Abstract
Background
Vitamin B12 deficiency is a common disorder. In circulation, vitamin B12 is bound to transcobalamin (holotranscobalamin), which is considered the active form of cobalamin. The objective of this study was to evaluate the analytical performance of the Roche Elecsys Active B12 immunoassay.
Methods
Limit of quantification and linearity were assessed according to CLSI EP17-A2 and EP-6A guidelines. Precision and bias of Roche Active B12 test against Architect ci8200 (Abbott) were performed according to CLSI EP-5 A3 guideline at three European laboratories. Precision requirements were set at <4% for within-run precision and <15% for intermediate precision. Requirements for method comparison were set at a slope of 1.00 ± 20%. In addition, 95% reference interval was conducted in European adults according to CLSI-EP28 guideline.
Results
The assay was shown to be linear and precise and met the requirements. Lot-to-lot variation and lab-to-lab variation both were ≤4%. Method comparison with Abbott Architect showed a significant bias of 9 pmol/L, which increases to up to 23 pmol/L around the current medical decision point. The 95% reference interval of the Roche Elecsys Active B12 test was determined at 37–188 pmol/L.
Conclusion
Although Roche Elecsys Active B12 immunoassay met the requirements, this test has a positive bias of 9 pmol/L, which led to an increase in lower limit of reference interval compared with Abbott test. This test first needs to be clinically validated before it can be applied as a first-line screening test in the diagnosis of vitamin B12 deficiency.
Introduction
Vitamin B12 deficiency is common and occurs particularly among the elderly. The diagnosis of vitamin B12 deficiency is hampered by the availability of good biomarkers. Clinical vitamin B12 deficiency with classical haematological and neurological symptoms also known as pernicious anaemia is relatively uncommon and is estimated to affect 2–3% of individuals of 65 years and older.1–3 More often, subclinical vitamin B12 deficiency occurs which, depending on the chosen cut-off value of vitamin B12, ranges from 3% to 26% in the general population. 4
Vitamin B12 is mainly derived from animal sources, and after ingestion, it is bound to haptocorrin in the stomach. In the duodenum, vitamin B12 is released from haptocorrin and binds to intrinsic factor after which it is taken up in the enterocytes of the distal ileum. In circulation, about 20% of vitamin B12 is bound to transcobalamin (holotranscobalamin, holoTC), which is the active form of vitamin B12 available to cells, while the remaining 80% is bound to haptocorrin.
The diagnosis of vitamin B12 deficiency is hampered by the availability of biomarkers with good diagnostic performance. Most frequently, total vitamin B12 is measured in serum or plasma as first-line test to estimate an individual’s vitamin B12 status. Several years ago, the active B12 assay became available, which enabled the measurement of holoTC on the Abbott routine platform. As this holoTC assay is not hampered by fluctuations in haptocorrin, it was expected to result in better diagnostic performance. Several studies have assessed the diagnostic performance of holoTC (or active B12) and have shown a marginally better performance of the active B12 test compared with total vitamin B12.5–7 In addition, homocysteine and methylmalonic acid (MMA) are frequently used as functional markers of vitamin B12.3,8,9 Most laboratories use total vitamin B12 or active B12 as first-line test, which is followed by MMA or homocysteine as second-line test 9 . The medical decision point is based at either an active B12 <20 pmol/L or in case active B12 is between 20 and 35 pmol/L by a follow-up test like MMA or homocysteine. For the diagnosis of vitamin B12 deficiency, it is of utmost importance to have access to biomarkers for vitamin B12 deficiency such as total vitamin B12 and active B12. At this moment, active B12 is available at Abbott Architect and was recently introduced by Siemens ADVIA centaur and Roche Diagnostics, which facilitates the availability of active B12 in clinical chemistry laboratories.
In this study, we performed a multicentre analytical evaluation of the Roche Elecsys® Active B12 immunoassay and assessed linearity, precision and bias by method comparison between
Material and methods
This study was executed between October 2016 and May 2017. Analytical performance of the Active B12 assay was evaluated on a
Analytical performance
Analytical performance of the Roche Active B12 immunoassay was assessed at
Limit of Blanc, limit of detection, limit of quantitation
Limit of Blanc (LoB) and limit of detection (LoD) were determined by measuring 10 times an analyte-free sample in six runs. LoB and LoD were calculated according to CLSI EP17-A2. Limit of quantitation (LoQ) was calculated according to CLSI EP17-A2; eight samples with low concentrations (between 3.2 and 8.7 pmol/L, respectively) were measured in five-fold on five days, and the coefficient of variation (CV%) was calculated per sample. LoQ is defined at the lowest concentration at which CV <10%.
Linearity
Linearity of the Active B12 assay was assessed by diluting three high analyte serum samples with diluent. Fifteen concentrations from above the upper limit throughout the measuring range and below the lower end of the specified measuring range were prepared (measuring range; between 3 and 150 pmol/L). The dilutions were measured in three-fold within a single run using one lot. Evaluation was performed according to CLSI EP6A guideline using a validated software tool provided by Roche Diagnostics Penzberg Biometry department. All the measured data of each dilution series were analysed in a respective regression analysis. Data were analysed using linear, quadratic and cubic order least square regression analysis according to CLSI guidelines. Specifications were predefined at ≤0.8 pmol/L at a concentration below 8 pmol/L and <10% for concentrations between 8 and 150 pmol/L.
Interference
Interference of the Active B12 assay for bilirubin, haemoglobin, intralipids and biotin was assessed by diluting a sample without interfering substance with a sample with interfering substance. For bilirubin interference, this was assessed at three concentrations 32.9, 74.2 and 145 pmol/L of active B12 using a serum sample with 66 mg/dL (1129
Sample stability
Sample stability at –20°C was determined by calculating the recovery of four human serum samples with different active B12 concentrations (i.e. 13, 44, 76 and 141 pmol/L) in triplicate for six months at –20 ± 5°C). Specification for recovery was set at ±10% for analyte concentrations >8 pmol/L.
Multicentre validation
Precision
Precision of the Elecsys® Active B12 immunoassay was assessed at each laboratory on a
Bias
At each laboratory, a minimal amount of 120 anonymized left-over human serum patient samples, in which active B12 was routinely measured by the Architect Active B12 assay (Abbott, Architect, Chicago, USA), were collected and stored at –20°C until measurement. Samples were collected at each laboratory according to predefined percentages covering the measuring range: ≥10% <25 pmol/L; ≥30% with active B12 between 25 and 50 pmol/L; ≥20% with active B12 between 50 and 100 pmol/L and ≥10% of active B12 >100 pmol/L. One reagents lot of Roche Elecsys immunoassay (MP lot, Roche Diagnostics, standardized against WHO 03/178), one level of Roche CalSet Active B12 calibrators consisting of two levels of calibrators and two levels of Roche PreciControls were used for method comparison). Two lots of Abbott active B12 reagents (both traceable against the WHO 03/178 [lot 10556UP00] [Erasmus MC] and lot 10557UP00 [Heidelberg]), four levels of controls (two commercial controls and two in-house serum pools) and two calibrator sets consisting each of six levels of calibrators were used in this study. Samples were thawed at room temperature and were homogenized gently by inverting the tubes and were centrifuged at 4000
Reference interval
Serum samples were collected from apparently healthy adults at one site operated by the German Blood Transfusion Service in Bavaria, Germany. Two hundred and fifty-two samples with approximately 50%–50% male–female distribution and with age from 20 to 79 had been selected. Homocysteine concentrations were determined in all samples using
Results
Analytical performance
In this experiment, the LoB was calculated to be 0.66 pmol/L and LoD was 0.78 pmol/L. The LoQ of this experiment demonstrated a CV of 2.9% at the lowest tested active B12 concentration of 3.2 pmol/L. From these results, the lower limits of measurements for the Elecsys Active B12 assay were decided to be set at: LoB = 2.0 pmol/L, LoD = 3.0 pmol/L and LoQ = 5.0 pmol/L. Samples were shown to be linear between 3.0 and 150 pmol/L active B12 with an absolute deviation of ≤0.8 pmol/L in the lower range (3–8 pmol/L) and a relative deviation of ≤10% in the range between 8 and 150 pmol/L. Interference for bilirubin was not detected up to a concentration of 66 mg/dL (1129
Precision
Two data points from the human serum pool (HSP05 were excluded from the analysis (Munich site, HSP05, 1 from MP Lot and 1 from PoQ lot), as they were above the measuring range. All other data points were used for precision analysis. Within-run precision of Roche Elecsys® Active B12 immunoassay was <4% at each centre, and intermediate precision was <6% at each centre (Table 1). Lot-to-lot variation was <4% and lab-to-lab variation was <3.1%.
Within precision and intermediate precision (IM) of Roche Elecsys® Active B12 assay at three sites.
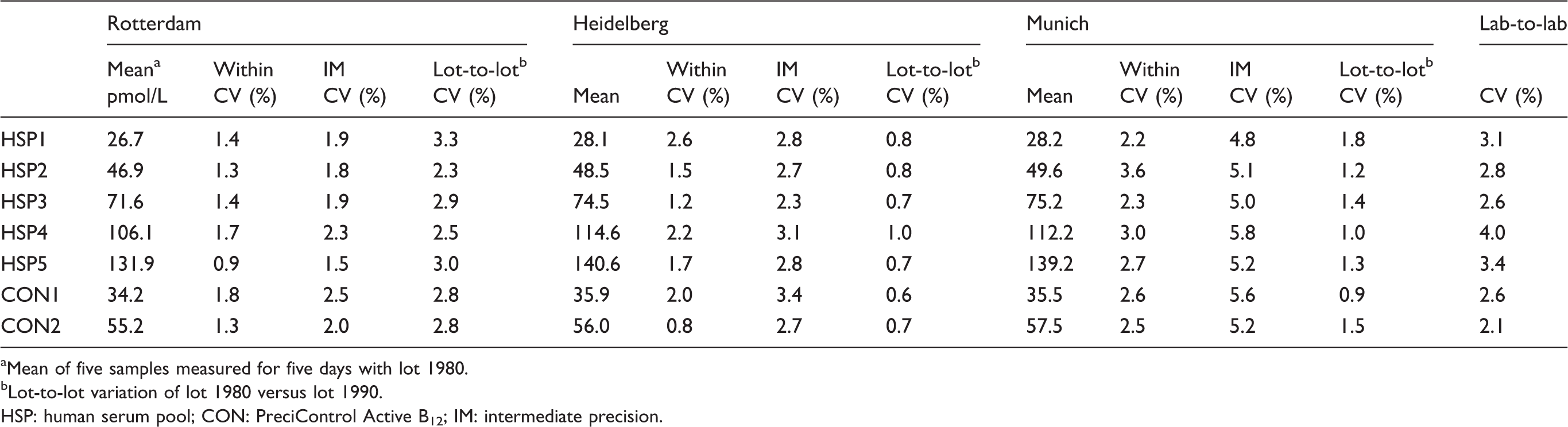
aMean of five samples measured for five days with lot 1980.
bLot-to-lot variation of lot 1980 versus lot 1990.
HSP: human serum pool; CON: PreciControl Active B12; IM: intermediate precision.
Bias
One hundred and forty-seven samples were used for method comparison at the Heidelberg site and 142 different samples were measured at the Erasmus MC site. Method comparison of
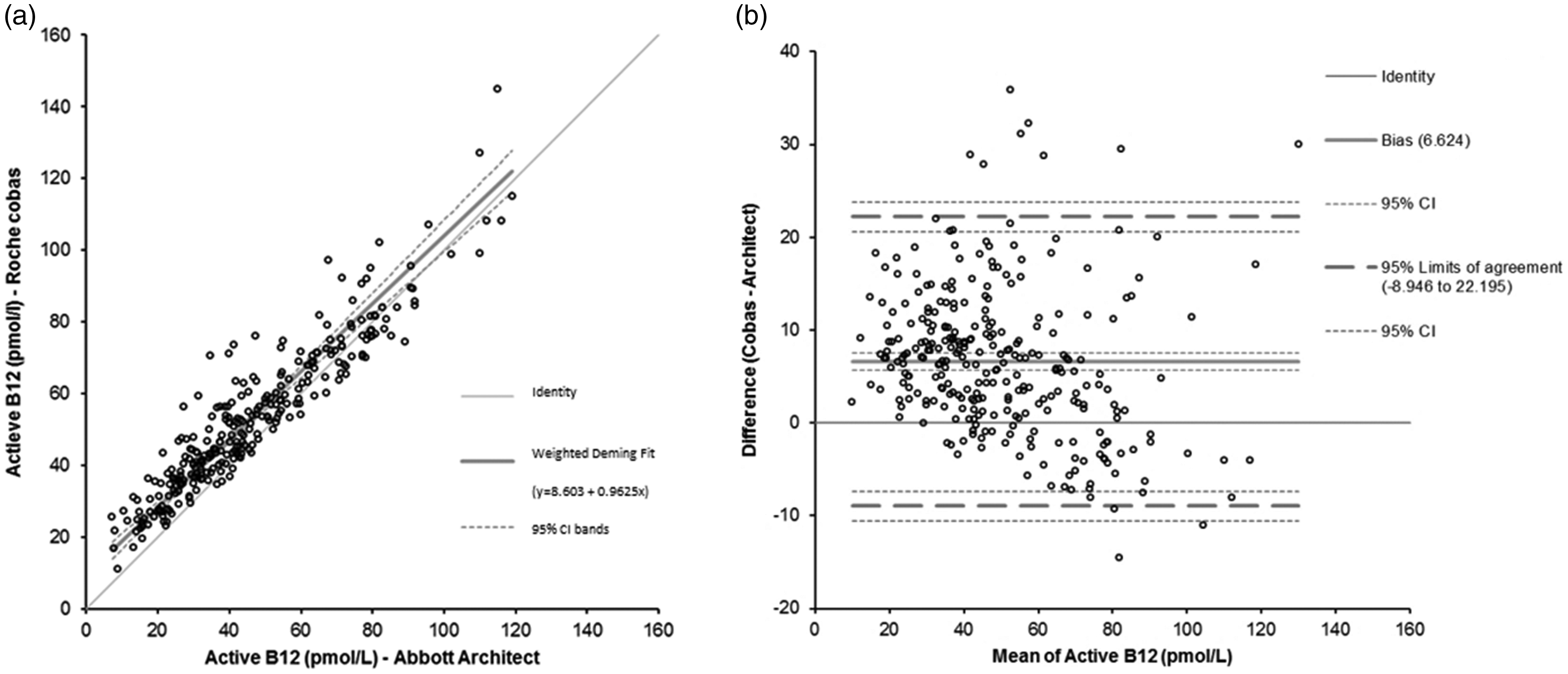
Bias plot of active B12 assay at Roche
Reference interval of active B12
Median active B12 concentration in serum of 214 samples was 77 pmol/L. The 95% reference interval of Roche Active B12 assay was calculated at 37–188 pmol/L.
Discussion
Active B12 is commonly used as biomarker in the diagnosis of vitamin B12 deficiency. In this study, we evaluated the new Roche® Elecsys Active B12 assay at the
The Roche® Elecsys Active B12 assay was shown to be precise with an intermediate precision lower than 6%, which is comparable to Abbott Architect intermediate precision of <6% (Abbott Architect Active-B12, package insert October 2015). Lab-to-lab variation and lot-to-lot variation both were small, enabling harmonization of test results and facilitating the use of general cut-off values in clinical algorithms. Method comparison showed a small constant bias of 9 pmol/L. Visual inspection of the bias plot shows that the observed bias is not constant over the measuring range. Roche Active B12 results within the lower range have a larger bias, which is expected to result in a higher active B12 test result (up to 22 pmol/L) around the current clinical decision point of 20–35 pmol/L.
Current diagnosis of vitamin B12 using active B12 is in most laboratories a two-step process, in which active B12 is applied as first-line test. If active B12 is <21 pmol/L, then the diagnosis of vitamin B12 deficiency is confirmed if clinical signs are present. However, if the test result is between 21 and 35 pmol/L, a follow-up test is performed by measuring MMA to confirm diagnosis because specificity of the active B12 test is too low within this range. Patients with active B12 > 35 pmol/L are unlikely to be diagnosed with vitamin B12 deficiency. However, these cut-off values are mostly based on the Abbott Active B12 test. In this study, however, we have shown that the reference interval of Roche Active B12 is considerably higher (37–188 pmol/L) making the current cut-off values not suitable. Novel cut-off values of Roche Active B12 should be established by calculating sensitivity and specificity of active B12 concentrations against MMA concentrations before this test can be applied as first-line screening test in the diagnosis of vitamin B12 deficiency.
In future, clinical algorithms would benefit from standardization against an active B12 reference method using commutable standards in the assay.
This multicentre evaluation of the Roche Elecsys Active B12 assay demonstrates excellent precision. A bias compared with Abbott Architect Active B12 assay was observed, which needs to be accounted by establishment of laboratory-specific reference intervals. Before this assay can be implemented on Roche routine platforms, a clinical validation of the Roche active B12 test is needed to establish reliable cut-off values to define clinical- and subclinical vitamin B12 deficiency.
Footnotes
Acknowledgements
The authors would like to acknowledge the project manager Ina van Gorp for assistance at the Erasmus MC site.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially sponsored by Roche Diagnostics as part of their multicentre evaluation study.
Ethical approval
The medical ethical committee of the Erasmus MC declared that this project is not subject to rules on medical research involving human subjects as anonymized left-over material is used (ref MEC-2016–599). In addition, approval has been received for collection of patient samples for reference interval study (Ethik Kommission der Bayerischen Landesärztekammer in Munich, Germany).
Guarantor
SGH.
Contributorship
SGH supervised the study at the Erasmus MC site, conducted data analysis, gained ethical approval and drafted the article. PB supervised the study at the Munich site. PF supervised the study at the Heidelberg site. SL developed the Roche active B12 assay and supervised the analytical measurements at Roche Diagnostics and was involved in data analysis and YS wrote the study protocol and supervised the study at Roche Diagnostics. YBdR contributed to data interpretation and writing of the article. All authors corrected and approved the final version of the article.
