Abstract
Background
Faecal immunochemical tests (FIT) for haemoglobin (Hb) are being used in the investigation of colorectal cancer. These tests use antibodies raised to the globin moiety of human Hb. Where the globin structure is abnormal or reduced, it is possible that antibody binding, and thus Hb-detection may be affected.
Methods
Lysates prepared from whole blood samples of patients with known variants were diluted in manufacturer-specific buffer to 10, 100 and 500 μg Hb/g faeces. These samples were analysed on four FIT analysers and the results compared with samples with no known variant present (normal samples).
Results
The results from this study show that of 20 variants tested, three showed a decrease in detection by all four analysers. These were β-thalassaemia major and two fetal cord blood samples.
Conclusions
Of 20 common Hb variants studied, 17 did not affect detection of Hb by the FIT systems tested. Hb variants leading to a reduction in the presence of a globin chain caused a reduction in Hb detection; in such cases, cancers could be missed.
Introduction
Faecal immunochemical tests for haemoglobin (FIT) are used worldwide for screening of asymptomatic patients, 1 ruling out cancers in patients with low-risk symptoms, 2 and are being studied for use in diagnosis and monitoring of irritable bowel diseases (IBDs). 3 These tests use polyclonal antibodies raised to the globin moiety of human Hb to detect blood in faecal samples. Human adult Hb is formed of four globin chains, two α-chains and two β-chains. This structure can be affected by a number of disorders characterized by synthesis of an abnormal globin chain (Hb variants), reduced synthesis of one or more globin chains (thalassaemias) or a mixture of both. 4 Occasionally, a patient will present with hereditary persistent fetal haemoglobinaemia (HPHF) 5 in which fetal Hb (α2γ2) production continues into adulthood with a simultaneous decrease in the production of the β-chain.
If the globin structure is abnormal or reduced, detection of Hb in faeces by FIT could be affected and polyps or cancers missed. Currently, the only data available describing assessment of the detection of Hb variants and thalassaemias by FIT systems are provided in the instructions-for-use from Sentinel which states that ‘The assay recognizes the following human Hb variants: Hb S, Hb C, HbA2 and Hb D’. 6 The other manufacturer’s instructions do not currently mention human Hb variants.
A challenge in the analytical evaluation of FIT systems is the difficulty surrounding acquisition of genuine patient samples. To overcome this, samples from patients presenting at a Regional Haemoglobinopathy service, for routine appointments were used. Whole blood lysate was used to enable the impact of the Hb variants to be assessed across the four analysers.
In this study, detection of a range of Hb variants was assessed. This included the most common frequently encountered Hb variants in the current UK population: HbC, HbD, HbE, sickle cell trait, β-thalassemia major and β-thalassemia carriers, sickle C, sickle cell disease. 4 Also included were patients with rarer conditions who were seen at the centre while samples were being collected (HbJ-Broussais, HbManitoba and HbQ-India), and cord blood in which there is a high concentration of HbF present. These samples cover mutations in alpha and beta globin chains and a reduction in the production of the beta chain.
The aim of this study was to assess the impact of Hb variants on detection of Hb by four FIT systems.
Four analysers and their appropriate buffers were assessed: HM-JACKarc (Kyowa Medex Co. Ltd), NS-Prime (Alfresa Pharma Corporation), OC-SENSOR PLEDIA (Eiken Chemical Co. Ltd) and SENTiFIT 270 (Sentinel Diagnostics).
Materials and methods
Samples
Anonymized whole blood samples from subjects with known Hb variants, attending the Regional Haemoglobinopathy service at Northwick Park Hospital, were used for this study. Variants were determined and their percentage abundance obtained using a Variant II – High Performance Liquid Chromatography machine (BioRad Laboratories Ltd, Watford, UK).
The numbers in brackets show the abundance of the variants detected in the samples studied, giving an indication of how much normal Hb (α2β2) is present in each sample, in some cases there are two samples and thus two values.
The samples used were: Normal (HbA2 = 2.6%); Hb C (35%/38%); Hb D-Punjab (36%/38%); Hb E (27%/25%); Hb J-Broussais (22%); Hb Manitoba (9.7%); Hb Q-India (17%); sickle cell trait (AS) (37%/39%); β-Thalassemia Major (BTM) (HbF = 100%) and β-Thalassemia carrier (BTC) (HbA2 = 4.7%/4.9%) Cord-blood (FA) (Hb F = 62%/78%); Sickle C (SC) ([HbS = 50%/HbC = 47%]; [HbS = 48%/HbC = 45%]); Sickle cell disease (SS) (95%/93%).
Sample preparation
Lysates were prepared for each sample and included a final 1-in-2 dilution in physiological saline. The Hb concentration of each diluted lysate was determined by measurement on an ADVIA 2120 (Siemens Healthcare Ltd, Frimley, UK). This concentration was used as a basis for determining the dilution required to give samples with concentrations in the range of 10, 100 and 500 μg Hb/g faeces. For each FIT system, each sample was diluted using the appropriate manufacturers’ buffer (from collection devices provided by the manufacturers).
Diluted samples were measured in sample cups, in duplicate using appropriate analysers. Analysis was run in two batches, with one normal sample in each batch.
Results
Of 22 samples studied, two were ‘normal’ samples (no known variant detected by liquid chromatography), and 20 were variant samples. Figure 1 shows the results of the variant samples at 10, 100 and 500 μg Hb/g faeces, presented as a percentage in comparison with the results of the normal Hb samples on the four analysers. We defined normal detection if the result was ±1.5× the interquartile range of the samples, 17 of the samples fell within acceptable limits with exceptions shown in Figure 1.
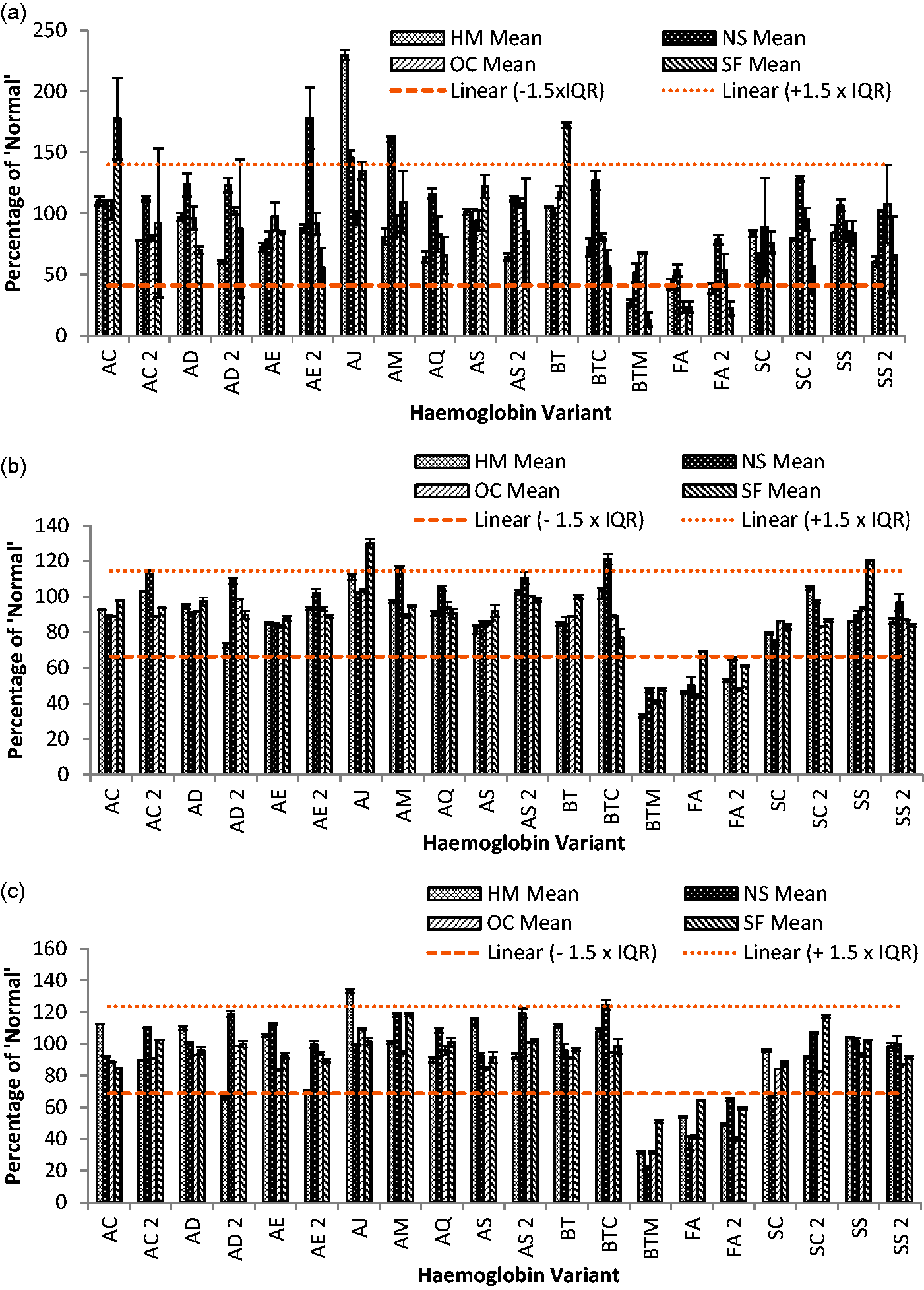
Each variant as a percentage of the normal sample run with each batch of tests at: (a) 10 μg Hb/g faeces, (b) 100 μg Hb/g faeces and (c) 500 μg Hb/g faeces. The results are separated by analyser, and the error bars show ± SD. The lines show ± 1.5× interquartile range.
The three samples classified as inadequately detected were the β-thalassaemia major (BTM), and two cord blood samples (FA and FA2). Results for these three samples ranged from 13% of normal – BTM on the SENTiFIT 270 at 10 μg Hb/g faeces to 69% of normal – FA on the SENTiFIT 270 at 100 μg Hb/g faeces (Figure 1).
Discussion
When evaluating FIT systems, ideally, genuine patient faecal samples should be used. However, these have limitations: it is impossible to know the original concentration of Hb released into the stool, making it difficult to compare the variant sample detection to that of ‘normal’ samples; the faecal matrix of every patient is unique and affects the degradation of the Hb individually. In combination, these limitations affect the ability to determine whether any disruption to detection is occurring. Thus, in this evaluation, we have used Hb lysate diluted in analyser-specific buffer (which contain products aimed at stabilizing Hb) as a surrogate, enabling comparison of how the four different analysers and their antibodies react analytically to the Hb variants assessed. 7
Of 20 Hb variants analysed, 17 measured as expected on the four analysers studied. Three main exceptions were the BTM (a sample collected before the patient was given their first blood transfusion) and two cord blood samples in which there are high concentrations of HbF and low levels of β-chain. The results in these samples were approximately 50% of the expected concentration at all concentrations studied, on all four analysers.
BTM (α2β2°) patients produce no β-chain, but are treated with regular blood transfusions which do contain the β-chain, and thus polyps, cancers and IBD should not be missed if tested with FIT.
Fetal Hb (at birth: α2γ2) is gradually replaced by adult Hb (α2β2) in the majority of people. A small percentage of people continue to produce fetal Hb (e.g. HPFH), 5 these patients if screened with FIT could have IBD, polyps or cancers missed. If diagnosed with IBD and FIT is used to monitor the patient, they will require a unique reference range to ensure flare-ups are not missed.
It is important to note that there are over 1700 different Hb variants identified, 4 and only 20 of these were investigated in this study.
Conclusions
This small study indicates that it is people with a missing globin chain that will potentially have a false low or negative FIT which could lead to cases of cancer and IBD being missed. The use of polyclonal antibodies appears to limit the disruption in Hb detection caused by other variants. Further work is required to investigate more Hb variants.
Footnotes
Acknowledgements
We thank FIT system suppliers (HM-JACKarc: Alpha Laboratories, Eastleigh, Hampshire, UK; OC-SENSOR PLEDIA: Mast Diagnostics Division, Bootle, Merseyside, UK; SENTiFIT 270: Sysmex UK Ltd, Milton Keynes, Buckinghamshire, UK; NS-Prime: Alere Ltd, Chester, Cheshire, UK) for supplying the analysers and consumables, and the Regional Haemoglobinopathy service for providing the blood samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Anonymized whole blood samples from patients with known variants were analysed. Ethical approval was not required.
Contributorship
SCB and NKD conceived the study. MRRC designed the study, processed specimens, analysed the data and prepared the manuscript. DB and CJ processed the specimens.
