Abstract
Background
This study aimed to investigate the long-term prognosis value of serum galectin-3, aquaporin (AQP)-1 and AQP-3 in young patients with colon cancer.
Methods
A total of 100 young patients with colon cancer, 100 cases of benign colon and 100 healthy people were collected. All colon cancer patients were followed up for 42 months.
Results
Compared with the benign lesion group and the control group, preoperative serum galectin-3, AQP-1 and AQP-3 concentrations were significantly increased in patients with colon cancer (P < 0.05). The immunohistochemistry scores of galectin-3, AQP-1 and AQP-3 in colon cancer patients were positively correlated with serum galectin-3, AQP-1 and AQP-3 concentrations (P < 0.05). Serum galectin-3, AQP-1 and AQP-3 concentrations were positively correlated with TNM staging (galectin-3: rPearson = 0.502, P < 0.001; AQP-1: rPearson = 0.415, P < 0.001; AQP-3: rPearson = 0.454, P < 0.001) and differentiation (galectin-3: rPearson = 0.377, P = 0.004; AQP-1: rPearson = 0.411, P = 0.001; AQP-3: rPearson = 0.483, P < 0.001). Receiver operator characteristic curve (ROC) analysis showed that the area under ROC curve (AUC) of the combination of galectin-3, AQP-1 and AQP-3 in distinguishing colon cancer was 0.907. The sensitivity in the parallel mode was 87.6%, and the specificity in the serial mode was 98.2%. Compared with the low galectin-3 group, low AQP-1 group and low AQP-3 group, the survival time of patients in the high galectin-3 group (χ2 = 13.929, P < 0.001), high AQP-1 group (χ2 = 10.157, P = 0.001) and high AQP-3 group (χ2 = 4.364, P = 0.037) were significantly shortened.
Conclusion
Galectin-3 combined with AQP-1 and AQP-3 had important value in the identification of young patients with colon cancer and was of great value in evaluating long-term prognosis.
Introduction
In 2018, the incidence of colon cancer in China accounted for 24.3% of the world, and the death rate accounted for 22.9% of the world.1,2 It is worth noting that among colon cancer patients, the proportion of young people under 30 years has increased to 15%. 2 In recent years, with the change in people's eating habits, the incidence of colon cancer has been increasing in China, especially for young people. The early symptoms of colon cancer are not typical. With the growth of the tumour, symptoms such as defecation habits, abdominal pain and bloody stools may appear. 3 The five-year survival rate of colon cancer is about 48.8%. 4 At present, the main screening methods for colon cancer include sigmoidoscopy or fiberscope, but the above methods are invasive, which makes people unwilling to conduct regular screening. Carcinoembryonic antigen (CEA) and cancer antigen 19–9 (CA19-9) have been used for the preliminary screening of colon cancer. However, the accuracy of them is still not satisfactory enough. 5
Proteomics technology has shown unique high sensitivity and specificity in the detection of molecular markers of solid tumours and plays an important role in the evaluation of tumour prognosis. 6 Recent studies have pointed out that the abnormal expression of galectin-3 is closely related to the occurrence and metastasis of various cancers. 7 Aquaporins (AQPs) are passive channels for water and small solute transport. Recent studies have shown that AQPs are related to the occurrence of different types of diseases, and AQPs play important roles in angiogenesis, cell migration and tumour growth. 8 Research evidence indicates that AQP-1 and AQP-3 have relatively higher concentrations in different types of tumours. 9 However, the expressions of galectin-3, AQP-1 and AQP-3 in the serum of young patients with colon cancer and their correlations with the long-term prognosis have not been reported. In this study, 100 young patients with colon cancer, 100 patients with benign colon disease and 100 healthy people were included to analyse the preoperative and postoperative concentrations of galectin-3, AQP-1 and AQP-3 in the three groups and explore the clinical value of galectin-3, AQP-1 and AQP-3 for young patients with colon cancer.
Materials and methods
Inclusion of colon cancer patients, benign colon disease patients and healthy people
A total of 100 consecutive young patients with colon cancer diagnosed from December 2015 to August 2016 were collected, and all were hospitalized for the first time and confirmed by endoscopic histological examination. The staging of colon cancer is based on the National Comprehensive Cancer Network (NCCN) guidelines version 2, 2018. 10 Among them, there were 37 patients with stage I–II, 63 patients with stage III–IV, 36 cases with well-differentiated cancer, 49 cases with moderately differentiated cancer, 15 cases with poorly differentiated cancer, 81 cases with adenocarcinoma, 9 cases with mucinous cancer and 10 cases with undifferentiated cancer. Before and after the operation, peripheral blood was collected from colon cancer patients, and serum was isolated. A total of 100 cases of benign colon diseases (including 25 cases of colon polyps, 29 cases of chronic specific colitis, 27 cases of fibroids and 19 cases of haemangiomas) were collected. In addition, 100 healthy people were selected as the control group.
Inclusion criteria: (1) Patients with colon cancer who did not receive radiotherapy and chemotherapy; (2) Subjects who signed the informed consent; (3) Patients who underwent gastroscopy and were diagnosed with benign colon diseases; (4) Healthy subjects who do not have underlying diseases. Exclusion criteria: (1) Persons with previous blood system diseases such as iron deficiency anaemia, aplastic anaemia, megaloblastic anaemia, etc.; (2) Patients with acute blood loss, history of blood transfusion or undergoing transfusion therapy; (3) Patients with pulmonary hypertension, acute coronary syndrome, ischaemic stroke, peripheral vascular disease (arteriosclerotic occlusive disease, arteriovenous thrombosis, aneurysm, etc.), acute and chronic vascular diseases such as heart failure, atrial fibrillation and hypertension; (4) Acute and chronic infections caused by bacteria, fungi and viruses; (5) Those with autoimmune diseases such as rheumatoid arthritis and systemic vasculitis; (6) Patients who did not complete follow-up or were lost to follow-up.
Ethical approval
The ethical approval, the review of informed consent, the implementation of experimental protocols and subsequent research of this study have been approved by the Clinical Medical Research Ethics Committee of the Second Affiliated Hospital of Anhui Medical University (Approval number: 2015317).
Collection of basic data and life history
Each subject completed a questionnaire before enrolment. The questionnaire mainly included age, gender, genetic history of colon cancer, staying up late, smoking history, drinking history, low fibre diet, high fat and high protein diet, history of nitrite intake, physical activity, etc.
Detection and quality control of serum galectin-3, AQP-1 and AQP-3
Serum galectin-3, AQP-1 and AQP-3 concentrations were detected by enzyme-linked immunosorbent assay. Serum specimens were diluted 200 times and were sequentially added to the microtitre plate and incubated with the antibody for 60 min at room temperature. After washing the plate five times, the substrate was added to the reaction for 30 min. Galectin-3, AQP-1 and AQP-3 kits were purchased from Shanghai Enzyme Biotechnology Co., Ltd.
In addition, we performed quality control on the galectin-3, AQP-1 and AQP-3 indicators testing and calculated the coefficient of variation (CV) of the intra- and inter-assay differences to evaluate galectin-3, AQP-1 and AQP-3 detection accuracy.
Follow-up of patients with colon cancer
Based on factors such as whether patients receive adjuvant treatment, financial status and convenience, a personalized follow-up plan is adopted. The general principles are as follows: Within one year after surgery, patients were reviewed every three months; within 1–2 years after surgery, patients will follow up every 3–6 months; within 2–5 years after surgery, patients will follow up every 6–12 months. Any suspicious signs that appear at any time after surgery should be consulted at any time. All patients in this study were followed up for 42 months, and their survival status was recorded during the follow-up.
Immunohistochemical detection and protein quantification
A total of five pairs of colon cancer patients' cancer tissues and adjacent tissues were collected for immunohistochemical detection of galectin-3, AQP-1 and AQP-3 in colon cancer tissues. Monoclonal antibodies galectin-3, AQP-1 and AQP-3 were added to the tissue samples, and the alkaline phosphatase secondary antibody was incubated at 4°C. ImagePro Plus was used to analyse the immunohistochemical results, and the percentage of positive cells and the staining strength of positive cells (x × y) were used, where the x represents the percentage of positive cells (x = 0, no positive cells; x = 1, positive cells were 1%–10%; x = 2, positive cells are 11%–50%; x = 3, positive cells are 51%–80%, x = 4 positive cells account for more than 81%), and the y represents the strength of positive cell staining (y = 0, negative; y = 1, weakly positive; y = 2, moderately positive; y = 3, strongly positive).
Statistical analysis
Statistical analysis was performed using SPSS 20.0. Comparisons between groups of normal distribution data (mean ± SD) were performed using one-way ANOVA or t-test. Pearson test was used to calculate the correlation between galectin-3, AQP-1 and AQP-3 and clinicopathological characteristics. Receiver operator characteristic curve (ROC) analysis calculated the area under ROC curve (AUC) of the serum galectin-3, AQP-1 and AQP-3 concentrations to discriminate colon cancer. The Kaplan–Meier method was used to calculate the survival analysis of serum galectin-3, AQP-1 and AQP-3 on colon cancer, and the Cox proportional regression model was used to analyse the potential factors affecting the prognosis of colon cancer patients. The α value was 0.05 in this study.
Results
Quality control and effectiveness evaluation of serum galectin-3, AQP-1 and AQP-3
As shown in Supplemental Table 1, the intra- and inter-assay CV values of galectin-3, AQP-1 and AQP-3 in the three groups were all less than 5%, suggesting that the sample detection system has good repeatability and accuracy.
Comparison of the immunohistochemistry scores of galectin-3, AQP-1 and AQP-3 in colon cancer tissues and preoperative serum galectin-3, AQP-1 and AQP-3 concentrations of subjects in the three groups
Compared with adjacent tissues, the immunohistochemistry scores of galectin-3, AQP-1 and AQP-3 proteins in colon cancer tissues were significantly higher (P < 0.05, Figure 1(a) to (c)). There were no significant differences in age, gender, genetic history of colon cancer, staying up late, smoking history, drinking history, low fibre diet, high fat and high protein diet, history of nitrite intake and physical activity (P > 0.05, Table 1). Univariate analysis of variance showed that the differences in preoperative serum galectin-3, AQP-1 and AQP-3 concentrations among the three groups were statistically significant (P < 0.05). Compared with the benign lesion group and the control group, preoperative serum galectin-3, AQP-1 and AQP-3 expression levels were significantly increased in patients with colon cancer (P < 0.05), as shown in Table 1 and Figure 1(d) and (e).
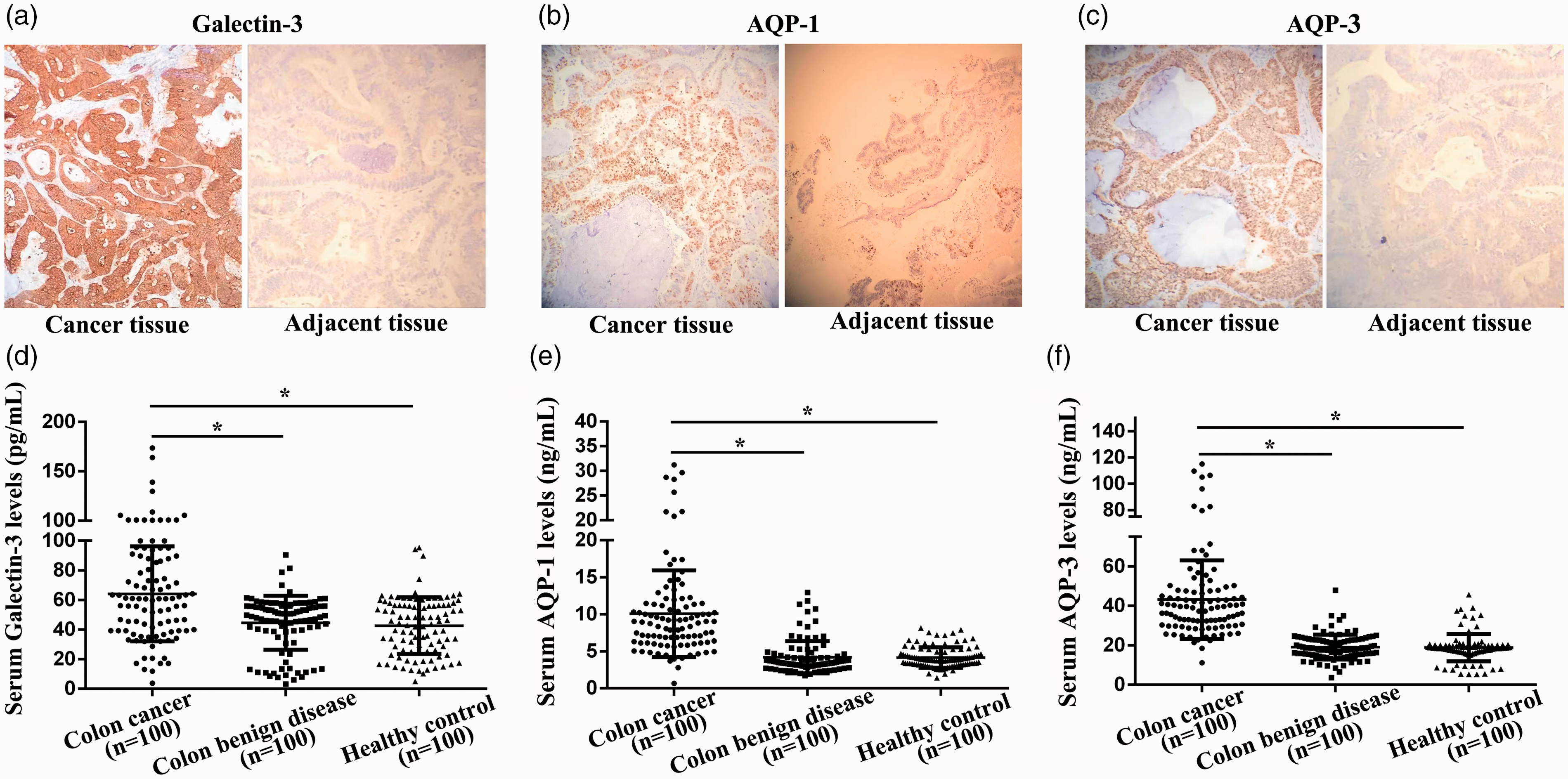
The immunohistochemistry of galectin-3, AQP-1 and AQP-3 in colon cancer tissues and serum galectin-3, AQP-1 and AQP-3 concentrations of subjects in young patients with colon cancer group, benign lesion group and the control group. (a) The immunohistochemistry of galectin-3 in colon cancer tissue. (b) The immunohistochemistry of AQP-1 in colon cancer tissue. (c) The immunohistochemistry of AQP-3 in colon cancer tissue. (d) Serum galectin-3 concentrations in the three groups. (e) Serum AQP-1 concentrations in the three groups. (f) Serum AQP-3 concentrations in the three groups. *Compared with colon cancer group, P < 0.05.
Comparison of clinical information and serum galectin-3, AQP-1 and AQP-3 concentrations in the three groups.
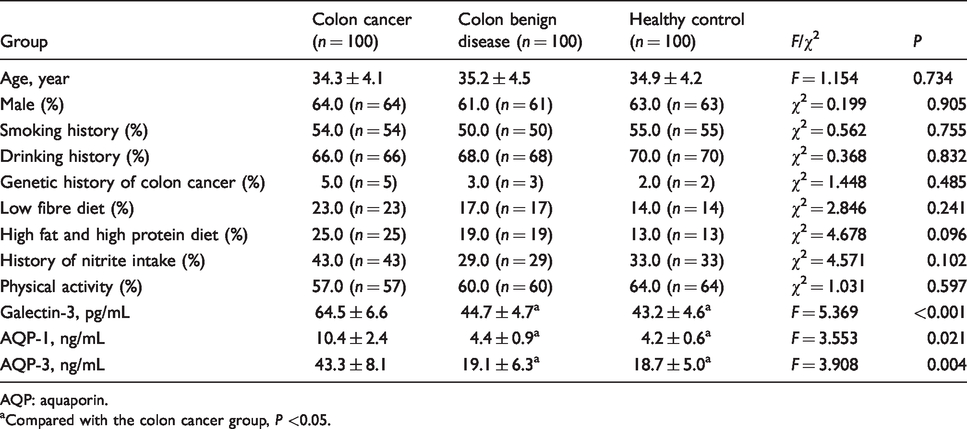
AQP: aquaporin.
aCompared with the colon cancer group, P <0.05.
Correlation of the immunohistochemistry scores of galectin-3, AQP-1 and AQP-3 in cancer tissues and preoperative serums of patients with colon cancer
Correlation analysis results showed that the immunohistochemistry scores of galectin-3, AQP-1 and AQP-3 in colon cancer patients were positively correlated with preoperative serum galectin-3 (rPearson = 0.481, P < 0.001, Figure 2(a)), AQP-1 (rPearson = 0.529, P < 0.001, Figure 2(b)) and AQP-3 (rPearson = 0.448, P < 0.001, Figure 2(c)) concentrations. Therefore, preoperative serum galectin-3, AQP-1 and AQP-3 concentrations can indirectly reflect the concentrations of galectin-3, AQP-1 and AQP-3 in cancer tissues and is expected to be used for the evaluation of patients' disease status and non-invasive screening.
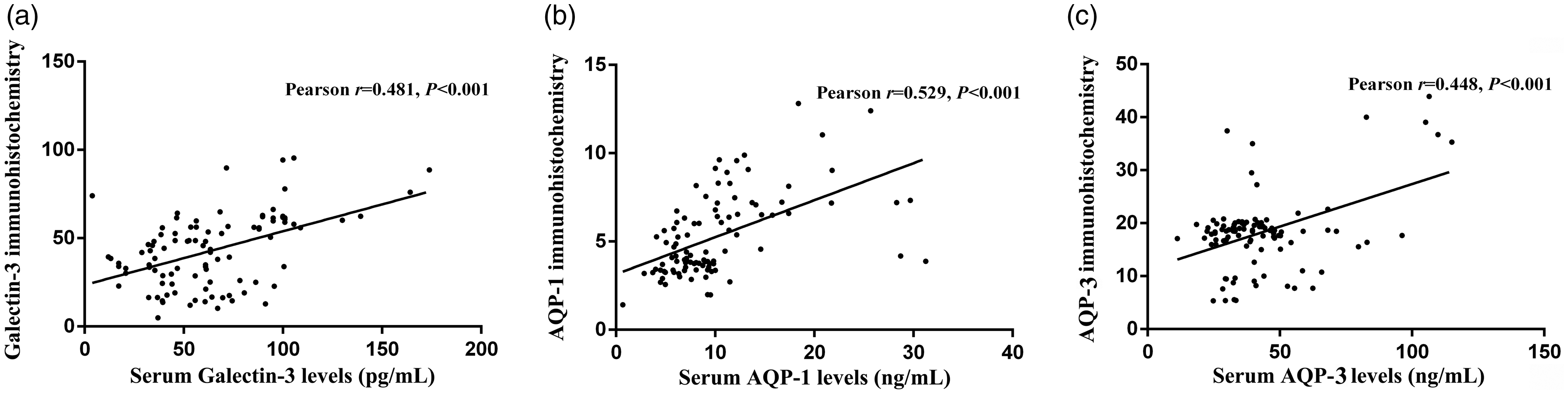
Correlation of the immunohistochemistry scores of galectin-3, AQP-1 and AQP-3 in colon cancer tissues and serums of patients with colon cancer. (a) The immunohistochemistry scores of galectin-3 in colon cancer patients were positively correlated with serum galectin-3. (b) The immunohistochemistry scores of AQP-1 in colon cancer patients were positively correlated with serum AQP-1. (c) The immunohistochemistry scores of AQP-3 in colon cancer patients were positively correlated with serum AQP-3.
Correlation of preoperative serum galectin-3, AQP-1 and AQP-3 concentrations with patients’ clinicopathological characteristics of colon cancer
Preoperative serum galectin-3, AQP-1 and AQP-3 concentrations were positively correlated with TNM staging (galectin-3: rPearson = 0.502, P < 0.001; AQP-1: rPearson = 0.415, P < 0.001; AQP-3: rPearson = 0.454, P < 0.001) and differentiation (galectin-3: rPearson = 0.377, P = 0.004; AQP-1: rPearson = 0.411, P = 0.001; AQP-3: rPearson = 0.483, P < 0.001). Moreover, galectin-3, AQP-1 and AQP-3 had the most significant increase in patients with advanced colon cancer and poorly differentiated colon cancer, as shown in Supplemental Tables 2 and 3.
Differential diagnosis of preoperative serum galectin-3, AQP-1 and AQP-3 in young patients with colon cancer
In this study, young patients with colon cancer were treated as the case group, and benign colon diseases and healthy controls were treated as the control group. The ROC curve analysis results showed that the AUC of preoperative serum galectin-3, AQP-1 and AQP-3 to distinguish colon cancer was 0.861 (95% CI: 0.823–0.899, P < 0.001), 0.804 (95% CI: 0.759–0.849, P < 0.001) and 0.780 (95% CI: 0.731–0.828, P < 0.001), respectively. The corresponding sensitivity and specificity when the Jordan index of each one is the maximum are 69.2%/92.4%, 80.2%/61.9% and 67.1%/75.7%, and the corresponding critical values are 57.1 pg/mL, 6.1 ng/mL and 29.2 ng/mL (Figure 3(a)). We used the logistic regression model technology to regression fit the preoperative serum galectin-3, AQP-1 and AQP-3 concentrations, and obtained the new variable ‘PRE-1’.Then, ROC analysis was performed on ‘PRE-1’. It was found that the AUC combined with the three indexes in distinguishing colon cancer was 0.907 (95% CI: 0.876–0.939, P < 0.001, Figure 3(b)). The sensitivity and specificity in the parallel mode are 87.6% and 84.4%, while the sensitivity in the serial mode is 70.7% and the specificity is 98.2%.
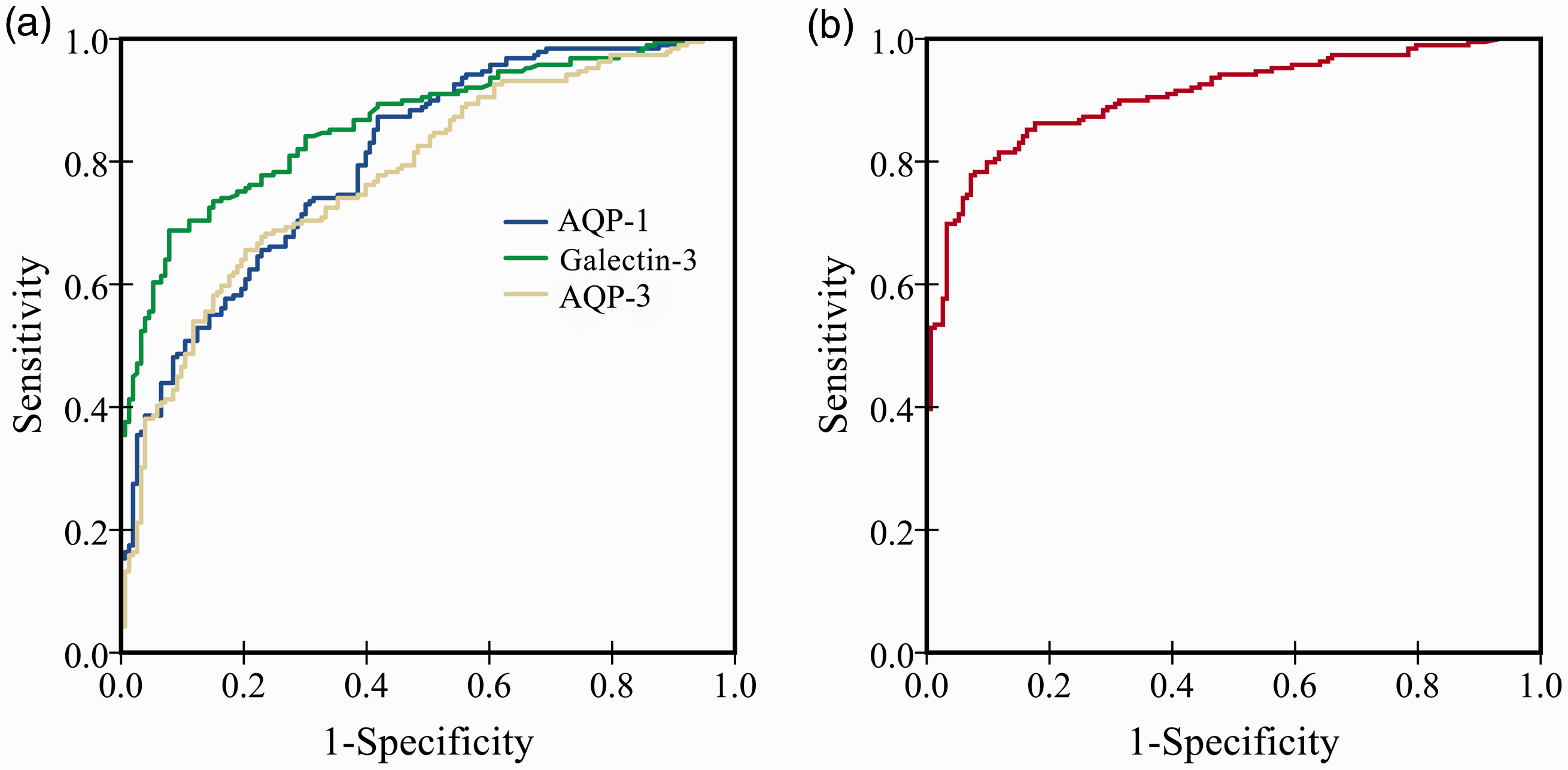
Differential diagnosis of serum galectin-3, AQP-1 and AQP-3 in young patients with colon cancer. (a) Differential diagnosis value of single index for young patients with colon cancer. (b) Differential diagnosis value of three indicators for young patients with colon cancer.
Survival analysis of postoperative serum galectin-3, AQP-1 and AQP-3 concentrations in young patients with colon cancer
A total of 37 colon cancer patients died during follow-up. According to the median postoperative serum concentrations of galectin-3, AQP-1 and AQP-3, 100 young patients with colon cancer were divided into high galectin-3 (n = 50) and low galectin-3 (n = 50) groups, high AQP-1 (n = 50) and low AQP-1 (n = 50) groups, and high AQP-3 (n = 50) and low AQP-3 (n = 50) groups. Compared with the low galectin-3 group, the survival time of patients in the high galectin-3 group was significantly shortened (χ2 = 13.929, P < 0.001, Figure 4(a)). Compared with the low AQP-1 group, the survival time of patients in the high AQP-1 group was significantly shortened (χ2 = 10.157, P = 0.001, Figure 4(b)). Compared with the low AQP-3 group, the survival time of patients in the high AQP-3 group was significantly shortened (χ2 = 4.364, P = 0.037, Figure 4(c)). Cox regression analysis showed that postoperative serum galectin-3 (HR = 1.763, P < 0.001), AQP-1 (HR = 1.265, P < 0.001) and AQP-3 (HR = 1.201, P < 0.001) were independent factors of poor prognosis in young patients with colon cancer (Table 2).
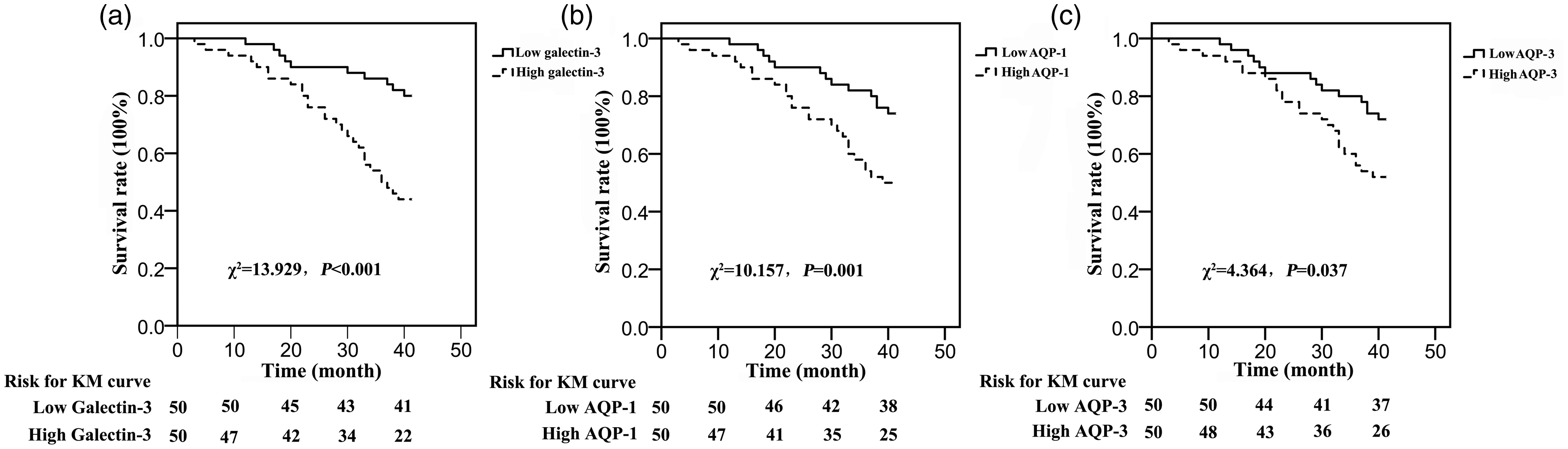
Correlation of serum galectin-3, AQP-1 and AQP-3 expressions with prognosis. (a) Compared with the low galectin-3 group, the survival time of patients in the high galectin-3 group was significantly shortened. (b) Compared with the low AQP-1 group, the survival time of patients in the high AQP-1 group was significantly shortened. (c) Compared with the low AQP-3 group, the survival time of patients in the high AQP-3 group was significantly shortened.
Cox regression analysis of serum galectin-3, AQP-1 and AQP-3 concentrations in assessing survival in young patients with colon cancer.
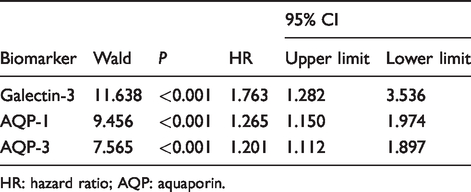
HR: hazard ratio; AQP: aquaporin.
Discussion
CEA and CA19-9 are important indicators for auxiliary diagnosis of colon cancer, and have great value for early diagnosis of colon cancer. 5 Recently, Attallah et al. 6 pointed out that the sensitivity of CEA and CA19-9 alone in the differential diagnosis of colon cancer and benign colon diseases and healthy people was 54.2% and 64.2%, and the specificity was 68.7% and 70.6%, respectively. Chen et al. 11 detected the serum CEA and CA19-9 concentrations in 60 patients with colon cancer and found that the combined diagnostic sensitivity of CEA and CA19-9 was only 71.7%. These results suggest that the specificity of serum CEA and CA19-9 in the differential diagnosis of colon cancer is low. Both CEA and CA19-9 are broad spectrum tumour markers, and both may be elevated in a variety of benign diseases such as enteritis, gastritis, hepatitis, etc., which is an important reason for the low specificity of the two in colon cancer diagnosis.
Recent studies have found that AQPs protein family exists in colon tissue. It is noteworthy that in the AQP protein family, AQP1 and AQP3 have been confirmed to participate in tumour cell migration and proliferation in multiple tumours. 12 Saadoun et al. 12 found that after subcutaneous injection of colon cancer cells, mice lacking AQP1 showed reduced tumour growth due to impaired neovascularization in the tumour. Compared with AQP +/+ endothelial cells, AQP–/– endothelial cells have a lower migration activity to chemotaxis. The above studies suggest that the expression of AQP1 and AQP3 in the colon plays an important role in the biological progress of colon cancer, especially in tumour metastasis and invasion, and thus affects the prognosis of patients. In recent years, there have been many reports on the relationship between galectin-3 and cancer.7,13,14 Liu et al. 13 found that galectin-3 overexpression in breast cancer cell lines can inhibit tumour cell apoptosis through the mitochondrial pathway and considered galectin-3 as a new biomarker for breast cancer. Lei et al. 14 pointed out that microRNA-107 can inhibit the post-transcriptional translation of galectin-3 by targeting galectin-3 gene expression to inhibit the proliferation, invasion and metastasis of osteosarcoma cells. The above studies suggest that the differential expression of galectin-3 is involved in the occurrence and development of tumours by interfering with various biological behaviours. Previous studies on the association between galectin-3 and colon cancer have also been reported. 15 Wang et al. 15 pointed out that galectin-3 gene expression was related to the clinical stage and differentiation of colon cancer patients, and with the increase of clinical stage and differentiation of colon cancer patients, the expression of galectin-3 gene in serum increased significantly. The above studies suggest that galectin-3, AQP-1 and AQP-3 are involved in the process of colon cancer. However, the analysis of serum galectin-3, AQP-1 and AQP-3 protein expression levels and prognosis of patients with colon cancer are not clear.
In this study, we tested the preoperative and postoperative serum galectin-3, AQP-1 and AQP-3 concentrations in young colon cancer patients, and explored the value of galectin-3, AQP-1 and AQP-3 as markers for diagnosis and prognosis of colon cancer in young people. Previous studies found that the expression level of galectin-3 in colon cancer tissues was significantly higher than that in adjacent tissues by using immunohistochemical techniques. 16 In this study, we detected high expression of galectin-3 in the serum of patients with colon cancer, echoing previous clinical studies. According to the above experiments, we speculated that the increase in galectin-3, AQP-1 and AQP-3 concentrations in the serum of patients with colon cancer may be caused by the release of galectin-3, AQP-1 and AQP-3 protein from colon cancer tissues into peripheral blood. In addition, we also found that serum galectin-3, AQP-1 and AQP-3 concentrations were linearly related to the stage and differentiation of young colon cancer patients, and galectin-3, AQP-1 and AQP-3 concentrations were most significantly increased in patients with advanced colon cancer and poorly differentiated colon cancer. These series of results suggest that the middle and advanced malignant tumours and less differentiated cancer cells will release more galectin-3, AQP-1 and AQP-3 into the peripheral blood, indicating that galectin-3, AQP-1 and AQP-3 concentrations in peripheral blood are closely related to the poor prognosis of colon cancer. The above results are basically consistent with the results of previous studies on the interference of galectin-3, AQP-1 and AQP-3 on tumour biological behaviour. ROC curve analysis showed that when galectin-3, AQP-1 and AQP-3 were combined, the specificity of distinguishing colon cancer from benign lesions and controls was 98.2%. It is suggested that the combination of the above three serum markers has higher clinical application value. Survival analysis found that patients with high galectin-3, high AQP-1 and high AQP-3 had significantly shorter survival times than patients with low galectin-3, low AQP-1 and low AQP-3. It is suggested that postoperative serum galectin-3, AQP-1 and AQP-3 concentrations of patients with colon cancer have potential value in assessing the prognosis of patients. However, this study has the following shortcomings: (1) Due to the limited number of included study subjects, the reference interval of galectin-3, AQP-1 and AQP-3 in young colon cancer patients needs to be further clarified. (2) The combined model of galectin-3, AQP-1 and AQP-3 is still to be verified by a larger sample and a multicentre cohort from the clinical application. (3) The potential mechanism of galectin-3, AQP-1 and AQP-3 in colon cancer needs to be clarified. (4) Due to the limited number of cases included, the correlation between traditional risk factors for colon cancer such as staying up late, smoking history, drinking history, low fibre diet, high fat and high protein diet, history of nitrite intake, physical activity with galectin-3, AQP-1 and AQP-3 needs to be further clarified.
In summary, we found that the combined detection of serum galectin-3, AQP-1 and AQP-3 has potential value for the diagnosis of young patients with colon cancer and confirmed that galectin-3, AQP-1 and AQP-3 are important markers for the prognostic evaluation of young patients with colon cancer.
Supplemental Material
sj-pdf-1-acb-10.1177_0004563220961752 - Supplemental material for Prognostic value of serum aquaporin-1, aquaporin-3 and galectin-3 for young patients with colon cancer
Supplemental material, sj-pdf-1-acb-10.1177_0004563220961752 for Prognostic value of serum aquaporin-1, aquaporin-3 and galectin-3 for young patients with colon cancer by Yanyan Hong, Zhendong Chen, Na Li and Mingjun Zhang in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to thank all laboratory members for their critical discussion of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Clinical Medical Research Ethics Committee of the Second Affiliated Hospital of Anhui Medical University (Approval number: 2015317). Written informed consent was provided in accordance with the Declaration of Helsinki.
Guarantor
ZC.
Contributorship
YH and ZC conceived this study. YH, NL and MZ were involved in gaining ethical approval, patient recruitment and data analysis. YH and ZC wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
