Abstract

Dear Editor,
It is not uncommon for clinical laboratories to change assay methods or analytical platforms. Deming or Passing–Bablok regression and Bland–Altman difference plots are used to determine whether the results from two different assays are consistent. 1 This assessment, however, is only of numerical value.
Similar results generated by two methods could lead to different clinical interpretation as a result of different reference ranges. This, however, is not considered in traditional method comparison studies and unlikely to be identified by internal quality control or external quality assurance data. It is generally inferred that between method differences, if any, are accounted for by method-specific reference intervals. 2
The derivation of in-house reference intervals is challenging for most laboratories 2 and repeating the process for analytes with a significant assay drift (e.g. with reagent lot change), or for the whole test repertoire with a change of analytical system is daunting. Most laboratories, including laboratories in our pathology network, therefore continue to use manufacturer-provided method-specific reference intervals. These, though valid when derived, may not remain valid with assay changes, such as drift, over time.
We recently reported a large clinical impact, a 56% clinical management discordance, on the diagnosis and management of subclinical hypothyroidism due to between method bias and manufacturer provided assay-specific reference intervals of the Abbott and Roche thyroid-stimulating hormone and free thyroxine assays. 3 The discordance resulted not only because Roche TSH results were approximately 35% higher than Abbott TSH results but also, despite that, Roche TSH upper reference limit (URL) is lower than Abbott URL (Figure 1). This striking clinical management discordance would not have been picked up by traditional method comparison experiments.
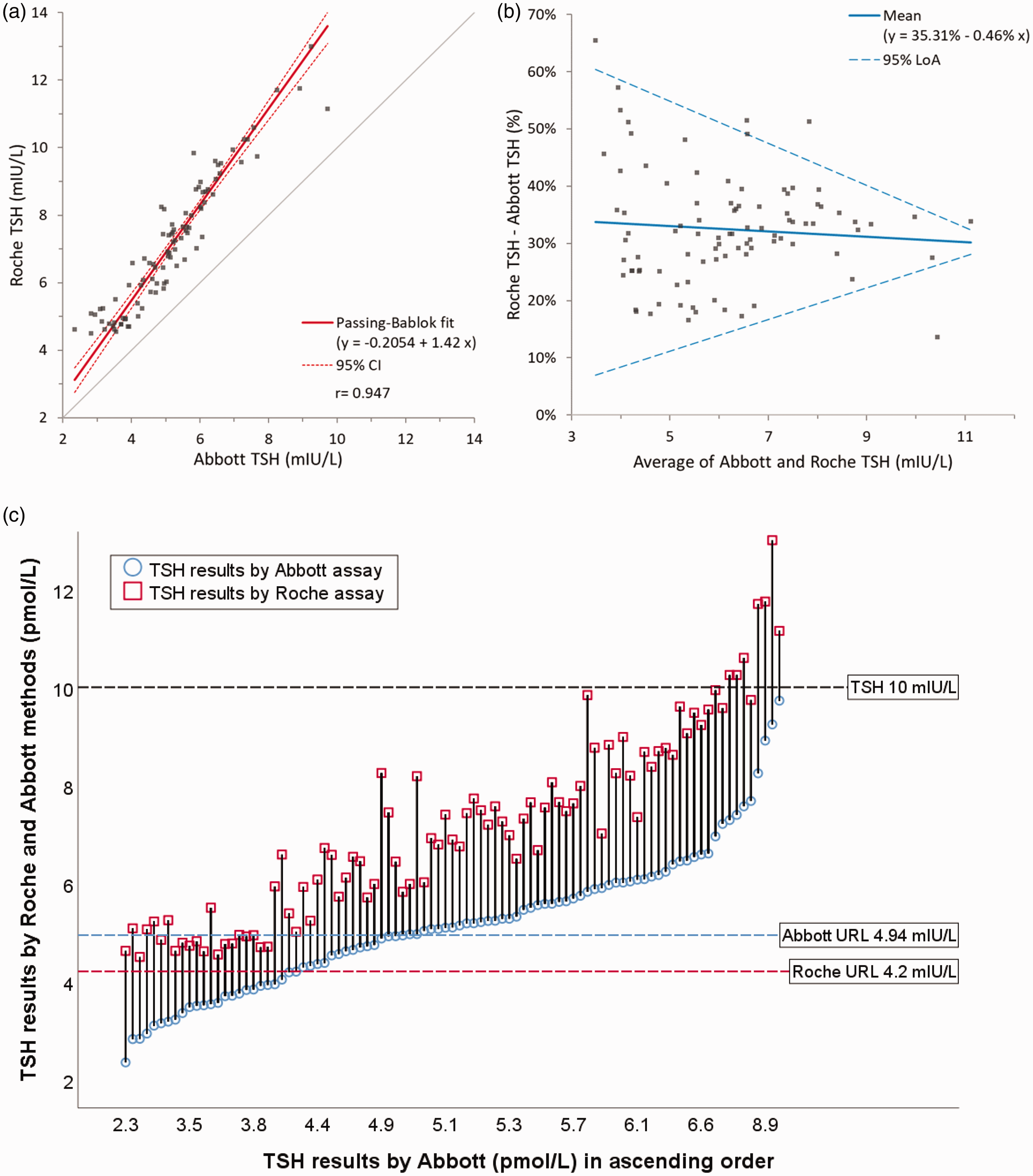
TSH results analysed by Roche and Abbott assays on samples with subclinical hypothyroidism on at least one of the two analytical platforms (n = 93). Passing–Bablok regression (a) and Bland–Altman percentage plot (b) were prepared using analyse-it. Discordance plot (c) was adapted with permission from Kalaria et al. 3 and was plotted in SPSS Statistics for Windows version 26. URL is upper reference limit.
The use of clinical management discordance figures (Figure 1(c)) or discordance tables 3 reviewing numerical results in the context of respective reference intervals or clinical decision thresholds could identify which patient management decisions may differ when transitioning from one method to another. Mitigation, depending on circumstances, includes harmonization of assays and reference intervals, re-derivation or adjustment of reference intervals by the manufacturer to reflect current assay performance, derivation of in-house reference intervals by the laboratory and communicating changes with service users. Reference intervals may be adjusted but universal clinical decision levels, such as TSH >10 mIU/L in subclinical hypothyroidism to initiate levothyroxine, cannot be changed. 1
Clinical discordance assessment requires additional resources while performing method validation. It, however, may only be required for selected assays. Examination of external quality assurance data of the methods while considering method-specific reference intervals could identify which assays may require this additional effort. Clinical discordance checks may be of value when changing assays not only for TSH3,4 and free T4, 3 but also parathyroid hormone, cortisol, growth hormone, 5 insulin like growth factor-1, 5 prolactin, testosterone, fructosamine, tumour markers and urine albumin.
In summary, analytical method comparisons that do not consider reference intervals may lead to differences in diagnosis and patient management. For selected analytes, this could be mitigated by using clinical discordance checks.
Footnotes
Acknowledgement
None.
Data availability
Data available from the corresponding author on request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Institution approved service evaluation project ID 5441 with the Royal Wolverhampton NHS Trust.
Guarantor
RG.
Contributorship
TK wrote the first draft of the manuscript. All authors critically reviewed, revised and approved the final version of the letter.
