the results.
Results
Linearity and specificity
Excellent linearity was observed in this study. Y is the signal ratio of Li/Ge, X is the concentration. A linear regression equation is established, with all all r-values > 0.9999. Blank reagents, with (38.3 CPS) and without (37.8 CPS) a germanium internal standard, were prepared and measured for lithium. The lithium signal intensities of the two blank samples were similar, and less than 0.5% of that in the low calibrator (32144 CPS). Therefore, serum sample preparation and germanium internal standard did not introduce lithium interference.
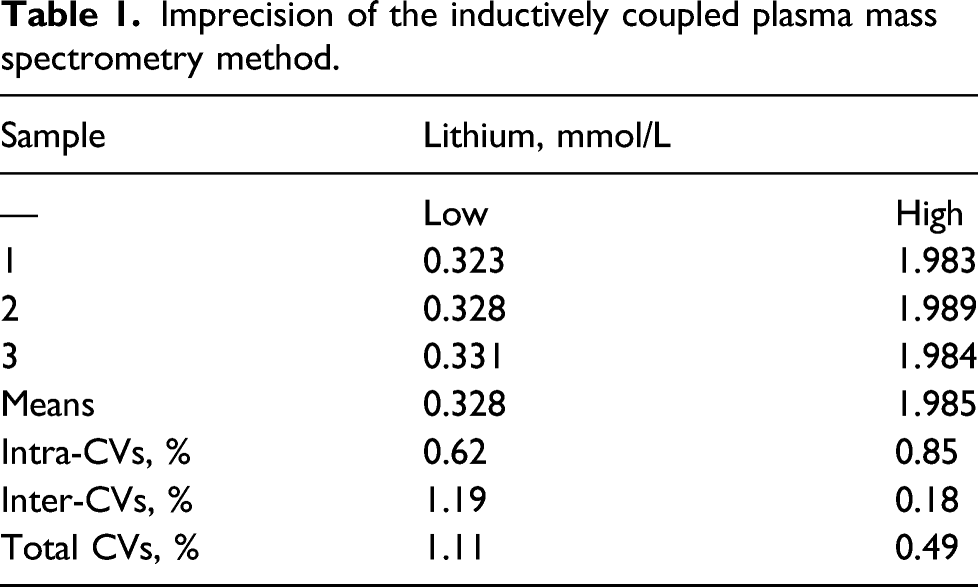
Imprecision
Imprecision of the inductively coupled plasma mass spectrometry method.
Trueness and accuracy
Measurement results for SRM 956d and Reference Laboratories in Laboratory Medicine comparison.
Method comparison
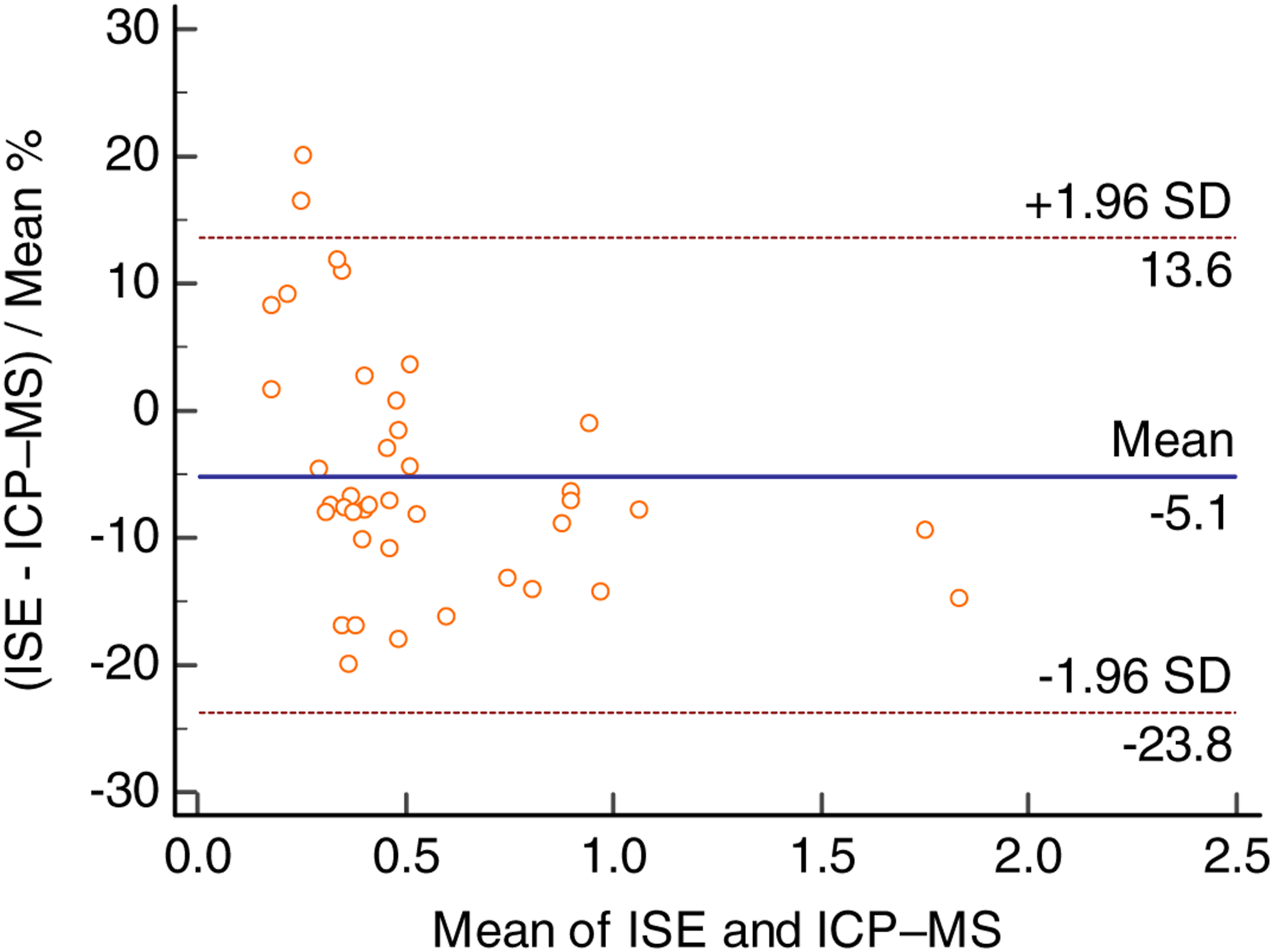
Forty clinical samples (minimum value 0.175 mmol/L, maximum value 1.971 mmol/L) were assayed and compared with the results of the routine ISE method. Passing-Bablok plots were used to evaluate the results as measured by the two methods. The correlation equation is as follows: lithium (ICP-MS, mmol/L) = 1.149 × lithium (ISE, mmol/L) − 0.031 (r = 0.9953). No significant deviation was found from linearity (p = 0.30) about the two methods. And for the reason for 1.149, see the Discussion section. The comparisons are shown in Figure 2. Bland–Altman plots showed that the differences appeared to be concentration-independent in Figure 3. Comparison of inductively coupled plasma mass spectrometry measurement results and results obtained by ISE for lithium Passing-Bablok regression analysis. Difference between inductively coupled plasma mass spectrometry measurement results and results of ISE for 40 serum lithium samples using Bland–Altman analysis.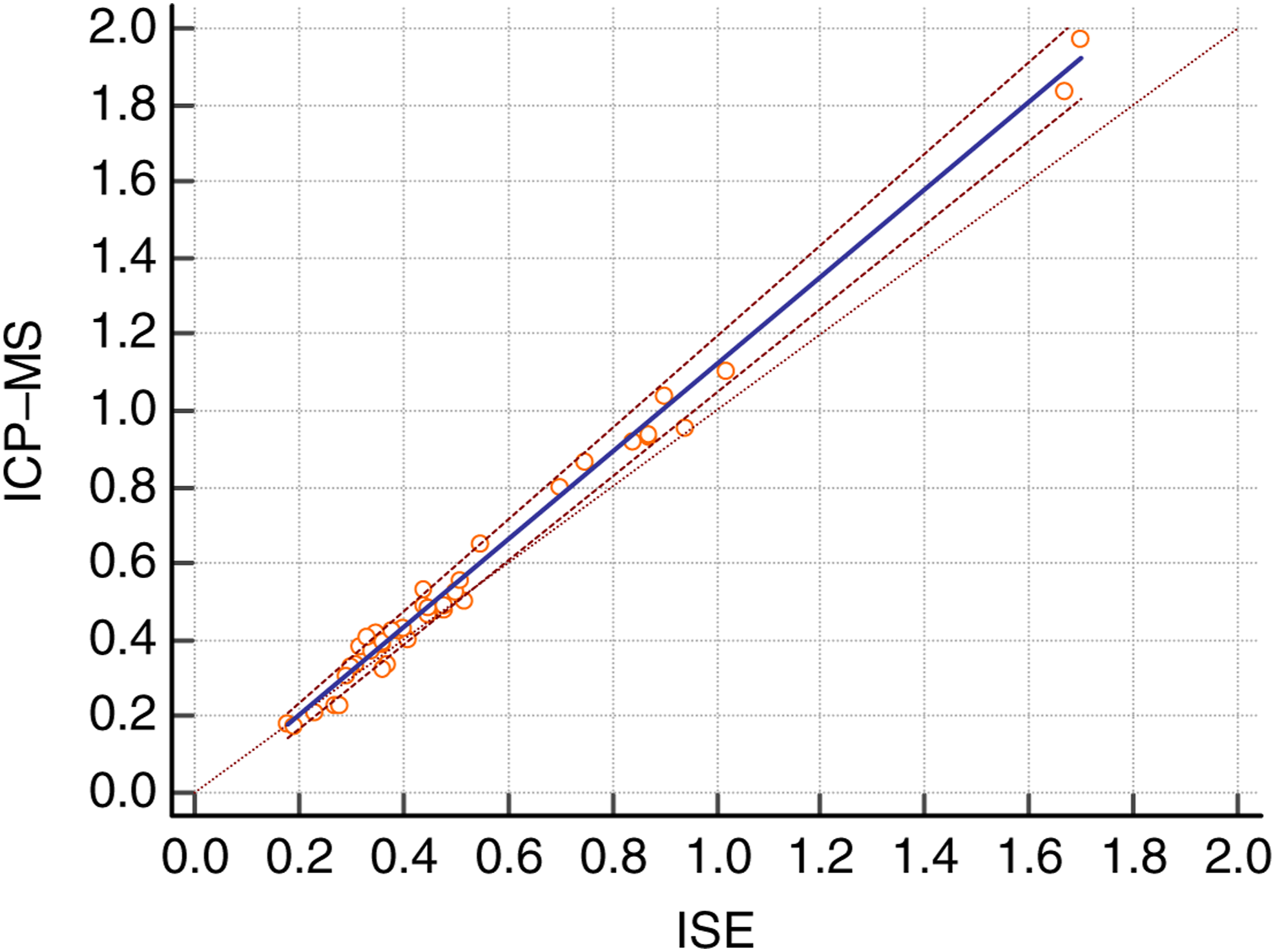
Discussion
In this work, quantitative method, pretreatment of the serum sample and processes of lithium measurement were studied. Because gravimetric procedures have much lower uncertainties than volumetric procedures, gravimetry was used instead of volumetry. The calibration is based on the standard addition method. The calibrators were made from higher order reference materials supplied by NIST and reference measurement procedures with traceability to SI according to ISO 17511:2020. 15
Due to the development of instrument technology, the ICP-MS method could provide a lower detection limit than flame atomic emission spectrophotometry 5 and much simpler than isotope dilution thermal ionization mass spectrometry. 6 The internal standard of lithium must be carefully selected for quantification. 6 Li was used as an internal standard for lithium by Kramer et al. 7 However, isotopes are rare and expensive and we need to look for common internal standard. Here, germanium was used as an internal standard because the serum level of germanium is very low and has no mass spectrometry interference and no interference with the lithium signal. Because a different matrix was used for the serum sample and the calibrator, there will be a matrix effect. 16 Grote-Koska et al. 8 used bracketing method quantification. However, samples should be digested. Here, the standard addition method 13 was used. In this method, sample does not need to be digested and can ensure the matrix of the calibrator is consistent with the serum sample. Moreover, the evaluation of accuracy based on analyzing SRM 956d and the RELA comparison all got satisfied results.
The results acquired with the ICP-MS method were well correlated with results acquired using the ISE method (r = 0.9953, p = 0.30) but with a positive slope (1.149). There may be the following reasons. Firstly, the ICP-MS method produces test results for lithium traceable to the NIST calibration solution SRM 3129a. But the ISE method produce test results for lithium traceable to the National Institute of Standard Materials (China, 360018-201701). Secondly, methodological difference and the ISE method are indirect potentiometry.17,18 Thirdly, the Nernst equation is the principle of the ISE, and the direct response is ionic activity. When in an ideal dilute solution, concentration equals ion activity. However, serum samples are not ideal dilute solutions because serum samples contain proteins. And a similar situation can be found in reference 19. So, a positive slope (1.149) is reasonable. The domestic routine ISE method needs to be improved to reduce the gap with the reference method. In the next step, we will carry out proficiency testing schemes of serum lithium in Shanghai area.
In conclusion, a new method to measure serum lithium was developed and validated. The method is based on ICP-MS analysis and standard addition method. And germanium is used as an internal standard. This new ICP-MS method has higher metrological traceability and is precise, accurate, simple, and suitable for a candidate reference method.
Footnotes
Acknowledgments
The authors are grateful to the Shanghai Center for Clinical Laboratory and Shanghai Mental Health Center, China, for the support of this project.
Author contribution
YJ designed the project and supervised the project. XF carried out the experiments and statistical analysis and wrote the manuscript. QL and ZJ assisted with statistical analysis. PL and MC collected clinical serum samples. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Key Research and Development Program of China (2019YFF0216500).
