Abstract
Background
The variability of Covid-19 severity between patients has driven efforts to identify prognosticating laboratory markers that could aid clinical decision-making. Procalcitonin is classically used as a diagnostic marker in bacterial infections, but its role in predicting Covid-19 disease severity is emerging. We aimed to identify the association between procalcitonin and Covid-19 disease severity in a critical care setting and whether bacterial co-infection is implicated.
Methods
We retrospectively reviewed Covid-19 patients with procalcitonin concentrations measured in a critical care setting at our institution between February and September 2020. Laboratory markers including peak procalcitonin values and a range of bacterial culture results were analysed. Outcomes were the requirement and duration of invasive mechanical ventilation as well as inpatient mortality.
Results
In total, 60 patients were included; 68% required invasive mechanical ventilation and 45% died as inpatient. Univariate analysis identified higher peak procalcitonin concentrations significantly associated with both the requirement for invasive mechanical ventilation (OR: 3.2, 95% CI 1.3–9.0, P = 0.02) and inpatient mortality (OR: 2.6, 95% CI 1.1–6.6, P = 0.03). Higher peak procalcitonin concentrations was an independent predictor of mortality on multivariate analysis (OR 3.7, 95% CI 1.1–12.4, P = 0.03). There was a significant positive correlation between increased peak procalcitonin concentrations and duration on invasive mechanical ventilation. No significant difference was found between peak procalcitonin concentrations of patients with positive and negative bacterial cultures.
Conclusions
Elevated procalcitonin concentrations in Covid-19 patients are associated with respiratory failure requiring prolonged invasive mechanical ventilation and inpatient mortality. This association may be independent of bacterial co-infection.
Introduction
The Coronavirus disease 2019 (Covid-19) pandemic, caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has taken a great humanitarian and economic toll in most countries and has spurred global efforts to develop and refine new therapeutic and diagnostic techniques. A significant proportion of patients with Covid-19 require hospitalization and treatment in high dependency (HDU) or intensive treatment units (ITU), where invasive mechanical ventilation is the cornerstone of treatment for respiratory failure in up to 88% of patients. 1 In high-demand settings where critical care resources including ventilatory equipment are constrained, establishing laboratory tests associated with severe Covid-19 and amplified risk of mechanical ventilation and mortality is crucial.
Procalcitonin (PCT) is a 116 amino acid precursor for the hormone calcitonin. Traditionally, PCT has been used in clinical practice as a diagnostic marker of bacterial infection, aiding clinical decisions surrounding the use of antibiotics. 2 Early data suggest that increased PCT concentrations may predict the severity of Covid-19. A meta-analysis of four studies from China showed that elevated PCT concentrations are associated with a five-fold higher risk of severe Covid-19, although the underlying pathophysiological mechanisms have not been elucidated. 3 Some authors have postulated that this positive association reflects bacterial co-infection, although currently, there is insufficient evidence to validate this hypothesis. 4 Additionally, guidelines from the National Institute for Health and Care Excellence (NICE) in the United Kingdom (UK) state that there is insufficient evidence combined to recommend PCT testing to guide decisions regarding antibiotics. 5 Consequently, further exploration in a variety of settings is required to fully establish the prognostication role of PCT in Covid-19 and whether bacterial co-infection is implicated.
Therefore, the aims of our study were to identify whether increased PCT concentrations are associated with use and duration of invasive mechanical ventilation and mortality in Covid-19 patients in a critical care setting and whether bacterial co-infection is implicated.
Materials and methods
All patients treated for Covid-19 in a critical care setting (HDU or ITU) at our institution in the UK who had PCT concentrations measured between February and September 2020 were identified retrospectively. Those who had ceilings of treatment were excluded. Patients with active Covid-19 were defined as those with positive real-time reverse transcriptase-polymerase chain reaction (RT-PCR) assay for severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) on nasopharyngeal swab or those being treated for suspected Covid-19 on clinical and radiological evidence of Covid-19 on chest radiograph or computed tomography. 6
Patient demographics and co-morbidities classified using the Charlson comorbidity index were extracted from patient records and our local clinical coding department. 7 The peak PCT concentration recorded during the critical care admission was used, with a cut-off value of 0.5 ng/dL as per local laboratory guidelines. Other prognostic biochemical and haematological investigations (platelet count, neutrophil count, lymphocyte count, C-reactive protein [CRP], D-dimer, ferritin, high sensitivity troponin T [hsTnT] and lactate dehydrogenase [LDH]) were recorded as close as possible to the time of peak PCT concentration. 8 Where values were above or below the range quantifiable by the laboratory, the highest or lowest quantifiable value was used, respectively.
Treatment of patients during the critical care admission with antibiotics, remdesivir, anti-IL6 and corticosteroids for any reason was recorded, as well as the proportion of patients receiving antibiotics for at least 24 h prior to critical care admission. The highest weight-adjusted thromboprophylaxis dose used during the critical care admission was recorded and classified as either prophylactic dose (use of low-molecular weight heparin or unfractionated heparin at prophylactic dose) or treatment dose (use of low-molecular weight heparin at treatment dose or an intravenous heparin infusion).
To identify concomitant bacterial infection, positive results for bacteria were considered from blood, sputum, bronchial lavage (either non-directed bronchoalveolar lavage or bronchoscopy with bronchoalveolar lavage), urine, line and wound cultures as well as urinary pneumococcal and legionella antigen tests. Blood cultures positive for coagulase negative Staphylococci which were deemed to be non-significant contaminants by clinicians at the time were classified as negative cultures.
Primary outcomes were defined as requirement of invasive mechanical ventilation and inpatient mortality. The secondary outcome was defined as duration of invasive mechanical ventilation using previously described methodology. 9 The number of days from initiation of invasive mechanical ventilation were counted up to a maximum of 28 days. The stop date was defined as the day when invasive mechanical ventilation was successfully stopped and not restarted within two days. Patients who died within 28 days of starting invasive mechanical ventilation were classified as receiving 28 days of invasive mechanical ventilation.
Statistical analyses
Kolmogorov-Smirnov and Shapiro-Wilk tests were used to confirm normality of continuous variables. When normally distributed, continuous variables were expressed as mean ± standard deviation and compared using the Student’s independent t-test. Non-normal data were presented as median (interquartile range: 25th–75th percentile) and compared using the Mann-Whitney U test. Categorical variables were summarized as frequencies and percentages and compared using either the Chi-squared test or Fisher’s exact test. Maximum PCT concentrations underwent logarithmic transformation to achieve normality (log10PCTpeak) and to allow for statistical analysis with parametric methods.
Covariates implicated in Covid-19 patient outcomes including age, ethnicity, gender, Charlson comorbidity index, log10PCTpeak as a continuous variable, neutrophils, lymphocytes, D-dimer, CRP, ferritin, hsTnT and LDH were fitted into a logistic regression model and comparative odds ratio (OR) with 95% confidence intervals (CI) was performed to identify predictors of mortality and invasive mechanical ventilation.8,10 A backward selection approach was employed into our multivariable model to identify independent risk factors. Spearman’s rank analysis was used to gauge correlation between the variables with peak PCT concentrations. All statistical analyses were performed using IBM SPSS Version 26 using a P-value of less than 0.05 to determine statistical significance.
Ethics
This study was approved by our hospital’s Research, Quality improvement and Audit Department with reference FH205. All data were collected locally, anonymized and handled in accordance with local data protection guidelines.
Results
A total of 60 patients (mean age 57 ± 12 years) were included into the study, 49 (82%) male and 11 (18%) female. Patient demographics and characteristics are summarized and compared in Table 1. This was a predominantly Caucasian (55%) population; other ethnicities included for analysis were Asian (40%) and Afro-Caribbean (5%). Diabetes mellitus was the most prevalent comorbidity in our population followed by cardiovascular diseases, with a median Charlson comorbidity index score of 3 (1–4). A total of 41 (68%) patients required invasive mechanical ventilation, while 27 (45%) deaths were recorded in our patient cohort.
Summary of patient demographics and comparison of data to analyse the use of invasive mechanical ventilation and mortality.
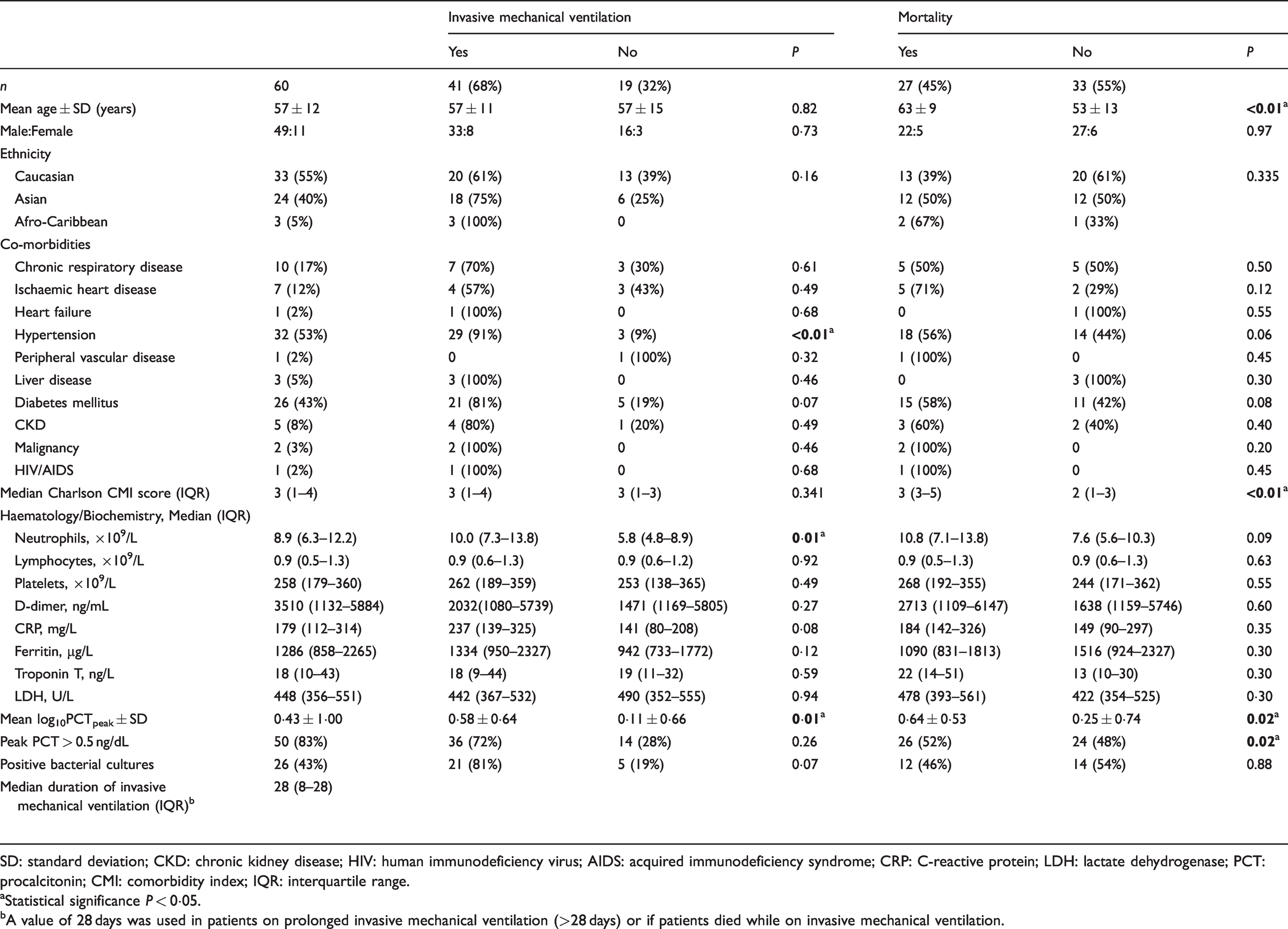
SD: standard deviation; CKD: chronic kidney disease; HIV: human immunodeficiency virus; AIDS: acquired immunodeficiency syndrome; CRP: C-reactive protein; LDH: lactate dehydrogenase; PCT: procalcitonin; CMI: comorbidity index; IQR: interquartile range.
aStatistical significance P < 0·05.
bA value of 28 days was used in patients on prolonged invasive mechanical ventilation (>28 days) or if patients died while on invasive mechanical ventilation.
Peak PCT concentrations were measured at an average of 5 ± 4 days after admission to the critical care setting. Higher peak PCT concentrations, neutrophil counts and hypertension were significantly associated with the use of invasive mechanical ventilation, while increasing age, higher peak PCT concentrations (particularly patients with peak PCT greater than 0.5 ng/dL) and Charlson comorbidity index scores were significant predictors of mortality. There was no significant association between gender, ethnicity, positive bacterial cultures, other specific comorbidities or other haematology/biochemistry measures with poor Covid-19 outcomes. There were 26 patients with positive bacterial culture results of which the most common source was bronchial lavage (40.5%), followed by blood (18.9%), central line (18.9%), urine (13.5%), sputum (2.7%), wound (2.7%) and pneumococcal antigen (2.7%). Peak PCT concentrations did not vary between patients who had evidence of positive bacterial cultures against those who did not (mean log10PCTpeak 0.6 ± 0.7 vs 0.3 ± 0.6, P = 0.10).
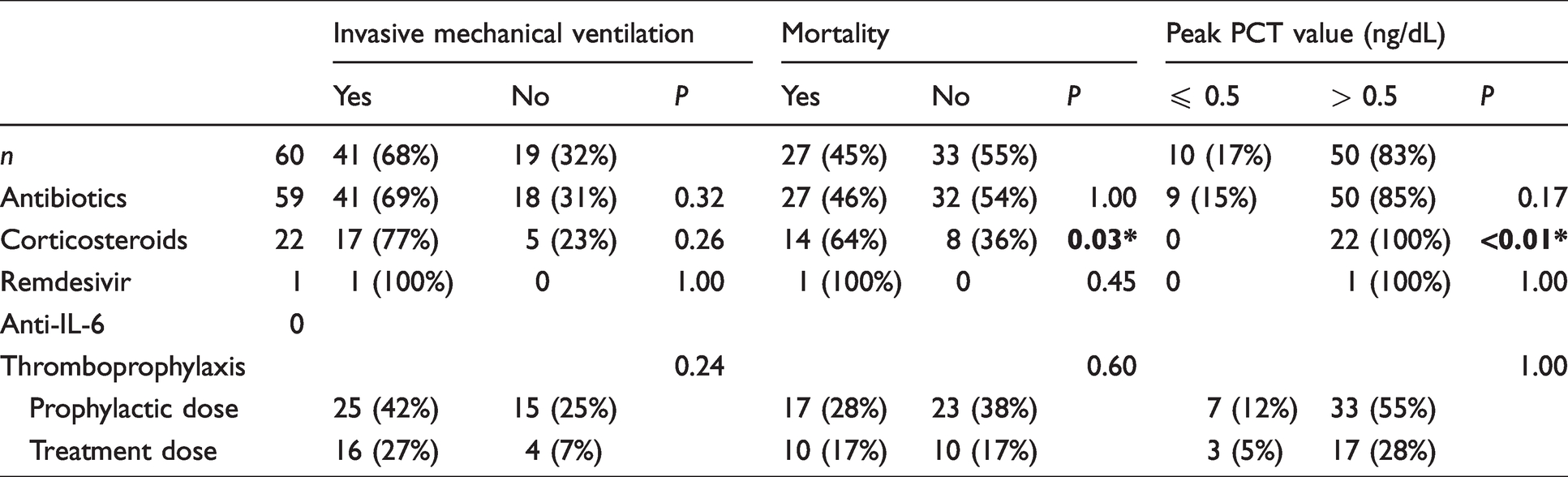
Table 2 summarizes different treatment strategies for patients. The addition of corticosteroid therapy appears to be associated with a higher mortality. A greater proportion of these patients also have peak PCT values of greater than 0.5 ng/dL. The most common indication for corticosteroid therapy was circulatory shock (59% of patients receiving corticosteroids). The overwhelming majority of patients received antibiotics during the critical care admission with 31 patients (52%) who received antibiotics for at least 24 h prior to admission. The majority of our patients received prophylactic doses of thromboprophylaxis and a smaller proportion received treatment doses. Use of antibiotics or thromboprophylaxis dose did not affect invasive mechanical ventilation or mortality outcomes.
Summary of treatment of patients according to invasive mechanical ventilation, mortality and peak procalcitonin values.
Our multivariable logistic regression model identified higher log10PCTpeak concentrations (OR 3.7, 95% CI 1.1–12.4, P = 0.03) and Charlson comorbidity index (OR 2.2, 95% CI 1.3–3.6, P < 0.01) to be independent predictors of mortality (Table 3). In patients who required invasive mechanical ventilation, higher log10PCTpeak concentrations (OR: 3.2, 95% CI 1.3–9.0, P = 0.02) and neutrophil counts (OR 1.2, 95% CI 1.1–1.5, P = 0.02) were significant risk factors on univariate analysis. There were no significant independent predictors of invasive mechanical ventilation identified within our multivariable model.
Univariable and multivariable logistic regression model to identify risk factors for mortality and invasive mechanical ventilation in Covid-19 patients.
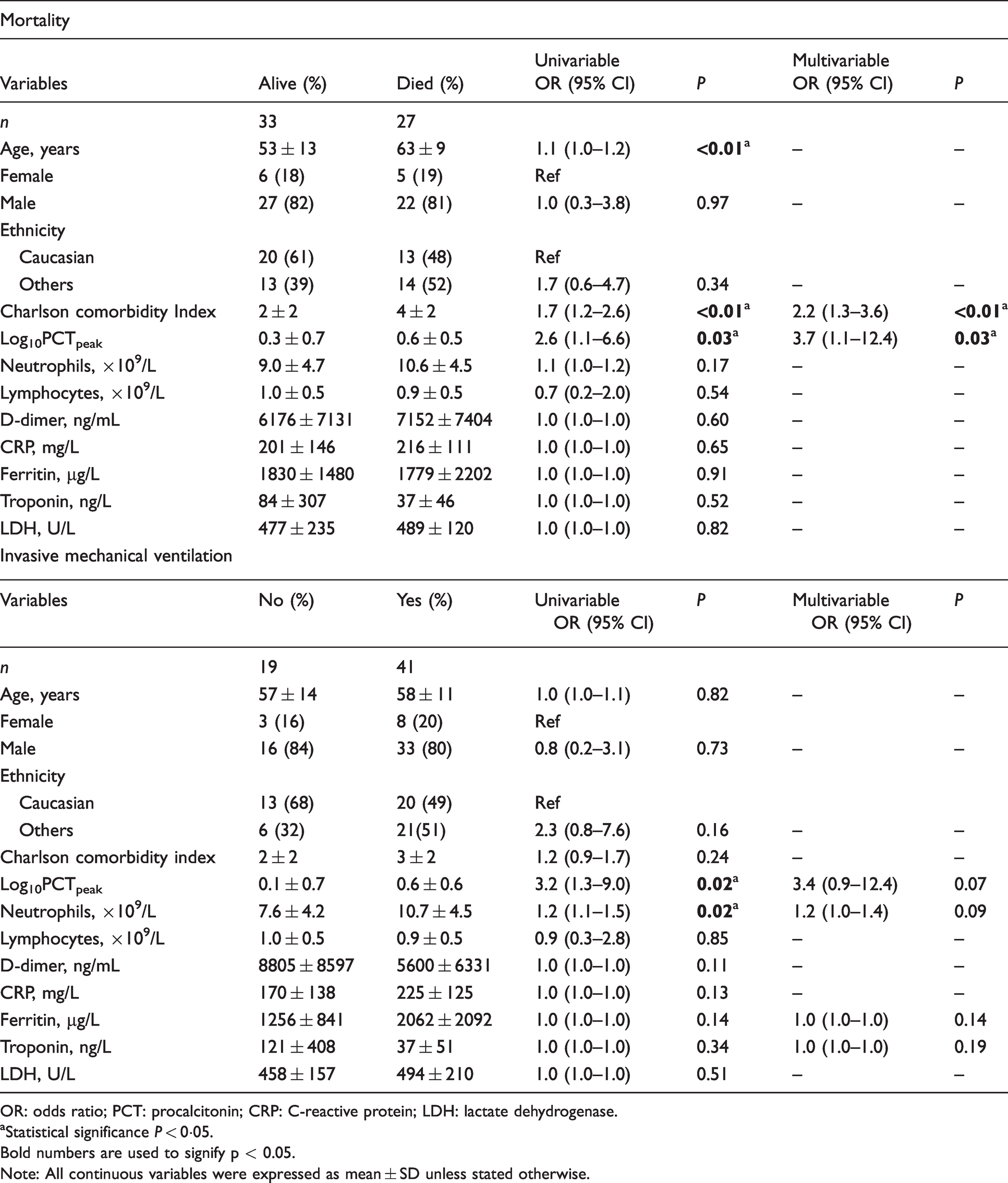
OR: odds ratio; PCT: procalcitonin; CRP: C-reactive protein; LDH: lactate dehydrogenase.
aStatistical significance P < 0·05.Bold numbers are used to signify p < 0.05.
Note: All continuous variables were expressed as mean ± SD unless stated otherwise.
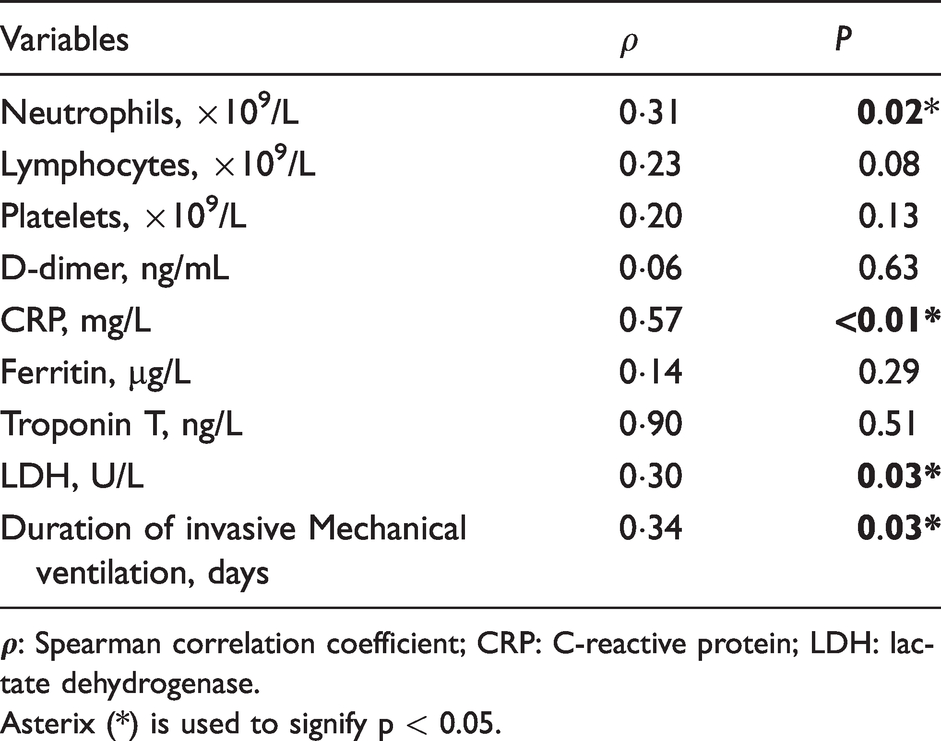
Spearman’s rank analysis identified a significant correlation between peak PCT and neutrophil counts (
Spearman’s rank correlation between peak PCT concentrations and haematology/biochemistry values and duration of invasive mechanical ventilation.
Discussion
Inconsistent presentation and variability in the course of Covid-19 between patients makes predicting those who will require invasive mechanical ventilation and who are consequently at heightened risk of mortality challenging. 11 Identification of biomarkers associated with these outcomes is therefore an adjunctive instrument to clinical decision-making. Consistent with other studies, 3 our findings demonstrated that increased serum PCT concentrations are associated with increased severity of Covid-19 as defined by respiratory failure necessitating invasive mechanical ventilation and in-hospital mortality. Notably, some studies have not found a statistically significant association with PCT and disease severity, perhaps explained by variations in sample sizes, definitions of severity and cut-off values for PCT. 4
Our study has demonstrated that increased PCT concentrations predict a longer duration of invasive mechanical ventilation. To our knowledge, our study is one of two to specifically assess this association. 12 These findings are particularly relevant, given the disparity in supply and demand of ventilatory resources in many areas during the first wave of the pandemic. Indeed, limited reserves may be associated with increased mortality from Covid-19 and the ability to predict the use of vital resources will allow for better planning. 13 Additionally, a prolonged duration of invasive mechanical ventilation is associated with complications such as ITU-acquired weakness/neuropathy, bacterial infection and mortality. 14 Identification of patients at risk of prolonged ventilatory support may allow better risk stratification and determination of patients who might require tracheostomy. Our study also supports that age and burden of co-morbidities are associated with mortality, findings which have been confirmed in multiple other studies. 10 Treatment with corticosteroids was also associated with mortality. However, this is likely a reflection of patients who were more critically unwell, given that the majority of patients in our cohort received corticosteroids for cardiovascular shock rather than for Covid-19 treatment itself.
Under normal circumstances, PCT is synthesized by thyroid parafollicular C cells, and serum concentrations remain low. In viral infections, it is thought that serum concentrations also remain low because of the action of interferon-γ which inhibits synthesis of PCT. 2 However, in bacterial infection, serum concentrations are greatly increased through synthesis by cells outside the thyroid gland. This is likely mediated by elevated concentrations of interleukin 6 and tumour necrosis factor-alpha (TNFα). 15 PCT concentrations are not found to be elevated in Covid-19 patients on admission or during early disease, but patients with worse outcomes demonstrate a progressive increase in PCT concentrations during the inpatient episode, 3 a phenomenon which this study aimed to capture through use of peak PCT value. It has been speculated that this increase could reflect bacterial co-infection, which likely occurs later in the course of illness, or the severity Covid-19.3,16 However, there is currently insufficient evidence to validate either hypothesis.
In our study, PCT concentrations were not increased in patients with positive bacterial cultures compared with those with negative bacterial cultures. Elevated PCT concentrations were, however, associated with increased neutrophil count, and patients requiring invasive mechanical ventilation had higher mean neutrophil count. Neutrophilia is traditionally associated with bacterial infection; however, in Covid-19, it is unclear as to whether neutrophilia (which has been linked to adverse outcomes) is related to added bacterial infection. 17 The variable use of corticosteroids during the pandemic according to current protocol may have contributed to neutrophilia, further accentuating the unreliability of neutrophilia as a marker of bacterial sepsis in Covid-19. Therefore, in the absence of an association with positive bacterial cultures, neutrophilia cannot be definitively associated with bacterial co-infection in our study.
We also demonstrated a positive association between PCT and both LDH and CRP, elevated levels of which are seen in both severe Covid-19 and bacterial sepsis.8,18,19 Increased PCT concentrations were not associated with other cited markers of cytokine storm characteristic of severe Covid-19, 8 potentially because these markers were not always measured on admission or at the peak of disease trajectory. Taken together, our findings do not support the hypothesis that elevated PCT concentrations in Covid-19 are due to bacterial co-infection.
To our knowledge, our study is one of few to report PCT concentrations according to the presence or absence of culture proven bacterial co-infection. 20 Previous studies have associated PCT with bacterial infection, while others have not and comparison between these studies is difficult due to varying definitions of bacterial co-infection. 4 Additional research is awaited to characterize the pathophysiological mechanisms through which PCT is synthesized, released and accumulated in severe Covid-19. Furthermore, at the time of writing, there is no consensus for the optimal cut-off value for PCT to predict worse outcomes in Covid-19. 4
Limitations
This study was performed in a single centre in South-East England with a limited sample size and population demographics and treatment guidelines that may not be comparable to other centres. As a retrospective study, exclusion of patients without measured PCT concentrations may have generated selection bias. Since there is no national guidance on the use of PCT in Covid-19, levels were measured at varying time points at the clinician’s discretion. As the use of PCT is restricted to critical care settings at our institution, comparison with patients treated outside this environment was not possible. Use of antibiotics may have resulted in false-negative bacterial culture results.
Conclusion
Our study complements the growing body of evidence linking increased serum PCT concentrations with worsened Covid-19 outcomes and opens avenues for future research surrounding the biochemistry and physiology of PCT. Specifically, our study has clearly demonstrated that patients with elevated PCT concentrations are more likely to develop respiratory failure requiring invasive mechanical ventilation, have a longer duration of ventilatory support and are at an increased risk of in-hospital death. Our findings do not support the hypothesis that this association necessarily represents bacterial co-infection and the role of PCT in antimicrobial stewardship within the context of Covid-19 remains unclear. We suggest that PCT functions well as an adjunctive tool to expectant observation and may facilitate timely intervention. Furthermore, PCT should be used as a surrogate marker for risk assessment and prognostication in patients with Covid-19.
Footnotes
Authors’ note
Constantinos G Missouris is also affiliated with University of Nicosia Medical School, Nicosia, Cyprus.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
As a retrospective analysis using clinically collected, non-identifiable data, this work does not fall under the remit of National Health Service Research Ethics Committees. This study was approved by our institution’s Research, Quality improvement and Audit Department with reference FH205. This statement is also present in the methods section of our article. It was not appropriate or possible to involve patients or the public in the design, or conduct, or reporting, or dissemination plans of our research.
Guarantor
CGM.
Contributorship
All eight authors contributed to the article. All were involved in the design of the study. RSH, JK, AKJM, DG, IALW and AKJM collected the data. JK was responsible for the statistical analysis. RSH, AKJM, JK, PS and CGM wrote the article and all authors were involved in the final approval of the article. AKJM and CGM are acting as guarantors of the submitted work.
