Abstract
Background
The aim of this work was to determine the frequency of ascorbic acid-positive urine samples and to evaluate the potential risk on patient safety.
Materials and methods
A total of 27,856 samples submitted for urinalysis in previous year were retrospectively analysed from the laboratory information system. Urinalysis was done on Iris IQ200 analyzer using 11-parameter iChem Velocity test strips. Risk analysis combined the impact of potentially erroneous results (due to ascorbic acid interference) on patient safety (severity – S) with their frequency (occurrence – O). Potential risk was identified as high, intermediate and low.
Results
One thousand one hundred and ninety-nine (4.3%) and 1646 (5.9%) samples were mildly (20 mg/dL, 1+) and highly positive (40 mg/dL, 2+) for ascorbic acid, respectively. Severity (S1 to S5) was assigned to four possible errors: false-negative glucose (U-Glc: S2), blood (U-Hb: S5), nitrite (U-Nit: S4) and bilirubin (U-Bil: S1). Based on the frequency of errors, occurrence was categorized as: O2 for U-Glc, and O3 for U-Bil, U-Nit and U-Hb. The risk analysis matrix revealed that false-negative U-Hb was associated with potentially high risk, false-negative U-Nit with intermediate risk, while false-negative U-Glc and U-Bil were associated with low risk on patient safety.
Conclusions
Positive ascorbic acid can cause potentially high risk for patient safety by missing positive blood in urine.
Introduction
Urine test strip analysis is one of the most commonly ordered tests in clinical chemistry. Even though limitations of this test are known, it is generally still considered a test with high negative predictive value. Negative results of proteins, leucocytes, nitrite and haemoglobin can pretty much exclude the existence of urinary tract inflammation or kidney disorder. In these cases, due to high workload and low risk of missing potential pathological elements in urine sediment, laboratories often do not perform sediment examination, 1 as proposed by the European urinalysis guidelines. 2 However, urinalysis results can be influenced by various preanalytical factors, including interferences.
Interferences are a major source of preanalytical errors in laboratory medicine. Presence of various exogenous or endogenous substances can interfere with the measurement and lead to false-positive or false-negative results.3,4 Interference of ascorbic acid (AA) on urine test strip analysis has been known for many years. 5 AA is a strong antioxidant which can cause false-negative results for analytes that are detected by peroxidase reaction. AA is eliminated with urinary excretion by simple first-order kinetics. 6 Bioavailability is complete after a single dose of 200 mg. At doses of 500 mg and higher, bioavailability decreases, which raises the amount of AA excreted in urine. Urinary excretion is detected even after a single dose of 100 mg, 7 and especially after ingestion of larger doses. Ko et al. have found that urine samples were positive for AA using urine test strip with a cut-off = 10 mg/dL 4 h after ingestion of 1000 mg of AA. 8 These results indicate that AA will be positive in most patients that are consuming increased doses of vitamin C during the flu and cold season. Therefore, clinical consequences of positive AA in urine have to be investigated.
Nowadays, manufacturers claim that test strips are completely or mostly resistant to AA interferences; however, our previously published work demonstrates that AA interference is still a major issue in urinalysis. 9 AA causes false-negative results for glucose, nitrite, bilirubin and blood for Combur-10M (Roche Diagnostics, Germany) and iChem Velocity (Iris Diagnostics, Germany) test strips. Intensity of interference differs significantly between manufacturers. 9 In order to maintain patient safety, all risks associated with laboratory testing have to be recognized and evaluated. Additionally, risk evaluation is a requirement for all laboratories accredited according to ISO 15189 standard. 10 Taking under consideration general increase of consumption of various vitamin supplements, 11 it is important to recognize patients that might be endangered by this practice.
Therefore, the aim of this study was to: (i) evaluate the frequency of positive urinary AA for inpatients and outpatient of a tertiary care hospital and to (ii) evaluate potential risk of positive urinary AA on results of urine test strip analysis.
Material and methods
Study design
This retrospective study was done at the Department of Clinical Chemistry of the Sestre Milosrdnice University Hospital Center, Zagreb, Croatia, by extracting data on patient gender, age, admittance (inpatient or outpatient) and results of urinalysis from the laboratory information system (LIS). All samples received for urinalysis from May 2017 to May 2018 were included in the study.
Methods
For urinalysis, samples were collected in VACUETTE® TUBEs 10 mL Z Urine No Additive, 16 × 100 yellow cap-yellow ring, Round Base, non-ridged (Greiner Bio-One, Kremsmuenster, Austria). Samples were analysed using the automatic analyzer Iris IQ200 (Iris Diagnostics, Chatsworth, USA) which includes modules for chemical test strip analysis (iChem Velocity Urine Chemistry System) and sediment examination (iQ200 Elite Urine Microscopy System). Chemical analysis was done using iChem Velocity urine test strips (Iris Diagnostics, Chatsworth, USA). This test strip performs an 11-parameter urine chemical analysis, including screening for AA in urine. AA reaction principle is based on Tilman’s reaction: in the presence of AA test-strip field changes colour from grey-blue to orange. Dehydroascorbic acid (oxidized form of AA) is not reacting with iChem Velocity test strip. Based on the manufacturers declarations, false-positive reaction for AA can be observed in the presence of 2-mercaptoethanesulphonate (MESNA) (1140 mg/dL), while pH of 9 can interfere with the test. Analytical sensitivity of AA is 17 mg/dL.
If urine test strip results were negative for blood, protein, nitrite, leukocytes and AA, sediment examination was not done. 1
Risk analysis
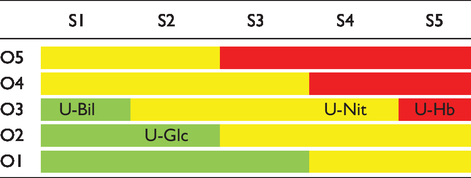
AA causes falsely negative results for glucose, blood, nitrite and bilirubin using iChem Velocity urine test strips. 9 With the exception of glucose, other parameters are affected at the lowest positive results for AA (20–40 mg/dL or 1+), while glucose is less sensitive to AA interference (> 40 mg/dL or 2+). Based on these results, four possible errors were identified due to AA interference: falsely negative glucose (U-Glc), blood (U-Hb), nitrite (U-Nit) and bilirubin (U-Bil). Each of these can lead to possibly erroneous medical decision. According to the possible harm that positive AA might cause to the patient, five different categories of severity (S) were identified ranging from the lowest (S1) to the highest (S5) possible impact. Severity was assigned by the consensus opinion of the authors of this manuscript: S1 for U-Bil, S2 for U-Glc, S4 for U-Nit and S5 for U-Hb. Frequency of errors (O – occurrence) was defined based on the results of the AA screening from the LIS data. Occurrence was also classified in five categories: O1 (<3%), O2 (3–10%), O3 (10–25%), O4 (25–50%) and O5 (>50%). Risk analysis was done according to ISO 14917 Medical Devices – Application of Risk Management to Medical Devices, 12 by combining the severity (S) and frequency of errors (O) in 5 × 5 matrix. Three types of potential risk for patient safety were identified: red area (highest risk), yellow area (intermediate risk) and green area (low risk).
Statistical analysis
Data are presented as count and percentages; age is presented as median and range. Comparison for qualitative variables was performed using the chi-square test. Difference in age according to results of AA was tested using Kruskal-Wallis test. Level of significance was set at 0.05. Analysis was done in Microsoft Excel (version 2010, Microsoft, USA) and MedCalc statistical software (v12.7.2.0, Ostend, Belgium).
Results
Urine test strip analysis was done for 27,856 samples in the investigated period. AA screening results were negative for 89.8%, mildly positive for 4.3% and highly positive for 5.9% samples (Table 1). Highly positive samples were more frequent in the inpatient than in the outpatient subgroup (6.1% vs. 4.4%,
Frequency of positive AA results and possible false-negative errors.
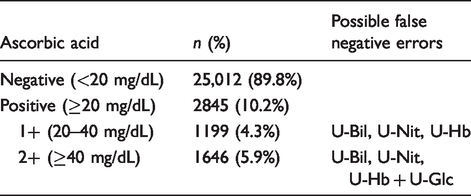
According to the frequency of positive AA in urine, categories of possible false-negative errors were O2 (5.9%) for U-Glc and O3 (10.2%) for U-Bil, U-Nit and U-Hb.
Potential risk was presented as combination of occurrence and severity (Table 2). False negative blood (U-Hb) was classified as an error with the highest potential risk, false negative nitrite (U-Nit) error with intermediate and false negative glucose and bilirubin (U-Glc and U-Bil) errors with the lowest risk for the patients’ health.
A 5 × 5 matrix for evaluation of potential risk as a combination of occurrence (O1–O5) and severity (S1 to S5) for false-negative results.
Discussion
This investigation proves that AA in urine may present potential risk factor for patients when blood in urine is evaluated. Due to the lower occurrence or severity, other affected parameters (nitrite, bilirubin and glucose) are associated with only moderate or low risk.
Urinalysis today continues to be a powerful tool in obtaining important medical information. Standard urinary test strip includes 10-parameter analysis (specific gravity, pH, proteins, glucose, ketones, bilirubin, urobilinogen, nitrite, blood and leukocytes) and mostly utilizes the same chemical reactions as at the time of the discovery.
13
Nitrite reaction is based on activity of nitrite reductase that is present in most gram-negative uropathogens such as
Based on our work, AA positivity in urine might result with severe risk of false-negative haemoglobin and moderate risk of missing nitrites. Missing blood in urine might seriously compromise patient’s health due to failure to diagnose renal diseases in very early stage, since negative result is probably not going to be investigated further. Negative nitrites might conceal
Usage of multivitamins and other dietary supplements has become growing trend among population. Generally, since vitamins are over the counter (OTC) preparations that can be bought without doctors prescriptions and are heavily marketed in media, people are often drown to increase consumption in order to boost their immune system. Additionally, vitamins are also prescribed as supportive therapy in various diseases and conditions. 16 In a large European study conducted recently, authors determined the frequency of consumption of various dietary products, vitamins or medicaments among the users of laboratory services. Vitamins were the most frequently used OTC product, percentage ranging from 20 to 58% of all surveyed outpatients in 18 European countries. When asked if it was important to disclose information about taking OTC preparations, patients were more inclined to report usage to their doctors than to laboratory staff (70 vs. 50%), leaving laboratory without important information on potential interference on laboratory test results. 11 In another study on preferences and attitudes toward the use of OTC drugs, authors have concluded that many OTC drug consumers did not always demonstrate responsible attitudes toward using these preparations, including informing themselves on possible effects on human health in case of high doses. 17 In this study also, vitamins were the most commonly used OTC preparations. Importance of disclosing data on usage of OTC drugs in medical charts has been emphasized by other authors, whereas lack of information is associated with a risk of overlooking interactions with prescribed drugs, especially in elderly patients. 18 This is especially true for laboratory measurements, since possible interactions and interferences cannot be recognized if the data are not available.
There are not many articles dealing with clinical consequence of positive AA results, even though high frequency of AA-positive urine samples has been a concern for a long time. In 1992, Brigden et al. have detected 22.8% urine samples positive for AA using test strip with cut-off of 37 mg/dL. 19 They have also discovered that even low doses of vitamin C or juices are sufficient to cause false-negative interference. Using URiSCAN 11 strips, Lee et al. have detected AA in urine samples of 18.1% of patients of their hospital (group undergoing medical check-ups 23.1%, inpatients 16.3% and outpatients 17.6%). 20 These results indicate higher frequency than discovered in our study; however, the threshold for positivity in their study was lower (10 mg/dL) than in ours (20 mg/dL). When AA was detected in clinical urine samples, 42.3%, 10.6%, and 8.2% of the glucose, haemoglobin, and leukocyte esterase urine strip tests were rated as false negative, respectively. 21 It is interesting to note that bilirubin and nitrite interferences were not recorded in their study, while our test-strip was free of interference for leukocytes.
Most of investigations cited in this article are rather old, since AA interference is a commonly known thing. Most manufacturers today declare that they have solved problem of AA interference in urine test strip analysis. However, we have previously demonstrated that manufacturers declarations are often not confirmed in the routine practice and those potentially important interferences have to be carefully investigated. 21 Based on the results of our previous work, AA can still interfere with the urinalysis. 9 Also, interferences differ between manufacturers. Based on the results of this work, these interferences can be associated with high potential risk for patients.
Laboratories should be aware that sensitivity to AA is very important factor when choosing test strips for routine use and should always aim to select test strips with the lowest sensitivity. Manufacturer’s declarations on AA interference should be carefully studied and investigated. All urine samples should be screened for AA and in case of AA positivity, supplementary analysis of automatic or manual sediment examination should be done. Laboratories also have to put effort to raise awareness among patients on importance of reporting usage of all OTC drugs and dietary supplements, when urinalysis is done. Additionally, strong demands on industry should be done to produce test strips not sensitive for AA or, if that is not possible, include AA indicator field on all test strips available on the market.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MM.
Contributorship
NNG, MM and AU conceived the study. All authors were involved in data collection and literature research. NNG has done data analysis and wrote the first draft of the article. All authors reviewed and edited the article and approved the final version of the article.
