Abstract
Background
The type of blood collection tube affects specimen quality and laboratory results. Because plasma specimens have a shorter processing time compared with serum specimens, emergency biochemistry tests use plasma. However, serum specimens remain stable after centrifugation and show more accurate results than plasma. Therefore, a quick-clotting serum separator tube is expected to be useful for shorter turnaround times and accurate results. We evaluated a new quick-clotting serum separator tube VQ-Tube™ (AB Medical, Korea) for clinical chemistry and thyroid hormone assays.
Methods
One hundred volunteers from four university hospitals were recruited, and peripheral blood samples were collected in quick-clotting serum separator tube VQ-Tubes™ and the commonly used serum separator tube V-Tubes™. The obtained specimens were used for 16 clinical chemistry assays and three thyroid hormone assays.
Results
The differences (%) in the test results obtained from the samples in each tube satisfied the allowable difference ranges (19 assays). The differences in the test results between the tubes satisfied the desired specifications for accuracy except for the glucose results (2.75%). The paired t-test revealed significant differences between the results of six assays, but each set of results showed a good correlation. Samples were visually inspected for serum clarity and gel barrier integrity, and incomplete clotting reactions and haemolysed serum were not observed.
Conclusions
The new quick-clotting VQ-Tube™ demonstrated reliable test results compared with the commonly used serum separator tube V-Tube™. This quick-clotting tube will provide fast test results with adequately separated serum specimens, especially for patients who need fast tests.
Introduction
The serum separator tube (SST) is the most commonly used tube in clinical laboratories for biochemical analyses. The use of SSTs has become more popular due to the advantage of rapid serum separation from cellular constituents, and the gel barrier resulting from centrifugation provides analytic stability from haemolysis and repetitive serum aliquoting. 1 Serum is the preferred matrix for immunoassays after optimal clotting, but delayed clot formation may result in fibrin clot interference in automated analysers. 2 Sometimes, the separator gels can affect the test results due to incorrect mechanical operations. 3 Also, the SSTs currently used in laboratories require about 20–30 min of clotting time and 10 min of centrifugation time before specimen analysis, which causes increased turnaround time (TAT) and the delayed delivery of medical services.4,5 In contrast to SSTs, heparinized tubes used to obtain plasma have a shorter processing and centrifugation time, but serum specimens remain stable longer after centrifugation and analytical differences in test results between plasma and serum have been reported.6–8 SST and heparinized plasma tubes each have strengths and weaknesses, and various improvements have been made. A heparin-containing plasma tube with a gel separator 9 and SSTs with reduced clotting time 10 were introduced and evaluated. The recently developed rapid clotting serum tubes are considered to provide a suitable alternative to lithium heparin plasma tubes. However, several factors need to be considered to improve the performance of SSTs. Latent clotting was observed in the blood of patients taking high-dose anticoagulant therapy, 10 and reduced lactate dehydrogenase (LD) concentrations10–12 have been reported.
The new quick-clotting SST VQ-Tube™ (AB Medical, Gwangju, Korea) contains a thrombin-based clot activator and provides a 5-min clotting time for serum separation. We previously reported the excellent analytic performance of the SST V-Tube™ (AB Medical) compared with two widely used SSTs. 13 In the present study, we evaluated the clotting performance and analytic accuracy of the quick-clotting SST VQ-Tube™ compared with the V-Tube™ in clinical chemistry and thyroid hormone assays.
Methods
This study was conducted from May to August 2019 at four hospitals (Eunpyeong St. Mary’s Hospital and Seoul St. Mary’s Hospital of the Catholic University, Seoul National University Hospital, and Soonchunhyang University Bucheon Hospital). A total of 100 adults participated in the study, with 25 participants each from the four hospitals. This study was approved by the Institutional Review Board of each of the four hospitals, and informed consent was obtained from all participants.
Specimen collection
Venous blood was collected by a phlebotomist according to each four hospital’s guideline for phlebotomy in the quick-clotting SST VQ-Tube™ (VQ-Tube) and the SST V-Tube™ (V-Tube). Healthy adults and outpatients who voluntarily participated in the study provided venous blood specimens for the two types of SSTs in a randomized drawing order. A BD needle holder (Becton-Dickson, Franklin Lakes, NJ, USA) and multi-draw 21 G needles (Greiner Bio-One, Monroe, NC, USA) were used for the blood collection. The tubes were filled to the capacity (5 mL) and smoothly mixed by five inversions according to the manufacturer’s instruction immediately after the blood specimens were obtained.
Sample preparation and observation of serum clarity
To calculate the exact clotting time, the tubes were sent to the laboratory within five min after specimen collection. Including the transport time to the laboratory, the VQ-Tubes were left 5 min at room temperature for clotting as the manufacturer recommended. After clotting, the tubes were centrifuged at 1500 g for 10 min without delay. The V-Tubes were left 20 min to clot and were centrifuged at 1500 g for 10 min. Before placing on the total laboratory automation system, the VQ-tubes and V-tubes were optically inspected for serum clarity to detect incomplete clotting products, haemolysis, and lipaemic and icteric serum.
After all test analytes were measured, the VQ-Tubes and V-Tubes were placed upright and visually re-checked for serum clarity and gel barrier integrity at the same time.
Analytic tests and instruments
After the optical inspections, the VQ-Tubes and V-Tubes were immediately placed in the TLA system for clinical chemistry and thyroid hormone assays. The Beckman Coulter 5800 system (Beckman Coulter, Brea, CA, USA) and the DXI800 (Beckman Coulter) analyser were used in Eunpyeong St. Mary’s Hospital for clinical chemistry and thyroid hormone assays, respectively. The Hitachi 7600 (Hitachi, Tokyo, Japan) and the ADIVA Centaur analyser (Siemens, Erlangen, Germany) were used in Seoul St. Mary’s Hospital. The Architect c16000 analyser (Abbott, Abbot Park, IL, USA) and the Architect i2000SR analyser (Abbot) were used in the Seoul National University Hospital. The Hitachi 7600 (Hitachi) and the Cobas e801 (Roche Diagnostics, Mannheim, Germany) were used in Soonchunhyang University Bucheon Hospital. Serum from the VQ-Tubes and V-Tubes were analysed simultaneously for 16 clinical chemistry and three thyroid hormone assays: aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), gamma-glutamyltransferase (GGT), total protein, albumin, blood urea nitrogen (BUN), creatinine, total bilirubin, direct bilirubin, total cholesterol, glucose, LD, sodium, potassium, chloride, triiodothyronine (T3), free thyroxine (free T4) and thyroid-stimulating hormone (TSH). In the ALP measurement, p-nitrophenyl phosphate with ethyl amino ethanol (EAE) buffer solution was used in Soonchunhyang University Bucheon Hospital (reference range: 120–370 IU/L). p-Nitrophenyl phosphate with EAE buffer solution was used in the other three laboratories.
Statistical analysis
The paired t-test was used for the parametric data, and the Wilcoxon signed-rank test was used for the non-parametric data to compare the statistical differences between the VQ-Tube and the V-Tube for each of the 19 measured analytes. Pearson’s correlation coefficient (r) was assessed to estimate the correlation between the tubes. Differences were considered statistically significant at a P-value of < 0.05. Bland-Altman plots were used to compare the differences measured in the two tubes. The differences in the mean value of each measured analyte were calculated to estimate the difference between the two tubes. The allowable difference range according to the Clinical Laboratory Improvement Amendments (CLIA) of 1988, 14 biological variability based on Westgard 15 and the Clinical and Laboratory Standards Institute guideline GP34-A 16 were used to evaluate the differences of the means. Statistical analyses were performed using Analyse-it ver. 4.95 (Analyse-it Software Ltd, Leeds, UK).
Results
Optical inspection for serum clarity
All tubes were visually reviewed for serum clarity and gel barrier integrity before and after analyte measurements. All blood samples in the VQ-Tubes were completely clotted. Incomplete clotting reaction products and/or fibrin were not detected in either the VQ-Tubes or the V-Tubes. The barrier formation in the two tubes was completed equally. The separated serum samples were inspected for haemolysis, lipaemia and icterus. Three lipaemic samples were observed in both the VQ-Tubes and the V-Tubes simultaneously.
The statistical results of the measured analytes in the VQ-Tubes and V-Tubes obtained from 100 participants are summarized in Table 1. The paired t-test of six analytes showed statistically significant differences between the VQ-Tubes and the V-Tubes: GGT (P = 0.001), creatinine (P < 0.001), glucose (P < 0.001), LD (P < 0.001), potassium (P < 0.001) and TSH (P = 0.004). The differences (%) in the parallel mean values between the VQ-Tube and the V-Tube were calculated. The differences in the 16 clinical chemistry and three thyroid hormone assays satisfied the allowable difference range. The difference in glucose (2.75%) was clinically significant when the desired specification based on biological variation was applied. The difference in LD (3.61%) between the two tubes met the desired specification but showed a relatively large difference. The results of the 16 clinical chemistry tests and three thyroid hormone assays in serum samples from the VQ-Tubes were in excellent correlation with those of the V-Tubes. Pearson’s correlation analysis showed linearity between the two tubes, and the r-value of the19 analytes was near 1.0.
Comparison of clinical chemistry tests and thyroid hormone assays between V-Tube and VQ-Tube (n = 100, four hospitals).
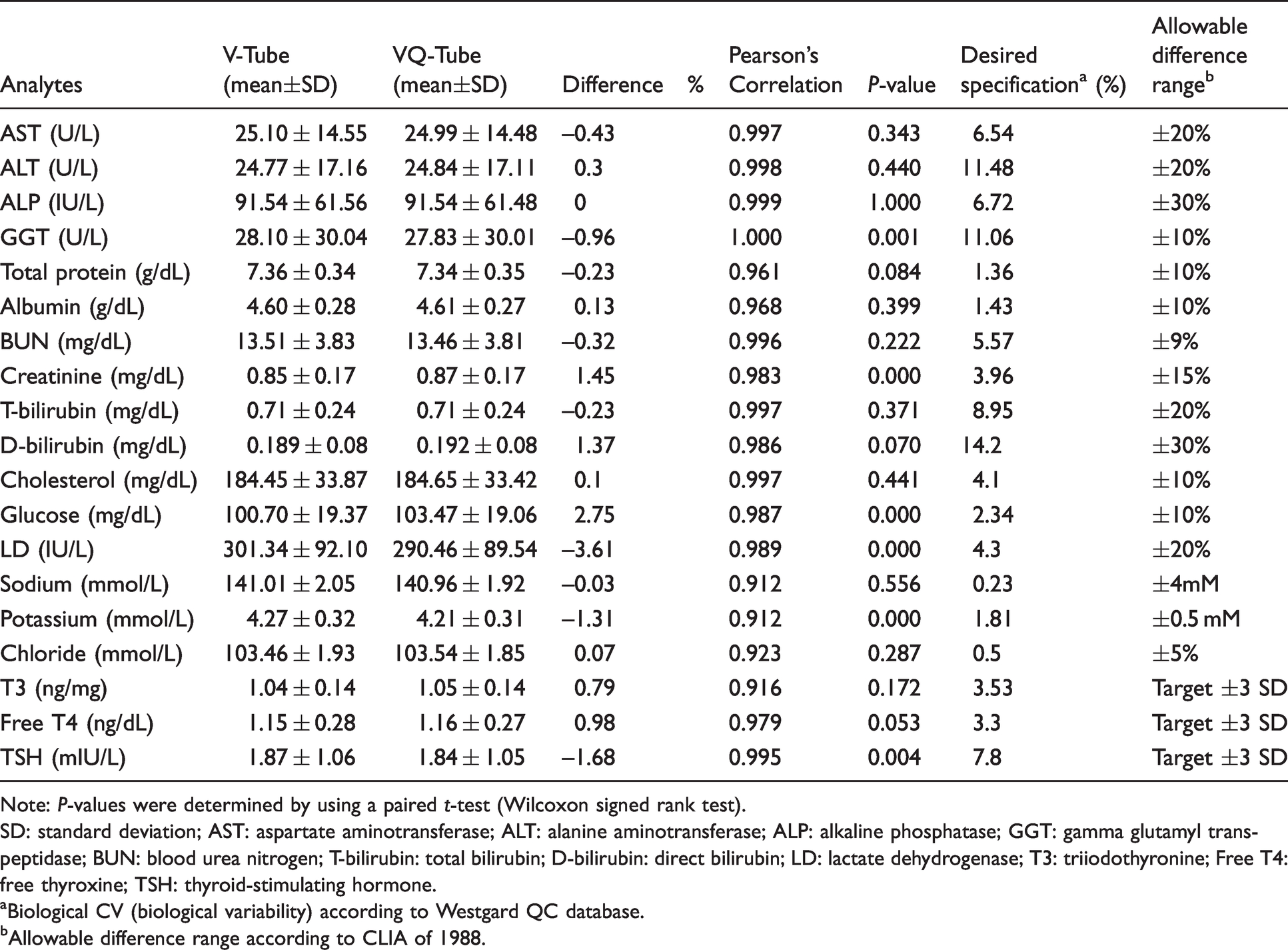
Note: P-values were determined by using a paired t-test (Wilcoxon signed rank test).
SD: standard deviation; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; GGT: gamma glutamyl transpeptidase; BUN: blood urea nitrogen; T-bilirubin: total bilirubin; D-bilirubin: direct bilirubin; LD: lactate dehydrogenase; T3: triiodothyronine; Free T4: free thyroxine; TSH: thyroid-stimulating hormone.
aBiological CV (biological variability) according to Westgard QC database.
bAllowable difference range according to CLIA of 1988.
Bland-Altman difference plots were generated for the 19 analytes (Supplemental Figures 1 to 3). The greatest mean bias was for glucose (Supplemental Figure 2(e)) and LD (Supplemental Figure 2(f)). High variations in ALP between the two tubes were observed for ALP concentrations >180 IU/L (Supplemental Figure 1(c)), and these results were notable in the ALP measurements using EAE buffer. The Bland-Altman plots showed an increased difference in creatinine concentrations > 0.9 mg/dL (Supplemental Figure 2(a)) and cholesterol concentrations >200 mg/dL (Supplemental Figure 2(d)). In the thyroid hormone assays, there were large differences between the two tubes in the overall measurement range (Supplemental Figure 3), and four values outside two standard deviations were found in the TSH comparison.
Table 2 summarizes the statistical results of the several analytes comparison between the VQ-Tubes and V-Tubes according to the four hospitals. Overall, the differences in the results obtained from each of the four hospitals all satisfied the allowable range difference. Five analytes (total protein, cholesterol, glucose, LD and potassium) showed statistically significant differences and two analytes (sodium and potassium) did not meet the desired specifications in Eunpyeong St. Mary’s Hospital (Supplemental Table 1). In Seoul St. Mary’s Hospital, four analytes (cholesterol, glucose, LD and TSH) showed statistically significant differences and glucose did not meet the desired specification (Supplemental Table 2). Four analytes (albumin, glucose, LD and chloride) showed statistically significant differences and glucose did not meet the desired specification in Soonchunhyang Bucheon Hospital (Supplemental Table 3). Eight analytes (total protein, creatinine, cholesterol, glucose, LD, sodium, T3 and free T4) showed statistically significant differences and glucose did not meet the desired specification in Seoul National University Hospital (Supplemental Table 4).
Comparison of analytes between V-Tube and VQ-Tube according to hospitals.
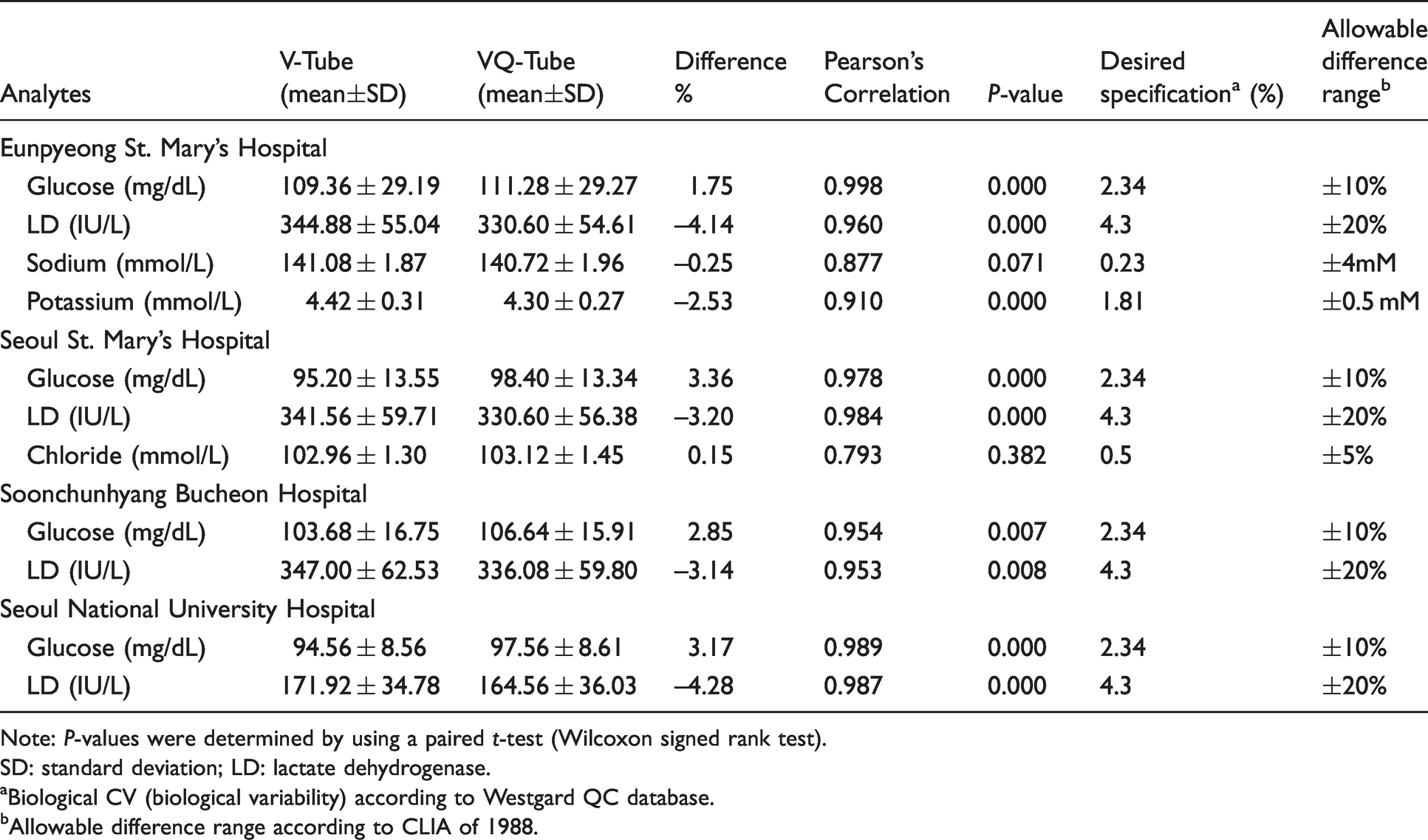
Note: P-values were determined by using a paired t-test (Wilcoxon signed rank test).
SD: standard deviation; LD: lactate dehydrogenase.
aBiological CV (biological variability) according to Westgard QC database.bAllowable difference range according to CLIA of 1988.
Discussion
This study performed a comparison of the quick-clotting SST VQ-Tube and the generally used SST V-Tube for routine chemistry and thyroid hormone assays. The 5-min clotting time of the VQ-Tubes reduced the TAT by 15 min compared with the V-Tubes. The differences in the evaluated analytes between the two tubes all satisfied the allowable range differences including those on different analytical platforms. The use of the quick-clotting VQ-Tube would be useful for clinical chemistry tests and thyroid hormone assays to reduce the TAT.
Increased serum glucose concentrations in the VQ-Tubes exceeding the desired specification were observed in all four participating hospitals. The serum glucose concentration decreased mainly due to red blood cell metabolism in the blood collection tubes, 17 as there were significant differences in glucose concentrations between the two tubes over time. Fast clot activation results in the more rapid separation of blood cells from serum. 2 Although serum glucose concentrations tend to be high in quick-clotting tubes than SSTs,10,12,18 it is measured at a lower concentration than in lithium heparin tube and gold standard. 19 In addition, blood collection tubes containing clot activator and lithium heparin are not suitable for glucose measurement;9,20 the use of VQ-tube alone should be avoided in order to accurately measure glucose concentrations. Previously reported studies suggested that shortened clotting time contributed bias to serum glucose as well as LD concentration between the tubes.2,21 Differences in concentrations of LD are frequently reported10–12,18 in rapid clotting serum tubes compared with general SSTs. Zhang et al. 22 reported that LD concentrations increased by serum–clot contact over time and suggested that prolonged contact with mechanical agitation between the cells and serum contributed to the substantial increase in LD concentrations. Bausset et al. 21 reported the tendency of LD concentrations to increase according to serum–clot contact time, but the mean bias percentage of LD concentration was below the measurement uncertainty until 4 h. In line with previous studies, LD concentration in quick-clotting VQ-tube is lower than SSTs. To assess the difference in LD concentration by clotting time between two tubes, serum quality evaluation is required. To evaluate preanalytic sample quality, clinical chemistry analyser provides automatic measurement of haemoglobin, lipaemia and icterus in the serum or plasma (HIL indices). 23 Preanalytic assessment of samples using HIL indices has now become an important process for quality control in the laboratory. 24 The HIL indices of the 25 samples, measured with Coulter 5800 set for clinical use were all zero. On the other hand, the assessment of 25 samples with Hitachi 7600 (Soonchunhyang University Bucheon Hospital) showed 5 lipaemic and 12 icterus results, but the results were concordant between the VQ-Tube and the V-tube. HIL indices measurement using Hitachi 7600 were not performed in Seoul St. Mary’s Hospital due to reduced throughput in clinical settings, and HIL indices results using Architect i2000SR analyser were not collected. As a result, evaluating the potential impact on the samples of quick-clotting SST using HIL indices was not sufficiently performed. For this reason, the preanalytic serum quality was evaluated using visual inspection. Although only optically inspected, incomplete clotting was not observed in the 100 samples. There were only three lipaemic serum samples, which were in the V-Tubes. In particular, no samples were observed to show haemolysis, which demonstrate the stability of the clotting process in the VQ-Tubes. To evaluate latent clotting, all samples were visually re-checked after testing and compared with the V-Tubes.
Latent clotting problems in rapid clotting tubes have been reported, especially in patients who received high-dose heparin (>7000 unit) therapy. 10 The limitation of this study was that we did not include a group of patients treated with anticoagulants and compared them to the normal group. Thus, incomplete clot formation may occur in certain samples of patients with delayed coagulation time. In addition, the number of samples used in the study was small, and no assessment was made of various substances that could react abnormally with separator gel matrix. Also, 19 analytes were used in the present study, which did not include various clinical chemistry tests and immunoassays. To complement this, research involving a larger number of patients group and analytes will need to be investigated further.
Unlike other participating hospitals, in Eunpyeong St. Mary’s Hospital using the Beckman Coulter 5800 system for chemistry analysis, the differences in sodium and potassium did not satisfy the desired specifications. The measurement of electrolytes in serum samples from the rapid serum tubes showed a variety of results depending upon the study. Ng and Yeo 25 reported the stability of electrolyte measurements using the Beckman Coulter DxC 800 analyser. However, other studies10,12,26 reported decreased serum potassium concentrations in serum samples from rapid serum tubes.
In the present study, we compared the performance of quick-clotting VQ-Tubes with the current SSTs. To calculate the exact clotting time, we established study conditions so that the specimen collection site and testing laboratory were as close as possible and included different analytical platforms. The results of this study indicate that the VQ-Tubes demonstrated reliable performance similar to SSTs for clinical chemistry tests and thyroid hormone assays. This makes rapid reporting of test results possible for patients with reduced hospital costs.
Supplemental Material
sj-pdf-1-acb-10.1177_00045632211018245 - Supplemental material for Evaluation of the quick-clotting serum separator tube, VQ-Tube™, for clinical chemistry and thyroid hormone assays
Supplemental material, sj-pdf-1-acb-10.1177_00045632211018245 for Evaluation of the quick-clotting serum separator tube, VQ-Tube™, for clinical chemistry and thyroid hormone assays by Sung Jin Jo, Hyojin Chae, Yong-Wha Lee, Jong Do Seo, Sang Hoon Song and Jehoon Lee in Annals of Clinical Biochemistry
Supplemental Material
sj-pdf-2-acb-10.1177_00045632211018245 - Supplemental material for Evaluation of the quick-clotting serum separator tube, VQ-Tube™, for clinical chemistry and thyroid hormone assays
Supplemental material, sj-pdf-2-acb-10.1177_00045632211018245 for Evaluation of the quick-clotting serum separator tube, VQ-Tube™, for clinical chemistry and thyroid hormone assays by Sung Jin Jo, Hyojin Chae, Yong-Wha Lee, Jong Do Seo, Sang Hoon Song and Jehoon Lee in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We greatly appreciate the support from all participated laboratory teams.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the domestic product user evaluation project fund of the Korea Health Industry Development Institute (KHIDI).
Ethical approval
This study was approved by Ethics Committee of four hospitals (Eunpyeong St. Mary’s Hospital, Seoul St. Mary’s Hospital, Seoul National University Hospital, and Soonchunhyang University Bucheon Hospital) and participants gave written informed consents.
Guarantor
JL.
Contributorship
SJ drafted the initial manuscript and organized data analysis. JL complete the study protocol and revised the article. HC, YL, SHS and JDS helped in designing the protocol, analysing data and gained ethical approval. All authors agreed on the final version of the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
