Abstract
Background
Vitamin D deficiency is a prevalent and important global health problem. Because of its role in growth and development, vitamin D status is likely to be particularly important in adolescent girls. Here, we explored the effects of high-dose vitamin D supplementation on cardio-metabolic risk factors.
Methods
We have examined the effects of vitamin D supplementation on cardio-metabolic risk factors in 988 healthy adolescent girls in Iran. Fasting blood samples and anthropometric measurements were obtained at baseline and after supplementation with high-dose vitamin D. All individuals took a capsule of 50,000 IU vitamin D/week for nine weeks. The study was completed by 940 participants.
Results
The prevalence of vitamin D deficiency was 90% at baseline, reducing to 16.3% after vitamin D supplementation. Vitamin supplementation was associated with a significant increase in serum concentrations of 25 (OH) vitamin D and calcium. There were significant reductions in diastolic blood pressure, heart rate, waist circumference and serum fasting blood glucose, total- and low-density lipoprotein-cholesterol after the nine-week period on vitamin D treatment, but no significant effects were observed on body mass index, systolic blood pressure or serum high-density lipoprotein cholesterol and triglyceride.
Conclusion
Vitamin D supplementation had beneficial effects on cardio-metabolic profile in adolescent girls.
Introduction
Vitamin D is an essential micronutrient that is important for various aspects of human health. Vitamin D deficiency is now a prevalent global health problem, and is an important risk factor in the aetiology of cancer, diabetes and cardiovascular disease (CVD). 1 The European Society for Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) has recently recommended a serum 25-hydroxy vitamin D (25-OH D) concentration >50 nmol/l (20 ng/mL) as the threshold value for vitamin D sufficiency. 2 Hypovitaminosis D is particularly prevalent in Asia 3 with a reported prevalence of 90% in the Middle East 3 and 79% in Iran. 4 The role of vitamin D is particularly important in adolescent girls, because vitamin D status influences various aspects of growth, development and puberty in this group.5–7 In Iran, the prevalence of vitamin D deficiency has been reported to be 79–81.3% in adolescents.8,9
Most chronic diseases, including CVD have their origins in childhood and adolescence, and the early control of their risk factors is important to reduce chronic diseases in adulthood. 10
Several observational studies have indicated that serum 25-OH vitamin D is inversely associated with body mass index (BMI), dyslipidaemia, inflammatory markers and hypertension in children11–14 and adults.14,15 Low serum concentrations of 25-OH vitamin D are proposed to be related to cardio-metabolic risk factors even in adolescence. 16 However, vitamin D supplementation trials are necessary to clarify whether a low serum 25-OH vitamin D is causally related to these cardio-metabolic risk factors. Several doses of vitamin D supplementation have been used previously in relation to affecting cardio-metabolic risk factors, 16 but the results from clinical trials have been inconsistent. Some clinical trials have suggested that vitamin D supplementation improves blood pressure, fasting blood glucose and lipid profile,17,18 while other studies have not reported any significant improvements in these parameters. 19
Given the importance of vitamin D in health and the high prevalence of its deficiency in the community, Iran's Ministry of Health has recently used high-dose supplements of vitamin D for reducing vitamin D deficiency in adolescents. In this intervention, approximately 100,000 adolescent girls took nine high-dose 500,00 IU of vitamin D supplements, over a period of nine weeks. We have investigated the effects of this high-dose vitamin D supplementation on cardio-metabolic risk factors in a random sample of this group of adolescent girls. To our knowledge, this study is one of the largest studies to date to examine the effects of high-dose vitamin D supplementation on cardio-metabolic risk factors in adolescent girls.
Methods
Study design and participants
This study was undertaken in the cities of Mashhad and Sabzevar, in northeastern Iran between January and April 2015. Participants were selected by using a randomized clustering method and computer-generated random numbers. Written consent was obtained from the girls and their parents. We excluded girls with any autoimmune diseases, cancer, metabolic bone disease, hepatic or renal failure, cardiovascular disorders, malabsorption or thyroid, parathyroid or adrenal diseases. Subjects who were taking anti-inflammatory, antidepressant, antidiabetic or antiobesity drugs, vitamin D or calcium supplement use and hormone therapy within the last six months were also excluded. A total of 1026 adolescents aged 12–18 years old were screened, of whom 988 met the inclusion criteria. All participants were provided with nine vitamin D capsules containing 50,000 IU vitamin D over nine weeks. Overall, 940 girls completed the intervention, with a dropout rate of 4.8%.
A validated food frequency questionnaire was used to evaluate dietary intakes.20,21 To estimate energy and nutrient intakes, the reported portion size in FFQ were converted to grams using household measures and then were entered to the Nutritionist IV software. Physical activity was assessed through a validated questionnaire 22 and provided as metabolic equivalents (METs) in hours per day. Demographic data, sun exposure and use of sunscreen were collected by an expert interviewer and by the use of a standard questionnaire. The ethical committee of Mashhad University of Medical Sciences approved the study, and informed written consent was completed by all participants.
Anthropometric and cardiac measurements
Anthropometric parameters were determined at baseline and after nine weeks of intervention. BMI was calculated as weight in kilograms divided by height in metres squared. Waist circumference (WC), Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured based on the standard procedure. Heart rate (HR) was measured to count the number of heart beats occurring over 60 s in sitting state and after 5-min rest.
Blood collection and routine biochemistry
Fasting blood samples were obtained early in the morning between 8 and 10 a.m. at baseline and after nine-weeks’ intervention, by venipuncture of an antecubital vein after a 14 h overnight fast. The samples were collected in vacuum tubes from subjects in a sitting position, according to a standard protocol. Blood samples were immediately centrifuged (Hettich model D-78532) at 1465 3 g for 10 min at room temperature to separate serum, or plasma (0.5 mL). Samples were stored at −80℃ at the reference laboratory in Mashhad University of Medical Science until analysis. An electrochemiluminescence method (ECL, Roche, Basel, Switzerland) was used for the measurement of serum 25-OH vitamin D. The limit of detection for the 25-OH vitamin D assay was 10 nmol/L for the ECL (Roche) and intra- and inter-assay variation were 5.7% and 9.9%, respectively. Serum calcium (Ca), phosphate (P), fasting blood glucose (FBG), triglyceride (TG), total cholesterol (TC) and high-density lipoprotein cholesterol (HDL-C) concentrations were measured using commercial kits (Pars Azmun, Karaj, Iran) and the BT-3000 auto-analyzer (Biotechnica, Rome, Italy). LDL-C was calculated using Friedewald formula if serum TGs concentrations were lower than 4.52 mmol/L. 23
Statistical method
Kolmogrov–Smirnow test was applied to ensure the normal distribution of variables. We categorized the participants into three groups by baseline serum concentrations of 25-OH D: deficient (<50 nmol/L), insufficient (50–74.9 nmol/L) and sufficient (>75 nmol/L). Significant differences in continuous variables across categories of 25-OH D were examined by use of the one-way analysis of variance (one-way ANOVA); this analysis was also applied to compare the dietary intakes of population across categories of serum 25-OH D. A chi-squared test was used to assess the distribution of categorical variables across three groups of 25-OH D status. Partial correlation analysis was applied to evaluate the associations between anthropometric, biochemical parameters and changes of serum 25-OH D concentration after adjustment for age. To examine the effects of vitamin D supplementation on 25-OH D and cardio-metabolic risk factors, we used paired t-tests. To control confounding factors (age, energy intake, dietary intake of vitamin D, menstruation, use of sunscreen, passive smoker, sun exposure, BMI and physical activity), we conducted analysis of covariance (ANCOVA). A P-value <0.05 was considered statistically significant. All statistical analyses were performed using SPSS-17 (SPSS Inc., Chicago, IL, USA).
Results
Baseline characteristics
Demographic characteristics of adolescent girls according to baseline serum of 25-OH vitamin D categories.
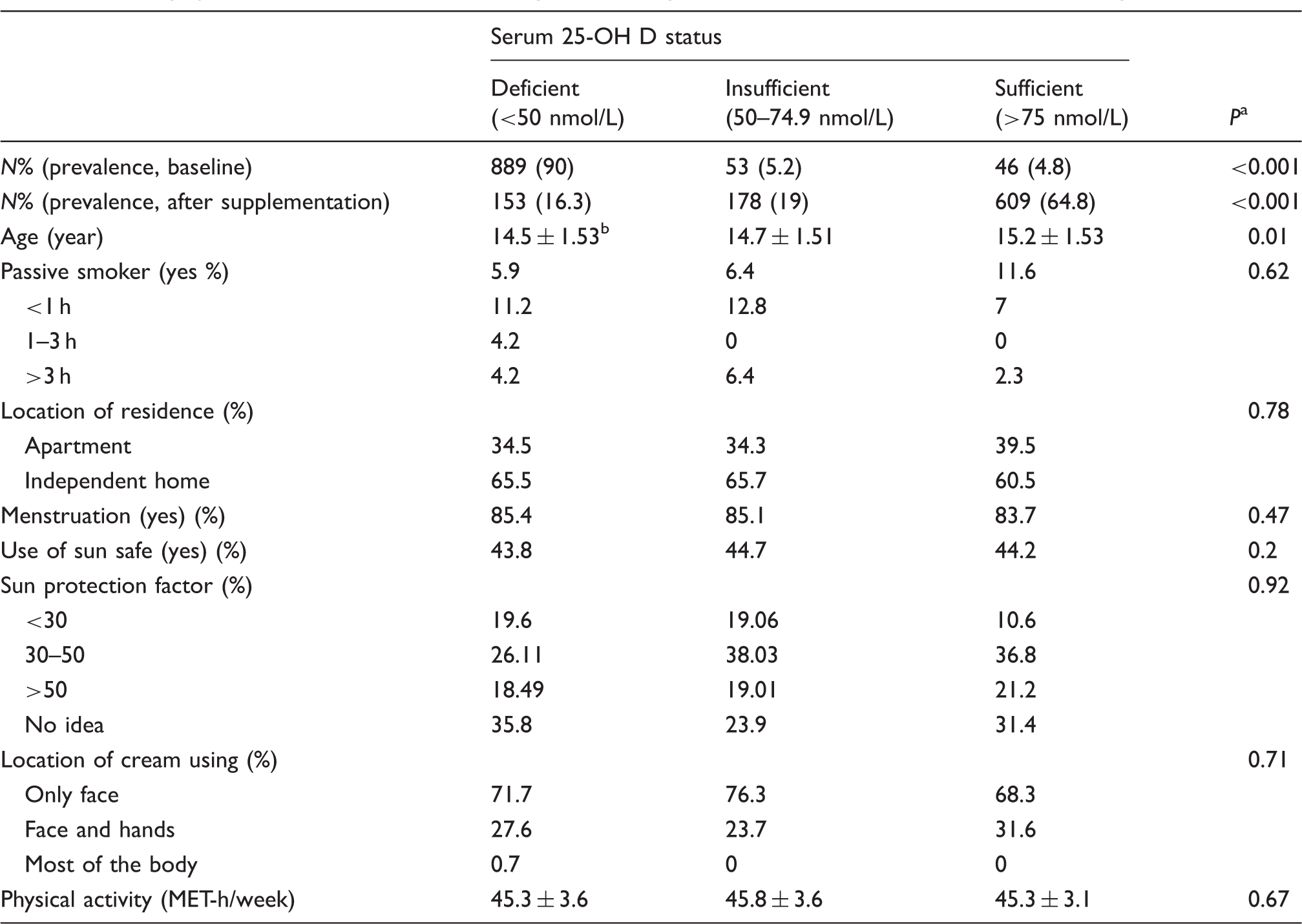
MET-h/week: metabolic equivalent of task-hour/week; 25-OH D: 25-hydroxy vitamin D.
Obtained from one-way ANOVA or chi-square test, where appropriate.
Mean ± standard deviation (SD).
Effects of vitamin D supplementation on anthropometric measurements in adolescent girls according to baseline serum of 25-OH vitamin D categories.
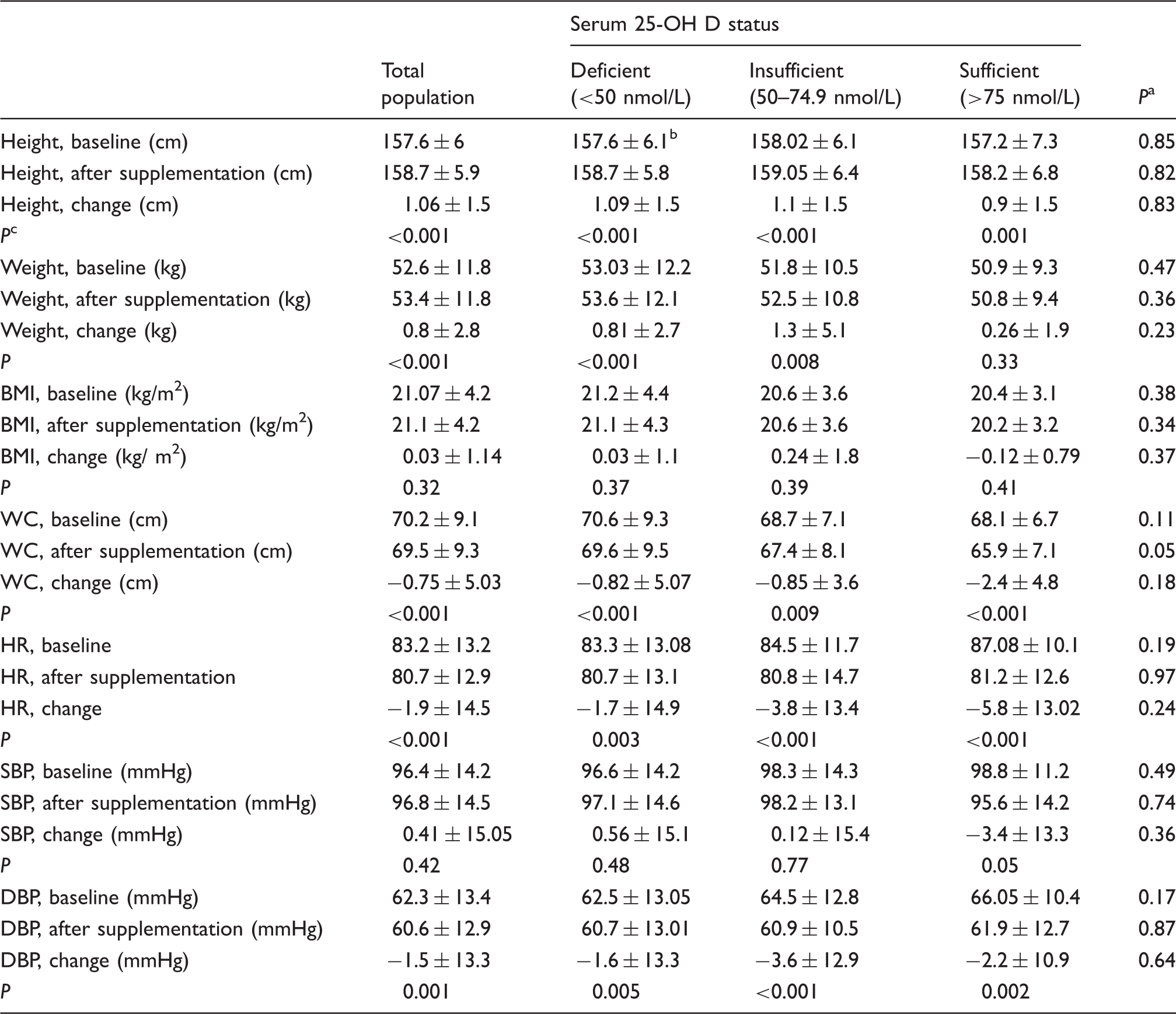
25-OH D: 25-hydroxy vitamin D; WC: waist circumference; BMI: body mass index; HR: heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure.
Obtained from one-way ANOVA to compare the differences between categories of 25-OH D status.
Mean ± standard deviation (SD).
Obtained from pair-samples t-test to examine the effects of vitamin D supplementation.
Effects of vitamin D supplementation on biochemical measurements in adolescent girls according to baseline serum of 25-OH vitamin D categories.
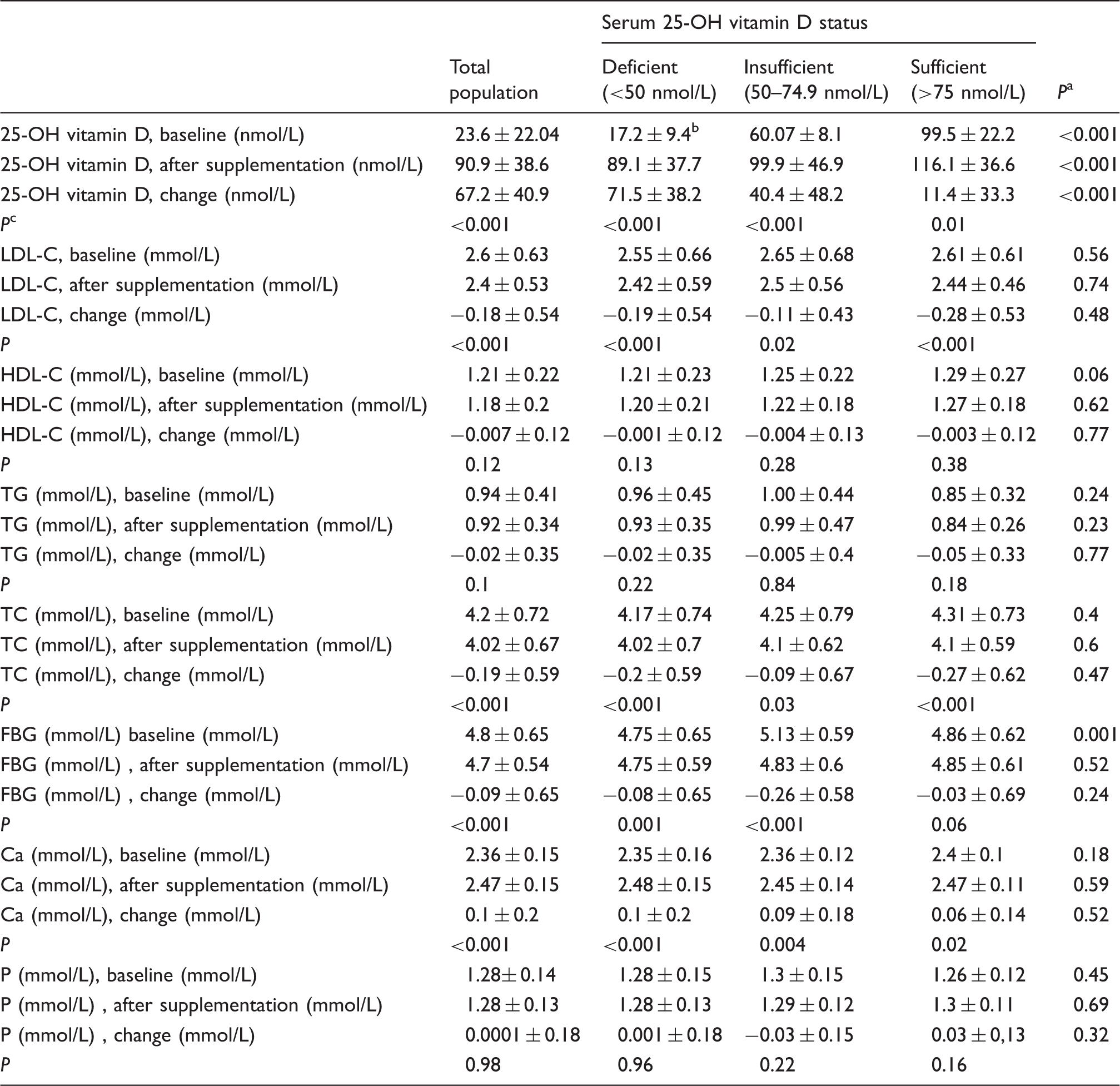
25-OH D: 25-hydroxy vitamin D; LDL-C: low-density lipoprotein-cholesterol; HDL-C: high-density lipoprotein-cholesterol; TG: triglyceride; TC: total cholesterol; FBG: fasting blood glucose; Ca: calcium; P: phosphate.
Obtained from one-way ANOVA to compare the differences between categories 25-OH D status.
Mean ± standard deviation (SD).
Obtained from pair-samples t-test to examine the effects of vitamin D supplementation.
Effects of vitamin D supplementation on anthropometric and biochemical parameters
Serum status of 25-OH D was classified based on the following threshold values (nmol/L): serum 25-OH D concentrations <50 deficiency, 50–74.9 insufficiency and >75 sufficiency. 1 Deficiency of vitamin D was present in 90%, while 5.2% and 4.8% of participants indicated insufficient and sufficient concentrations of 25-OH D at the baseline, respectively. After intervention, the prevalence of vitamin D deficiency was reduced to 16.3%, while insufficiency and sufficiency concentrations were increased to 19% and 64.8%, respectively.
The effects of vitamin D supplementation on anthropometric measurements (Table 2) and biochemical profiles (Table 3) for the total population and for the three separate baseline categories of 25-OH vitamin D are shown in the respective tables. The findings for the total population were in line with findings in each baseline categories of 25-OH D. A significant reduction in WC (69.5 ± 9.3 vs. 70.2 ± 9.1, P ≤ 0.001), HR (80.7 ± 13.2 vs. 83.2 ± 12.9, P < 0.001) and DBP (60.6 ± 12.9 vs. 62.3 ± 13.4, P = 0.001) were seen after intervention compared with baseline, while no statistically significant differences were found for BMI and SBP.
Serum concentrations of 25-OH vitamin D (90.9 ± 38.6 vs. 23.3 ± 22.04, P ≤ 0.001) and Ca (2.36 ± 0.15 vs. 2.47 ± 0.15, P ≤ 0.001) were increased significantly by the end of the study compared with at the baseline. The high-dose vitamin D supplementation resulted in a significant reduction in serum TC (4.2 ± 0.72 vs. 4.02 ± 0.67, P < 0.001), LDL-C (2.6 ± 0.63 vs. 2.4 ± 0.53, P < 0.001) and FBG (4.8 ± 0.65 vs. 4.7 ± 0.54, P < 0.001). We did not find any significant differences in serum concentrations of phosphate, TG and HDL-C before and after supplementation.
We adjusted the effects of vitamin D supplementation on anthropometric and biochemical measurements (adjusted for age, energy intake, dietary intake of vitamin D, menstruation, use of sunscreen, passive smoker, sun exposure, BMI and physical activity). We did not obtain any significant differences between the crude and adjusted model. After adjustment, the differences between before and after supplementation values for DBP, HR, WC, 25-OH D, Ca, TC, LDL-C and FBG, remained statistically significant. No significant differences were observed for other variables including SBP, phosphate, HDL-C and TG (Supplementary Table 2).
Discussion
We analysed data from a large interventional study with the purpose of determining whether high-dose vitamin D supplementations have beneficial effects on cardio-metabolic risk factors. Taking vitamin D supplements appeared to have beneficial effects on DBP, HR, serum 25-OH vitamin D, Ca, TC, LDL-C and FBG. The prevalence of vitamin D deficiency was 90% at baseline, while it was decreased to 16.3% after intervention. To the best our knowledge, this study is one of the first of its kind in the adolescent girls group. Taking high-dose 50,000 IU-vitamin D for eight weeks is recommended for vitamin D deficiency. 24 In our study, we prescribed nine high-dose vitamin D pearls (50,000 IU/week cholecalciferol) over a period of nine weeks. At the end of the study, the mean of 25-OH vitamin D was raised to 90.9 nmol/L; it has been suggested that the health benefits of vitamin D are seen for serum a 25-OH vitamin D of between 75 and 100 nmol/L. 25
In our study, vitamin D supplementation improved serum 25-OH vitamin D. Similar results were found in relation to the effect of vitamin D supplementation on serum 25-OH vitamin D in previous studies.26–28 It has been reported that serum 25-OH vitamin D can be increased by approximately 1.5 to 2.5 nmol/L for every 100 IU of vitamin D ingested. 29 When the serum 25-OH D is less than 37.4 nmol/L, it is expected that serum 25-OH vitamin D would increase by 5 to 7.5 nmol/L. 1
An inverse association has been reported between serum 25-OH vitamin D and obesity in previous studies 3 ; also a recent meta-analysis confirmed that low concentrations of vitamin D are associated with higher concentrations of BMI. 30 We observed that treatment with vitamin D supplements was associated with an improvement in WC in our population. It is possible that this was associated with changes in diet over the intervention period. However, higher serum concentrations of PTH are associated with increasing lipogenesis and decreasing lipolysis. PTH reduction following vitamin D intake, might be result in an improvement in some anthropometric indexes. 31 Vitamin D can also reduce adipogenesis through reduction in the expression and activity of peroxisome proliferator-activated receptor-gamma in adipocytes. 32
We found that vitamin D supplements were associated with a significant reduction in DBP, but no effect on SBP. There is some evidence that vitamin D supplements may improve blood pressure,33–35 although other studies using relatively short treatment periods or low doses of vitamin D supplements have reported no significant effects 36 particularly in individuals with sufficient serum concentrations of 25-OH vitamin D before supplementation. 37 A meta-analysis has reported a reduction in SBP of 2.44 mmHg in vitamin D-treated subjects, but no significant effect on DBP. 38 Wamberg et al. 39 have reported that higher dose of vitamin D supplementation did not have a significant effect on blood pressure, although the results of their study may have included a small sample size. 39 The control of renin–angiotensin system by decreasing renin gene expression and regulation of parathyroid hormone (PTH) production by parathyroid cells has been suggested as one of the biological mechanisms for the effects of vitamin D on blood pressure.40,41 Moreover, vitamin D through increment of calcium absorption improves blood pressure via altering cellular concentrations of sodium and calcium ions. 42 Further randomized controlled trial studies are needed to clear the actual effects of vitamin D on blood pressure.
Vitamin D deficiency is identified as a risk factor for CVD. 1 We found that taking vitamin D supplements caused a significant reduction in HR. This finding is in agreement with a previous study on healthy subjects. Vitamin D deficiency may be associated with a suppression of resting cardiac autonomic activity. 43 Parasympathetic nerve fibres or vagus nerve are known as regulators of the HR; activity of these nerves is related to slow the HR. 44 In individuals with low level of vitamin D in the serums, cardio-protective vagal tone declined in response to an acute vascular stressor 45 ; it seems that 1,25-dihydroxy vitamin D may act as an important mediator in reducing vagal tone and therefore HR. 46
We observed that vitamin D supplementation led to significant change in serum calcium, but not in serum phosphorus. Mozaffari-Khosravi et al. 34 confirmed our findings in terms of effects of vitamin D on serum calcium and phosphorus. The absorption of calcium and phosphate is increased by vitamin D through various pathways. 47 Intake of excessive vitamin D can cause hypercalcaemia and hyperphosphataemia, 48 but this may be related to baseline status.
Vitamin D deficiency is considered as a potent risk factor for the development of impaired glucose metabolism and type 2 diabetes. 49 Vitamin D deficiency or insufficiency has been reported to cause a 2–5-fold higher risk of enhanced blood glucose concentration in children. 50 We found that vitamin D supplementation led to a significant reduction in FBG. Several studies have demonstrated a favourable effect of vitamin D on glycaemic control,51,52 while some others could not find any significant effect53,54 which may be due to small sample size, normal FBG of study participants at baseline or relatively short treatment period.
In has been reported that calcium–vitamin D co-supplementation resulted in a significant reduction in FPG, serum insulin concentrations and HOMA-IR. 55 Nikooyeh et al. found that vitamin D alone and vitamin D plus calcium yogurt drink caused a reduction in HOMA-IR, FPG and haemoglobin A1C in individuals with type 2 diabetes. 56 The exact mechanisms that are involved in terms of effect of vitamin D in glucose metabolism are unclear. Vitamin D supplementation may improve glucose metabolism via more production of 1, 25-dihydroxy vitamin D, which, in turn, leads to increased expression of insulin gene and then enhanced insulin action, synthesis and release. 57 Moreover, low 25-OH vitamin D concentrations may be associated with an increased production of PTH, which has been related to insulin resistance. 58 Furthermore, improved calcium status and increased local production of 25-OH D may result in higher insulin sensitivity. 59
Vitamin D status is known as an important factor in the pathogenesis of CVD. Serum 25-OH D and 1, 25-dihidroxy vitamin D concentrations are inversely associated with the presence of coronary artery diseases.
In present study, a reduction in serum TC and LDL-C were associated with vitamin D supplementation, but no significant effect was observed for HDL-C and TG. In line with our findings, some studies have previously demonstrated significant beneficial effects on lipid profiles,60,61 while others did not report any improvement.55,62 These inconsistent results may be due to different characteristics of population, study design, difference in dosage supplementation and confounder variables. The mechanism of the effect of vitamin D on lipid profiles is largely unknown. Vitamin D intake can improve lipid profile by reduction in PTH concentration. 63 Increment of insulin sensitivity is considered as one of the potential mechanisms in relation to the effects of vitamin D on lipid profiles. Insulin decreases the biosynthesis of cholesterol via increased β-hydroxy-β-methylglutaryl coenzyme A reductase activity. 64 It has also been proposed that vitamin D might correct the lipid profile via increasing calcium absorption. 65 It seems that long-term interventions are required to show the effects of vitamin D supplementation on lipid profiles.
The main strength of the present study is the large sample size for intervention. The next strength of the present study design was that it was performed in apparently healthy adolescent girls aged 12–18 years. However, some limitations need to be considered in the interpretation of our findings. Also, we were unable to measure PTH in our population, which supports the need for the evaluation of this marker before and after vitamin D supplementation. Owing to advice of our ethics committee, we were not able to have a control group in the present study. The relatively short duration of supplementation was another limitation in our study. The short intervention and bolus dose may have resulted in some of the null effects obtained in this study compared with previous studies of vitamin D supplementation.
In conclusion, high-dose vitamin D supplementation with 50,000 IU/week for nine weeks in apparently healthy adolescent girls led to improvement in WC, DBP, HR, 25-OH D, FBG, LDL-C, TC, Ca, but it did not affect BMI, SBP, TG, HDL-C and phosphate. Future clinical trial studies are recommended to clear the effects of vitamin D supplementation on cardio-metabolic risk factors in adolescents.
Footnotes
Acknowledgements
The authors are grateful to all study participants, volunteers and study personnel.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was jointly funded by the Mashhad and Sabzevar University of medical sciences. We appreciate Al Zahra University for providing a part of funds.
Ethical approval
The ethical committee of Mashhad University of Medical Sciences approved the study (IR.MUMS.fm.REC.1395.12).
Guarantor
MGM.
Contributorship
The current manuscript was produced from a large study and therefore several persons were involved in this project. The paper was drafted by SSK with contributions from all authors. SJM, MGM, GAF and HBT designed the study; AA, PH, ZA, MF, MT and SSK participated in field implementation and sampling. Also, AA, MGM, ZA, HBT were involved in clinical examination and patient confirmation. FR and AJ performed biochemical analysis. SSK and MT contributed to statistical analyses. MGM and HBT supervised the study. All authors contributed to the development of, and read and approved the final version of, the manuscript.
