Abstract
Background
The current study aimed to explore the value of cytotoxic T-lymphocyte-associated protein 4 (CTLA4)-associated microRNAs combined with inflammatory factors in the differential diagnosis of non-small cell lung cancer (NSCLC).
Methods
A retrospective study including 245 NSCLC patients and 245 healthy controls was conducted on a testing group. A regression formula for NSCLC prediction was established based on the testing group. Two validation groups from two centres were used to assess the novel logistic regression model including 144 NSCLC patients and 144 healthy controls recruited from the Wuchang Hospital Affiliated to Wuhan University of Science and Technology, and 128 NSCLC patients and 128 healthy controls recruited from the Zhongnan Hospital of Wuhan University.
Results
Predictive software and dual-luciferase reporter assays showed that miR-155-5p and miR-630 could target CTLA4 expression. The miR-155-5p and miR-630 concentrations in the NSCLC patients were significantly lower, and the neutrophil to lymphocyte ratio, hypersensitive C-reactive protein (hs-CRP), interleukin 6, cytokeratin-19-fragment (CYFRA21-1), squamous cell carcinoma antigen (SCCA) concentrations and the smoking rate were significantly higher than that in healthy controls (P < 0.05). A logistic regression model that included smoking, neutrophil to lymphocyte ratio, hs-CRP, interleukin 6, CYFRA21-1, SCCA, miR-155-5p and miR-630 was performed. This model presented a high discriminating value (AUC: 0.830, sensitivity/specificity: 74.6%/89.7%) than any single indicator. In the validation groups, this model still showed a high discriminating value (AUC = 0.838 with the internal validation group; AUC = 0.851 with the external validation group).
Conclusion
The current model has potential significance for the non-invasive diagnosis for NSCLC.
Keywords
Introduction
Lung cancer is the leading cause of cancer-related deaths worldwide. According to the Global Cancer Statistics report, 2,093,876 new lung cancers were diagnosed in 2018, and 1,761,007 deaths were due to lung cancer, accounting for 11.6% of all new cancers. 1 Non-small cell lung cancer (NSCLC) is the main type of lung cancer, accounting for about 85% of all lung cancers. 2 Although the treatment strategies of NSCLC have made some progress, the prognosis is still very poor, and the five-year survival rate is only about 15.6%. 3 Early diagnosis is the key to achieve satisfactory clinical outcomes.
In recent years, with the deepening of the understanding of the antitumour effect of the immune system, researchers have found that after identifying the tumour-associated antigen, the body could induce the activation, proliferation and migration of effector cells such as cytotoxic T cells, to kill the tumour cells.4,5 In contrast, tumour cells have evolved multiple mechanisms to reduce this killing effect from the immune system through immune escape. 5 The activation of T cells is critical for the antitumour effect of immune system. Activation of T cells is regulated by the first and second signals. 6 The first signal is derived from the recognition of antigen by T cells. The tumour-specific antigen is recognized by the major histocompatibility complex on the surface of the antigen-presenting cell and presented to the T-cell receptor on the T cell surface, producing the first signal. The second signal is produced by the interaction of the co-stimulatory molecule CD28 on the surface of the T cell with the B7 ligand on the surface of the antigen-presenting cells. After the activation of T cells, the expression of synergistic inhibitory molecules appears. The synergistic inhibitory molecules and the synergistic promoting molecules compete with each other. By combining with the ligands, respectively, the stimulation signal and the inhibition signal are generated. The two signals are activated at the same time and are in a subtle dynamic equilibrium state, so that the immune system implements the antitumour effect without being over-activated to avoid the accidental injury of normal tissues. In this dynamic regulation process, cytotoxic T lymphocyte-associated antigen 4 (CTLA4) is one of the important synergistic inhibitory molecules.7,8 Tumour cells inhibit the activation of T cells by promoting CTLA4 expression, achieving the purpose of immune escape and avoid the killing of the immune system. Therefore, CTLA4 is an important target for antitumour immunotherapy drugs.
MicroRNA (miR), a 22-nucleotide endogenous small non-coding RNA molecule, regulates cell behaviour and modulates gene expression at the post-transcriptional level by binding to mRNA and suppressing translation. 9 Aberrant gene expression patterns resulting from aberrant expression profiles of miRs are associated with numerous pathologies and diseases including cancer. 10 In the current study, firstly, we identified and validated miRs that are targeted for down-regulating CTLA4 expression through the luciferase reporter gene assay. Secondly, we designed a survey and evaluated the significance of CTLA4-associated miRs combined with inflammatory factors in distinguishing NSCLC from healthy controls. Corresponding receiver operating characteristic (ROC) curves were also generated to evaluate the diagnostic potentials. Finally, a logistic regression prediction model was built for NSCLC based on the testing group and was validated by internal and external validation groups, respectively.
Materials and methods
Inclusion and exclusion criteria
In the testing and internal validation groups, we analysed data from patients with NSCLC at the Department of Clinical Laboratory, Wuchang Hospital Affiliated to Wuhan University of Science and Technology between June 2016 and May 2019. In the external validation group, we analysed data from patients with NSCLC from the Zhongnan Hospital of Wuhan University between July 2018 and May 2019. The inclusion criteria for subjects are: (1) pathological diagnosis of NSCLC; (2) subject who has provided informed consent; (3) the general condition is good, no serious heart, liver, kidney and other complications; (4) the patient was conscious and had no history of mental illness. Subjects with any of the followings were excluded: (1) history of other cancer diseases; (2) history of radiotherapy and chemotherapy treatment; (3) not willing to participate in the research; (4) patient's clinical pathology data are incomplete. In the testing, internal and external validation groups, the healthy controls matched the NSCLC patients in terms of age and gender.
Testing group, internal and external validation groups
A total of 524 patients, who were suspected of having NSCLC, were chosen in this study. Among them, 517 patients were defined as NSCLC according to microscopic or macroscopic tissue examination. The diagnosis of NSCLC was based on the histological result. Testing group: 245 patients with NSCLC and 245 healthy controls were included. Internal validation group: 144 NSCLC patients and 144 healthy controls were included. External validation group: 128 NSCLC patients and 128 healthy controls were included. Our study was approved by the Ethics Committee of the Wuchang Hospital Affiliated to Wuhan University of Science and Technology, Wuhan, China (20160117) and the Ethics Committee of the Zhongnan Hospital of Wuhan University, Wuhan, China (2018–019). Written informed consent was provided in accordance with the ethical principles of the Declaration of Helsinki.
Logistic regression model establishment and validation
A regression formula for NSCLC prediction was established based on the testing group. The formula is: Logit (P) = A0 + A1B1 + A2B2 + A3B3 + ⋯ + AnBn = ln[p/(1−p)], ‘p’ means the incident probability (NSCLC), ‘n’ means the number of interference variables, ‘A’ means the influence coefficient of each interference variable, ‘B’ means the value of each interference variable. The internal and external validation groups were used to assess the above logistic regression model.
Collection of clinicopathological and life history variables
The clinicopathological data, including age, sex, hypertension, diabetes, histologic type, differentiation status, metastatic status, size of tumours were collected. Life history variables, including smoking, drinking, were also collected.
Blood and tissue specimen collection and biomarkers detection
Whole blood (EDTA-K2 anticoagulation tube) and serum (coagulant separation gel tube) samples from NSCLC patients were separately collected before being admitted to hospital for treatment. Biomarkers tested in this study include inflammatory factors (neutrophil to lymphocyte ratio [NLR], monocytes to lymphocyte ratio [MLR], hypersensitive C-reactive protein [hs-CRP], interleukin 6 [IL-6]) and tumour markers (cytokeratin-19-fragment [CYFRA21-1], squamous cell carcinoma antigen [SCCA]). NLR and MLR were tested by BC-5000 blood cell analyser (Mindray, China). hs-CRP was tested by AU5400 (OLYMPUS, Japan). IL-6, CYFRA21-1 and SCCA were tested by E411 (Cobas, Switzerland). In addition, four pairs of cancer and adjacent tissues (>3 cm from the edge of cancer tissue) from NSCLC patients who underwent surgical treatment were enrolled.
Quantitative real-time polymerase chain reaction
This study complies with the Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE) guidelines 2009 (Supplementary Table 1). 11 MiRs concentrations were detected by quantitative real-time polymerase chain reaction (qRT-PCR). The total RNAs were reverse transcribed to cDNA using PrimeScript™ RT reagent Kit with gDNA Eraser (Takara, Japan, Lot number: RR064C), following: 42°C for 2 min, and then 37°C for 15 min, 85°C for 5 s. The quality of extracted RNAs was tested by Nanodrop ND 8000 (Invitrogen, Thermo, New York, USA). The primers sequences were as follows: miR-155-5p (forward: 5′-CGGCTCGGATCCGTTAG-3′ and reverse: 5′-CGACTACCGTTGAGCTAGA-3′); miR-630 (forward: 5′-CCGTCAACAGCTAGTCAGCAG-3′ and reverse: 5′-GGCACCAGTACTAGACA-3′); U6 (forward: 5′-CGCTCCAGTCCGACGATCAC-3′ and reverse: 5′-AAGGCTAGGTCCAAGTACT-3′). The total reaction volume was 20 μL, which contained 2 μL cDNA, 10 μL SYBR-Green Supermix, 1.3 μL gene-specific forward and reverse primers and 6.4 μL nuclease-free water. The specificity of the amplified products was detected by melting curve analysis and DNA sequencing. Relative expression was calculated by 2−△Ct.
Cell culture and transfection
H1975 cells (Institute of Biosciences Cell Resource Center, Chinese Academy of Sciences, Shanghai, Lot number: CASS1812023) and human normal lung epithelial cell line BEAS-2B were grown in RPMI-1640 medium containing 10% fetal bovine serum at 37°C, 5% CO2. Each miR-mimic or miR-control was transfected into H1975 cells using Lipofectamine 2000. Si-CTLA4 and si-NC were also transfected to cells using Lipofectamine 2000. After transfection for 48 h, the target miR and protein were detected.
Western blot analysis
Proteins from tissues were separated by SDS-PAGE. The membrane was incubated overnight with one of the following antibodies: rabbit monoclonal CTLA4 (1:2000; Cell Signaling Technology, USA, Lot number: 5616288–7) and rabbit monoclonal β-actin (1:3000; Cell Signaling Technology, USA, Lot number: 153156–01). Then, the membrane was incubated with a secondary antirabbit antibody (1:4000; Affinity, USA, Lot number: 0912764–15) for 1 h. Finally, the membrane was visualized by ECL-PLU (Amersham Biosciences, Sweden).
Immunohistochemical staining
Cells were blocked with goat serum for 1 h at room temperature and incubated with CTLA4 monoclonal antibody (rabbit monoclonal, 1:2000; Cell Signaling Technology, USA, Lot number: XY15638-2) at 4°C overnight, then incubated with alkaline phosphatase secondary antibody (rabbit monoclonal, 1:1000; Cell Signaling Technology, USA, Lot number: XY-37831) for 1 h. Immunohistochemical staining score was calculated according to the staining intensity of positive cells. The formula was X × Y. X represents the percentage of positive cells: x = 0, no positive cells; x = 1, positive cells are 1%–10%; x = 2, positive cells are 11%–50%; x = 3, positive cells are 51%–80%; and x = 4 positive cells account for more than 81%. Y represents positive cell staining intensity: y = 0, negative; y = 1, weakly positive; y = 2, moderately positive; y = 3, strong positive.
Luciferase reporter gene assay
Wide type (WT)-CTLA4 3′UTR-WT luciferase reporters and mutant type (Mu)-CTLA4 3′UTR-Mu luciferase reporters were co-transfected with pRL-SV40 (Invitrogen, New York, USA) and corresponding miR-mimic/miR-control into H1975 cells, and luciferase activity was determined 48 h later.
Statistical analysis
All statistical analyses were performed using SPSS version 19.0 (SPSS, Chicago, IL). Data were presented as the mean ± standard deviation (SD, normally distributed numeric variables), or median (interquartile range [IQR], non-normally distributed variables), or number of cases (%, counting data). Difference among normally distributed data was evaluated by t-test; otherwise, Mann-Whitney U-test was used. Univariate and multivariate models were performed to investigate the relation of various variables with NSCLC in testing group. P < 0.05 was considered significant.
Results
Inclusion of subjects and research process
From June 2016 to May 2019, 1324 eligible subjects were identified at the Wuchang Hospital Affiliated to Wuhan University of Science and Technology and Zhongnan Hospital of Wuhan University. After exclusion of subjects who did not meet the inclusion criteria, 1034 subjects were eventually included in the final analysis. The research process is presented in Figure 1.
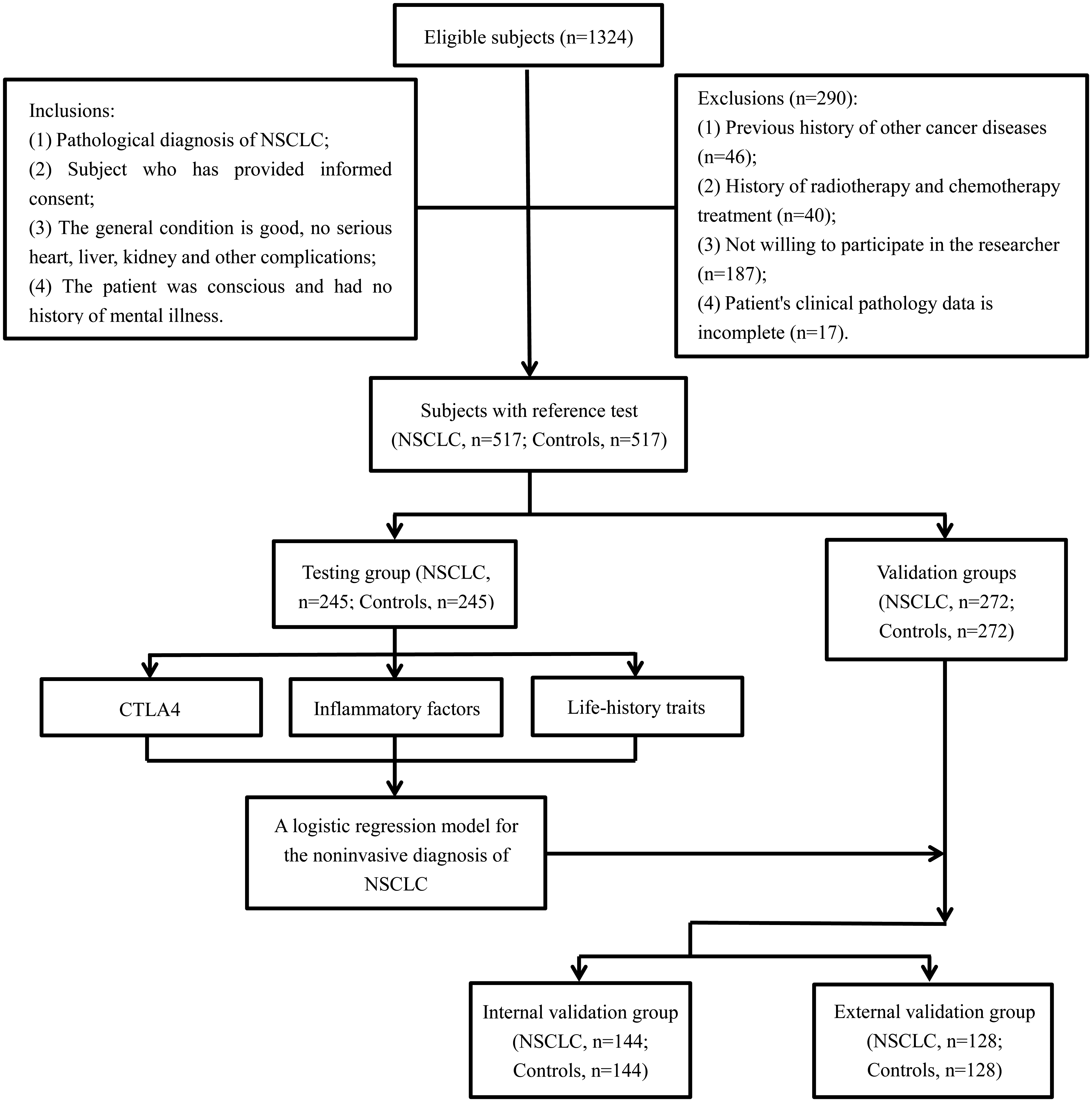
Flow diagram showing the inclusion and exclusion of eligible subjects.
Expression of CTLA4 protein in NSCLC patients and the prediction and validation of miRs regulating CTLA4 expression
The results of immunohistochemical staining are shown in Figure 2(a) to (c). Compared with adjacent tissues (7.2 ± 2.4, Figure 2(b)), the expression concentration of CTLA4 in cancer tissues (26.8 ± 7.9, Figure 2(c)) increased significantly (P < 0.05). The immunohistochemical result of using PBS instead of primary antibody as a negative control is shown in Figure 1(a). The validation of the anti-CTLA4 antibody is shown in Figure 2(d). Our results showed that the anti-CTLA4 antibody does have cross-reaction with other proteins. Results of Western blot analysis of four pairs of NSCLC patients are shown in Figure 2(e). Compared with adjacent tissues (1.6 ± 0.5), CTLA4 protein concentrations in cancer tissues (5.8 ± 1.1) were significantly increased (P < 0.05).
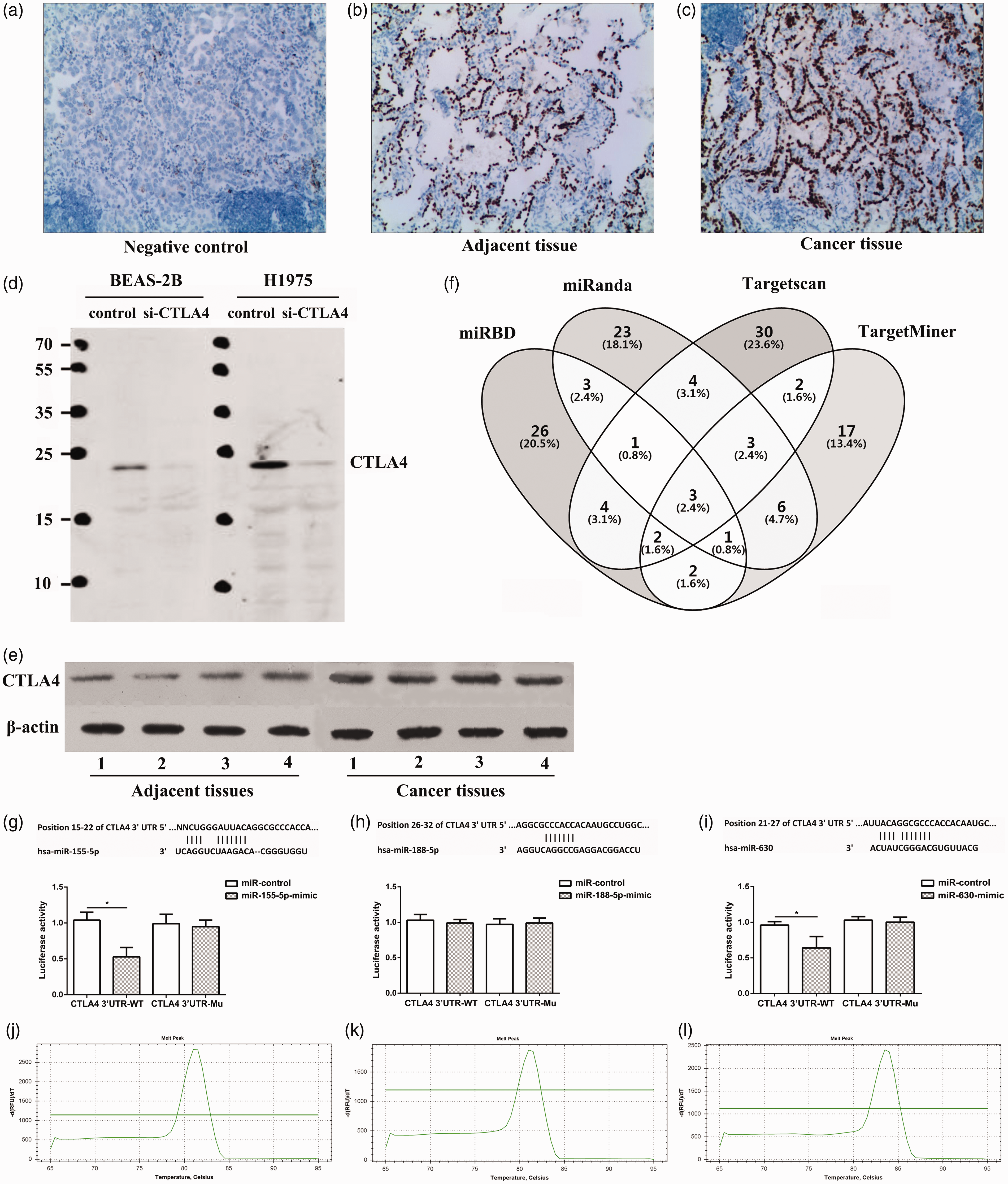
Expression of CTLA4 protein in NSCLC patients and prediction and validation of miRs regulating CTLA4 expression. (a to c): Detection of protein concentrations in tissues of NSCLC patients by immunohistochemistry: (a) negative control; (b): adjacent tissue; (c): cancer tissue; (d and e) detection of protein concentrations in tissues of NSCLC patients by Western blot: (d): the validation of the anti-CTLA4 antibody; (e): results of Western blot analysis of four pairs of NSCLC patients; (f) Targetscan, miRanda, miRDB and TangetMiner software to predict the miRs targeting CTLA4; (g to i): luciferase reporter gene assay: (g) miR-155-5p; (h): miR-188-5p; (i): miR-630; (j): the melting peak of miR-363-5p; (k): the melting peak of miR-188-5p; (l): the melting peak of miR-630. *P < 0.05.
Using Targetscan, miRanda, miRDB and TangetMiner to predict the miRs targeting CTLA4, a total of three miRs (miR-155-5p, miR-188-5p and miR-630) were simultaneously predicted, which may be involved in the targeted regulation of CTLA4 (Figure 2(f)). The luciferase reporter gene assay showed that the luciferase activities in miR-155-5p mimic group (0.51 ± 0.09) and the miR-630 mimic group (0.63 ± 0.13) were significantly lower than that in the negative control group (0.90 ± 0.08, 0.93 ± 0.04) of the wild-type CTLA4 (P < 0.05), but there was no significant difference in the mutant CTLA4 (P > 0.05, Figure 2(g) to (i)). The melting peaks of miR-155-5p, miR-188-5p and miR-630 were single, indicating that there was no non-specific amplification (Figure 2(j) to (l)).
Patient characteristics in the testing group
There were 490 subjects in the testing cohort. The subjects’ clinicopathological characteristics, inflammatory factors, tumour markers and life history traits are presented in Table 1. There was no significant difference in age, sex, hypertension, diabetes, drinking and MLR between the NSCLC patients and healthy controls (P > 0.05). However, the miR-155-5p and miR-630 concentrations in the NSCLC patients were significantly lower, and the NLR, hs-CRP, IL-6, CYFRA21-1, SCCA concentrations and the smoking rate were significantly higher than the healthy controls (P < 0.05, Table 1).
Clinicopathological characteristics, inflammatory factors, tumour markers and life history traits in the testing group.
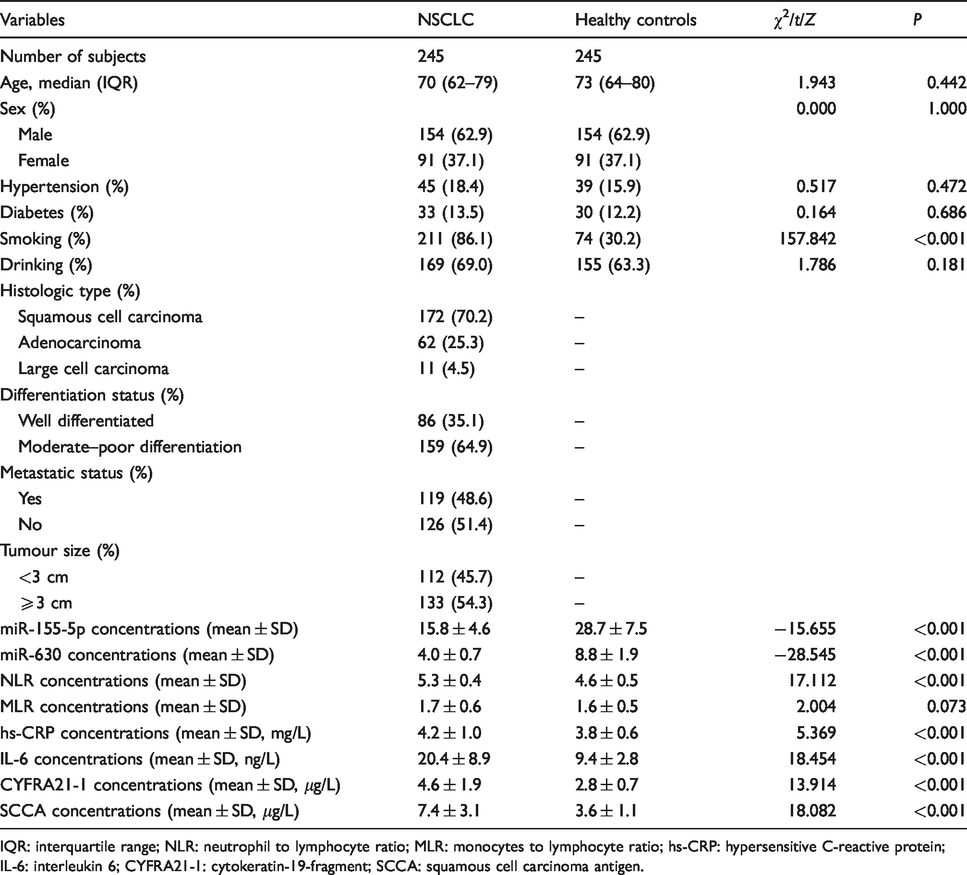
IQR: interquartile range; NLR: neutrophil to lymphocyte ratio; MLR: monocytes to lymphocyte ratio; hs-CRP: hypersensitive C-reactive protein; IL-6: interleukin 6; CYFRA21-1: cytokeratin-19-fragment; SCCA: squamous cell carcinoma antigen.
A novel logistic regression model predicting NSCLC in the testing group
Results of logistic regression analyses are presented in Table 2. Smoking, NLR, hs-CRP, IL-6, CYFRA21-1, SCCA, miR-155-5p and miR-630 had significant influences on NSCLC (all P < 0.05). Results of ROC analyses showed that the area under curve (AUC) of NLR, hs-CRP, IL-6, CYFRA21-1, SCCA, smoking, miR-155-5p and miR-630 were 0.694, 0.644, 0.560, 0.803, 0.681, 0.582, 0.798 and 0.808, respectively (Figure 3 and Table 3).
Univariate and multivariate logistic regression analyses for NSCLC.
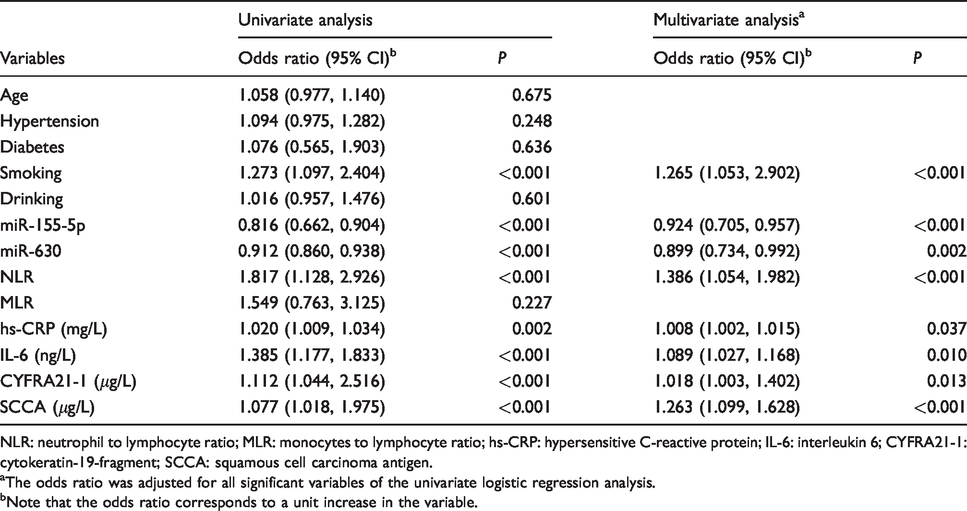
NLR: neutrophil to lymphocyte ratio; MLR: monocytes to lymphocyte ratio; hs-CRP: hypersensitive C-reactive protein; IL-6: interleukin 6; CYFRA21-1: cytokeratin-19-fragment; SCCA: squamous cell carcinoma antigen.
aThe odds ratio was adjusted for all significant variables of the univariate logistic regression analysis.
bNote that the odds ratio corresponds to a unit increase in the variable.
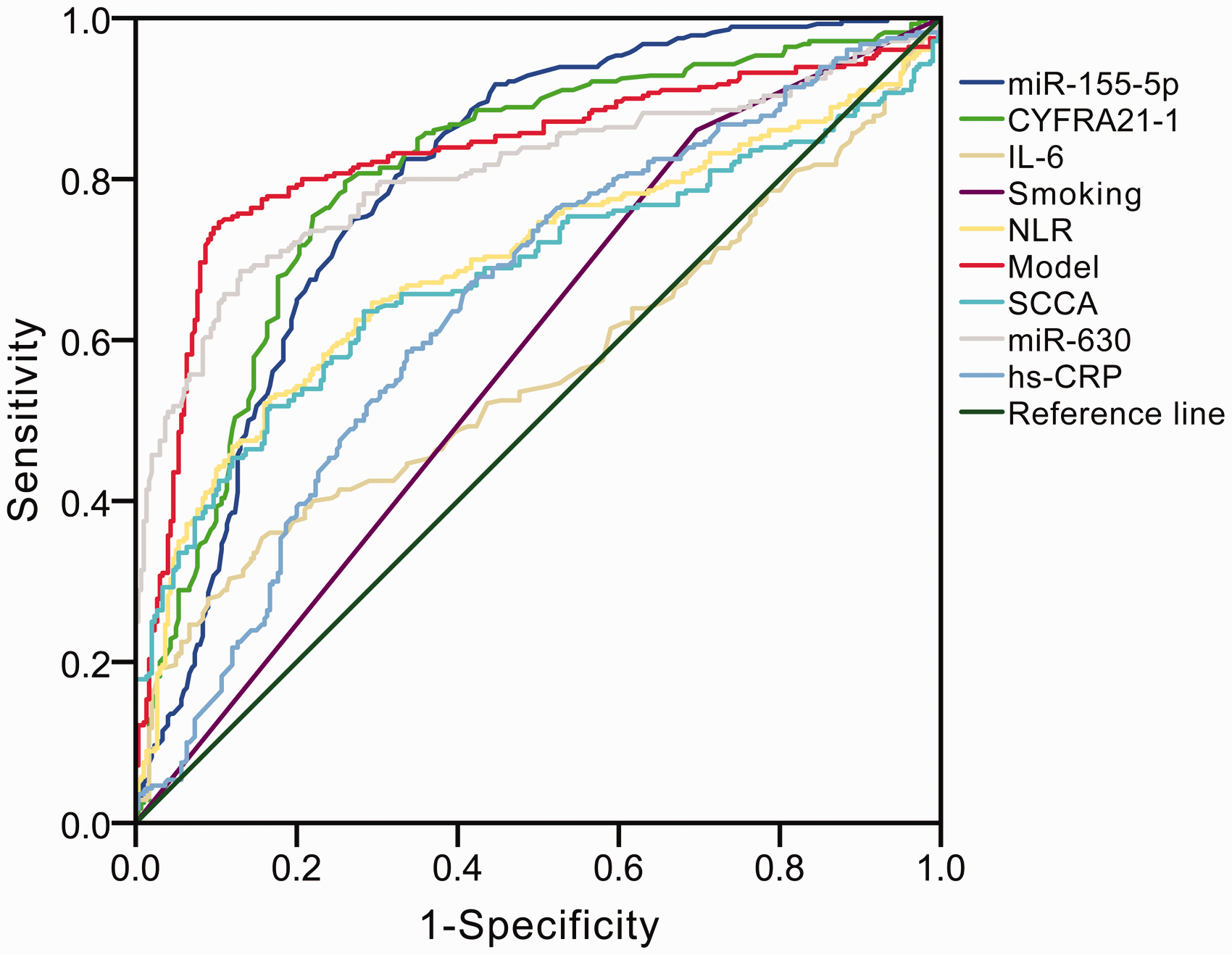
Receiver operating characteristic (ROC) curve analysis for the differential diagnosis values in the eight independent factors (neutrophil to lymphocyte ratio [NLR], hypersensitive C-reactive protein [hs-CRP], interleukin 6 [IL-6], cytokeratin-19-fragment [CYFRA21-1], squamous cell carcinoma antigen [SCCA], miR-155-5p and miR-630 and smoking) for NSCLC.
The calculated performance values for different variables in the testing group.
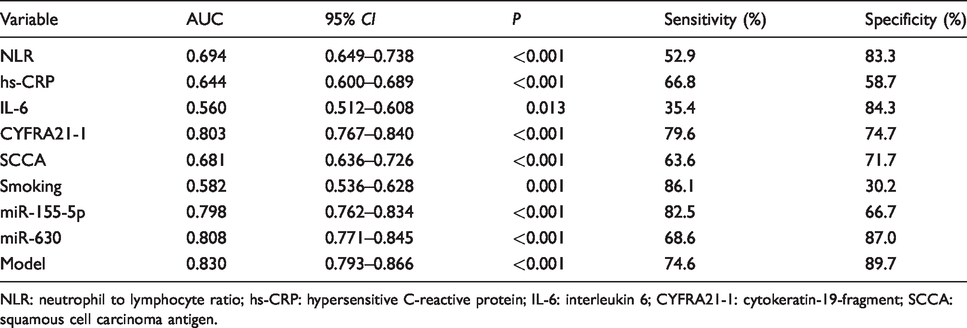
NLR: neutrophil to lymphocyte ratio; hs-CRP: hypersensitive C-reactive protein; IL-6: interleukin 6; CYFRA21-1: cytokeratin-19-fragment; SCCA: squamous cell carcinoma antigen.
Each of the abovementioned factors with a significant difference was included in the logistic regression model (Supplementary Table 2). The final predicting model for NSCLC prediction was: Logit(P)=75.283–0.607(NLR)−19.078 (hs-CRP) −3.756 (IL-6)–3.209 (CYFRA21-1)–0.215 (SCCA) + 0.036 (smoking, Yes = 1; No = 0)–3.462 (miR-155-5p) + 0.009 (miR-630). This model has a high value to identify NSCLC with AUC of 0.830 (Figure 3 and Table 3), and the estimated probability was 0.457, which means if the probability is lower than 0.457, it will be classified into NSCLC; on the contrary, it will be classified into the non-NSCLC.
The internal and external validation of the novel logistic regression model
The validity of our logistic regression model was assessed in an internal group and an external validation group. The subjects’ clinicopathological characteristics, inflammatory factors, tumour markers and life history traits are presented in Supplementary Table 3 (internal validation group) and Supplementary Table 4 (external validation group), respectively. In the internal validation group, the probabilities of 111 (out of 144) NSCLC patients were lower than 0.457, and the probabilities of 127 (out of 144) healthy controls were higher than 0.457 (Figure 4(a)). The sensitivity and specificity for NSCLC were 77.1% and 88.2%, respectively. In the external validation group, the probabilities of 96 (out of 128) NSCLC patients were lower than 0.457, and the probabilities of 116 (out of 128) healthy controls were higher than 0.457 (Figure 4(b)). The sensitivity and specificity for NSCLC were 75.0% and 90.6%, respectively.
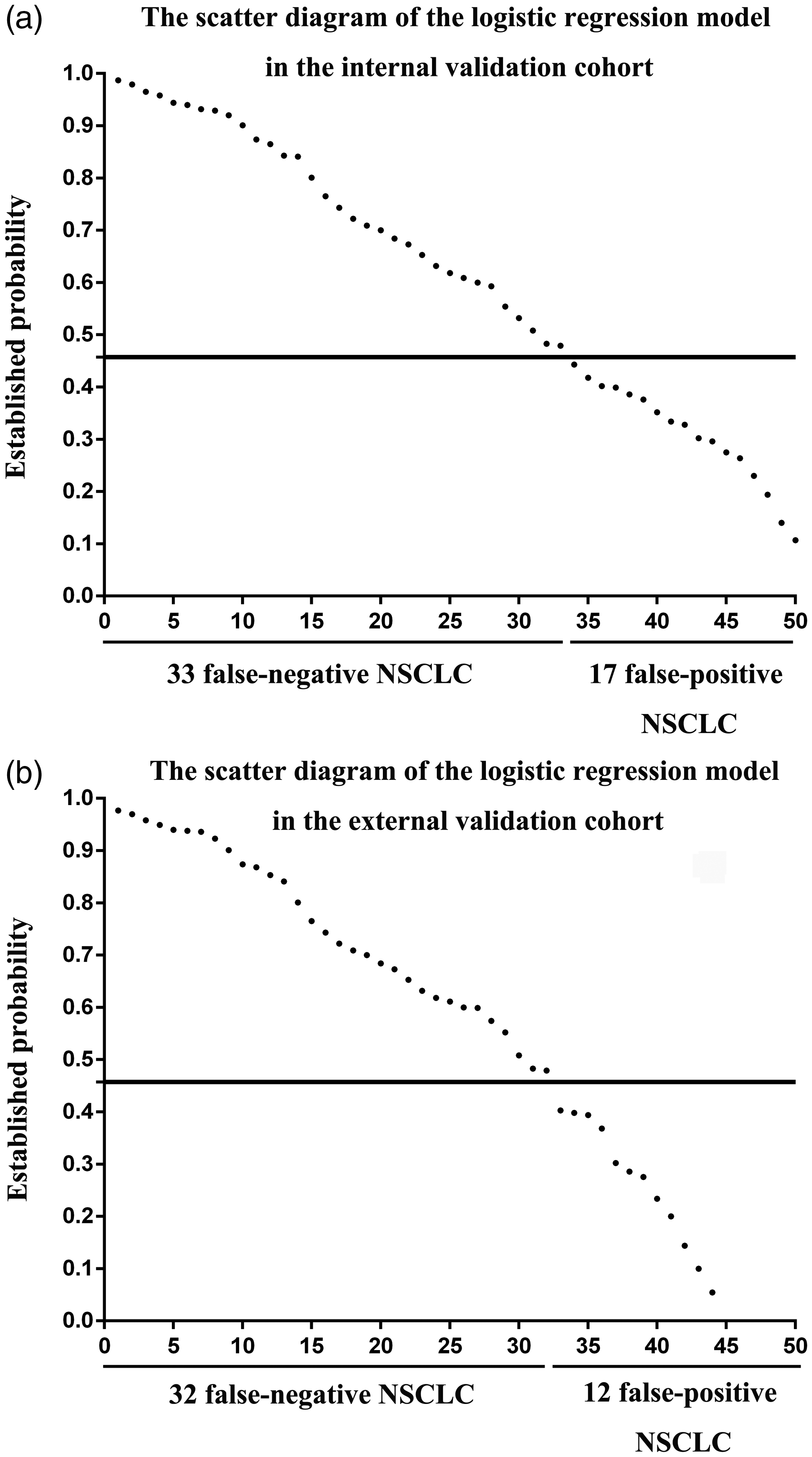
The scatter diagrams of the logistic regression model in two validation groups. (a) Thirty-three false-negative NSCLC and 17 false-positive NSCLC in the internal validation group. (b) Thirty-two false-negative NSCLC and 12 false-positive NSCLC in the external validation group.
Discussion
Lung cancer is one of the most common malignant tumours in the world, accounting for 18.2% of the world's malignant tumour mortality. 12 It has also become the leading cause of malignant tumour death among Chinese urban residents. So far, although patients can obtain a range of effective treatments, including surgical resection, chemotherapy, targeted drug therapy and other methods, the prognosis of patients with NSCLC is still poor, the report pointed out that its five-year survival rate is only 11–15%. 3 Metastasis and recurrence are the leading causes of morbidity and mortality in patients with NSCLC. The reason for the poor prognosis of patients with NSCLC is largely due to the fact that about 75% of NSCLC patients are in advanced stage of cancer at the time of diagnosis, resulting in poor efficacy of various treatments. 13 Therefore, early detection and diagnosis of NSCLC is a key way to improve the prognosis of patients and improve survival rate. The commonly used examination methods for the diagnosis of NSCLC are X-ray examination, bronchoscopy and cytology, but the sensitivity of X-ray examination for NSCLC is only about 45%–50%, and the sensitivity of other methods is also unsatisfactory, especially the detection of patients with early NSCLC.14,15 This forces us to establish a more sensitive and specific method for NSCLC diagnosis.
CTLA4 is closely related to the formation of NSCLC. CTLA4 binds to the B7 ligand, produces a synergistic inhibitory signal and inhibits the proliferation and activation of T cells, and thus reduces the body's antitumour immune response.7,8 As an immune regulation factor, CTLA4 is also an important target for NSCLC immunotherapy. 16 Since 1863, the German pathologist Coussens has proposed a hypothesis that tumour cells might originate from chronic inflammation based on the phenomenon of a large amount of leukocyte infiltration found in tumour tissues, 17 which has opened up the exploration of the relationship between inflammation and tumour. In recent years, a large number of studies have found that the occurrence and development of 15% of human tumours were related to chronic inflammation caused by infectious factors such as bacteria and viruses.18,19 The interrelationship between tumours and inflammation has been the focus of long-term controversy. Epidemiological studies have also confirmed that chronic inflammation increased the risk of developing tumours and promoted tumour progression. 20 When the body is infected or repairing damage, a large number of white blood cells will be activated and accumulate in the injury site. These cells form an inflammatory microenvironment by releasing a large amount of soluble media such as cytokines. At the same time, tumour growth and infiltration accompanied by cell apoptosis and necrosis can also release a large number of inflammatory mediators, which in turn induce angiogenesis and inflammation. 21 These inflammatory mediators, inflammatory cells, interact with tumour cells and induce each other to form an inflammatory cascade reaction. The persistent microenvironment of inflammation accelerates tumour progression by promoting the formation of new blood vessels and plays an important role in the occurrence, development and metastasis of tumours.
In this study, we assessed the diagnostic values of CTLA4-associated microRNAs combined with inflammatory factors for NSCLC. Our results showed that compared with healthy controls, miR-155-5p and miR-630 concentrations were significantly lower, while NLR, hs-CRP, IL-6, CYFRA21-1, SCCA concentrations and the smoking rate were significantly higher in NSCLC patients. Among them, the elevation in NLR could be the consequence of increased neutrophil counts and decreased lymphocyte counts. Up-regulation of peripheral neutrophils is thought to reflect the intrinsically aggressive nature of tumour cells because they are induced by cytokines produced by tumour cells. 22 Hs-CRP and IL-6 are also considered to be important markers of inflammation. MiR-155-5p and miR-630 have been found to play essential roles in tumour suppression in both clinical and basic researches of lung cancer. Lin et al. 23 found that the miR-155-5p suppressed the migration and invasion of lung adenocarcinoma A549 cells by targeting Smad2. Zhu et al. 24 found that the knockdown of miR-155-5p attenuated radiation suppressed of NSCLC cell proliferation. Cao et al. 25 found that miR-630 reduced apoptosis by attenuating several apoptotic modulators such as PARP3, DDIT4, EP300 and EP300. In our research, although miR-630 had better effects (AUC = 0.808) of diagnosis for NSCLC than NLR (AUC = 0.694), hs-CRP (AUC = 0.644), IL-6 (AUC = 0.560), CYFRA21-1 (AUC = 0.803), SCCA (AUC = 0.681), smoking (AUC = 0.582) and miR-155-5p (AUC = 0.798), but its sensitivity (68.6%) was unsatisfactory. Therefore, we built a novel logistic regression model including multiple parameters containing NLR, hs-CRP, IL-6, CYFRA21-1, SCCA, smoking, miR-155-5p and miR-630. It showed higher diagnostic efficiency (AUC = 0.830, sensitivity = 74.6%, specificity = 89.7%) than any single parameter. To avoid the occurrence of selection bias as much as possible, we selected NSCLC patients and healthy controls from two different branches. This model showed good diagnostic efficiency in both validation cohorts (AUC = 0.838 with the internal validation group; AUC = 0.851 with the external validation group), indicating it predicts NSCLC effectively.
In summary, the current model that combined multiple parameters (NLR, hs-CRP, IL-6, CYFRA21-1, SCCA, smoking, miR-155-5p and miR-630) might improve the diagnostic efficiency of NSCLC. However, our results are required to be verified by multicentre studies with larger sample sizes.
Supplemental Material
ACB901564 Supplemental material1 - Supplemental material for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer
Supplemental material, ACB901564 Supplemental material1 for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer by Xiaohong Liu, Chunlian Tang, Xianda Song, Lanfang Cheng, Ying Liu, Fan Ding, Chun Xia, Lian Xue, Jun Xiao and Baorong Huang in Annals of Clinical Biochemistry
Supplemental Material
ACB901564 Supplemental material2 - Supplemental material for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer
Supplemental material, ACB901564 Supplemental material2 for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer by Xiaohong Liu, Chunlian Tang, Xianda Song, Lanfang Cheng, Ying Liu, Fan Ding, Chun Xia, Lian Xue, Jun Xiao and Baorong Huang in Annals of Clinical Biochemistry
Supplemental Material
ACB901564 Supplemental material3 - Supplemental material for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer
Supplemental material, ACB901564 Supplemental material3 for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer by Xiaohong Liu, Chunlian Tang, Xianda Song, Lanfang Cheng, Ying Liu, Fan Ding, Chun Xia, Lian Xue, Jun Xiao and Baorong Huang in Annals of Clinical Biochemistry
Supplemental Material
ACB901564 Supplemental material4 - Supplemental material for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer
Supplemental material, ACB901564 Supplemental material4 for Clinical value of CTLA4-associated microRNAs combined with inflammatory factors in the diagnosis of non-small cell lung cancer by Xiaohong Liu, Chunlian Tang, Xianda Song, Lanfang Cheng, Ying Liu, Fan Ding, Chun Xia, Lian Xue, Jun Xiao and Baorong Huang in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Wuhan Municipal Health and Family Planning Commission Research Project (WX16C29 and WX17D02).
Ethical approval
The ethics committee of the Wuchang Hospital Affiliated to Wuhan University of Science and Technology (20160117) and the ethics committee of the Zhongnan Hospital of Wuhan University (2018–019) approved this study.
Guarantor
BH.
Contributorship
BH and XL researched literature and conceived the study. XL, CT, XS, YL, FD, CX and LC were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. XL, LX, JX and BH wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.
Supplemental material
Supplemental material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
