Abstract
Background
Triglyceride-rich lipoproteins are considered to be independent predictors of atherosclerotic cardiovascular disease. The molecular basis of its atherogenicity is uncertain. Here, we aim to identify molecular species of phosphatidylcholine hydroperoxides (PCOOH) in triglyceride-rich lipoproteins. For comparison, copper-oxidized triglyceride-rich lipoproteins were investigated as well.
Methods
A fasting EDTA blood sample was collected from six healthy human volunteers to isolate two major triglyceride-rich lipoproteins fractions – very low-density lipoproteins (VLDL) and intermediate-density lipoproteins (IDL) using sequential ultracentrifugation. Triglyceride-rich lipoproteins and plasma samples were studied for PCOOH by liquid chromatography (LC) coupled with Orbitrap mass spectrometry.
Results
Twelve molecular species of PCOOH in triglyceride-rich lipoproteins and/or plasma were identified using the following criteria: (1) high-resolution mass spectrometry (MS) with mass accuracy within 5 ppm, (2) retention time in LC and (3) fragmentation pattern in MS2 and MS3. PC36:4-OOH was most often detected in VLDL, IDL and plasma. The ratio of total PCOOH to phosphatidylcholine progressively increased with the duration of oxidation in both VLDL and IDL.
Conclusion
This study demonstrated the presence of 12 molecular species of PCOOH in native triglyceride-rich lipoproteins. The frequent detection of PCOOH in triglyceride-rich lipoproteins provides a molecular basis of the atherogenicity of triglyceride-rich lipoproteins. PCOOH in triglyceride-rich lipoproteins might serve as a molecular basis of the atherogenicity of triglyceride-rich lipoproteins.
Keywords
Introduction
Plasma triglyceride is a promising candidate for the risk stratification of coronary heart disease (CHD), which is predominantly carried by triglyceride-rich lipoproteins (TRL). However, the specific role of TRL towards the progression of CHD is uncertain and has long been controversial. 1 Alterations of TRL are linked with endothelial dysfunction and considered to be atherogenic. 2 Hence, detection of lipid oxidation products in TRL might provide a basis of the its atherogenicity. The surface of lipoproteins is primarily covered by phosphatidylcholine and is susceptible to oxidative modification in exposure to reactive oxygen species (ROS). Phosphatidylcholine hydroperoxides (PCOOH) are one of its oxidation products and are linked with the pathogenesis of various diseases including atherosclerosis, Alzheimer’s disease, Parkinson’s disease, multiple sclerosis, rheumatoid arthritis, diabetes, oxidative stress and chronic alcoholism.
In the present study, we aimed to identify and characterize molecular species of PCOOH in TRL and plasma. Moreover, we also observed a time course generation of PCOOH during its copper-catalysed oxidation.
Materials and methods
EDTA plasma was collected from six healthy human volunteers after fasting for 14–16 h. Sequential ultracentrifugation was carried out to isolate very low-density lipoproteins (VLDL) and intermediate-density lipoproteins (IDL) from the plasma as described earlier. 3 Oxidized VLDL and oxidized IDL were prepared by incubating the isolated lipoprotein fraction with Cu2+. For this, EDTA-free native VLDL and IDL were incubated with CuSO4 in PBS (Final concentration – 0.5 mg lipids, 5 μM CuSO4) at 37 °C for 0, 0.5, 1, 2, 3, 4 and 8 h. Oxidation of lipoproteins was evaluated by agarose gel electrophoresis (Helena Laboratories, Beaumont, TX, USA). The oxidized lipoproteins were also examined for thiobarbituric acid reactive substances (TBARS) (Cayman Chemical Company, MI, USA).
For the LC/LTQ Orbitrap mass spectrometer (Thermo-Fisher Scientific Inc., San Jose, CA, USA) analysis, total lipids were extracted from native or oxidized lipoproteins, and plasma samples as described earlier. 4 The lipid extracts were injected to a reversed-phase Atlantic T3 C18 HPLC column (150 mm × 2.1 [i.d.] mm; particle size 3.0 μm) (Waters, Milford, MA, USA), maintained at 40°C. Gradient elution was performed with a mobile phase composed of 5 mmol/L aqueous ammonium acetate, isopropanol and methanol. Synthetic standards of PCOOH were used for assignment of equivocal peaks. PCOOH in TRL and plasma samples were identified based on retention time (RT) on LC, elemental composition and typical fragmentation pattern in the mass spectra. The limit of detection for each PCOOH was 0.1 pmol at signal-to-noise ratio above 6. The relative concentration of PCOOH was expressed as a ratio of its peak intensity to corresponding molecular species of phosphatidylcholine. Sum of the relative concentration of all detected species of PCOOH is designated as total PCOOH.
Results
We identified 12 molecular species of PCOOH in TRL and/or plasma using the following criteria: (1) elementary composition and high-resolution m/z with mass accuracy within 5 ppm, (2) RT in LC and (3) fragmentation pattern in MS2 and MS3.
A typical LC/MS extracted-ion chromatogram (EIC) and the corresponding chromatograms of PC34:2-OOH obtained from the standard, VLDL and IDL, and their MS2 and MS3 spectra are provided in Supplementary Figure 1. The major fragment ions of PCOOH observed in MS2 spectra were a result of the loss of H2O from hydroperoxide group, fatty acids (FA) from sn-1 carbon and FAOOH from sn-2 carbon (Supplementary Figure 2). In contrast, the fragment ions of phosphatidylcholine in MS2 spectra were formed by the natural loss of the phosphatidylcholine head-group and FA. All characteristic ions of phosphatidylcholine and PCOOH in VLDL and IDL, and the corresponding RT and fragment ions can be found in the Supplementary Table 1. In VLDL, 12 species of PCOOH were identified, among which, 11 species were also detected in IDL, and 10 species in plasma. PC36:4-OOH was most often detected in VLDL, IDL and plasma. PC34:2-OOH, PC36:2-OOH and PC36:4-OOH were detected in all plasma samples. PC34:2-OOH, PC36:2-OOH, PC36:4-OOH, PC38:4-OOH, PC38:6-OOH and PC40:6-OOH were major PCOOH species detected in TRL (Supplementary Table 2).
Oxidized VLDL and IDL showed the increase in electrophoretic mobility, indicating the loss of positive charge at the amino groups of apolipoprotein B100 due to the Schiff-base formation with the aldehyde groups from oxidized acyl chains (Figure 1(a) and (b)). Accordingly, TBARS for VLDL and IDL increased progressively and peaked at 4 h during oxidation. The ratio of total PCOOH to phosphatidylcholine progressively increased with the duration of oxidation in both VLDL (Figure 1(a’)) and IDL (Figure 1(b’)). Based on the ratio of PCOOH to its corresponding phosphatidylcholine, the phosphatidylcholine with polyunsaturated FA particularly, PC34:3, PC36:3, PC 36:5 and PC38:5 were comparatively more susceptible to oxidation (Supplementary Figure 3).
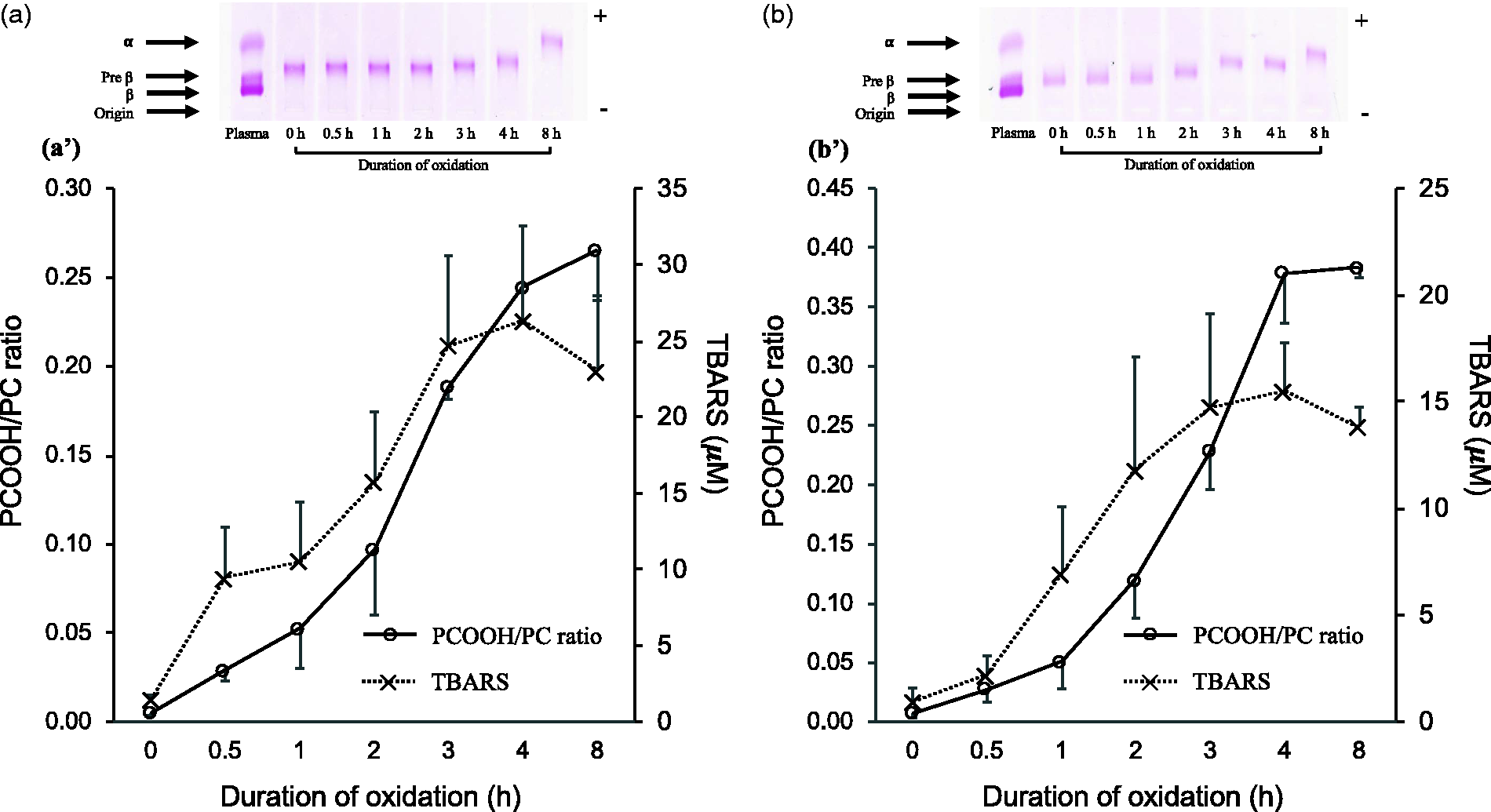
Agarose gel electrophoresis of copper-oxidized VLDL (a) and IDL (b). Lipoproteins (3 μg lipid) was electrophoresed in 50 mmol barbital buffer (pH 8.6) for 30 min at 100 V followed by staining with fat red 7B. Time course of generation of phosphatidylcholine hydroperoxides (PCOOH) in terms of its ratio to phosphatidylcholine (PC) and TBARS during oxidation of VLDL (a’) and IDL (b’).
Discussion
This is the first study to identify molecular species of PCOOH in TRL in its native form. The use of a high-sensitivity and high-resolution Orbitrap mass spectrometer, coupled with a reversed-phase liquid chromatograph and synthetic PCOOH standards, enabled us to perform a high-quality detection and identification of PCOOH molecular species in TRL. PC34:2-OOH, PC36:2-OOH, PC36:4-OOH, PC38:4-OOH, PC38:6-OOH and PC40:6-OOH were frequently identified in native TRL. The relative contents of PCOOH significantly increased with the increase in the degree of artificial oxidation (Figure 1). The time course of PCOOH generation in TRL is similar to that of LDL and HDL published previously. 5 Similar to oxidized LDL, the majority of PCOOH species show lag phase up to 1 h followed by rapid propagation phase (Supplementary Figure 3).
Although the generation mechanism of PCOOH in TRL is not determined, the oxidation of phosphatidylcholine in TRL might occur due to the interaction with ROS in the circulation, as phosphatidylcholine resides in the outer layer of the lipoproteins and are susceptible to oxidative damage. Moreover, we have identified that both VLDL and IDL contain hydroperoxides of triglycerides and choletserylester.3,4 These FA are liberated from the TRL by the action of lipoprotein lipase during its metabolism. It is known that the metabolism of TRL is accompanied by the release of a significant amount of potentially toxic oxidized FA. 6 Therefore, the capillaries endothelial cells are constantly exposed to hydroperoxides, as TRL deliver triacylglycerol to tissues including cardiac muscle. Perhaps, this phenomenon may result in endothelial dysfunction and inflammation which are the crucial features in early atherogenesis. Furthermore, various molecular species of oxidized phospholipid molecules have been detected in both oxLDL and human atherosclerotic plaque. Therefore, it is of significance to measure PCOOH in TRL for the better understanding of the pathogenicity of these lipoproteins.
The present study revealed the molecular species of PCOOH, which in turn become the target of future quantitative study. Our study was primarily limited to identify PCOOH in the native lipoproteins. Further works are needed to find the association of PCOOH in the atherosclerotic process. Nevertheless, our demonstration of the existence of PCOOH would provide a new insight to understand the pathogenicity of TRL.
Conclusion
This study demonstrated the presence of 12 molecular species of PCOOH in native TRL. The existence of PCOOH in TRL provides a molecular basis of the atherogenicity of TRL, and its measurement might contribute to early prediction and risk assessment of CHD.
Supplemental Material
ACB880932 Supplemental Material - Supplemental material for Identification of molecular species of phosphatidylcholine hydroperoxides in native and copper-oxidized triglyceride-rich lipoproteins in humans
Supplemental material, ACB880932 Supplemental Material for Identification of molecular species of phosphatidylcholine hydroperoxides in native and copper-oxidized triglyceride-rich lipoproteins in humans by Rojeet Shrestha, Zhen Chen, Yusuke Miura, Yusuke Yamamoto, Toshihiro Sakurai, Hitoshi Chiba and Shu-Ping Hui in Annals of Clinical Biochemistry
Footnotes
Acknowledgments
We would like to thank entire laboratory member of Laboratory for Advanced Lipid Analysis (LALA) for their assistance and guidance.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the Regional Innovation Strategy Support Program, Sapporo Health Innovation ‘Smart-H’, of The Ministry of Education, Culture, Sports, Sciences and Technology, Japan and partly by Grant-in-Aid for Scientific Research from the Japan Society for Promotion of Sciences, Japan.
Ethical approval
The study was carried out according to The Code of Ethics of the World Medical Association (Declaration of Helsinki). Informed consent was obtained from all human volunteers and study was approved by the Ethical Committee of Faculty of Health Sciences, Hokkaido University, Sapporo, Japan (Approval Number 08–57-2).
Guarantor
SPH.
Contributorship
RS, SPH and HC researched literature and conceived the study. RS, YM, YY and TS were involved in sample collection, lipoprotein separation, lipid measurements and data analysis. RS and ZC performed the mass spectrometric analysis. RS wrote the first draft of the article. All the authors have made a substantial contribution to the conception and design of the study, and acquisition, analysis and interpretation of data. The article has been reviewed and approved by all authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
