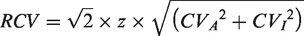Abstract

In their recent letter to another journal, 1 Flowers et al. highlight the importance of ensuring that analytical precision (CVA) is sufficient to meet the requirements of the European Society of Cardiology (ESC) guidelines 2 for rule-out of non-ST-segment elevation myocardial infarction (NSTEMI) using measurement of high-sensitivity cardiac troponin T (cTnT). They conclude that the CVA of both the Roche Cobas e602 and Cobas e801 is adequate to reliably detect the delta change required, subject to appropriate use of low concentration internal quality control (IQC).
The method of Flowers et al., using a single serum pool, has the effect of removing any difference in cTnT concentration that is due to intraindividual biological variation (CVI). Conceptually, we should expect cTnT CVI to be very low in healthy individuals because they should by definition have very low cTnT. However, any detectable cTnT (≥5 ng/L) has been shown to be associated with increased risk of future cardiac events; 3 therefore, it could be argued that patients in whom NSTEMI is ruled out by the ESC algorithm with baseline cTnT <12 ng/L and Δ0–1 h <3 ng/L are not ‘healthy individuals’. It is probably prudent to take into account CVI because we are concerned with ruling out a significant change and hence diagnosing a stable low-level elevation in cTnT.
A recent review has summarized published data on biological variation.
4
Estimates vary widely according to the subject population, the analytical platform and the timeframe. If we limit ourselves to healthy individuals, using the more precise Cobas e602 and looking at short-term variation (over a period of hours), the CVI is 1.2%. In routine clinical use of the cTnT assay to exclude NSTEMI, this component of variation remains, and it should be incorporated when calculating the total variation allowable to meet the delta change target of the guideline, using the equation for a reference change value (RCV)
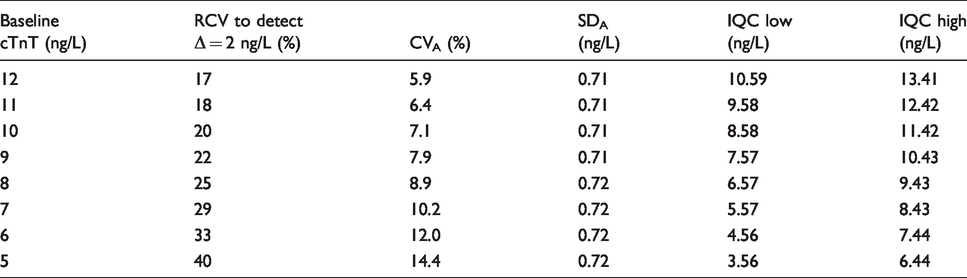
In our laboratory, we looked at this question the other way around and attempted to define the CVA required to meet the ESC requirement at 95% certainty, i.e. with z = 1.96. The ESC guidelines use Δ0–1 h <3 ng/L; this implies that we need to be confident in Δ0–1 h = 2 ng/L. We calculated the maximum allowable RCV in order to detect a delta change of 2 ng/L for each baseline cTnT concentration from 12 ng/L down to 5 ng/L. By substituting these values and CVI=1.2% into the above equation, we were able to calculate the required CVA at each cTnT concentration. From this figure, we calculated the acceptable standard deviation (SDA), and hence defined IQC low and high limits as ±2 SD from the target (Table 1). Definition of acceptability for performance of the low-level IQC is important, because commercial IQC suppliers often quote ranges that are too wide.
QC limits as defined using the ESC 1-hour algorithm for ruling out non-ST-segment elevated myocardial infarction.
It should be appreciated that while the study of Flowers et al. suggests that this analytical precision is attainable, these are still ambitious targets for routine clinical use and require close monitoring of the IQC performance.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable
Guarantor
OCM.
Contributorship
OCM conceived and executed the work.