Abstract
Background
High doses of Eltrombopag have been previously reported to cause bilirubin interference. Following receipt of a sample from a patient receiving high-dose Eltrombopag therapy, the laboratory decided to investigate the effect of this drug on routine chemistry testing.
Methods
Interference studies were performed by spiking Eltrombopag into aliquots of a serum pool to give concentrations ranging from 0 to 500 µg/mL. The following analytes, namely albumin, alkaline phosphatase, alanine transaminase, aspartate transaminase, Urea, total calcium, cholesterol, triglycerides, glucose, high-density lipoprotein cholesterol, iron, magnesium, inorganic phosphate, creatinine, bicarbonate, transferrin, ferritin, electrolytes, total and direct bilirubin and serum indices (hemolysis, icterus and lipaemia) were then measured on the Roche Cobas 6000 chemistry analyzer (Roche, Indianapolis, USA).
Results
Eltrombopag interference (>10% change of the baseline value) was observed for total cholesterol, triglycerides, inorganic phosphate and high-density lipoprotein cholesterol. Clinical significant interference was observed for total cholesterol, inorganic phosphate and high-density lipoprotein cholesterol
Conclusions
Presence of high Eltrombopag concentrations in blood samples has been demonstrated to cause interference in the measurement of certain spectrophotometric-based assays on the Roche Cobas 6000 analyzer.
Keywords
Background
The clinical chemistry laboratory received a routine sample for biochemical testing. Following centrifugation of the sample, on gross inspection it was observed that the plasma was a deep brownish red colour.
On further investigation, it was determined that the sample was from a 55-year-old female with aplastic anemic and accompanying thrombocytopenia. Following discussion with the referring clinician and review of her medical record, it was noted that the patient was on a clinical protocol receiving high-dose Eltrombopag therapy. Eltrombopag or Promacta [3′-{(2 Z)-2-[1-(3,4-dimethylphenyl)-3-methyl-5-oxo-1,5-dihydro-4H-pyrazol-4-ylidene]hydrazino}-2′-hydroxy-3-biphenylcarboxylic acid-2-aminoethanol (1:2)] is a small molecule thrombopoietin (TPO) receptor agonist that interacts with the transmembrane domain of the TPO receptor to increase platelet production.1,2 The current US Food and Drug Administration (FDA) maximal dose for Eltrombopag is 75 mg daily, while patients on the high-dose therapy protocols receive 200–300 mg daily.3,4 Although therapeutic monitoring of Eltrombopag is not routinely performed, the FDA describes a therapeutic Eltrombopag serum concentration of 10 µg/mL at the dosage of 75 mg daily. It has been suggested that the drug’s concentrations can be possibly exceeding 100 µg/mL if linear pharmacokinetic behavior is assumed. 5 Negative interference for the total bilirubin assay (diazo method, Beckman Coulter DxC 800 analyzer) has been previously described with an Eltrombopag spike in of 100 µg/mL. 5
The objective of our study was to investigate the possible interference effect of Eltrombopag on commonly ordered biochemical tests on the Roche Cobas 6000 analyzer.
Methods
Spike-in experiments with different concentrations of Eltrombopag were performed as described by CLSI (formerly NCCLS). 6 A drug-free plasma pool was prepared using plasma samples with chemistry results within normal reference intervals. A concentrated solution of Eltrombopag in the plasma pool was made. The Eltrombopag solution was then spiked into the plasma pool to give final concentrations of 10–500 µg/mL, with the solution being less than or equal to 10% of the final sample volume. An aliquot of the plasma pool without addition of the Eltrombopag solution was used as the blank sample. The following assays were then measured on the Roche Cobas 6000 chemistry analyzer (Roche, Indianapolis, USA): albumin, alkaline phosphatase (ALP), alanine transaminase (ALT), aspartate transaminase (AST), Urea, total calcium, cholesterol, triglycerides, glucose, high-density lipoprotein cholesterol (HDL), iron, magnesium, phosphate, creatinine, bicarbonate, transferrin, ferritin, electrolytes, total bilirubin, direct bilirubin and serum indices (haemolysis, icterus and lipemia, HIL). Results for spiked-in samples were then compared to the baseline values obtained from the unspiked specimen. The interference was calculated as percent change: Interference% = 100 × (measured value−baseline value)/baseline value. Significant interference was defined when the change of the analyte value exceeded 10% of the baseline value. Additionally, clinically significant interference was also determined by calculating the reference change value (RCV) for each test and comparing this to the percentage difference between the baseline and spiked samples at different concentrations of Eltrombopag. The clinically significant interference is defined as a result that in the presence of the interferent differs from the result without the interferent more than 1.96 × (CV2analytical + CV2within subject biological variation)1/2 at 95% significance, i.e. where the percentage was greater than the RCV for the assay the interference was determined to be clinically significant. 7
Results
Tests affected at different concentrations of Eltrombopag.
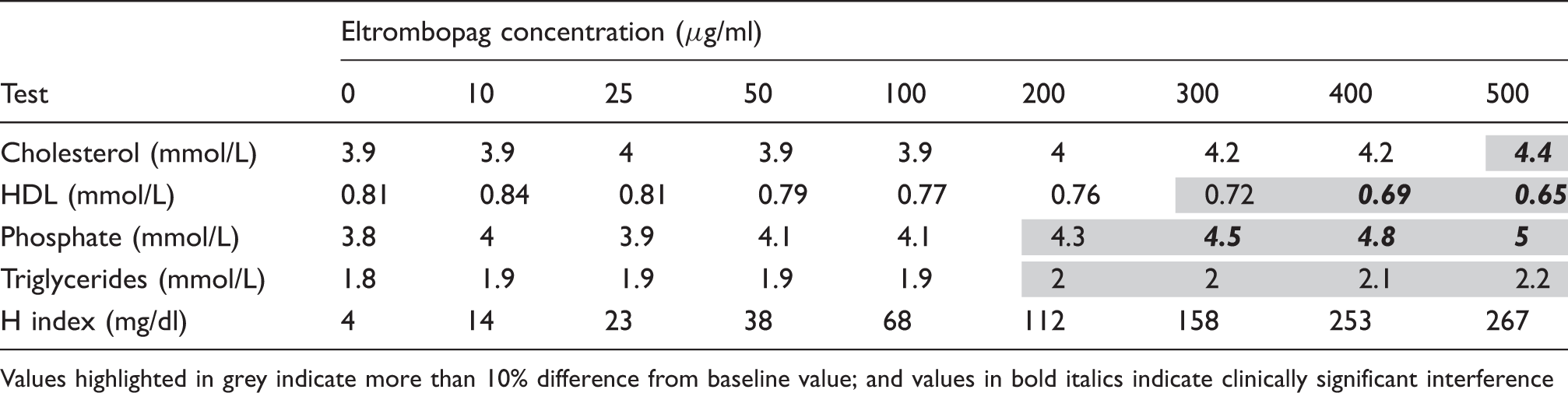
Values highlighted in grey indicate more than 10% difference from baseline value; and values in bold italics indicate clinically significant interference
Calculation of reference change values (RCVs) for clinical significant interference determination.
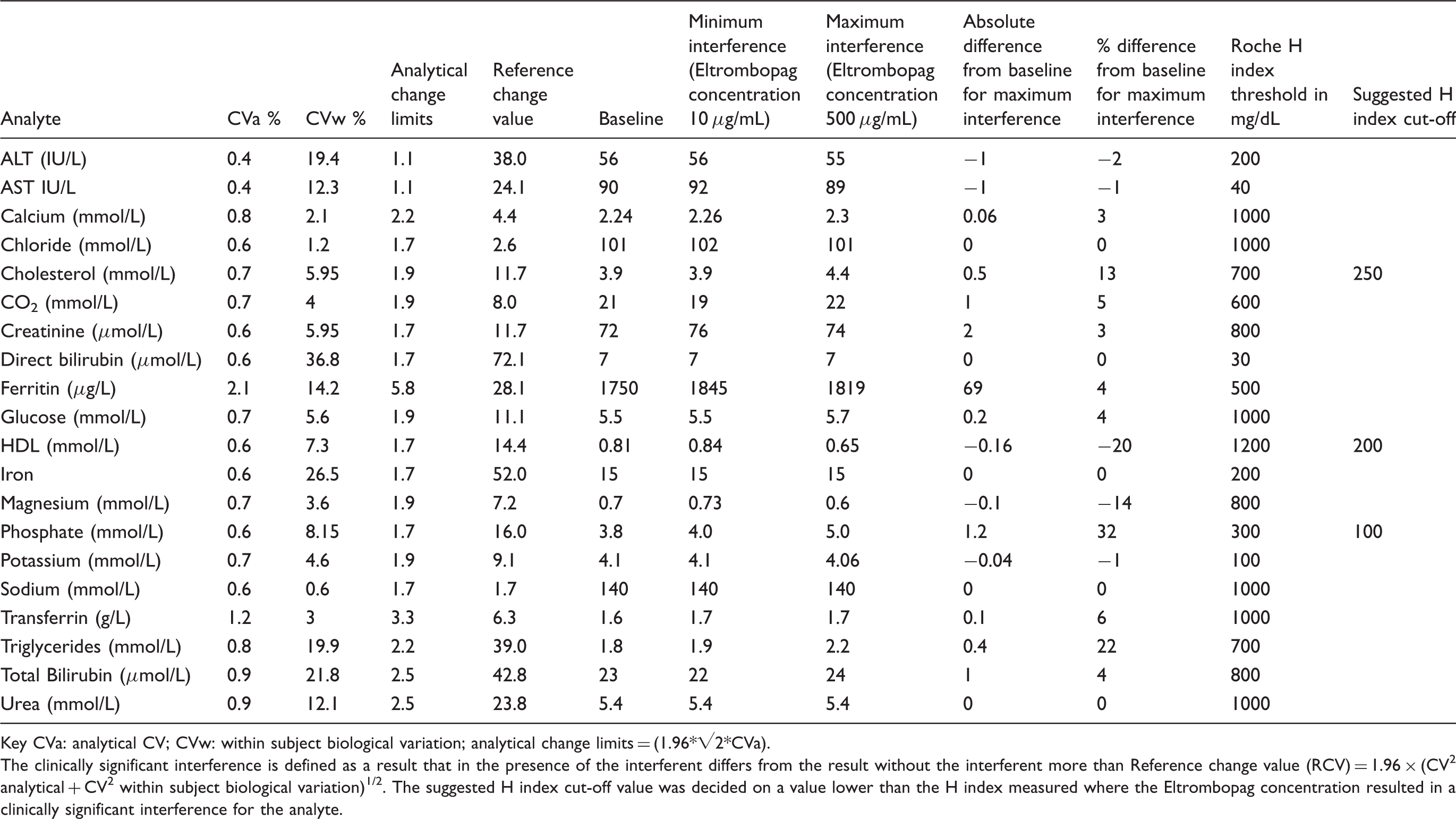
Key CVa: analytical CV; CVw: within subject biological variation; analytical change limits = (1.96*√2*CVa).
The clinically significant interference is defined as a result that in the presence of the interferent differs from the result without the interferent more than Reference change value (RCV) = 1.96 × (CV2 analytical + CV2 within subject biological variation)1/2. The suggested H index cut-off value was decided on a value lower than the H index measured where the Eltrombopag concentration resulted in a clinically significant interference for the analyte.
Additionally, both lipemic and hemolysis indices showed positive changes in values with the increase of Eltrombopag concentrations. Icteric indices did not show any change in values up to an Eltrombopag concentration of 500 µg/mL, when compared to the baseline value.
Discussion
In this brief study, we have demonstrated significant interference with high-dose Eltrombopag concentrations for phosphate, total cholesterol and triglycerides. We did not observe the previously reported interference of the total bilirubin assay measured on the Beckman Coulter DxC 800 analyzer. These data suggested that the Eltrombopag inference appears to be analyzer specific. The analytical interference of Eltrombopag should be recognized and explored by the laboratories on the specific analyzers. Specifically, it is important that the laboratory using the Roche Cobas 6000 analyzer is aware of such issues and inform requesting clinicians with patients on high-dose Eltrombopag therapy that results for these affected analytes may be unreliable. In these cases, clinicians should be advised that these affected analytes should be tested prior to initiation of therapy or after discontinuation. Furthermore, for all three affected analytes (phosphate, total cholesterol and triglycerides), the HIL indices were still within the manufacture suggested acceptable limits at Eltrombopag concentrations that resulted in significant interference. Keen observation by technologists when loading samples is also important in alerting to such an interference as the HIL indices provided by the analyzer may not indicate the presence of the interference or for certain assays a lower threshold may be considered for use on the automated analyzer when Eltrombopag interference is suspected (see our recommendations in Table 2).
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Intramural Research Program of the National Institutes of Health.
Ethical approval
The Institutional Review Board at the National Institutes of Health approved this study (STUDY NUMBER: 93-CC-0094).
Guarantor
ZZ.
Contributorship
Both authors contributed to the conception, performance and preparation of the manuscript and approved the final version.
