Abstract
Background/aims
The addition of further tests to patient samples, whether by reflex (automated) methods or reflectively after result review, remains an important function of the diagnostic laboratory. This can assist clinicians with interpretation of results and aid further management. We aimed to investigate how this is performed within Clinical Biochemistry laboratories across the United Kingdom (UK).
Methods
A questionnaire, consisting of 16 clinical scenarios, was circulated to Clinical Biochemistry laboratories across the UK via the Association for Clinical Biochemistry and Laboratory Medicine (ACB) office. The survey sought to examine opinion on what tests are considered reflex and reflective, which staff members are involved in addition of further tests, whether clinicians are contacted and how abnormal results are handled.
Results
Responses were received from 74 laboratories, and data were compiled for each scenario surveyed. The majority of laboratories are adding calcium and magnesium, reflectively, to confirm hyperkalaemia or hypocalcaemia, and a number of additional endocrine tests are also commonly performed in particular scenarios. However, there are variations in what is regarded as reflex and reflective testing, in how this testing is conducted within the laboratory and in the level of communication with the requesting clinician.
Discussion
We present the consensus views of the survey participants for a number of commonly used additional tests within Clinical Biochemistry and a discussion on current reflex and reflective testing practice based on the survey results and evidence, where it exists. There is a need for harmonization and national guidance in this area.
Introduction
In laboratory medicine, there has been increasing focus on governance in the preanalytical and analytical aspects of service delivery, particularly since the introduction of ISO15189 standards, and there has been a drive to show ongoing improvements in these areas. However, a key area where laboratories can provide further ‘added value’ is in the postanalytical phase where additional testing is actioned by laboratory professionals. This is regarded as an important function of the diagnostic laboratory to assist with an interpretation of a patient’s results to aid diagnosis, management or further investigations. 1 However, despite the importance of this role, it is an area which is not well established or defined, where practice relies on individual judgments, resulting in significant variations in practice. 2
Two main methods are employed for the addition of further tests to a sample in Clinical Biochemistry laboratories. Recent advances in laboratory information systems and automated analysis have allowed rules and algorithms to be developed to add tests automatically if a number of conditions are met. Such ‘reflex’ testing is commonplace, where automated rules are used to assist with the identification of critical results, expediting the patient journey. 3 ‘Reflective’ testing is a procedure where the laboratory specialist adds additional tests. 4 , 5 This is a more complex process incorporating clinical judgement and review of previous results, if available. Previous studies in this area have shown that requesting clinicians appear in favour of the concept of reflective testing. However, it was noted that there was likely to be interindividual variations in how this was performed. 6 Although it was not the main focus of this survey, reflective testing is also often associated with the addition of interpretative comments by senior clinical staff, which are written comments added to laboratory reports which provide guidance on result interpretation to the requesting clinician. 1
Previously, it has been noted that that there is little information about the range of reflective testing practised in different laboratories and that formal advice on the matter is missing. 4 , 7 The Association for Clinical Biochemistry and Laboratory Medicine (ACB) published best practice guidelines for providing interpretative comments on laboratory medicine reports in 2014, 8 which detailed guidelines for the standardization of interpretative comments and provided some guidelines on the ethical issues surrounding reflective testing. Certainly, both patients 9 and requesting clinical colleagues 6 appear to be broadly comfortable with the concept of tests being added unprompted. However, if the reflective test could lead to the identification of a disease not originally considered by the requestor or unrelated to the initial test, then consent should be sought from the patient, usually via the requesting clinician. Reflex and reflective testing form part of a wider spectrum of activity that ‘add value’ to high-volume processing of laboratory samples. The NEQAS Interpretative Comments scheme enables assessment of the practitioner against best practice, and peers, but this is largely a result interpretation scheme.
This survey sought to assess the practice in UK clinical laboratories of common reflex and reflective tests added to routine samples within clinical biochemistry and what level of variation in practice currently exists. In addition, we aim to provide some discussion of the findings, in line with the current evidence base.
Methods
The questionnaire consisting of 16 clinical scenarios, each involving the addition of a specific test, designed by DBF and GCM, is shown in Table 1. The scenarios chosen were ones which the authors had commonly experienced in their practice. The questionnaire examined whether the test would be reflex or reflective and, if reflective, what staff group added the additional test. Also, the questionnaire sought information on whether the requestor was contacted before addition of the test, whether comments were added to the report and also whether the requestor was telephoned with the result.
The clinical/laboratory scenarios in the questionnaire (listed by their question number).

Note: Question 1 asked for the respondent details. For full questionnaire, please see online supplementary material.
EDTA: ethylenediaminetetraacetic acid; (F)T4: free thyroxine; TSH: thyroid-stimulating hormone; HCG: human chorionic gonadotrophin; LH: luteinizing hormone; FSH: follicle-stimulating hormone; TPO: thyroid peroxidase; (F)T3: free triiodothyronine; PSA: prostate-specific antigen; ALP: alkaline phosphatase; HFE: hemochromatosis gene.
The questionnaire was distributed electronically, using SurveyMonkey®, on 11 November 2016 to 208 hospital laboratories within the UK. Using the ACB members database and with assistance from the ACB Office, the survey was sent to Heads of Department or, if this information was not available, to the most senior Clinical Scientist/Chemical Pathologist in each laboratory. It was requested that only one response per hospital was submitted. A reminder to complete the survey was sent to recipients on 8 December 2016 and a final reminder was sent on 14 December 2016. The survey was closed on 16 December 2016. Results from returned questionnaires were collated electronically onto an Excel spreadsheet by SLH.
Results
The survey was sent out to 208 laboratories from whom 66 responses were returned. We attempted to improve the response rate by re-circulation of the survey on two occasions, and the final response rate was 32%. Some of the responses covered more than one laboratory, and the 66 responses represented 74 individual laboratories. A summary of the responses for each survey question are detailed in Table 2.
Summary of results for Questions 2–17 of the Reflex and Reflective Testing Survey.
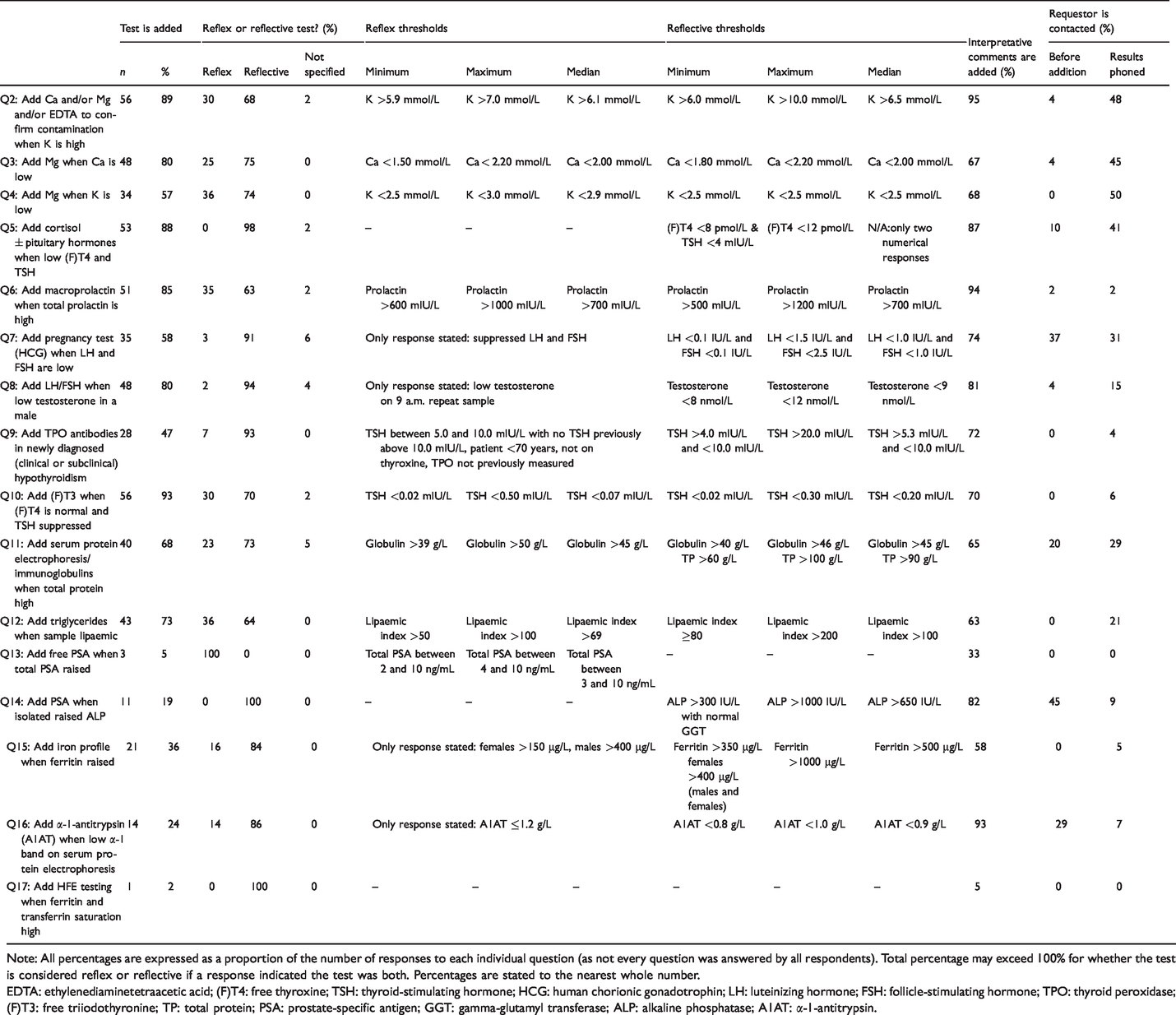
Note: All percentages are expressed as a proportion of the number of responses to each individual question (as not every question was answered by all respondents). Total percentage may exceed 100% for whether the test is considered reflex or reflective if a response indicated the test was both. Percentages are stated to the nearest whole number.
EDTA: ethylenediaminetetraacetic acid; (F)T4: free thyroxine; TSH: thyroid-stimulating hormone; HCG: human chorionic gonadotrophin; LH: luteinizing hormone; FSH: follicle-stimulating hormone; TPO: thyroid peroxidase; (F)T3: free triiodothyronine; TP: total protein; PSA: prostate-specific antigen; GGT: gamma-glutamyl transferase; ALP: alkaline phosphatase; A1AT: α-1-antitrypsin.
Two of the first survey questions (Q2 and Q4) dealt with deranged potassium results (both high and low) and how these would be further investigated. The majority of laboratories considered additional testing in these cases as reflective (68% and 74% for Q2 and Q4 respectively), with analysis of calcium and/or magnesium being performed at the point of clinical validation. Various thresholds were quoted as a trigger of further testing in hyperkalaemia, commonly >6.5 mmol/L, with all laboratories using a threshold potassium concentration of <2.5 to 3 mmol/L for reflexing magnesium in hypokalaemia. The addition of serum magnesium when investigating hypocalcaemia (Q3) is commonly performed when calcium or adjusted calcium concentration is <2.0 mmol/L, and variably this is reflectively added at the point of clinical validation (75%). It is not felt necessary to contact the patient’s clinician before proceeding with such reflective testing in these scenarios, although between 45% and 50% of laboratories will contact the requestor with the results.
A number of endocrine-based questions were provided for comment in survey questions 5 to 10. The majority of laboratories’ thresholds for the addition of cortisol to rule-out hypopituitarism are based on when both free thyroxine ([F]T4) is below the lower limit of the reference interval and thyroid-stimulating hormone (TSH) is low or within the reference interval. All respondents considered this to be a reflective approach with the vast majority adding interpretative comments, such as ‘Suggest endocrine referral’, and some (41%) communicating the results to the requestor.
Most laboratories will add macroprolactin analysis, if this has not been undertaken previously, when the serum prolactin is >700 U/mL, although there are some higher prolactin thresholds in use. Almost all laboratories (94%) add interpretative comments to such reports. Interestingly, macroprolactin analysis is added both reflectively and by reflex testing. Only 58% of laboratories actually added a pregnancy test (human chorionic gonadotrophin, HCG) with a finding of suppressed follicle-stimulating hormone (FSH) and luteinizing hormone (LH), with the majority being added reflectively by a senior clinical professional (Clinical Scientist/Medical). The thresholds for such testing are suppressed LH and FSH results (lower limit of detection for LH and FSH) and are dependent on clinical information (e.g. if not on oral contraceptive). The majority add interpretative comments to the reports, and some laboratories (37%) will make contact with the requestor before performing such additional testing.
Most laboratories surveyed (80%) add LH/FSH when testosterone is below the reference interval on a morning sample (9 a.m.) and sex hormone-binding globulin (SHBG) is added where available in the repertoire. Almost all of these consider this a reflective test and 81% add interpretative comments to the reports. Less than half (47%) of laboratories added antithyroid peroxidase (TPO) antibodies on newly diagnosed clinical or subclinical hypothyroidism and the majority were added reflectively. A range of TSH thresholds were quoted for prompting such additional testing (Q9, Table 2). The majority of laboratories add free triiodothyronine ([F]T3) if patient is not on thyroxine or any other medication which suppresses TSH, and/or the value for TSH is less than reference interval with a (F)T4 that is within reference interval. Again, as with the other endocrine scenarios, interpretative comments are added to the majority of such reports.
The latter part of the survey included some questions where additional testing can be used to assist with the diagnosis of malignancy (Q11, 13–14) or the detection of important conditions such as α-1-antitrypsin (A1AT) deficiency (Q16), haemochromatosis (Q15, Q17) or lipid disorders (Q12). The addition of serum protein electrophoresis (SPE) testing was a common reflective test, with laboratories using thresholds based on total protein (TP) and globulin results that range from >60 to >100 g/L and >40 to >46 g/L, respectively, with raised TP >90 g/L being most commonly applied. Interpretative comments are added by 65% of laboratories, although only a small number of laboratories noted that they would contact the requesting physician. Only a small number of respondents noted the use of free prostate-specific antigen (FPSA) testing in their laboratory alongside total PSA (TPSA) and all considered such additional testing as reflex. Different TPSA thresholds ranging from 2 to 10 ng/L were noted for this, some with age restrictions (<75 years). No contact with the requester and no communication of results were noted. The addition of TPSA analysis in the presence of elevated alkaline phosphatase (ALP) was slightly more common, yet only conducted by 19% of respondents. Only 45% of these would contact the requestor before the reflective addition of this test. The majority who responded had no set reflective threshold (>300 to >1000 IU/L were quoted) and noted that it would be performed at the discretion of senior clinical staff after review of clinical information.
A number of those who responded (73%) will perform additional triglyceride testing in lipaemic samples, with the majority considering this a reflective approach. The decision limits ranged from visible lipaemia to cut-offs set by manufacturer or grossly lipaemic samples, when a number of other test results are affected. Some laboratories (63%) add interpretative comments, and the vast majority do not contact the requestor. The addition of a full iron profile in the presence of a raised ferritin is performed by 36% of laboratories who responded, with the majority also considering this a reflective test. No laboratories contact the requester before performing the test and 58% add appropriate comments to the report. It was noted that this would be performed by medical haematologists in some laboratories. Various ferritin action limits were noted by laboratories, ranging from >150
Finally, 24% of those who responded noted that they would add A1AT testing when a low alpha-1 band is detected during protein electrophoresis, with the majority of these laboratories considering this reflective. Only 29% of these would contact the requester before performing A1AT analysis. Decision thresholds were based on clinical information, although limits ranging from < 0.8 to 1.0 g/L on the alpha-1 region or low/absent alpha-1 band were quoted.
Discussion
In the recent publications about harmonization in laboratory medicine, it has been emphasized that despite years of advances in laboratory quality, which has been primarily focused on the analytical phase, we are still struggling to harmonize significant areas of the total testing process, which includes areas such as reference ranges, report formats, decision limits and criteria for interpretation. 10 , 11 The practice of adding on tests is widespread in the UK; however, this survey has shown that significant variation exists in how this is performed and whether additional testing falls under reflex or reflective testing. Here, we aim to provide some discussion and proposals based on the various scenarios surveyed and the current evidence base around these topics.
A number of laboratories have automated algorithms in place for reflexing tests to rule out contamination, such as EDTA. It seems a sensible approach, if suitable IT infrastructure is in place, to allow such offline calcium or magnesium testing to flag potential problem samples. The majority of those who responded take a more reflective approach to this, where staff review the result, taking into account previous results, and make such decisions. Where EDTA, or other additive, contamination is found, a comment should be added to the report stating suspected contamination of the sample and requesting a repeat sample. It is important to assess what other biochemistry results may be influenced by EDTA contamination, as per assay manufacturer sampling recommendations, and ensure that these are also removed from the final report.12–15 Laboratories should also have a standard operating procedure (SOP) to guide those reporting on interpretation.
The addition of magnesium testing in the presence of hypocalcaemia (Q3) is useful; however, normal serum magnesium (total or ionized) concentrations must not be used to exclude deficiency, since magnesium is predominantly an intracellular moiety. 3 , 16 , 17 It could be argued that implementing a reflex testing strategy for magnesium testing in hypokalaemia (Q4) may be a more standardized approach and may lead to an increased absolute detection of hypomagnesaemia. Certainly, Jones and Twomey demonstrated that both reflex and reflective testing had similar ‘number of add on tests needed to obtain a diagnosis’ (NNDs), and potentially lead to a reduction in delayed detection of aetiology and correction of hypokalaemia. 18 Across those surveyed, magnesium is predominantly added reflectively in these scenarios where consideration is given to previous results and other relevant test results (such as parathyroid hormone [PTH] and vitamin D) before additional testing is performed. Relevant comments should be added to reports to highlight that the hypocalcaemia may be due to hypomagnesaemia and, if a viable sample still exists, one should consider adding PTH and vitamin D, where these have not previously been tested. Significantly abnormal results should be communicated as appropriate. 3 , 16 , 17
Many of the laboratories surveyed are engaged in reflex and reflective testing strategies involving endocrinology and endocrine tests. It is interesting that while the published thyroid guidelines 19 point out the likely pattern of thyroid function tests (TFTs) in central hypothyroidism and suggest that referral to endocrinologists for further pituitary function tests (prolactin, FSH, LH, cortisol) may be required to make a diagnosis of hypopituitarism, they do not suggest laboratory initiated reflective testing. A recent study by Elnenaei et al. has highlighted the clinical value of interventions by laboratory staff, by reflective addition of pituitary hormone testing. This aided the early detection of pituitary dysfunction, thereby reducing the risk of delayed management. 20 Further work on this area has focused on using a TSH-only initial approach to thyroid testing, followed by reflex testing for (F)T4, where TSH is abnormal. The TSH cut-offs used for such reflex testing have been discussed, with no clear obvious consensus, although a recent study has shown that applying TSH cut-offs wider than the TSH reference range for reflex (F)T4 testing can reduce the number of (F)T4 tests performed, with minimal impact on detection of overt hypo- and hyperthyroidism. 21 Biochemistry laboratories should have a formal SOP in place for further investigation of abnormal TFTs and need to consider having an SOP for reflex testing, especially where a TSH-only approach is in use. The quality of clinical details given on the request form are extremely important, and a TSH-alone approach is not appropriate for certain patient groups with known or suspected pituitary disease or established thyroid disease. 21
Cortisol plus or minus anterior pituitary hormones are added reflectively in all laboratories surveyed (Q5), and interpretative comments should be added. Each laboratory may need a policy agreed with their Endocrinology Department, which may include referral of particular laboratory reports if the general practitioner desires, and results should be telephoned to the requestor. 19 , 22 , 23
Current best practice recommends that serum samples with elevated total prolactin concentrations with subfractionality using polyethylene glycol precipitation provide a more meaningful clinical measurement of the bioactive monomeric prolactin content (Q6). Clinical laboratories should introduce screening procedures to exclude macroprolactinaemia in all patients identified as having hyperprolactinaemia,24–26 with the exception of known cases of prolactinoma. It is important for patients with elevated monomeric prolactin concentrations to undergo routine evaluation for hyperprolactinaemia to identify the exact pathological state and introduce adequate treatment regardless of the presence of macroprolactin. Suitable comments should be added to reports to highlight why this testing has occurred and where abnormal macroprolactin results are reported. 27 , 28
Regarding testing for pregnancy (Q7), only some laboratories would make contact with the requesting clinician in advance. It is felt that best practice should be that the requestor is contacted when a positive pregnancy test result has been obtained. 29 There are potential ethical issues with the addition of such testing, and this should be given due consideration in any laboratory policies. Suitable comments should be added to highlight the reason why LH/FSH have been added in the presence of a low testosterone in males (Q8), and significantly abnormal results should be communicated as appropriate to the requestor. It is good practice to withhold testosterone results until LH/FSH results are available. 29 Interpretative comments linked to the reflective addition of anti-TPO antibodies in new clinical or subclinical hypothyroidism (Q9) should be in accordance to the ACB and British Thyroid Association guidelines regarding repeat interval and progression. 19 , 22 , 30 , 31 (F)T3 may be added either reflectively or by reflex testing (Q10), and the clinical interpretation depends on the other results and clinical details. If (F)T3 is high, endocrine referral should be suggested.
The addition of SPE testing when elevated TP and/or globulin fraction is detected (Q11) is a common example of additional reflective testing that can potentially lead to the uncovering of a disease not originally considered when the sample was drawn. The value of such testing has been discussed previously noting the ethical dilemmas and that more data on the clinical usefulness of discretionary requesting of SPE in this situation were needed. 32 A previous audit reviewed screening criteria (or cut-off) of globulin concentrations >45 g/L and noted that one in 20 samples where additional testing was performed produced a clinically relevant finding. The arguments for and against such discretionary reflex addition of SPE in this scenario have been discussed. 32 , 33 It is difficult to recommend such routine reflex testing without review of the larger clinical picture. Additional reflective SPE testing should be considered in the presence of raised TP and/or globulins where a B-cell malignancy is suspected, especially if no SPE has been performed in the preceding year, and there is no evidence of infection, liver disease or inflammation. A decision limit of TP >90 g/L or globulins >45 g/L could be used. It is recommended that that there should be consultation with the patient’s requesting clinician before proceeding with this reflective testing. 8 A suitable comment should be added to highlight the reason for SPE testing (e.g. raised TP/globulins), who approved by, and abnormal results should be communicated as appropriate, where haematology review is indicated. The finding of a low or absent alpha-1 band during SPE should prompt consideration of likelihood of undiagnosed A1AT deficiency, where an early and correct diagnosis is important to allow for effective disease management, including appropriate lifestyle modifications and therapeutic options. 34 , 35 Performing A1AT testing is important, although the requestor and patient (through the clinician) need to be contacted in advance to highlight that a potential genetic diagnosis may be identified. Depending on how the current sample has been handled, a suitable comment could be added to the report to suggest that A1AT testing should be performed in a repeat sample.
With regard to the analysis of triglyceride concentrations in the presence of lipaemia (Q12), commonly the degree of lipaemia does not correlate directly with triglyceride concentration, and the relationship is non-linear and complex. Iatrogenic factors are the most common cause of severe lipaemia, including lipid-containing medications used for parenteral nutrition and antidotes for poisonings. 36 Few guidelines exist for performing such testing. There may be value in the reflex assessment of triglyceride in grossly lipaemic samples; however, consideration may be given to other test results (such as liver function tests [LFTs], glucose, amylase) before deciding to report or follow-up with the requesting clinician. For automated methods measuring a lipaemic index, an action threshold that equates to a clinically significant triglyceride concentration could be used. Suitable comments should be added, and significantly raised triglyceride results should be communicated as appropriate, depending on review of clinical risk.
Question 13, reflexing of FPSA when TPSA elevated, is not commonly performed, although this may be linked to the demographic of those who responded. As this was considered a reflective test by all respondents, such FPSA testing could be performed in those presenting with TPSA between 4 and 10 ng/mL and no recent PSA result and an FPSA <20% requires further investigation for the possibility of malignancy.37–40 Due to the stability challenges for FPSA (1 day at 2–8°C), a fresh sample should be considered. 41 In situations where PSA testing is being considered due to the presence of an isolated raised ALP (over twice the upper reference limit) in a male (Q14), thought should be given to the age of the current sample and whether a fresh sample for TPSA analysis should be collected. Such reflective testing should be based on the likelihood of prostate cancer and appropriate clinical review by a senior clinical professional, and prompt a discussion with the patient’s physician where the addition of PSA analysis should be discussed. 42 , 43
Iron profile testing when ferritin is elevated (Q15) is a reflective test based on the clinical likelihood of a patient having haemochromatosis, which should not be missed.
44
A recently published report
45
has highlighted the value of the reflective addition of iron studies based on elevated serum ferritin (>291
It is imperative for laboratories to involve clinical colleagues in the establishment of reflex algorithms as has previously been stressed. 50 Although reflex and reflective testing is considered to be a useful way to improve the process of diagnosing (and treating) for both General Practitioners and other clinicians, 4 there is no consensus yet on the point when additional testing should be indicated, for which tests, or for what type of results. 51 In addition, no quality indicators (QIs) or performance criteria in additive testing have been set. 52 The ACB best practice guidelines for providing interpretative comments on laboratory medicine reports is one of the few publications that aims to provide some guidance on how to conduct additional testing. If the reflex test can lead to the identification of disease not originally considered by the requestor or unrelated to the initial test(s), then consent should be sought from the patient, usually via the test requestor. 8 However, from this survey, it is evident that a number of laboratories will perform some further testing with limited clinical liaison. This could be an area which is targeted for improved harmonization.
As Ian Young stated, ‘reflective testing is a valued part of the practice of clinical biochemistry and as a profession, we should seek to educate clinical colleagues and patients about this aspect of work’. 7 He cited that we ought to ‘benchmark’ reflective testing, and to some extent, this is what this survey has undertaken, but we do need to establish some standards by which we can judge whether this practice is clinically useful and cost-effective and, importantly, ethically acceptable. Laboratory professionals need to ensure that they are not going beyond what the patient has given consent to or indeed been made aware of. In such cases, there should be further discussion with the clinician before the test is performed. 53
With regard to clinical effectiveness, the NND has been used. Previous studies have found favourable NNDs for both reflex and reflective testing in scenarios such as diagnosis of vitamin D deficiency but less so for the diagnosis of HH. 3 It has been highlighted that the efficiency of both reflex and reflective testing depends critically on the reflex thresholds applied. The use of ‘tight’ thresholds will detect most or all of the diagnoses, at the expense of efficiency (leaving reflective testing largely unnecessary), whereby the use of ‘looser’ thresholds will favour efficiency, at the cost of effectiveness. Reflective testing, by vigilant laboratory staff, is then required to pick up some of the missed diagnoses. 54 The NND will be influenced by thresholds used for reflex testing and the individual reporting practice in reflective testing. The selection of thresholds, ideally, should be evidence based, but unfortunately, this evidence rarely exists. It is thought that reflex testing is best suited to those scenarios where there is high efficiency (i.e. where a low NND can be readily achieved). However, the contribution of reflective testing is comparatively greater where more complex factors need to be considered. 3 In such circumstances, the quality of clinical details provided by the requestor, and access to further test results, would have a major influence on the NND and the more information, the more added value can be provided. 5 A recent article demonstrated that implementing reflex testing of direct bilirubin in infants with biliary atresia costs New Zealand the equivalent in US Dollars of $3200 for each newly diagnosed case of biliary atresia, compared with an estimated cost of $175,000 for paediatric liver transplant with an annual cost of $14,000. 55
All laboratories need to ensure that they are performing further tests in a timely manner and that samples have been stored appropriately for optimum sample stability. This can be a challenge in a busy laboratory where fluctuations in workload and analyser downtime/maintenance schedules can lead to situations where samples are spending excessive periods at room temperature (instead of refrigerated). In some cases, it may be necessary to ask for a fresh sample if result quality cannot be guaranteed, and a reflective test is deemed essential. In all laboratory handbooks there should be a statement regarding reflex and reflective testing with a procedure to support these. This may have to be discussed with the client clinicians. It is evident from the survey responses that senior clinical laboratory staff (clinical scientist and medical) are those mostly involved with deciding on when to implement further testing, with the exception of ruling out sample contamination (undertaken by Biomedical Scientists) (Q2). Automated (reflex) methods are used for additional testing in Q2, Q10 and Q12, as well as reflective methods. It is evident that there is variation between laboratories on how to conduct this practice of adding value with no clear consensus on what testing can be performed automatically and what requires a more subjective approach. Ideally, we need national guidance in the UK to try to obtain consensus opinion and use of evidence of best practice where it exists. This was certainly the view of the respondents of this survey. Perhaps, this could be approached by the profession in the same way as the ‘National Minimum Retesting Interval’ project was managed. 30 There may implications for other healthcare settings outside the UK with regard to additional testing policies, which would be an area for discussion within those individual health economies.
We acknowledge that the low response rate is a possible limitation to this work. As we asked for only one response from each hospital, it is possible that, as more laboratories are now working across networks with shared policies and procedures, we have captured a greater snapshot of current practice than the statistics suggest. Our response rate of 32% is identical to that obtained by Darby and Kelly. 6 In addition, we acknowledge that there may be local IT limitations that prevent a preferred reflex approach to some of the scenarios surveyed and that some of the additional tests may fall under the responsibility of Immunology laboratories (e.g. anti-TPO and SPE), some of which may not have been captured with this survey.
In conclusion, reflex testing appears to be relatively well established in the UK, and reflective testing should be seen as a new dimension in the service of the clinical chemistry laboratory, both to primary and secondary care. However, there is a need for additional research to deliver further scientific proof of the clinical effectiveness for patient management. It is hoped that the European Federation of Clinical Chemistry and Laboratory Medicine Working Group on the postanalytical phase will be addressing some of these issues. In the short term, it is hoped that some of aspects discussed here will allow the start of a more harmonized approach to this important clinical utility for effective patient care.
Supplemental Material
ACB888541 Supplemental Material - Supplemental material for Reflex and reflective testing practice in Clinical Biochemistry in the United Kingdom – a national survey
Supplemental material, ACB888541 Supplemental Material for Reflex and reflective testing practice in Clinical Biochemistry in the United Kingdom – a national survey by Gareth C McKeeman, Sally L Hall and Danielle B Freedman in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We would like to thank Mike Lester at the ACB office for his assistance, Dr David Housley for reading the article and, importantly, all the respondents of the survey. This survey was commissioned on behalf of the ACB Clinical Practice Section.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
DBF.
Contributorship
DBF and GCM designed the survey and SLH collated the survey findings. Initially, DBF wrote the review for survey responses 2 to 10 and GCM wrote the review for survey responses 11 to 17. GCM drafted and edited the final article and all authors had input into layout and content of the final version.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
