Abstract
Disorders of vitamin D metabolism have only recently become more widely recognized. In 2011, a series reported six children with familial idiopathic infantile hypercalcaemia, a condition in which patients develop hypercalcaemia following bolus vitamin D supplementation due to mutations in CYP24A1, formerly known as 24-hydroxylase. This is the chief enzyme in the catabolism of 1,25-dihydroxyvitamin D3 (calcitriol) to inactive 1,24,25-(OH)3D3. Isolated cases of loss of function CYP24A1 mutations have been reported across a wide spectrum of ages, including three cases first identified during pregnancy in the context of vitamin D supplementation. We describe a family in which two siblings had hypercalcaemia due to a disorder of calcitriol catabolism as a result of compound heterozygous loss of function mutations of CYP24A1, including a novel mutation K351Nfs*21. The index case, who has kindly given written informed consent for this report, was a female in her mid-20s presenting with symptomatic hypercalcaemia precipitated by vitamin D supplementation in her first pregnancy. In a subsequent pregnancy, she remained normocalcaemic in the absence of supplementation. Her asymptomatic brother was identified through cascade screening. Upregulation of calcitriol production in pregnancy, particularly when combined with vitamin D supplementation, can unmask previously unidentified disorders of vitamin D metabolism. This report emphasizes the importance of screening of family members and the need for caution with vitamin D supplementation in pregnancy.
Case description
A previously healthy primigravida in her mid-20s presented with hyperemesis and hypercalcaemia at 13 weeks’ gestation and was referred for endocrinological assessment. Prior to the pregnancy, her general practitioner had diagnosed mild vitamin D deficiency (25-OH-D3 30 nmol/L (reference interval [RI] 50–150) and prescribed supplementation (cholecalciferol 50,000 units monthly). Examination was as expected for gestation, and there was no clinical suspicion of malignancy, infection or an inflammatory process.
Laboratory investigations revealed elevated albumin-corrected plasma calcium 2.9 mmol/L (RI 2.2–2.6) (11.6 mg/dL), normal phosphate 1.1 mmol/L (RI 0.8–1.5), (3.41 mg/dL), low parathyroid hormone 0.7 pmol/L (RI 1.6–7.0) (6.6 pg/mL) and normal renal function. 25-OH-D3 was normal at 116 nmol/L. Urine calcium excretion was markedly elevated at 2.09 mole ratio (RR 0.06–0.45). Plasma 1,25-(OH)2D3 (calcitriol) was twice the upper limit of the reference range at 380 pmol/L (RI 65–175). Parathyroid hormone-related peptide (PTHrP) was undetectable at 20 weeks’ gestation. Abdominal ultrasound showed a 5 mm asymptomatic renal calculus and normal uterine anatomy appropriate for gestation. Chest X-ray was normal, serum angiotensin converting enzyme was normal and a QuantiFERON-TB Gold test was negative. DEXA bone densitometry performed two months prior to pregnancy was normal for age.
Vitamin D supplementation was discontinued. A trial of prednisone (20 mg daily for 14 days) had no effect on plasma calcium which remained between 2.9 and 3.3 mmol/throughout the remainder of her pregnancy. At 36 weeks’ gestation, she developed idiopathic cholestasis of pregnancy (ICP) and delivered a healthy normocalcaemic infant at term. Hypercalcaemia resolved by four weeks’ post-partum, although hypercalciuria persisted.
A diagnosis of disordered calcitriol catabolism was considered, and samples were sent to a specialized international laboratory for the analysis of vitamin D metabolites. The results are shown in Table 1 and Figure 1. Quantitative assay of vitamin D metabolites was performed by modified liquid chromatography-tandem mass spectrometry (LC-MS/MS) technology using a chromatographic approach that resolves 24,25(OH)2D3 from 25,26(OH)2D3. The ratio of plasma 25-OH-D3 to 24,25-(OH)2D3 was markedly elevated. In addition, 1,24,25(OH)3D3 concentrations were undetectable confirming the lack of functional CYP24A1.
Summary of biochemical investigations undertaken for each consenting family member.
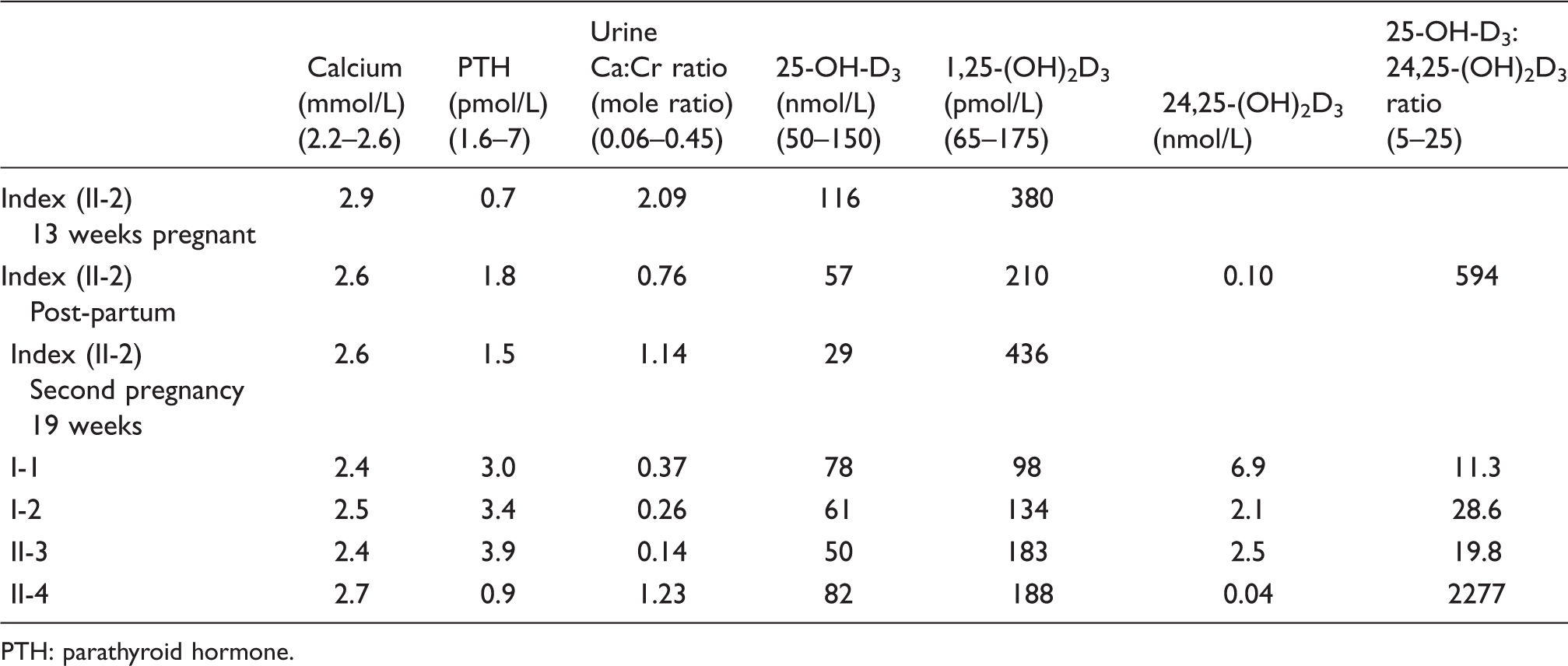
PTH: parathyroid hormone.
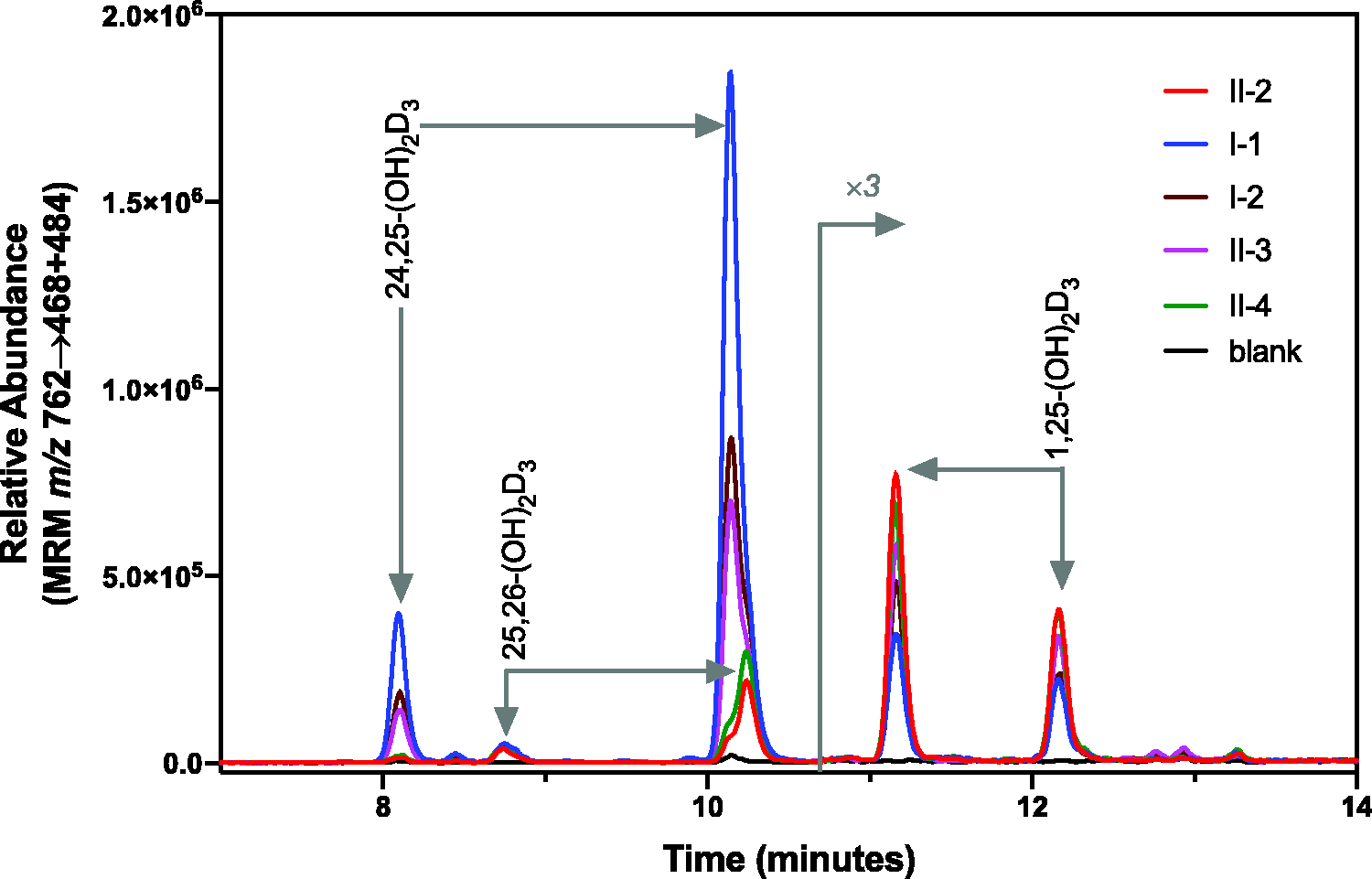
LC-MS/MS analysis of 24,25-(OH)2D3 and 1,25-(OH)2D3. Quantitative assay of vitamin D metabolites including 24,25-(OH)2D3 and 1,25-(OH)2D3 involves dilution with specific deuterated internal standards, protein precipitation, extraction and derivatization with DMEQ-TAD prior to LC-MS/MS analysis. A representative chromatogram of m/z 762→468 + 484 is shown, based on immunoextraction with anti-1,25-(OH)2D3 antibody. Derivatization of vitamin D metabolites with DMEQ-TAD yields both 6S and 6 R isomers, which are chromatographically resolved. As 24,25-(OH)2D3 and 1,25-(OH)2D3 share the same molecular mass and certain fragment ions, both metabolites can be observed on the m/z 762→468 + 484 trace.
Genetic analysis using PCR and DNA sequencing of the entire genome coding region and splice junctions was performed. Compound heterozygous loss-of-function mutations of CYP24A1 were detected (Figure 1). The E143del mutation has previously been reported in association with the condition.1–3 and K351Nfs*21 is novel and reported as likely pathogenic.
Cascade screening of immediate family members was carried out (Figure 2 and Table 1) and identified that her brother, aged in his early teens, was also affected. On review, he was asymptomatic but was found to have an albumin-corrected plasma calcium of 2.7 mmol/L (10.8 mg/dL) and hypercalciuria (1.23 mole ratio [RI 0.06–0.45]). Like his sibling, the ratio of plasma 25-OH-D3 to 24,25-(OH)2D3 was also markedly elevated. Renal tract ultrasound identified no calculi or nephrocalcinosis. He was made aware that he was at increased risk of stone disease and advised to avoid vitamin D supplementation.
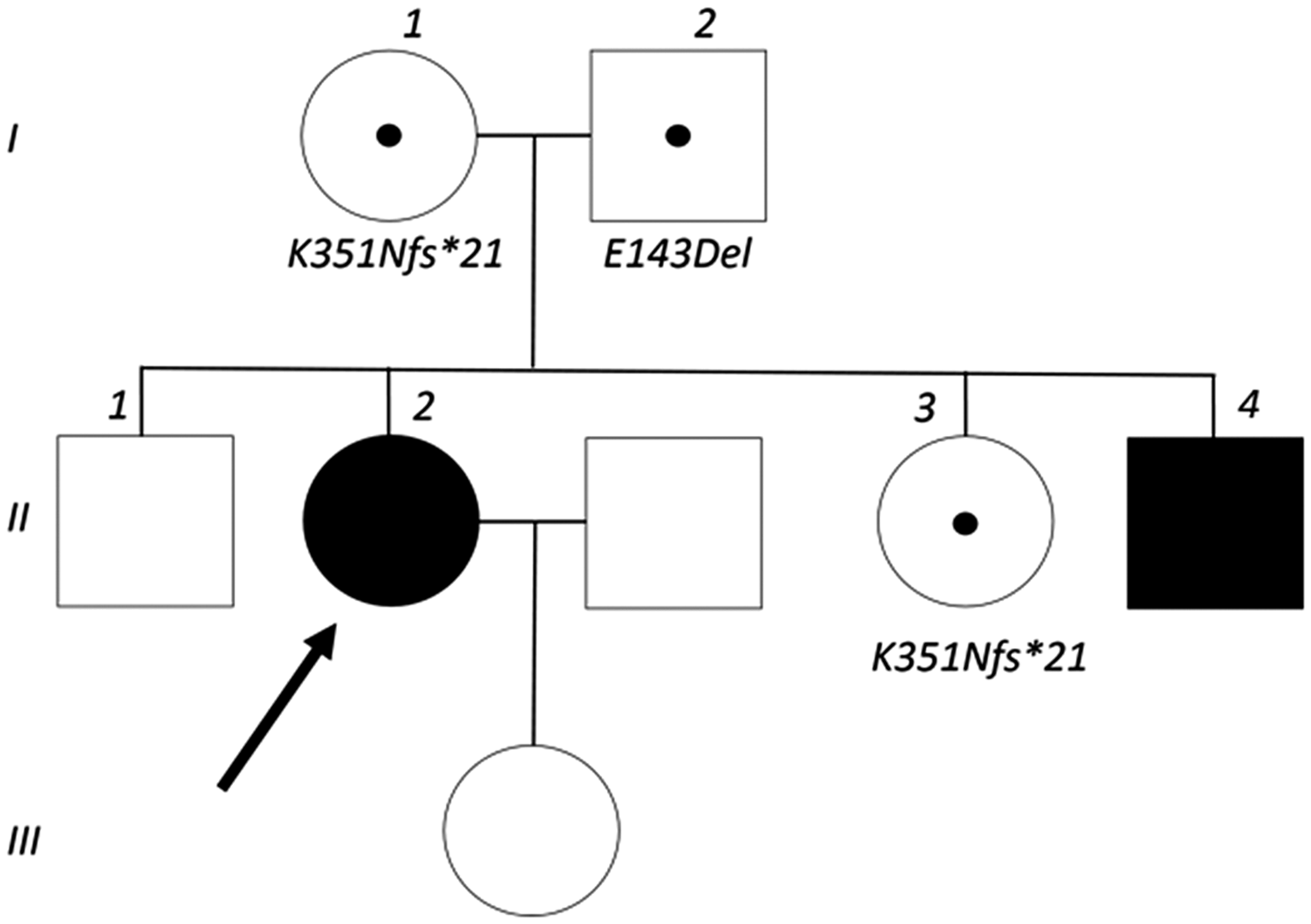
The index case (II-2) is indicated by the bold arrow. Affected family members are shown as solid black symbols. Family members carrying either mutation are indicated with a black dot. The patient’s brother (II-1) refused consent for mutation testing.
The index case had a subsequent pregnancy three years later, during which serum calcium was monitored monthly and remained within the reference range (maximum corrected calcium 2.6 mmol/L) in the absence of vitamin D supplementation. She developed recurrent ICP and delivered another healthy infant at term.
The final diagnosis for this family was of familial hypercalcaemia due to abnormal vitamin D metabolism as a result of heterozygous CYP24A1 loss-of-function mutations, often referred to as idiopathic infantile hypercalcaemia (IIH)/CYP24A1-hypercalcaemia. Symptomatic hypercalcaemia was unmasked in the index case by vitamin D supplementation. Physiological changes of pregnancy may also be relevant; however, given she remained normocalcaemic in a subsequent pregnancy (without vitamin D supplementation), these are not likely to be the dominant precipitant.
Discussion
Disordered vitamin D metabolism and hypercalcaemia due to CYP24A1 mutations
Vitamin D is metabolized through a series of hydroxylation steps, first in the liver to 25-OH-D3 and then in the kidney by 1α-hydroxylase to the active form 1,25-(OH)2D3 (calcitriol). Calcitriol is in turn catabolized to inactive 1,24,25-(OH)3D3 and calcitrioic acid, by cytochrome P450 enzyme CYP24A1, formerly known as 24-hydroxylase, which is also responsible for the inactivation of 25-OH-D3. The importance of inactivation of 1,25-(OH)2D3 in the regulation of vitamin D metabolism and therefore calcium balance has only recently been recognized. In 2011, hypercalcaemia following bolus vitamin D supplementation was described in a series of six children. The authors described this condition as ‘familial idiopathic infantile hypercalcaemia’. 1 Affected individuals were identified as harbouring bi-allelic loss-of-function mutations in CYP24A1, and thus demonstrated increased sensitivity to vitamin D loading. Since then, cases of inactivating CYP24A1 mutations have been reported across a wide spectrum of ages, including three cases first identified during pregnancy, all of which received prenatal supplements containing vitamin D. 2 , 4 , 5
IIH/CYP24A1-hypercalcaemia classically presents as symptomatic hypercalcaemia in infancy; however, a recent review found that more than a third of currently reported patients with bi-allelic CYP24A1 mutations presented later in life. 3 Many affected individuals are asymptomatic but hypercalciuria and renal calculi are common, 3 both of which were identified in our index patient. Chronic kidney disease, with a decline in glomerular filtration rate, has also been reported, although it is unclear if this is a primary complication of the disease or a result of calculi and/or associated intervention. 2 , 6 The variable penetrance is thought to be due to a combination of genetic and environmental factors with the cumulative dose of exogenous vitamin D playing a critical role. The influence of sunlight exposure, and thus cutaneous vitamin D, may also be important with higher concentrations of serum and urinary calcium seen during summer months. 6
Quantification of vitamin D metabolites by liquid LC-MS/MS provides a sensitive and specific method for identifying IIH/CYP24A1-hypercalcaemia 7 and can be carried out by 150–200 specialist laboratories worldwide at a cost of around US$40 per sample. 8 A chromatographic approach that resolves 24,25(OH)2D3 from 25,26(OH)2D3 as used in this case, is recommended to avoid misleading results due to the contamination of the 24,25(OH)2D3 peak by 25,26(OH)2D3. 9 In affected individuals, concentrations of 25-OH-D3 may be elevated and 1,24,25-(OH)3D3 and 24,25-(OH)2D3 are very low or undetectable. As the measurement of 1,24,25-(OH)3D3 is technically difficult, the measurement of the ratio of 25-OH-D3 to 24,25-(OH)2D3 is recommended with the result being markedly elevated (>140 using the method described above). 9 This ratio correctly predicted both affected cases in our family despite the postpartum return to normocalcaemia in the index case.
Diagnosis is confirmed by genetic analysis. A spectrum of mutations along the entire CYP24A1 gene has been identified 10 , 11 ; however, the K351Nfs*21 mutation identified in this case has not previously been reported. The prevalence of IIH/CYP24A1-hypercalcaemia is unknown; however, almost 0.7% of the general European population have been found to be heterozygous carriers of one of four common CYP24A1 loss-of-function alleles. 10 As there are now over 20 mutations reported, the true carrier frequency is likely to be significantly higher.
Management of IIH/CYP24A1-hypercalcaemia includes avoidance of vitamin supplementation, a low calcium-diet (if appropriate) and advice regarding sun-protection and maintenance of hydration. For symptomatic hypercalcaemia, usual treatments including hydration, glucocorticoids, calcitonin and bisphosphonates have been used successfully. 10 If hypercalcaemia is refractory, consideration can be given to medication that induces surrogate vitamin D catabolism. 12 Treatment with rifampicin, a potent inducer of CYP3A4, successfully treated hypercalcaemia in two cases of IIH, the mechanism postulated to be increased degradation of 1,25-(OH)2D3 or its precursor 25(OH)D3 in the absence of CYP24A1. 13 Inhibition of vitamin D-synthesizing enzymes by low-dose fluconazole has also been successfully used in a small number of reported cases. 14
Calcium physiology during pregnancy
Maternal calcium metabolism adapts to accommodate the demands of the placenta and growing fetal skeleton, which contains around 30 g of calcium by full term. 15 Although the majority of the calcium is accumulated during the third trimester, changes to calcium homeostasis begin to occur early in pregnancy. Expansion of circulating plasma volume reduces serum albumin concentrations and thus total serum calcium; however, albumin-corrected and ionized free calcium concentrations remain within the normal non-pregnant reference range. Renal and placental upregulation of calcitriol production, which may lead to a several fold increase in serum concentrations by the third trimester, 15 contributes to this effect. Consequently, fractional absorption of calcium from the small intestine increases two-fold during the first trimester, a change that is sustained throughout the remainder of pregnancy. 15 This in turn leads to increased renal filtration and urinary calcium excretion which may reach the hypercalciuric range. 16
A further consequence of the upregulation of calcitriol is that parathyroid hormone concentrations are suppressed during the first trimester to levels that may even fall below the lower limit of normal. 15 PTHrP, produced by placental and lactating mammary tissue, rises gradually throughout pregnancy peaking in the postpartum period and being sustained by lactation. 17
To ensure there is adequate vitamin D to meet this demand and to prevent fetal vitamin D deficiency, guidelines for vitamin D supplementation during pregnancy have recently been introduced in a number of countries. Guidance in Australia, 18 New Zealand 19 and United States 20 suggests consideration of vitamin D measurement for women at high risk of deficiency (those with darker skin, who avoid sun exposure, take photosensitizing medications, or have liver or kidney disease) 21 and supplementation is advised for pregnant women with low concentrations. As the index patient wore a hijab, she would be considered at high risk for deficiency. Australia and New Zealand guidelines also suggest that supplementation should be considered for all pregnant women at higher risk, without testing vitamin D concentrations. 18 , 19 In the UK, it is recommended that all women be advised early in pregnancy to supplement vitamin D. 22 These recommendations are based on evidence that, depending on geographical location and climate, a sizeable proportion of women of child bearing age (over one third in NZ23) have vitamin D concentrations below the recommended range, and there is correlation between maternal vitamin D concentrations and vitamin D deficiency in the newborn. 21 Widespread blood testing for vitamin D deficiency is not encouraged because the cost of testing is far greater than the cost of treatment, and the potential harms from treatment have traditionally been considered small. This case demonstrates that harms from treatment can be significant as, in the setting of disordered calcitriol metabolism, maternal vitamin D supplementation can unmask significant maternal hypercalcaemia. There is also evidence of harm with increased rates of infantile hypercalcaemia being reported in in Australia since the introduction of guidelines in 2006. Of first measured serum calcium in infants under six months at a single laboratory, hypercalacaemia was detected in 1.1% in 2005–2007 and increased to 8.7% in 2011–2013. 24 During the latter period, 13 infants were diagnosed with IIH with raised 1,25-(OH)2D3 in keeping with an abnormality of CYP24A1. Infants had associated significant morbidity including symptomatic hypercalcaemia and nephrocalcinosis. Of these infants, 12 had mothers who had received prenatal vitamin D3 supplementation.
Overall, hypercalcaemia in pregnancy is rare. Primary hyperparathyroidism (PHPT) accounts for the majority of cases but still affects less than 0.1% of reproductive age women. 6 In our patient, PHPT was excluded based on suppressed parathyroid hormone. There are a broad range of other causes of hypercalcaemia in pregnancy including malignancy, granulomatous disease and excessive calcium and alkali ingestion. 25 In this case, as clinical assessment was not suggestive of malignancy and the response to steroids was not consistent with granulomatous disease, further investigation was carried out. The very high calcitriol concentration early in the pregnancy raised the possibility of a disorder of vitamin D metabolism. Hypercalcaemia due to abnormalities of vitamin D metabolism include vitamin D intoxication, IIH due to mutations of SLC34A1 (encoding renal proximal sodium-phosphate co-transporter Na-Pi-IIa) and, as identified in this case, IIH due to loss-of-function mutations of CYP24A1.
Conclusion
Hypercalcaemia due to disorders of vitamin D metabolism have previously been described but remain poorly recognized. IIH due to loss-of-function mutations in CYP24A1 should be considered in the differential diagnosis of hypercalcaemia or hypercalciuria in all age groups. Pregnancy (when there is an upregulation of calcitriol production) and, perhaps more importantly, a trend to supplement vitamin D, can precipitate symptomatic hypercalcaemia in affected patients and result in significant morbidity to both the mother and infant.
The ratio of 25-OH-D3 to 24,25-(OH)2D3 provides a sensitive and specific method for predicting the presence of CYP24A1 mutations even during periods of normocalcaemia, and diagnosis is confirmed through genetic analysis. Cascade screening of family members of individuals with CYP24A1 mutations is important and should be considered at adulthood, or earlier if symptomatic. Affected asymptomatic individuals benefit from diagnosis through prevention of unnecessary investigation of hypercalcaemia and awareness of the importance of avoiding vitamin D supplementation, particularly in pregnancy.
Footnotes
Acknowledgements
We would like to thank the index case for her consent to publish and the Molecular Genetics Laboratory at Labplus, Auckland for performing the genetic analysis in this case. We acknowledge the loan of the LC-tandem mass spectrometer used in these studies as part of a Waters-Queen’s University Research Agreement.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
CM.
Contributorship
CM and TU reviewed the literature and wrote the article. All authors reviewed and edited the article and approved the final version.
