Abstract
Background
The faecal immunochemical test (FIT) detects the presence of haemoglobin (Hb) in faeces. It is used as a screening tool for colorectal cancer (CRC) and increasingly to triage patients presenting with symptoms of CRC. A number of quantitative point-of-care (POC) FIT systems marketed for professional use and intended for use in a clinical setting are available. Here we reviewed the POC FIT systems available; three (Eurolyser Cube, OC-Sensor iO and QuikRead go) were evaluated to assess their performance against manufacturers’ claims and suitability for use in a clinical setting.
Methods
The analytical evaluation of the POC FIT systems was undertaken using Hb lysates, patient samples and an external quality assessment sample. The evaluation focused on linearity, recovery, imprecision, prozone effect, Hb variant detection and suitability for use in a clinical setting.
Results
All three POC FIT systems performed to their manufacturer’s claims and demonstrated good analytical performance with acceptable linearity, recovery, within- and between-run imprecision. The QuikRead go and OC-Sensor iO were able to accurately detect samples with results above their measuring range. However, because of a prozone effect the Eurolyser Cube gave falsely low results when using high concentrations of Hb. The QuikRead go performed best in the usability assessment due to portability and timeliness of result.
Conclusion
Each system performed according to their manufacturers’ claims. The QuikRead go and OC-Sensor iO are suitable for use. The Eurolyser Cube is not recommended because of the risk of falsely low results.
Keywords
Introduction
The presence of haemoglobin (Hb) in faeces is a risk factor for colorectal cancer. Testing for the presence of faecal haemoglobin (f-Hb) has been used in screening programmes across the world to identify people at highest risk to stratify them for further investigation. 1 Historically, f-Hb was detected using the qualitative and relatively non-specific guaiac method. More recently, the faecal immunochemical test (FIT) has been used to detect the presence of haemoglobin by utilizing polyclonal antibodies that bind to the globin moiety of the human haemoglobin molecule. Four laboratory-based quantitative assays have recently been evaluated and demonstrated to provide high-quality analytical results. 2
f-Hb has been recommended for use in patients presenting with lower gastrointestinal symptoms that may be suggestive of colorectal cancer (CRC). 3 This has led to the increased use of FIT in primary care as a diagnostic tool for symptomatic patients. FIT has also proved very useful during the COVID-19 pandemic to help triage patients for colonoscopy and is increasingly being used in primary and secondary care pathways as per NHS England and NHS Scotland guidance.4,5
For the symptomatic patients, laboratory-based immunoassay methods are typically used. NICE specifically recommends the use of three FIT systems as per the DG30 guidance for colorectal referrals. 3 These three systems are recommended because at the time of writing of the guidance, only three analysers had clinical evidence to support their use in symptomatic populations. We have demonstrated that a fourth quantitative FIT analyser performs equally as well analytically, but to our knowledge lacks clinical studies. 2
A FIT system consists of an analyser, reagents, calibration materials, QC material and sampling devices. The sampling device has a probe with grooves or dimples attached to a lid. This probe is either scraped along the surface of a faecal sample or dipped into multiple sites within the same faeces. The probe is then re-inserted into the device, through a collar to remove excess faeces into a preservative buffer solution. This is then traditionally returned to a laboratory for analysis.
There are an increasing number of FIT methods available for use at the point of care (POC). Both quantitative and qualitative methods are available and these include methods marketed for health-care professional use, for example in a clinic environment, as well as those for use by the general public. In 2017, Daly et al. provided a summary of the vast number of FITs available in the USA. The study highlighted the lack of both clinical and analytical data for the majority of these tests. 6 The majority of the FIT tested were manual qualitative lateral flow cassettes. Only two were quantitative automated systems, both manufactured by Eiken, and these were shown to have the best sensitivities and specificities for detection of advanced adenomatous polyps and CRC. To our knowledge, there have been no published studies to date on the analytical performance of any quantitative POC FIT devices, and there are limited clinical outcome studies assessing comparability of diagnostic outcomes between different POC FIT tests.7–9
With FIT being increasingly used in the primary and secondary care setting, there is growing interest in alternative ways to carry out the testing. As a research group, we are being frequently asked for our opinions on POC FIT systems. With no analytical evaluations available and very few clinical studies, it is not possible to provide an objective opinion on these. To address this gap and to provide some evidence base on which people can make informed decisions to implement POC FIT in clinical settings, we have carried out an analytical evaluation of three quantitative FIT systems and compared with the manufacturers’ claims (online Appendix 1). These are marketed for professional use and have the potential to be placed in a clinic environment.
Aim
The aim of this study was to carry out an analytical evaluation of quantitative POC FIT systems marketed for use in a clinic environment. This included an assessment of the ease of use of each system and suitability for a clinical environment.
Materials and method
FIT system selection
An internet search was carried out to identify as many quantitative POC FIT systems as possible. In addition, the companies who have quantitative FIT systems for laboratory use were contacted to enquire whether they had any analysers they would consider marketable for POC use. Five were identified that were marketed for quantitative health-care professional use and further to email contact with the manufacturers and/or suppliers of each system, all were considered for this study.
Two of the systems were excluded from the study early on; QuantOn Hem (Immundiagnostik AG, Bensheim, Germany) measures both haemoglobin and haptoglobin, and as such could not be fully assessed in this study. The EasyReader (Veda-lab, Alençon, France) reports semi-quantitative results, with no quantitative quality control material being available at the start of the evaluation. For these reasons, both analysers were excluded from this study.
Three systems proceeded to the full evaluation (Table 1).
Quantitative POC FIT analysers, sample collection devices, manufacturers and suppliers.
Evaluation samples
Aqueous Hb samples
Human Hb lysate was prepared from residual blood from a patient, with Hb in the reference range, which was obtained from the Blood Sciences Laboratory, Royal Surrey County Hospital. The Hb concentration of the lysate was determined by measurement as part of the full blood count on an ADVIA 2120 (Siemens Healthcare Ltd, Frimley, UK). The lysate samples were frozen at –20°C until used. To dilute the Hb lysate, buffer from the relevant sample collection device was removed and dilutions were made into 2 mL plastic bottles. Aqueous samples were pipetted either directly in to the cuvette or an analyser-specific sampling cup depending on the FIT system.
Hb variants: anonymized whole blood samples from subjects with known Hb variants, attending the Regional Haemoglobinopathy Service at Northwick Park Hospital, were used for this study. Variants were determined and their percentage abundance obtained using a Variant II – high-performance liquid chromatography machine (BioRad Laboratories Ltd, Watford, UK). Six Hb variants were used for this study: Hb D-Punjab (AD), Hb E (AE), sickle cell trait (AS), foetal cord blood (FA), sickle-beta thalasaemia0 (SB0) and sickle C (SC). Variant Hb samples were stored at –20°C on arrival and were prepared by the same method as the non-variant samples prior to analysis.
Faecal samples
Patient samples sent to Berkshire and Surrey Pathology Services for calprotectin measurement were used. The samples were anonymized and divided into plastic bottles; each sample was then homogenized for 2 min using a clean wooden stick. The initial Hb concentration in the samples was determined using a laboratory FIT system to identify the positive samples for future use. The OC-Sensor Pledia was selected, as it was in routine use in our laboratory. The faecal samples were frozen at –20°C until required. Defrosted samples were further homogenized for 2 min before sampling with the POC devices.
External quality assessment
External quality assessment (EQA) material was provided by SKML (Nijmegen, Netherlands). This material was chosen as it is lyophilized faeces spiked with human Hb, which once reconstituted is in aqueous form. Using aqueous EQA limits the preanalytical variation which can arise from loading the sampling devices with faecal-like material used by some EQA schemes. 10 Samples were stored at 4°C upon receipt and brought to room temperature for at least 30 min before reconstitution following the manufacturer’s instructions.
Methods
Linearity
The measurement range of each assay quoted by the manufacturer was checked using serial dilutions of Hb lysate in manufacturer-specific buffer encompassing the expected range. Doubling dilutions were prepared in two series, one starting at the upper limit of the manufacturers quoted detection range and one starting at approximately 75% the upper detection limit. The highest concentration was designated a dilution factor of 1, and the dilution factors were calculated against this value. The results for each of the FIT systems for both series were combined, and R2 values were calculated.
Recovery
Two series of Hb lysate in manufacturer-specific buffer were prepared at the lower end of the measurement range of the FIT systems. For series one, increasing volumes ranging from 0 to 50 μL, were replaced with a prepared high concentration Hb lysate solution, and for series two, identical volumes were replaced with buffer. No volume replacement exceeded 10% of the final volume. The samples were measured in duplicate and the mean calculated. The differences between the corresponding mean values from series one and two were calculated. Recovery was calculated for each sample as
[(Sample series 1 – sample series 2)/(amount of analyte added)]×100. The mean of the recovery results equals the percentage recovery for that FIT system.
Imprecision
Within-run imprecision was calculated by measuring each quality control (QC) solution provided by the manufacturers 20 times each in one run.
Between-run imprecision was assessed by measuring three replicates of two concentrations of Hb lysate (10 µg Hb/g faeces and 50 µg Hb/g faeces, diluted in manufacturer-specific buffer and stored at –20°C). These were measured on five consecutive days using separate aliquots.
The mean, standard deviation (SD) and coefficient of variation (CV) were calculated.
A CV of <10% was considered satisfactory in accordance with recommended acceptance criteria. 11
Prozone detection
To test whether very high concentrations of Hb can cause artificially false low results due to antigen excess, we examined three concentrations of Hb lysate diluted with manufacturer-specific buffer. The Hb concentrations of the lysate were measured predilution on the ADVIA 2120 with the approximate value of the samples postdilution being between 1000 µg Hb/g faeces and 10,000 µg Hb/g faeces which greatly exceeds the measurement ranges of all the FIT systems. These samples were measured in duplicate to check whether erroneous results were blocked and reported with a clear error message.
Haemoglobin variant detection
Six Hb variants (AD, AE, AS, FA, SB0 and SC) and one ‘normal’ sample were diluted in manufacturer-specific buffer to give an approximate concentration of 20 µg Hb/g faeces; each variant was examined once on the relevant FIT system. The results for each variant are expressed as a % value of the normal ‘non-variant’ sample result for that analyser. Then, the median and IQR was calculated across all variants and analysers. We defined normal detection if the result was ±1.5 the interquartile range of the variant samples from the median. This is in keeping with our previous study when using the laboratory-based analysers. 12
Sample comparison
Patient samples: five Hb-positive faecal patient samples were examined using the collection devices specific to each FIT system. Prior to sampling, the faecal material was homogenized for 2 min using a clean wooden stick. For each FIT system, four collection devices were used to sample the specimens according to the manufacturers’ instructions. The samples were stored in the dark at room temperature for 24 h. All samples were mixed by inversion and allowed to stand for 10 min before analysis on their respective FIT system.
External quality assessment: one concentration of lyophilized EQA material was reconstituted according to the provided instructions and a single dilution made so that it was within the analytical range of all the systems. The correct amount of matrix was pipetted into system-specific cuvettes or a FIT system-specific sampling cup. The EQA material was analysed five times on each FIT system.
Ease of use
To investigate the suitability for analyser use in a clinic environment, we performed a usability analysis. A decision matrix was developed for each of the POC FIT systems (online Appendix 2). The categories were divided into six components for the use of the device in a clinic setting. These included initial machine set up, starting a session, the sample devices for the patient, the processes in running an individual test, the results display and the portability of the device. A clinic session to review colorectal patients will typically allow 10 min for review of each patient; therefore, the highest weighting was given to the ease and time to perform a test. Clinics often occur in different rooms and therefore increased weighting was also given to the portability of the machine. Each category was broken down into subcategories and the weighting for each then determined the overall score.
Two individuals performed the usability assessment of each POC FIT system. One was a laboratory technician that specializes in FIT and the other was a colorectal clinician with no previous laboratory experience. The scoring was based on the consensus between both parties for each subcategory. To avoid bias, the individuals were unaware of the analytical evaluation results at the time.
Results
Analysers
All three FIT systems work on the principle of immunoturbidimetry. Small particles are coated with antibodies specific for human Hb. These antibodies bind to the Hb resulting in an increase in turbidity. This change is measured by the FIT system and converted in to a numerical value.
The Cube is a small portable bench top analyser which operates through a dedicated Android App. It has dimensions of 160 × 130 × 145 mm and weighs 2.4 kg. It takes 9 min to analyse one sample and only one sample can be processed at a time.
The OC-Sensor iO is a bench top analyser that has dimensions of 360 × 560 × 425 mm and weighs 35 kg. This analyser is not portable. It takes 7 min to analyse one sample. The iO has capacity to load 20 samples at once and can run 88 samples an hour.
The QuikRead go is a small portable bench top analyser with dimensions of 270 × 155 × 145 mm and weighs 1.9 kg. It takes 2 min to analyse one sample and only one sample can be processed at a time.
Linearity was R2>0.97 for all FIT systems within their quoted measurement range; spike recovery ranged was 93% on the Cube, 73% on the iO and 108% on the QuikRead go. It is important to highlight that the measurement range of the Cube is small, up to 80 µg Hb/g faeces. All three methods had a CV <5% for the within-run imprecision on all concentrations tested and <10% for between-run imprecision.
Prozone detection
The iO and QuikRead go detected the prozone appropriately and both reported an error code; the iO gave error code OR (over-read) and a numerical result which was above 200 µg Hb/g faeces. There is a risk that the number reported on the iO could be misunderstood to be an f-Hb concentration which would be falsely low. The QuikRead go gave error code E16422 (cannot measure the Hb concentration of the sample) without any numerical result.
The Cube reported an incorrect (falsely low) numerical result with no indication that these results could be wrong. The manufacturer does state that there is a prozone effect with this method. This was also demonstrated as part of the linearity assessment in which the Cube showed very clear signs of prozone effect (as the concentration increases the detected result decreases) at much lower concentrations.
Figure 1 shows the linearity assessment of the Cube using the initial expected concentrations based on the Hb results from the Advia 2120.
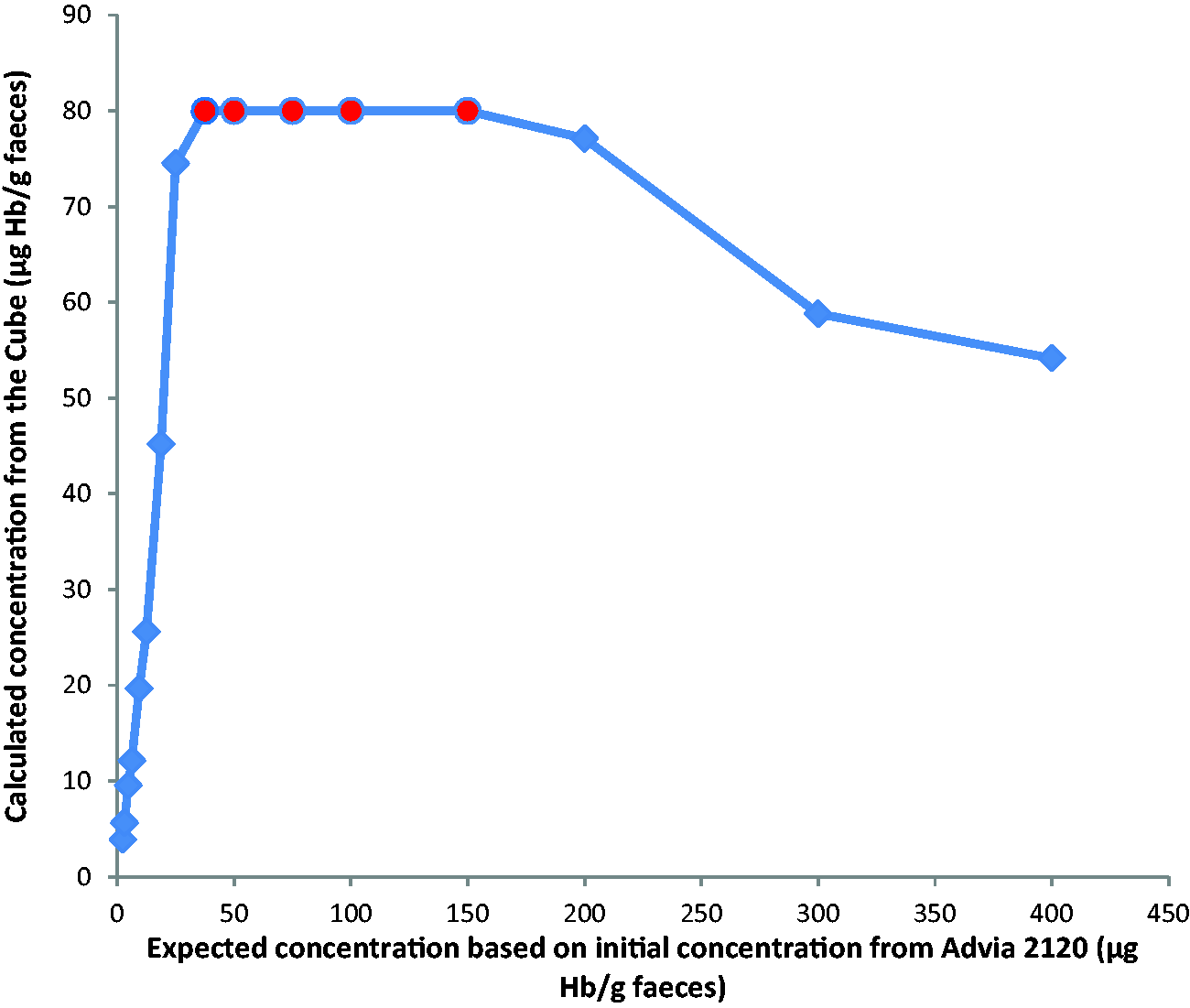
Linearity and prozone of the Cube showing expected concentrations based on results from Advia 2120.
Haemoglobin variant detection
The specificity of the methods is shown in Figure 2. Four results fell outside of the normal detection range – one above (AS for QuikRead go) and three below (FA for Cube and iO; SB0 for Cube).
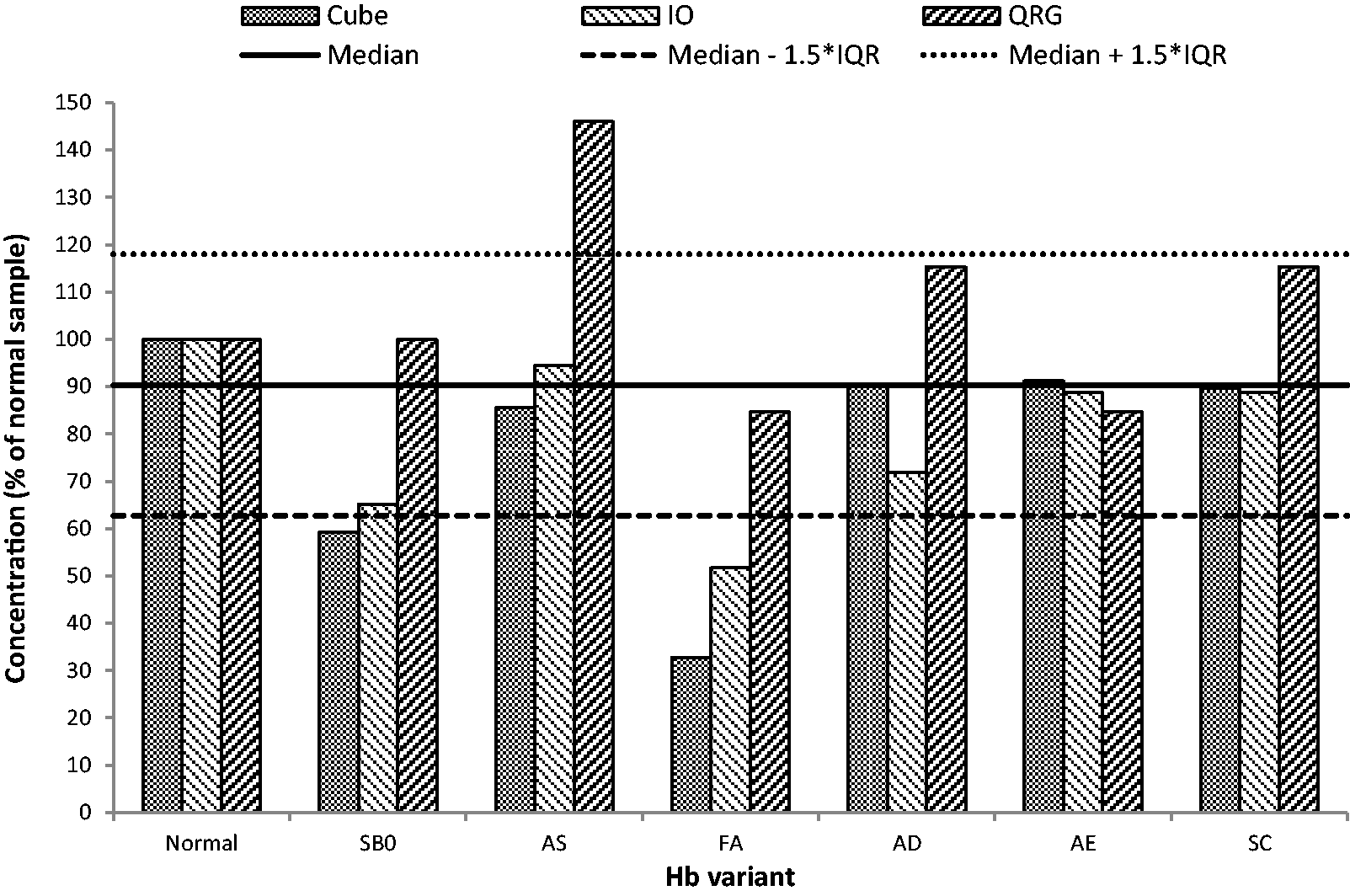
Specificity of the quantitative POC FIT methods for Hb variants (QRG – QuikRead go).
POC method comparison
The results of the sample comparison are shown in Table 2. The positive patient samples are labelled A-E, the SKML EQA labelled F. The Cube gave results >80 µg Hb/g faeces for three out of four repeats for sample C (not shown in Table 2). The iO gave results <10 µg Hb/g faeces for sample A, and both the iO and QuikRead go gave results <10 µg Hb/g faeces for sample D. Results outside of the measurement range stated by the manufacturer have not been shown on the table. The Cube tends to give higher results than the other two FIT systems for the patient samples. This is clear with sample D where the iO and QuikRead go both give results of <10 µg Hb/g faeces, while the Cube gave a mean result of 16 µg Hb/g faeces. For samples B and E, the Cube gives results over twice as high as both the iO and the QuikRead go. Both the iO and QuikRead go gave similar results for all samples except sample A where the iO was below the measurement range. Any variation due to the collection devices is negated in sample F where a reconstituted lyophilized EQA sample was used. All results for sample F are similar ranging from 18 to 23 µg Hb/g faeces.
Method comparison of Hb measured by quantitative FIT POC systems: patient samples (n = 4 for each sample A-E) and EQA samples (n = 5 for sample F).
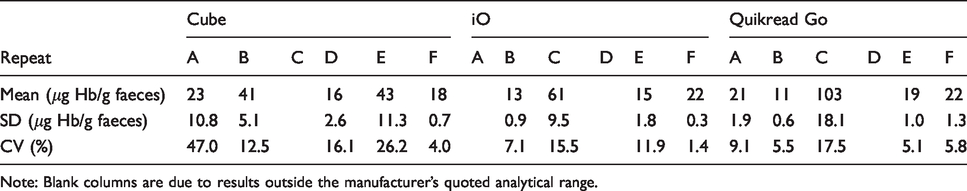
Note: Blank columns are due to results outside the manufacturer’s quoted analytical range.
Ease of use
Table 3 shows the results of the ease of use study. The scoring matrix and weighting for each parameter were devised by a clinician based on perceived importance for a POC system in a clinical setting.
Ease of use of quantitative POC FIT systems.
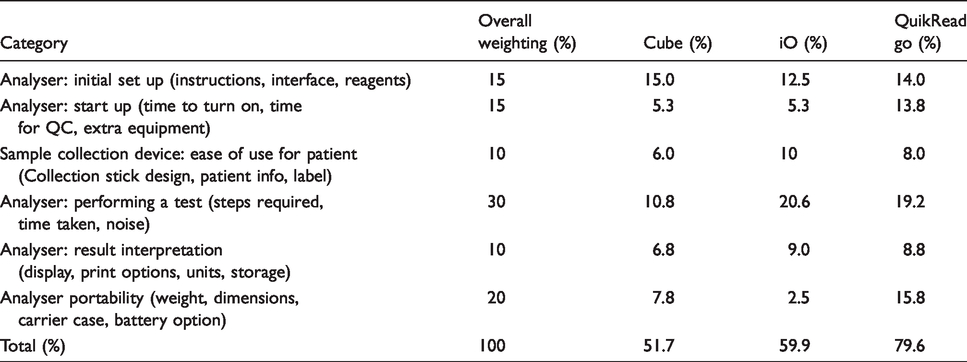
The QuikRead go scored higher than the other two systems in the ease of use scoring matrix due to its portability and speed to set up and run a patient sample.
Discussion
To our knowledge, this is the first study to consider the analytical validity of quantitative FIT systems that are marketed as suitable for use at the point of care in a clinic environment. With the increasing use of FIT, especially since the COVID-19 pandemic, there is a greater need to offer services that make clinical pathways more efficient. POC testing is often considered by clinical teams, and it is important to ensure that the analytical characteristics of any devices used are fully understood.
We immediately identified that two systems, the QuantOn Hem and EasyReader, were not fit for the purpose. One of these measures a combination of f-Hb and faecal haptoglobin through a polyclonal antibody system but does not report these results individually giving a single numerical value. These results cannot be interpreted in line with current FIT guidelines which rely on f-Hb alone. The other was a semi-quantitative method with no quantitative quality control material available and so was unable to proceed through the evaluation process for a quantitative POC FIT.
Of the three systems tested, the Cube, the iO, and the QuikRead go, each manufacturer provided details of their own assessment of their FIT systems, and the results from this evaluation were either compared with these claims, or they were within the recommended guidelines (online Appendix 1). All three systems performed according to the manufacturer claims, and the evaluation of the QuikRead go and the iO revealed no concerns based on their analytical evaluation.
We have not independently verified the limit of detection (LOD) and limit of quantitation (LOQ) for each of these FIT systems. The manufacturer of the Cube provided information for the LOD and LOQ; however, this was calculated using the Mindray BS-300 analyser and not the Cube FIT system. The Quikread go manufacturer quotes an LOD of 2.5 µg Hb/g faeces and LOQ of 9.5 µg Hb/g faeces, but the system does not report numerical results below 10 µg Hb/g faeces, so we were unable to verify these data. The iO quote an LoD of 2 µg Hb/g faeces and an LoQ of 4 µg Hb/g faeces, but the recommendation is only to report results between 10 and 200 µg Hb/g faeces.
The Cube has an analytical range up to 80 µg Hb/g faeces. In principle, this small analytical range could be deemed acceptable because in symptomatic populations, a very low threshold of 10 µg Hb/g faeces is most commonly used as a clinical decision point. 3 During this evaluation, we found that the Cube reported results of diluted lysate much higher than expected based on the initial results of the Advia 2120 and also much higher than the other two systems. As there is no standardization of FIT, it is not possible to determine at which concentration the prozone effect begins, although Eurolyser have shown the prozone effect starting at 112 µg Hb/g faeces. This work was completed using the Cube materials but was analysed using the BS-300 (Mindray). 13 The Cube is unable to accurately report a very high sample as high, and our study demonstrates that it reports a falsely low result, which poses a clinical risk. Indeed, currently during the COVID-19 pandemic, thresholds of 100 µg Hb/g faeces (England) and 400 µg Hb/g faeces (Scotland) have been published as concentrations requiring urgent investigation.4,5 There is a risk that a high f-Hb would be reported as <80 µg Hb/g faeces using the Cube system.
In addition to this, the Cube had the highest variation in Hb results when sampling with multiple devices from the same homogenized patient sample or aqueous EQA material directly pipetted in to the cuvette (CV range 7.2–47%). The iO (CV range 0.9–15.5%) uses the OC-Sampling Bottle 3 which previous work has shown has a well-designed internal collar to remove excess faeces; this study showed the mass of faeces loaded on to the probe did not affect the concentration of Hb captured by the device. 14 The QuikRead go (CV range 2.6–17.5%) has a similar design with a tight fitting collar to remove excess faeces. The Cube has a collar but it is not as tight as the other two devices which could lead to increased variation in the amount of faeces in the buffer. Further studies would be required to assess the effectiveness of the collar in the sampling devices of the Cube and Quikread go.
A limitation of this study, we concede that it contains a small number of patient samples. Ideally, we would have included at least 20 for our assessment, but unfortunately, this was not possible. Also due to the lack of standardization of FIT, we have been unable to determine the accuracy of the systems using EQA materials.
In the usability assessment, the QuikRead go outperformed the other devices. It was easily portable, and a result was available in 2 min. The iO performed less well because it is essentially a smaller form of the laboratory-based analysers. Although smaller than the laboratory instrument from the same manufacturer, it is much less portable than the QuikRead go and the Cube, although it did offer a quick analysis, taking 7 min to generate a result. The Cube was deemed to perform the least well on usability as it took approximately 10 min to generate a result after sample analysis was started. Although the device was light and small, there was no carrying case for the analyser and the reagents.
The iO requires several extra steps before the instrument can be used, such as preparing the wash solution and filling the deionized water tank, and in our opinion, the iO would be more suitable in a laboratory environment. The iO may also have a higher throughput than is required in a POC setting, which may lead to wastage of reagents. The packaging of the Cube and the QuikRead go reagents lend themselves to a clinical setting where staff are less likely to have experience running analytical systems. All three systems have the potential to print out the results immediately after analysis which may be an important requirement for a clinician. However, the iO is the only system with an integrated printer; the Cube and the QuikRead go require an additional printer which were not provided for this study.
One consideration for the QuikRead go is that only one level of QC is currently available. Should this system be implemented, then it is our belief that additional levels should be produced and made available. We also believe that there is a need for independent QC to be made available for all FIT systems. 15
The FIT systems were also reviewed in context of traceability of the patient results and the reagents, calibrator and QC lot information (online Appendix 3). All three FIT systems tested have the potential to be integrated into hospital results systems and for a barcode reader to scan a barcode on the device. The Cube and the QuikRead go record reagent, calibrator and QC lot information, but the iO does not have this facility. The Cube and the QuikRead go also have the potential to set up individual user ID’s on the FIT system, whereas the iO does not have this option.
As there is no international standardization of FIT, 16 we have been unable to assess the accuracy of the methods. As such, clinical studies are required to validate the results in a clinical pathway, ideally against diagnostic outcomes, e.g. colonoscopy or at least as a method comparison against an established laboratory method.
The QuikRead go has previously been considered in a study by Gies et al. looking at the diagnostic accuracy of a range of quantitative FIT tests, four of which were POC. 17 The study was in a bowel cancer screening population and they adjusted positivity thresholds to give a predefined positivity rate. The analysers performed equally well supporting our study that indicates that the QuikRead go has potential for use in the clinical environment. The Gies study did not include an analytical evaluation. A further study by the same group considered the diagnostic performance characteristics of six qualitative immunochemical faecal occult blood POC tests. 7 The study highlighted the different diagnostic performances of each test and advised that careful evaluation of each test is required. While these qualitative POC FIT are different to the quantitative ones that we have evaluated, it does highlight the potential risks of easy access to POC FIT and therefore the need for independent evaluation to enable us to understand the analytical characteristics of each method before it is implemented in to routine use. We are not aware of any study of a direct comparison of the qualitative and quantitative POC FIT systems. Quantitative systems have the advantage in providing a numerical result allowing a specific and user defined threshold to be applied. This facility is not applicable to a qualitative system which will have a fixed threshold.
This study is an analytical evaluation of POC FIT systems and we have not addressed several other factors which may be important when choosing a POC FIT system. We have not considered the requirements for verification of the FIT assays such as the ISO standards of the UK Accreditation Service (UKAS). The cost for each of the systems is also outside the remit of this evaluation. While we have provided information regarding the practicalities of the ease of use of each FIT system, we have not addressed when and how a patient would take a sample as there are several options.
In summary, we have identified two quantitative FIT systems that are potentially suitable for quantitation of f-Hb at the POC. In addition, we have highlighted some weaknesses of other systems marketed as quantitative FIT for POC to avoid inappropriate use of these instruments and hence clinical risk to patients. Clinical studies are now required to validate the diagnostic accuracy of these methods.
Supplemental Material
sj-pdf-1-acb-10.1177_0004563220986595 - Supplemental material for Assessment of the analytical performance of point-of-care faecal immunochemical tests for haemoglobin
Supplemental material, sj-pdf-1-acb-10.1177_0004563220986595 for Assessment of the analytical performance of point-of-care faecal immunochemical tests for haemoglobin by Shane O’Driscoll, Magdalen Carroll, William Maclean, Carolyn Piggott, Iain Jourdan and Sally C Benton in Annals of Clinical Biochemistry
Supplemental Material
sj-pdf-2-acb-10.1177_0004563220986595 - Supplemental material for Assessment of the analytical performance of point-of-care faecal immunochemical tests for haemoglobin
Supplemental material, sj-pdf-2-acb-10.1177_0004563220986595 for Assessment of the analytical performance of point-of-care faecal immunochemical tests for haemoglobin by Shane O’Driscoll, Magdalen Carroll, William Maclean, Carolyn Piggott, Iain Jourdan and Sally C Benton in Annals of Clinical Biochemistry
Supplemental Material
sj-pdf-3-acb-10.1177_0004563220986595 - Supplemental material for Assessment of the analytical performance of point-of-care faecal immunochemical tests for haemoglobin
Supplemental material, sj-pdf-3-acb-10.1177_0004563220986595 for Assessment of the analytical performance of point-of-care faecal immunochemical tests for haemoglobin by Shane O’Driscoll, Magdalen Carroll, William Maclean, Carolyn Piggott, Iain Jourdan and Sally C Benton in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to thank the charity “GUTS- fighting bowel cancer” based in Guildford, for providing funding to enable us to recruit staff to support this study. We thank the FIT system manufacturers and suppliers (Eurolyser Cube: Eurolyser Diagnostica GmbH, Salzberg, Austria and Oxford Biosystems Ltd, Oxon, UK; OC-Sensor iO: Eiken Chemical Co., Ltd, Tokyo, Japan and Mast Diagnostics Division, Bootle, UK; QuikRead go: Aidian Oy, Espoo, Finland; QuantOn Hem: Immundiagnostik AG, Bensheim, Germany; EasyReader: Vedalab, Alençon, France and Quadratech Diagnostics Ltd, Lewes, UK). We would also like to thank the Regional Haemoglobinopathy service for providing the haemoglobin variant samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the charity ‘GUTS – Fighting bowel cancer’ a charity based in Guildford, UK.
Ethical approval
Not applicable.
Guarantor
SCB.
Contributorship
SCB conceived the study. MC planned the study. SOD undertook the laboratory FIT analysis. WM created the ‘ease of use’ parameters from a clinical perspective and completed the assessment on each FIT system with SOD. SOD, MC and SCB created drafts of the article. SCB and CP reviewed all drafts. All authors contributed towards the writing of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
