Abstract
Background
Repeated phlebotomy for laboratory diagnostic testing is a known cause of iatrogenic anaemia and in critically ill neonates often leads to blood transfusion being required. This study has developed a spreadsheet clinical decision support tool to allow neonatal staff to determine the true minimum blood volume required to analyse groups of blood tests and modelled its potential benefit compared with the existing system in use.
Methods
The tool calculates the minimum blood volume accounting for novel factors including the current patient haematocrit for plasma/serum samples, instrument minimum test and dead volumes (including those where shared) and sharing of samples within/between laboratory departments. A year of neonatal unit laboratory requests were examined comparing the volumes and containers of blood recommended by the hospital information system with both the amount actually collected by staff and that recommended by the tool.
Results
A total of 463 patients had 8481 blood draws for 23,899 tests or test profiles over the year. The hospital information system recommended collecting 11,222 mL of blood into 18,509 containers, while 17,734 containers were actually received (10,717 mL if fully filled). The tool recommended collecting 4915 mL of blood into 15,549 containers.
Conclusions
This tool allows neonatal intensive care unit staff to objectively determine the minimum blood volume required for a combination of tests and is generalizable between laboratory instruments. Compared with the hospital information system, use of the minimum blood volume clinical decision support tool could maximally reduce the volume of blood collected from this neonatal unit by more than a half. Neonatal intensive care unit staff had apparently already gone some way to determining their own minimum volumes required.
Introduction
Anaemia among babies being treated in neonatal intensive care units (NICUs) is common, especially among those weighing less than 1 kg, with 80% of this low birth weight group requiring at least one blood transfusion during their admission. 1 , 2 The cause of the anaemia is multifactorial, but phlebotomy losses for diagnostic testing is a well-recognized major contributor.3–9 As the blood volume of neonates is approximately 85 to 105 mL/kg, even a single 1 mL blood sample could represent more than 1% of its total blood volume. 7
Initiatives to reduce the blood volumes for testing have included the increased use of point of care testing (POCT) instruments, which often have lower sample volume requirements than traditional laboratory instruments.2–5 However, use of the laboratory is often still the mainstay, either to measure tests unavailable on POCT platforms or to check results obtained in this way.
Laboratory handbooks have been the traditional means of communicating the container and volume requirements for individual tests or test profiles. More recently, laboratory or hospital information systems (HISs) increasingly guide healthcare staff on the sample containers that require to be filled in order to complete any requested combination of tests from a single phlebotomy draw. However, both approaches have limitations when trying to establish the absolute minimum volume required for laboratory testing. Since handbooks and HISs tend to only deal with individual tests or the commonest test profiles, they are unable, for example, to convey the knowledge that if a single plasma sodium test requires 0.1 mL blood, then a request for sodium, potassium and chloride on most instruments will also require just 0.1 mL rather than 0.3 mL. The same applies should instrument dead volumes be shared between a number of tests. Even quoting ‘0.1 mL’ may have been decided through expediency due to limitations to the volumes that can be used by the HIS or to ensure there is sufficient sample to repeat an analysis, as opposed to stating the volume most closely reflecting the actual requirements of the laboratory instrument. Assuming this ‘0.1 mL’ were truly accurate, some HISs will then only recommend the type and number of containers required for the test(s) without qualifying that the tube(s) need not necessarily be fully filled. Laboratory handbooks may also struggle to document how the organization can or cannot share the same container for different tests within or between a particular laboratory’s departments. Lastly, one of the most crucial aspects of determining the minimum blood volume required for a serum or plasma test is the haematocrit of the individual patient. Without this information, quoted test requirements have to accommodate the possibility of prodigiously high haematocrit values when, for most patients, the required amount of serum/plasma could be retrieved from a lower blood volume.
We set out to develop an informatics ‘minimal blood volume’ clinical decision support (MBV-CDS) tool that would be able to overcome all these and other limitations and be simple to use by NICU staff in order to accurately determine, for individual patients, the minimum blood volume and containers required for any combination of commonly requested laboratory tests or test profiles. We then sought to establish the impact this MBV-CDS tool could have on the volume of blood taken for laboratory tests from patients in our NICU over the period of a year.
Methods
Spreadsheet tool development
An Excel spreadsheet was constructed where one tab contained the data for the tests and test profiles. Table 1 contains the information fields in that tab which are initially required to be entered by the laboratory on a single occasion. The data entered for this study were for the instruments and containers in use for 67 of the most commonly requested blood tests and test profiles within the Clinical Biochemistry, Haematology, Transfusion, Immunology and Serology divisions of Pathology at Sidra Medicine, Doha, Qatar. This also tailored how tests with the same sample types were or were not routinely shared between divisions. The laboratory testing instruments were from Beckman Coulter DxC, DxI and DxH (Beckman Coulter Inc, Brea, CA, USA), ACLTOP 700 (Instrumentation Laboratory Company, Bedford, MA, USA), Diasorin Liaison XL (DiaSorin, Saluggia, Italy), BioRad Variant II (BioRad, Hercules, CA, USA) and the ESR STAT PLUS (HemaTechnologies, Lebanon, NJ, USA). Sample and dead volume information was obtained mainly from manufacturers’ instrument manuals and test information sheets. For example, the DxC instrument has two sections, a ‘modular’ section sharing the same dead volume between all ion selective electrodes and a ‘cartridge’ section sharing a dead volume between other general chemistries. The containers used were BD microtainers (Becton, Dickinson and Company, Franklin Lakes, NJ, USA).
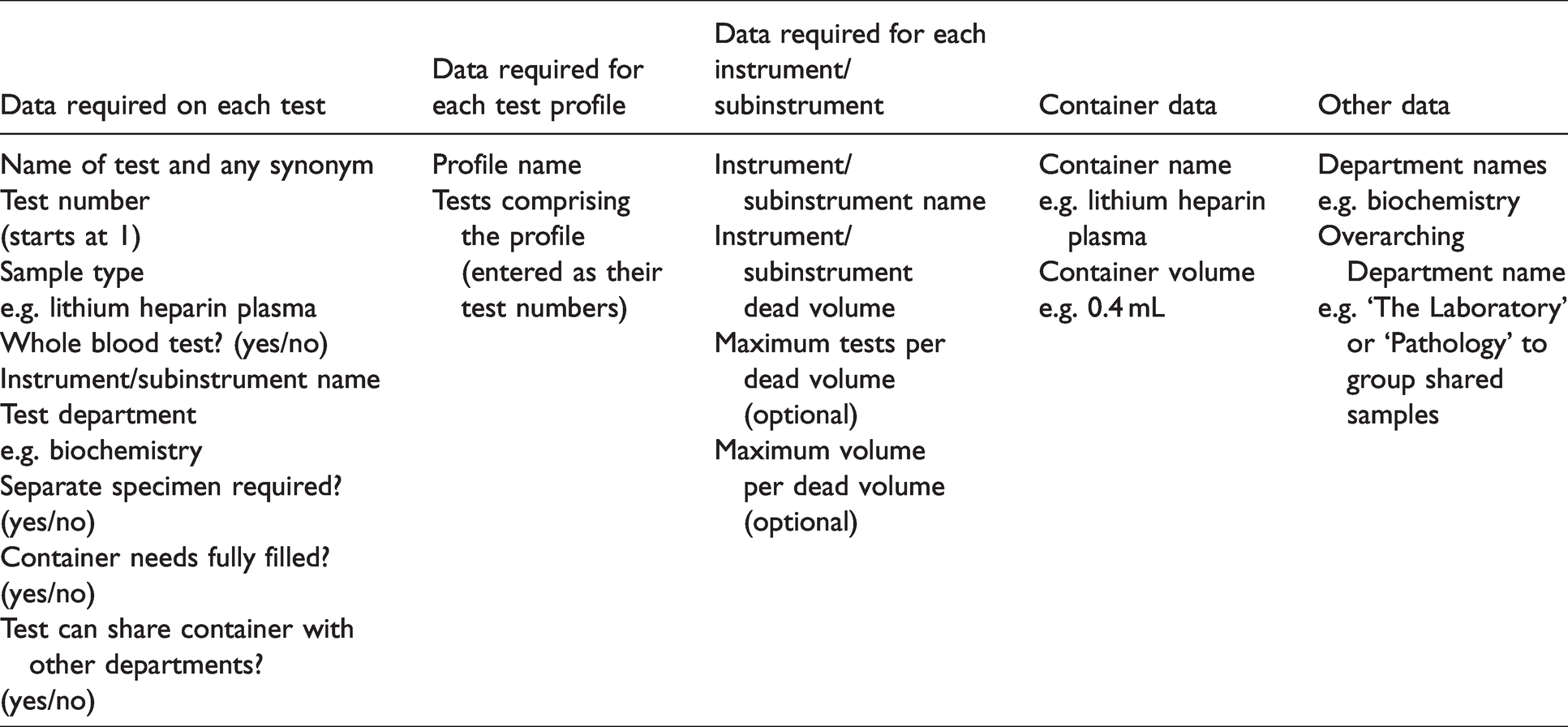
An input interface in a separate tab allowed the test requester to enter the test name(s) for which the minimum blood volume(s) were desired. An example is shown in Figure 1. Clicking on ‘Search’ allows a test or test profile name to be searched for and added to the list of tests, with the output providing the minimum volume required for the patient hematocrit manually entered and the containers required to fulfil this. Further individual tests/profiles can be added by searching again; individual tests can be removed by clicking on the cell with its name and deleting to see the effect this would have on reducing the required blood volume.
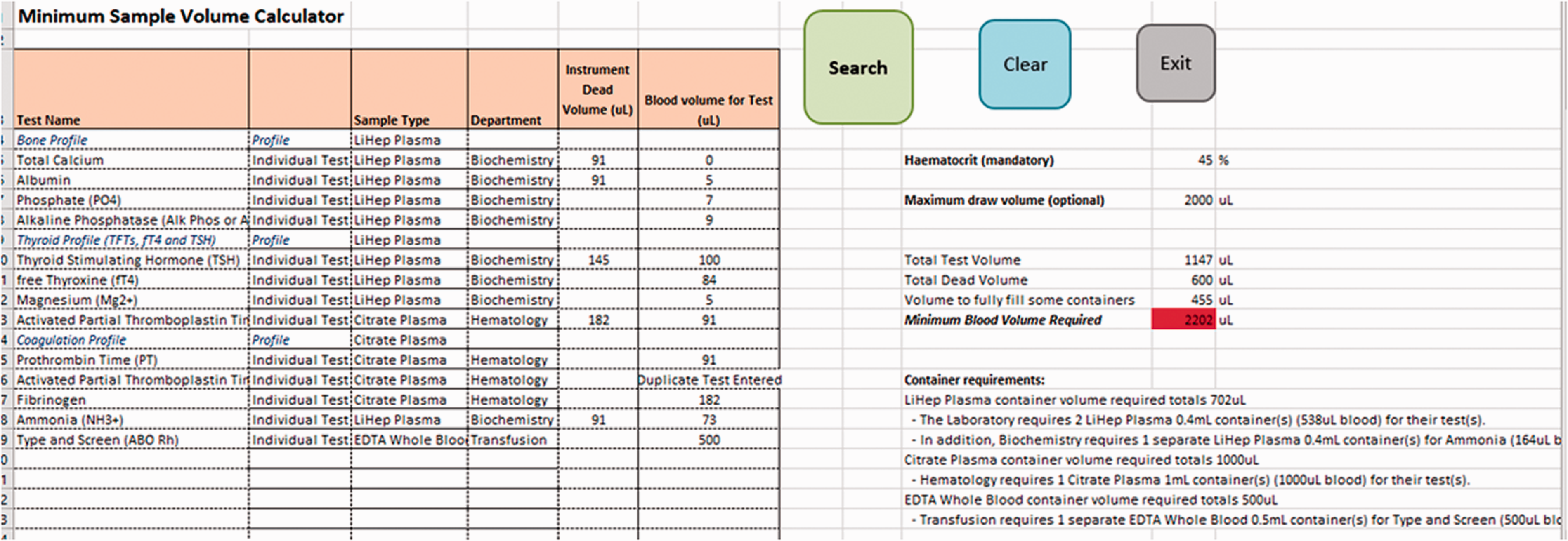
Screenshot of the spreadsheet tool.
The commonest requested tests and profiles were validated to ensure that the sample volumes recommended by the tool reflected amounts which could be analysed by the instruments.
Blood volume comparisons
To determine the potential effect implementation of the MBV-CDS tool could have in changing the volume of blood being drawn, one year of anonymized NICU laboratory requesting data from our single centre institution was extracted from the enterprise HIS (Cerner Corp., North Kansas City, MI, USA) over the period 1 June 2019 to 31 May 2020. Only data relating to the 67 blood tests/test profiles included in the spreadsheet were used; point of care test requesting was excluded.
Several comparisons between different blood volumes were made. The first set of volumes were derived from those recommended by the HIS itself which is able to calculate the number and type of blood containers required for a particular set of test requests based on test volume and container data entered into the HIS by the laboratory. The number and type of containers recommended for each blood draw (which is communicated to the collector during the specimen collection process) were recorded.
It is common for intensive care nursing staff to informally establish their own MBVs through experience (known as an informal process heuristics) to minimize blood loss associated with phlebotomy, 10 so the total number of containers submitted to the laboratory for ordered tests was also established.
To establish the volumes recommended by the MBV-CDS tool, every NICU test request was also individually entered into the MBV-CDS tool to establish the recommended blood volume, container type and number(s) of containers. The haematocrit for each patient during their hospitalization was derived from assessments from a total of 2730 full/complete blood counts by the laboratory and 26,101 blood gas samples collected on patients during the study period (GEM 4000, Instrumentation Laboratory, Werfen, Barcelona, Spain). For the purpose of the study, the average haematocrit for each patient’s stay was used for all of their blood volumes calculated by the tool. In clinical use, if a recent haematocrit was unavailable then the spreadsheet could default to a chosen percentage.
Since NICU staff were unlikely to be able to collect and decant blood into containers with the millilitre accuracy suggested by the MBV-CDS tool, an additional analysis was performed based on the knowledge that the BD microcontainers used in the study hospital had only two levels of markings: one for their maximum volume and one for either half this volume (e.g. 400 and 200
Institutional Review Board review was obtained and deemed this study protocol with anonymized data to be of exempt status.
Results
Figure 1 shows features of the MBV-CDS tool which allows the minimum test volume to be calculated more accurately than by a typical HIS. Appendix 1 provides examples of these features in relation to the figure.
The NICU test requests over a 12-month period showed that 463 patients were bled 8481 times to obtain 23,899 tests/test profiles (median 19/patient, interquartile range 9–50) and comprising 81,542 individual tests (n.b. a full/complete/blood count counted as one test). Table 2 shows the number of containers and test blood volumes recommended by the HIS, the containers actually collected by staff and the containers recommended by the MBV-CDS tool, either with exact blood volumes or volumes rounded up to the nearest level marking on a container, as described in Methods section. There was a 16% reduction in the container count using the MBV-CDS tool compared with the HIS recommendations and a reduction in the volume of blood recommended to be taken, either by 56% if blood volumes recommended by the MBV-CDS tool were used or by 36% if containers were filled to the next nearest container level, as described in Methods section. Compared with what was being actually taken by NICU staff, the reduction in container count using the MBV-CDS tool was 12%. A potential blood volume reduction of 54% could be achieved when sampled blood volumes recommended by the MBV-CDS tool were followed; if containers were filled to the next nearest level, as described in Methods section, a potential blood volume reduction of 33% could still be realized when using the MBV-CDS tool.
Number of blood containers and blood volume either collected or recommended for 463 NICU patients over a 12-month period.

aAssumes no container was under/overfilled.
Figure 2 shows the distribution of blood collection volumes for individual patients in each of these scenarios.
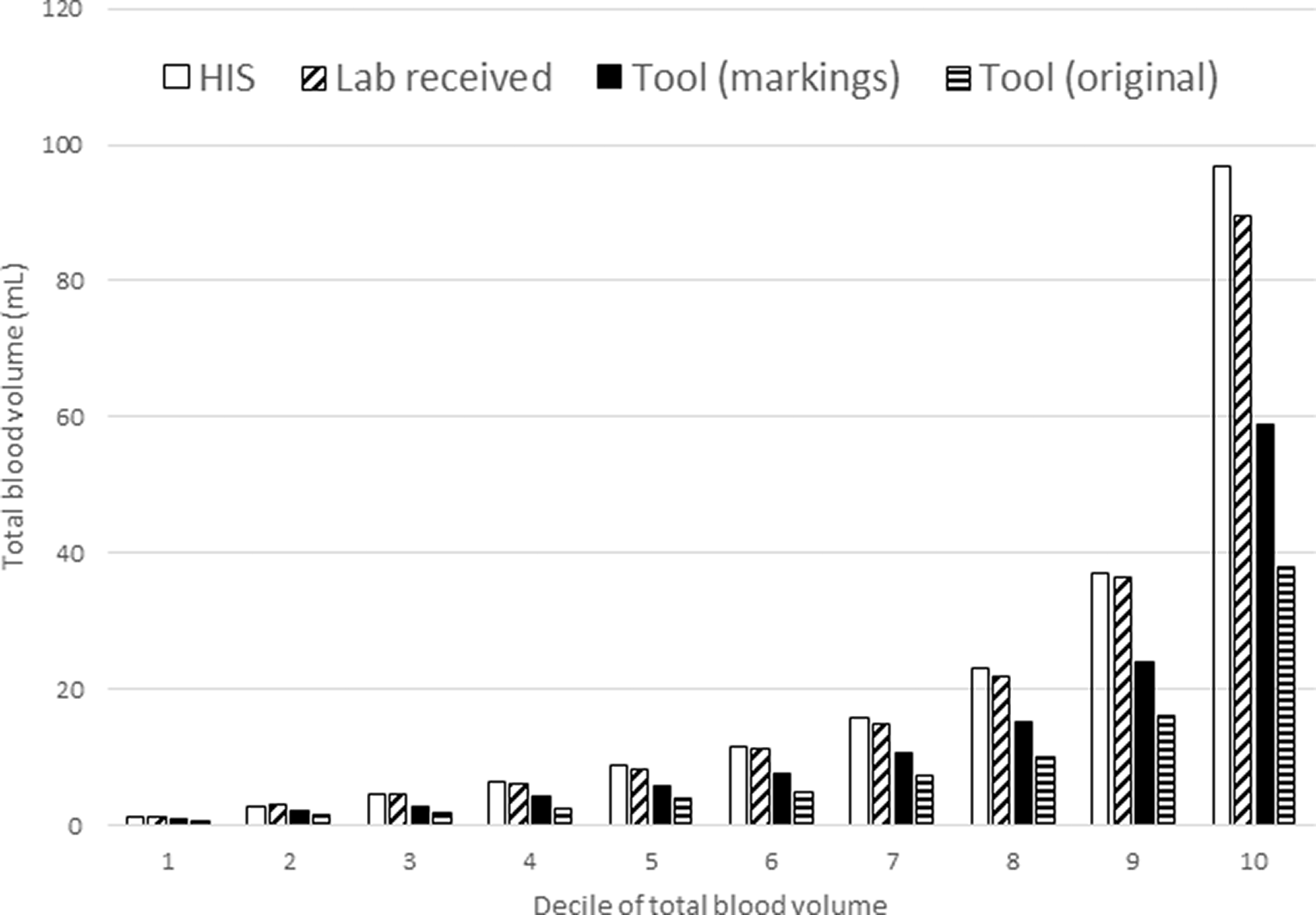
Deciles of blood volumes recommended or taken on NICU patients during the study period. Median of each decile shown. Tool (original): minimum volume from MBV-CDS tool. Tool (markings): minimum volume from MBV-CDS tool rounded up to the nearest level marking on a container.
Discussion
It is common for clinical laboratories to be asked what the smallest amount of blood is required in order to report results on a number of tests, especially when the patient has limited circulating blood volumes, such as a neonate; furthermore, variable situational circumstances of patients, especially critically ill neonates, make such queries likely to be given the answer ‘it depends’. This study describes and validates a novel informatics clinical decision support tool which can incorporate many of the key variables of these situational circumstances, including contemporary haematocrit concentrations, concurrent testing requests, and laboratory instrument test and dead space requirements, to provide a simple and clear answer to the query of the minimal blood volume and container number required for the ordered tests. Furthermore, this MBV-CDS tool was developed to be adaptable for use by laboratories utilizing instruments different from those in our institution.
Compared with the standard HIS recommendations for specimen collection, the MBV-CDS tool was able to recommend a reduction in blood sampling of up to 54% in our study population of critically ill NICU patients. However, no phlebotomist could collect and transfer blood volumes as accurately as the tool recommends but even using the conservative and pragmatic approach of filling up to lines already on blood containers there could still be a 33% reduction in blood collection volumes. Filling to these lines might also minimize any issues that could arise from a tube, such as those with additives, requiring a minimum blood volume, and reinforces the need to follow tube manufacturer’s guidance no matter what volume the tool recommends. In further respect of these fill lines, it is unfortunate that the marks on the paediatric containers used in the study hospital only started either half-way or two-thirds the way up to the maximum fill level and not before, otherwise the gains by this simple technique could have been greater; this represents an opportunity for improvement in the manufacturing of such collection tubes.
We are not aware of another CDS tool which been used to minimize neonatal phlebotomy losses, as the preponderance of publications in this area have rightly focused on initiatives such as increasing the use of POCT, reducing the blood discarded at the start of a draw and standardizing physician protocols.2–5 However, the incorporation of a novel MBV-CDS tool, such as the one presented in this study, with a suite of strategies to reduce overall phlebotomy blood loss in patients should be considered in future efforts. Of note, this MBV-CDS tool was primarily designed to be used by NICU staff but its informatics foundation allows it to be easily adapted and adopted for use in a range of healthcare environments within an enterprise-level HIS. Regarding integration into a HIS, this could further refine the spreadsheet used here by potentially being able to use electronic health record information such as the latest patient haematocrit (from laboratory or point of care sources) to automatically populate the tool and ensuring only the latest version of it was in use. Even without considering the haematocrit of an individual patient, there may be elements of our spreadsheet, such as using exact test volumes or accounting for shared dead volumes, which could still be incorporated into an integrated tool to improve its accuracy for all ages of patients.
It is of interest that it appears the innate informatics of human problem-solving among our institution’s NICU staff had already created informal heuristics to reduce required blood volumes beyond those recommended by the existing enterprise HIS system, as the container count and blood volumes extracted by staff were less than that those recommended by the HIS, albeit not reduced to the degree advocated by the MBV-CDS. The latter means that, even for experienced NICU staff, this novel MBV-CDS tool could allow them to confidently go further in reducing blood sample volumes. Alternatively, if staff knew they had collected too small a sample then, before sending it to the laboratory, they could use the MBV-CDS tool to calculate and proactively prioritize the most urgently required tests to be analysed from the sample volume they have managed to obtain. This would likely reduce clarification ‘phone’ calls between NICU and the laboratory and might even reduce the need for rebleeding.
This study is not without limitations. Being retrospective in nature, it is not possible to definitively claim that use of the MBV-CDS tool would result in clinically significant reductions in phlebotomy-induced anaemia and blood transfusion requirements for intensive care patients; such a study design, however, is being planned. Secondly, the blood volumes for each test recommended by the HIS are those of the study institution and so could be different with those used elsewhere, although they were determined in the knowledge they were for use in a paediatric hospital. Lastly, there is the problem that the volume calculated by the tool is genuinely the minimum required, which means that if an analysis is required to be repeated, either because of an analytical problem or to verify an unexpected result, or if additional tests are asked to be added on, then there would be a lack of sample remaining with which to do so. This would be less of an issue if filling were to the next container line but if the absolute minimum volume is being aimed for then it may be that this option should be reserved for patients where phlebotomy losses are likely to be most pronounced, such as those who are critically or chronically ill.
In conclusion, this novel ‘minimum blood volume’ clinical decision support tool can provide the smallest blood volumes required for the most commonly requested tests in a way which is superior to that calculated by a generic enterprise HIS or by nurses’ informal human heuristic; furthermore, this MBV-CDS can provide recommendations individualized to the haematocrit of the patient and laboratory instruments used by the testing centre. When applied to the intensive care area, this MBV-CDS also has the potential to empower staff to objectively and substantially reduce the phlebotomy losses of their patients.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Exempted by Sidra Medicine IRB (IRB00009930).
Guarantor
Eric Kilpatrick.
Contributorship
ESK and EG conceived the idea, collected and analysed the data. The tool was developed by ESK with assistance from EG and BL. BL helped develop the idea and obtained ethical exemption. ESK wrote the paper draft and EG and BL amended and approved the final version.
