Abstract
Background
Plasma viscosity is an important biomarker both in diagnostics and treatment monitoring of plasma cell dyscrasias and other disorders with hyperviscosity syndrome as a clinical manifestation. Here, we investigate the performance of a new microfluidic-based viscometer for clinical use, establish a new reference range to be used with this instrument and determine the importance of sampling temperature.
Methods
The microVisc™ viscometer was evaluated for within-run and between-run imprecision and bias using standardized reference material (Paragon controls) and Seronorm™ control material. The reference range was established for the adult population using EDTA-plasma from 120 healthy blood donors. Sampling temperature was investigated by drawing and transporting blood at room temperature and 37°C and comparing the viscosity between the two sampling methods.
Results
The microfluidic-based viscometer performed well, and imprecision was comparable to ReoRox® G2 free oscillation rheometer. A new reference range for the adult Danish population was established as 1.2–1.5 mPa s at 37°C. Furthermore, sampling temperature at room temperature and 37°C was investigated, and there was no difference in results obtained.
Conclusions
MicroVisc™ is suitable for measuring plasma viscosity in a clinical setting and results can be evaluated using the established reference range. Blood sampling for viscosity analysis can be performed as a standard procedure at room temperature.
Introduction
Hyperviscosity syndrome is a range of clinical manifestations that can occur in patients with, e.g. multiple myeloma (MM), leukaemia, rheumatoid arthritis (RA) or Waldenström macroglobulinemia (WM). Patients with one of these disorders might present with increased viscosity of plasma, due to the presence of monoclonal paraproteins (MM, WM) or polyclonal increase in gamma globulins (chronic lymphocytic leukaemia or RA). 1 The protein concentration is a direct determinant of viscosity 2 and appearance of clinical symptoms, but the biochemical properties of the proteins also play a role, e.g. protein structure or ability to aggregate with self or other proteins. Due to the ability of IgM molecules to form pentameric structures and unspecifically bind other proteins, plasma viscosity is greater at any given protein concentration than for IgA and IgG paraproteinaemia. 2 Since hyperviscosity syndrome does not manifest at a specific protein concentration that being, e.g. a monoclonal component or polyclonal gammaglobulins and thus cannot be directly determined based on protein concentration,1,3 it is important both in diagnostic workup as well as the management of these patients that plasma viscosity can be measured reliably and quickly in a clinical setting. Several different types of viscometers have been in clinical use over the years, 4 with one of the latest measuring methods being the free oscillation rheometer ReoRox® G2. However, this viscometer is no longer technically supported and can no longer reliably be used for clinical routine measurements. We therefore chose to test a completely new viscometer utilizing microfluidic chip technology for clinical measurements of plasma viscosity. The microVisc™ instrument measures viscosity as the drop in pressure over a given distance that occurs when the plasma is forced through a rectangular flow channel that is made of glass, has a depth of 50 µm and several pressure sensors positioned along the flow channel. Pressure drop is a linear function of position and the slope generated from the graph is used together with width and depth of the flow channel in calculation of shear stress. Shear rate is calculated from flow rate, width and depth of the flow channel, and plasma viscosity is calculated as shear stress divided by shear rate. 5 The measuring range of the instrument is 0.2–100 mPa s, thus allowing the measurement of highly viscous samples. This measuring method is very different from our previous instrument that used free oscillation rheometry. In this short report, we investigate the clinical use of the microfluidic viscometer microVisc™ from RheoSense, Inc. and establish a new reference interval to be used with the microVisc™ method. Furthermore, we assess whether sampling temperature is a critical preanalytical factor that impacts on the results.
Methods
Plasma from healthy donors
EDTA-plasma from 120 Danish healthy blood donors was obtained from the Department of Clinical Immunology, Odense University Hospital. The donors were ≥18 years of age and of both genders. Blood donors in Denmark are selected according to the Transfusion Medical Standard from the Danish Society for Clinical Immunology. 6 The donors gave their informed consent, and all samples were used fully anonymized. Since in our current study the reference interval is not partitioned based on age and gender, the blood donor samples for the new reference interval were used without knowledge of the age or gender of the individual blood donor. EDTA-plasma from donors was run in triplicate on the microVisc™ viscometer (Rheosense Inc., Biolab A/S, Risskov, Denmark), and the mean value from each participant was used for establishment of the reference range.
Bias and imprecision
Imprecision and bias was established using the standardized glycerol solutions MGVS20-100 (target value: 1.468 mPa s), MGVS40-100 (target value: 2.769 mPa s) and MGVS100-100 (target value: 6.337 mPa s) (Paragon Scientific, Ltd, Prenton, UK). Imprecision was further assessed using the Seronorm™ Human control (#200805, SERO, Billingstad, Norway). For within-run imprecision, samples were analysed consecutively in a single day. For between-run imprecision and bias samples were analysed on different days by different operators.
Sampling procedure and temperature
EDTA-plasma samples were obtained using BD Vacutainer® tubes with k2-EDTA (7.2 mg/4.0 mL) (REF 368861, BD Biosciences, USA), transported to the laboratory and centrifuged at 2000 × g for 10 min. Phlebotomy is routinely performed at room temperature. However, for viscosity measurements, the procedure has been performed at 37°C with prewarmed utensils and transported to the laboratory in a heated container. This is to maintain the physiological condition of the blood until measurement. To investigate the necessity for this elaborate sampling procedure, we obtained two EDTA-plasma samples during routine sampling (n = 5) for viscosity such that one was sampled and transported at 37°C and the other using the standard procedure at room temperature. The samples were anonymized and both were measured at 37°C in the microVisc™ instrument. The 120 healthy blood donor samples were obtained according to standard procedure at room temperature.
Statistical analysis
All data obtained were analysed in Microsoft Excel 2010 for Windows (Microsoft Corp., Redmond, Washington USA) or GraphPad Prism version 8.3.0 for Windows (GraphPad Software, San Diego, California, USA). The distribution of the healthy donor samples as well as establishment of the reference range was performed using Analyse-it® version 4.92.4 for Excel (Analyse-it Software, Ltd, Leeds, UK). The Anderson-Darling test was performed to test for normality distribution. Paired t-test was performed to test the significance of sampling temperature. A P-value <0.05 was considered statistically significant.
Results
Imprecision
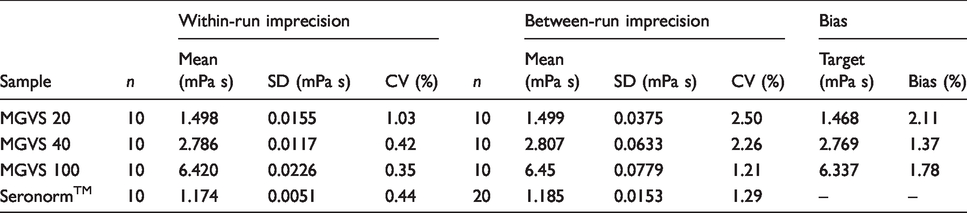
Imprecision was established within-run and between-run by analysing the three Paragon glycerol control materials and Seronorm™ control material. For between-run analysis, Seronorm™ was analysed on 20 consecutive days, whereas the Paragon controls were evaluated over 10 days during the same period. All measurements were performed in triplicate. For within-run analysis, Seronorm™ and Paragon controls were analysed as 10 replicates on the same day. Within-run imprecision ranged from 0.35 to 1.03% and between-run imprecision from 1.21 to 2.50% (Table 1).
Between-run imprecision, within-run imprecision and bias.
Bias
Bias was established using the same Paragon standardized glycerol controls as above. Mean value from 10 runs was used to calculate bias based on target value of the Paragon controls. Bias ranged from 1.37 to 2.11% (Table 1).
Sampling temperature
Standard sampling at room temperature versus sampling at 37°C was evaluated and showed a minimal bias of –0.2% for sampling at 37°C with no statistically significant difference between the two sampling methods (paired t-test, P = 0.62, n = 5).
Reference range
The Anderson-Darling normality test showed that plasma viscosity measurements in healthy blood donors were normally distributed (P = 0.16; skewness =–0.2). The reference range was established as 1.2–1.5 mPa s, corresponding to the 2.5 and 97.5 percentiles (%) of the normal quantile range. The reference range was calculated using Analyse-it® for Excel. The median of the distribution was 1.3 mPa s. The reference range was established according to the C28-A3c guideline from Clinical and Laboratory Standards Institute.
Discussion
Plasma viscosity is still a useful biomarker in the diagnosis and monitoring of hyperviscosity syndrome. 7 It is therefore important that we maintain a high-quality method, so we continuously can provide clinically useful results. We have previously reported that the free oscillation rheometer ReoRox® G2 performed well in accordance with the ICSH recommendations. 8 However, this instrument has been discontinued and a suitable replacement must be implemented. For this purpose, we have validated the new microfluidic-based instrument microVisc™ for clinical use with patient plasma samples. Our validation shows that microVisc™ can satisfactorily be used on EDTA-plasma samples with a CV ≤2.50% and a bias ≤2.11%. Moreover, the instrument is very simple to operate, uses a small sample volume (400 µL in the syringe, 55 µL for measuring) and is easily maintained. Disposable utensils (syringe) minimizes the risk of carry-over issues; however, this instrument needs cleaning with sodium hypochlorite (<5%) followed by 3×MilliQ water following a highly viscous sample before continuing measurements. Furthermore, every time the instrument has been in use this cleaning procedure should be performed following the final measurement. This is necessary to prevent clotting and residue build-up in the flow channel. The cleaning procedure is checked by running a water sample. If the water sample viscosity is too high according to the measuring range determined for water, the cleaning procedure is repeated. We have observed that CV% increases if residue is allowed to build-up (data not shown). The imprecision of the microVisc™ instrument is lower than our previous ReoRox® G2 (between-run CV%: 1.9–3.2). 8 The turnaround time of the samples is also within a similar time range, but due to the more time-consuming cleaning procedure, the total technician time spent per analysis is increased with the microVisc™ instrument. However, this is fully acceptable, considering the superior stability and precision of the instrument. We therefore conclude that the performance of the microVisc™ viscometer is acceptable according to the ICSH guidelines. 9
Historically, blood sampling for viscosity measurements has been performed at 37°C with prewarmed utensils and phlebotomy equipment and transported at 37°C to the laboratory. In this study, we assessed whether this elaborate procedure is necessary, or whether standard blood sampling at room temperature is adequate. We conclude that sampling temperature in this setup has a negligible impact on the results with an overall bias of –0.2%, when sampling at 37°C compared with room temperature. We therefore recommend that standard phlebotomy procedure is performed for viscosity; however, the actual measurement of plasma viscosity still needs to be performed at 37°C with prewarmed utensils. For the microVisc™ instrument, this is done by keeping the instrument in a heating box that maintains instrument temperature at 37°C. All utensils as well as samples are preheated prior to measurement. In addition, freezing of the samples must be avoided, since this results in particle formation, which obscures viscosity measurement (data not shown).
Due to the different measuring principles of microVisc™ and ReoRox® G2, we chose to establish a new reference range for the microVisc™ analysis and with standard phlebotomy procedure at room temperature. This was done according to the CLSI guideline C28-A3c using plasma from 120 healthy blood donors. We established a range from 1.2 to 1.5 mPa s with a median of 1.3 mPa s, whereas our previous reference range was 1.24–1.44 mPa s with a median of 1.32 mPa s. The difference between the reference ranges is very small, and it could be argued that the ReoRox® G2 reference range could have been transferred. However, in our view, it is important to establish a new, updated reference range when the method changes radically as in this case. It should be noted that the reference range is for adults from 18 years of age, not gender specific and may not be applicable to children.
Footnotes
Acknowledgements
We thank Lone McManus, Birgit Pedersen and Christian Munck, Department of Clinical Biochemistry and Pharmacology, Odense University Hospital, for technical assistance. We thank the Department of Clinical Immunology, Odense University Hospital for providing the blood donor samples.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was conducted as a quality assurance project and did not require ethical approval as advised by The Regional Committee on Health Research Ethics for Southern (20192000–11).
Guarantor
LHJ.
Contributorship
LHJ analysed data and wrote the article, LHJ, VSM and JR conceived the study and developed the assay; LHJ and JR obtained donor samples and ethical guidance. All authors read, edited and approved the article.
