Abstract
Background
Tolvaptan is converted to major metabolites including three monohydroxylates (DM-4110, DM-4111 and DM-4119), an oxidate (MOP-21826) and a carboxylate (DM-4103) in humans. This study developed a simultaneous quantitative method for tolvaptan and its five major metabolites in human plasma using liquid chromatography coupled to tandem mass spectrometry.
Methods
Deproteinized plasma specimens using acetonitrile were separated using a 3-μm particle size octadecylsilyl column with 250 mm length and a simple linear gradient program at a flow rate of 0.3 mL/min with a total run time of 15 min. This method was applied to the determination of plasma samples collected from 20 heart failure patients treated with 3.75–15 mg tolvaptan.
Results
No interfering peak was found in drug-free plasma specimens. The calibration curves of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were linear over the concentration ranges of 3.125–1000, 0.3125–100, 1.25–400, 0.625–200, 0.125–40 and 31.25–10,000 ng/mL, respectively. Their pretreatment recovery rates and matrix factors were 94.1–113.9% and 86.9–108.0%, respectively. The intra- and inter-day accuracies and imprecisions were 91.6–106.5% and 0.9–10.9%, respectively, for all analytes. The plasma concentration ranges of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were 9.37–280, 1.91–16.3, 3.43–88.9, 1.43–10.4, 0.160–1.01 and 40.2–1471 ng/mL, respectively, in heart failure patients.
Conclusions
This validated method with acceptable analytical performance can be utilized for evaluating the pharmacokinetics of oral tolvaptan, including the determination of its major metabolites, in heart failure patients.
Keywords
Introduction
Tolvaptan, a potent, highly selective and orally effective non-peptide vasopressin V2 receptor antagonist, is commonly used for the treatment of water retention caused by heart failure.1–3 It suppresses the synthesis and insertion of aquaporin-2 water channels into the luminal plasma membrane of collecting tubule principle cells, resulting in the restriction of water reabsorption in the hypertonic medulla without renal function deterioration.1,4 Tolvaptan has a beneficial effect on patients in whom other diuretics, including loop diuretics, are ineffective; however, the US Food and Drug Administration (FDA) has issued a warning about the adverse effects of severe hypernatraemia and potentially fatal liver injury. 5
Oral tolvaptan is rapidly absorbed from the intestine and its bioavailability is approximately 60% due to first-pass metabolism.6,7 Tolvaptan is metabolized to three monohydroxylates (DM-4110, DM-4111 and DM-4119) and an oxidate (MOP-21826) primarily by cytochrome P450 (CYP) 3A4 (Figure 1). MOP-21826 is ring-opened by CYP3A4, and further metabolized to a carboxylate (DM-4103) through an unrevealed pathway.8,9 The monohydroxylates have relatively weak V2 receptor antagonistic activity, and likely contribute to the diuretic effect of tolvaptan. 10 DM-4103 exists in human plasma at the highest concentration among the tolvaptan metabolites. 11 DM-4103, with no diuretic effect, recently has been reported to potentially cause hepatic disorders through an inhibition of hepatic transporters and bile acid transporters.12–15 Few studies on the pharmacokinetic characteristics of tolvaptan metabolites in humans have been published. 11
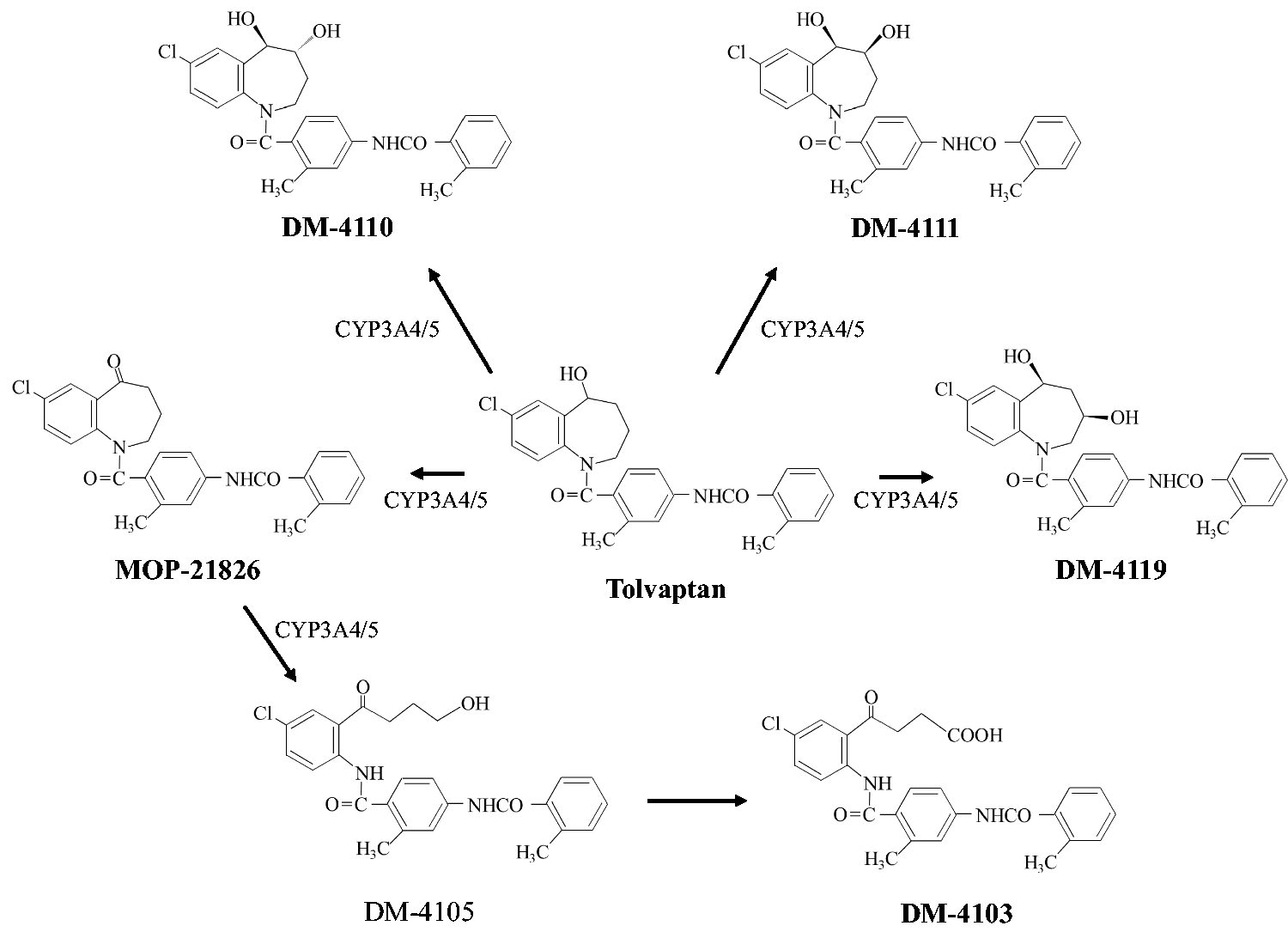
Possible metabolic pathways of tolvaptan in humans. 8
A few methods using a liquid chromatography (LC) coupled to tandem mass spectrometry (MS/MS) have been developed for determination of tolvaptan in human plasma.11,16,17 However, tolvaptan metabolites in human plasma have been only rarely measured to date. 11 They reported simultaneous quantitation method of tolvaptan and two carboxylates including DM-4103 using samples taken from volunteers who received tolvaptan. The quantitation of tolvaptan metabolites requires MS/MS detection from the view point of selectivity and sensitivity. The LC separation of tolvaptan (molecular weight, 448.94) and MOP-21826 (molecular weight, 446.93), both possessing a chlorine with stable isotopes, is also needed due to their almost identical molecular masses. Furthermore, a challenge also exists in the LC separation of the monohydroxylates, which have theoretically identical molecular masses since they are mutual structural isomers with each other. MS/MS detection still necessitates the selection of a suitable mobile phase in order to minimize ion suppression caused by co-eluting compounds in biological specimens to overcome the extremely low sensitivity of MOP-21826 in particular. 8 The above difficulties are most likely responsible for the lack of available simultaneous quantitative methods for tolvaptan metabolites, including monohydroxyrates and oxidates, in human plasma. 11 To the best of our knowledge, no validated LC-MS/MS methods for quantitation of DM-4110, DM-4111, DM-4119 and MOP-21826 in human plasma have been reported.
Effective and validated chromatographic methodologies for the determination of tolvaptan metabolites in human specimens are needed for clinical use. This study aimed to develop a simultaneous quantitative method for tolvaptan and its five major metabolites in human plasma using LC-MS/MS. In addition, the present method was validated with respect to pharmacokinetic evaluation in heart failure patients.
Materials and methods
Materials
Tolvaptan and tolvaptan-d7 as an internal standard (IS) were obtained from LKT Laboratories, Inc. (St Paul, MN, USA) and Medical Isotopes Inc. (Pelham, NH, USA), respectively. MOP-21826 was purchased from Toronto Research Chemicals Inc. (Toronto, Ontario, Canada). DM-4110, DM-4111, DM-4119 and DM-4103 were donated by Otsuka Pharmaceutical Co., Ltd (Tokyo, Japan). All other reagents were of analytical grade and commercially available.
Solutions
Stock solutions of tolvaptan (100 μg/mL), DM-4110 (20 μg/mL), DM-4111 (20 μg/mL), DM-4119 (20 μg/mL), MOP-21826 (50 μg/mL), DM-4103 (50 μg/mL) and IS (20 μg/mL) were prepared in methanol. Standard solutions of all analytes were obtained by the dilution of stock solution with methanol. Calibration standards were prepared in drug-free pooled plasma (Kohjin-Bio Co., Ltd, Saitama, Japan). The final concentrations were the following: 3.125, 6.25, 12.5, 25, 50, 100, 500 and 1000 ng/mL for tolvaptan; 0.3125, 0.625, 1.25, 2.5, 5, 10, 50 and 100 ng/mL for DM-4110; 1.25, 2.5, 5, 10, 20, 40, 200 and 400 ng/mL for DM-4111; 0.625, 1.25, 2.5, 5, 10, 20, 100 and 200 ng/mL for DM-4119; 0.125, 0.25, 0.5, 1, 2, 4, 20 and 40 ng/mL for MOP-21826 and 31.25, 62.5, 125, 250, 500, 1000, 5000 and 10,000 ng/mL for DM-4103. Spiked in drug-free human plasma, the concentrations of quality control (QC) samples were set as follows: 7.5, 75 and 750 ng/mL for tolvaptan; 0.75, 7.5 and 75 ng/mL for DM-4110; 3, 30 and 300 ng/mL for DM-4111; 1.5, 15 and 150 ng/mL for DM-4119; 0.3, 3 and 30 ng/mL for MOP-21826 and 75, 750 and 7500 ng/mL for DM-4103.
Sample pretreatment
Blood specimens collected into a tube containing ethylenediaminetetraacetic acid dipotassium salt (EDTA-2K) were immediately centrifuged at 1670 g at 4°C for 10 min. The upper layer was isolated into a microtube as a plasma sample and then stored at –80°C until sample pretreatment. To 100 μL aliquots of plasma, 600 μL of acetonitrile and 100 μL of IS solution (1 ng/mL) were added. The mixtures were ultrasonicated and centrifuged at 18,000 g at 4°C and then 650 μL of the supernatants were dried by rotary vacuum evaporation at 40°C. The residues were dissolved in 150 μL of mixture of 2.5 mmol/L ammonium acetate and acetonitrile (59:41, v/v), and then centrifuged at 18,000 g at 4°C, and 100 μL of the supernatants were filtrated with a Millex-LH syringe filter (0.45 μm, 4 mm, Merck Millipore Ltd, Billerica, MA, USA) before injection into an LC system for analysis.
Chromatographic conditions
Tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826, DM-4103 and IS in human plasma were separated using an LC system (Nexera X2, Shimadzu Corporation, Kyoto, Japan). The LC system consisted of a CBM-20A system controller, DGU-20A5R degasser, two LC-30ADXR pumps, SIL-30AC autoinjector, CTO-20AC column oven and FCV-20AH2 valve unit. The chromatographic separations were carried out on a 3-μm particle size octadecylsilyl column (TSKgel ODS-100V, 250 × 2.0 mm inner diameter, Tosoh, Tokyo, Japan) with a guard column (TSK guardgel ODS-100V, 3 μm particle size, 10 × 2.0 mm inner diameter, Tosoh). The mobile phase consisted of a gradient mixture of 2.5 mmol/L ammonium acetate and acetonitrile. The LC gradient program was: 0–8.3 min, 41% acetonitrile; 8.3–8.7 min, linear gradient 41–80% acetonitrile; 8.7–10.3 min, 80% acetonitrile; 10.3–10.7 min, linear gradient 80–41% acetonitrile and 10.7–15 min, 41% acetonitrile. The flow rate was 0.3 mL/min and the column temperature was set at 40°C, and the autoinjector was set at 4°C. The injection volume was 2 μL.
Mass spectrometer conditions
The column effluent was monitored using a triple quadrupole tandem mass spectrometer (LCMS8050, Shimadzu Corporation) equipped with an electrospray probe in positive ionization mode for tolvaptan, DM-4110, DM-4111, DM-4119 and IS; and in negative ionization mode for MOP-21826 and DM-4103. It was controlled by LabSolutions LCMS ver. 5.91 (Shimadzu Corporation). The ion transitions were monitored using a dwell time of 100 ms for the compounds: tolvaptan, m/z 448.90/119.10; DM-4110, 464.95/252.05; DM-4111, 464.95/252.05; DM-4119, 464.95/252.05; MOP-21826, 445.05/194.05; DM-4103, 477.05/224.15 and IS, 455.90/259.05 (Figure 2). Samples were introduced to the interface through a turbo ion spray with the temperature set at 350°C. Voltages applied to the interface were 4.0 kV in positive ion mode and –3.0 kV in negative ion mode. Collision-induced-dissociation gas, drying gas, nebulizer gas and heating gas were set at 270 kPa, 10 mL/min, 3 mL/min and 10 mL/min, respectively. Collision energies for tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826, DM-4103 and IS were –25, –20, –23, –20, 29, 40 and –20 volts, respectively.
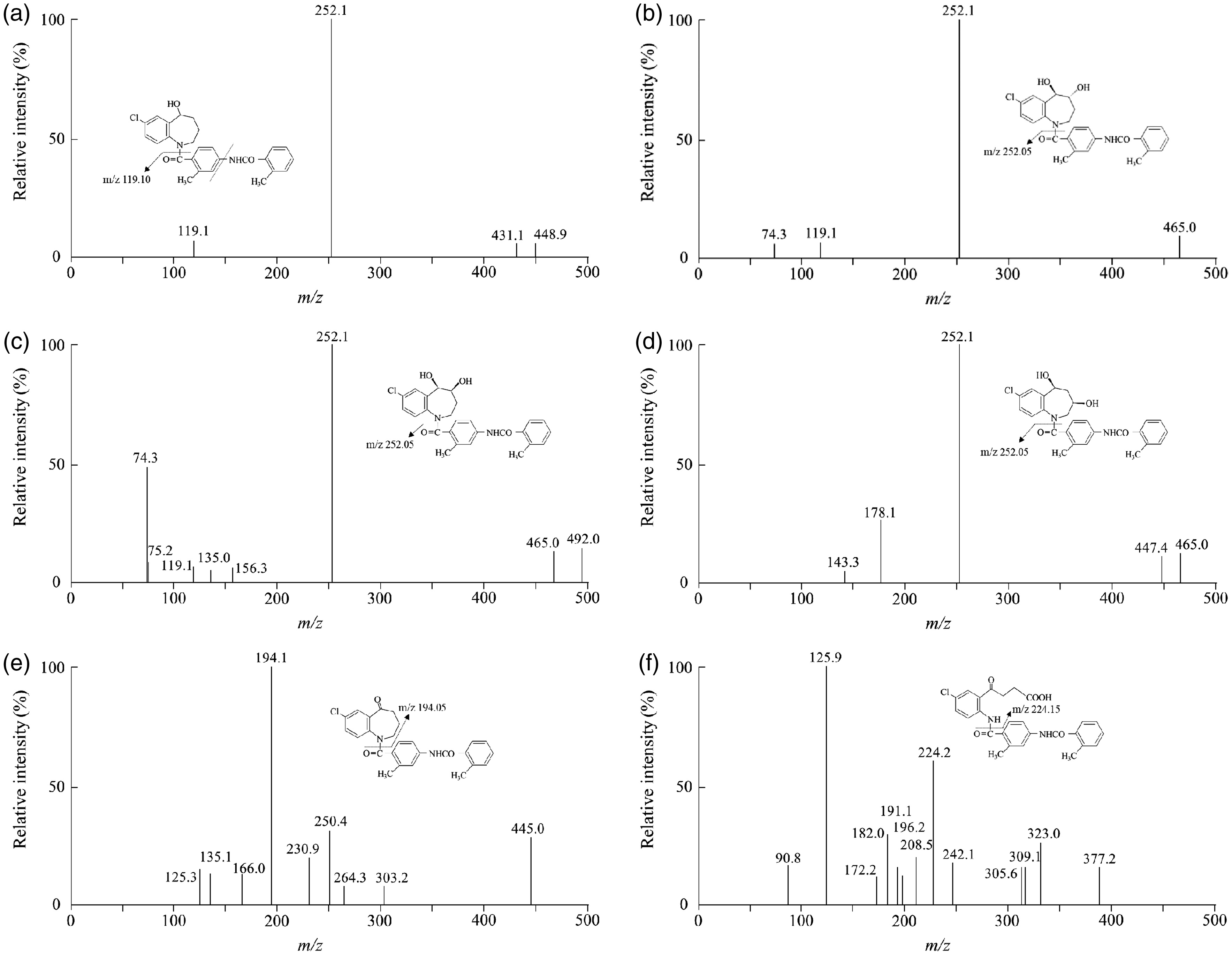
Mass spectra and mass-to-charges (m/z) of tolvaptan and its five major metabolites. (a) Tolvaptan, (b) DM-4110, (c) DM-4111, (d) DM-4119, (e) MOP-21826 and (f) DM-4103. Tolvaptan, m/z 448.90/119.10; DM-4110, 464.95/252.05; DM-4111, 464.95/252.05; DM-4119, 464.95/252.05; MOP-21826, 445.05/194.05; DM-4103, 477.05/224.15.
Method validation
This method was validated in the US FDA bioanalytical method validation guidance. 18 Selectivity of the method was evaluated by analysing six independent drug-free plasma samples from heart failure patients. Calibration curves were obtained with the weighting factor 1/x2 by plotting the measured peak area ratios of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 to IS. The linearities of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were observed at concentration ranges of 3.125–1000, 0.3125–100, 1.25–400, 0.625–200, 0.125–40 and 31.25–10,000 ng/mL, respectively. The lower limit of quantification (LLOQ) was defined as the concentration at which the relative standard deviation (RSD) did not exceed 20%. Four replicates of each QC sample were used to evaluate that the imprecision of pretreatment recovery and that of matrix effect were within 15%. Accuracy was expressed as the measured value divided by the nominal value, and imprecision was shown as the RSD. The intra- and inter-day accuracies and imprecisions were calculated for six replicates of each QC sample in plasma. Carry-over was assessed by measuring blank plasma with 0 ng/mL of tolvaptan and its five major metabolites after a high-concentration sample at the upper limit of quantification; the nominal concentrations for tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were 1000, 100, 400, 200, 40 and 10,000 ng/mL, respectively.
The stabilities of the analytes were assessed using three replicates of each QC sample. Short-term stabilities were evaluated by comparing initial peak areas with: peak areas after 24 h of storage at 4°C and at room temperature, respectively, in plasma; and peak areas after 24 h of storage at 4°C in an autoinjector, in injection solution. Long-term stabilities were evaluated by comparing initial peak areas with: peak areas after one month of storage at –80°C, in plasma; and peak areas after three months of storage at 4°C, in stock solution. Freeze–thaw stability was evaluated by comparing the initial peak areas with peak areas freeze–thawed twice, in plasma.
Ethics
This study was performed in accordance with the Declaration of Helsinki and its amendments. The study protocol was approved by the Ethics Committee of Hamamatsu University School of Medicine. Each patient received information about the scientific aim of the study, and each provided written informed consent.
Patients and pharmacokinetic evaluation
A total of 20 Japanese heart failure patients treated with 3.75–15 mg tolvaptan at Hamamatsu University Hospital were enrolled: 3.75 mg for six patients; 7.5 mg for eight patients; and 15 mg for six patients (Supplemental Table 1). The patients received tolvaptan tablets (Samsca, Otsuka Pharmaceutical Co., Ltd) after breakfast for volume overload. No patient was co-treated with strong potent enzyme inducers including rifampicin and carbamazepine, or inhibitors such as azole antifungal agents and macrolides. Blood specimens (2 mL) drawn from a forearm were collected in tubes containing EDTA-2K just before dosing on the seventh day after starting medication or later. Variations in plasma exposure of tolvaptan and its metabolites were evaluated as the absolute plasma concentration adjusted with the tolvaptan dose and body weight. The tolvaptan metabolism was defined as metabolic ratio: the plasma concentration ratio of the metabolites to tolvaptan.
Results
Separation and selectivity
Figure 3 shows the LC-MS/MS chromatograms of tolvaptan and its metabolites in human plasma. No peaks interfering with tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826, DM-4103 or IS in six independent tolvaptan-free plasma specimens in heart failure patients were observed. Tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826, DM-4103 and IS were eluted at 12.6, 10.8, 8.8, 9.7, 13.0, 6.1 and 12.6 min, respectively, with a total run time of 15 min.
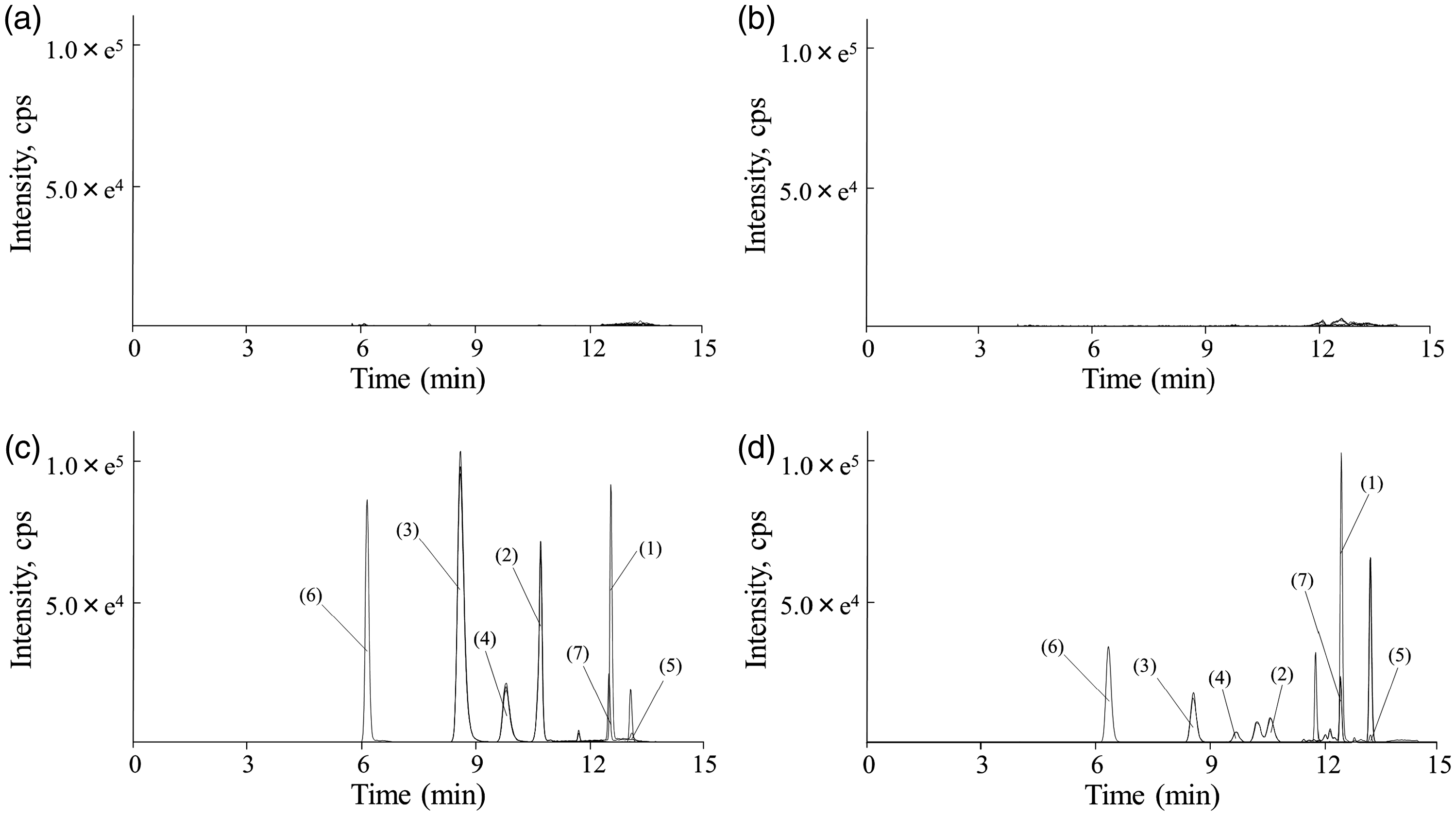
LC-MS/MS chromatograms of tolvaptan and its metabolites in human plasma. (a) human drug-free plasma, (b) a plasma specimen obtained from heart failure patients treated without tolvaptan, (c) human drug-free plasma spiked with 75 ng/mL tolvaptan, 7.5 ng/mL DM-4110, 30 ng/mL DM-4111, 15 ng/mL DM-4119, 3 ng/mL MOP-21826 and 750 ng/mL DM-4103, (d) a plasma specimen obtained from heart failure patients treated with 7.5 mg tolvaptan just before dosing on the seventh day after initiation of therapy or later. (1) Tolvaptan, (2) DM-4110, (3) DM-4111, (4) DM-4119, (5) MOP-21826, (6) DM-4103 and (7) tolvaptan-d7 as internal standard.
Calibration curve and sensitivity
The calibration curves of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 in human plasma were linear over the concentration ranges of 3.125–1000, 0.3125–100, 1.25–400, 0.625–200, 0.125–40 and 31.25–10,000 ng/mL, respectively. Their correlation coefficients were greater than 0.999. The LLOQs of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 in human plasma were 3.125, 0.3125, 1.25, 0.625, 0.125 and 31.25 ng/mL, respectively (n = 6).
Pretreatment recovery, matrix effect and carry-over
The pretreatment recovery rates including deproteinization of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826, DM-4103 and IS were mean ± standard deviation (SD), 101.8 ± 3.0%, 99.1 ± 3.8%, 102.5 ± 11.1%, 102.2 ± 4.5%, 102.0 ± 4.1%, 101.1 ± 3.6% and 99.2 ± 4.7%, respectively, in the middle QC samples (n = 4). The analytes and IS did not exhibit any matrix effects in human plasma: mean ± SD, 92.9 ± 5.4% for tolvaptan; 94.0 ± 7.0% for DM-4110; 98.9 ± 3.5% for DM-4111; 97.0 ± 3.0% for DM-4119; 86.9 ± 2.5% for MOP-21826; 102.3 ± 1.8% for DM-4103 and 94.2 ± 5.1% for IS, in the middle QC samples (n = 4). The pretreatment recoveries and the matrix effects of the analytes in the low and high QC samples (n = 4) were also validated (Supplemental Table 2). No peak area was observed in the blank plasma when measured after the high-concentration sample at the upper limit of quantification.
Assay accuracy and imprecision in human plasma
Table 1 shows the intra- and inter-day accuracies and imprecisions in human plasma. The intra- and inter-day accuracies of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were 96.5–106.5% and 97.0–100.4%, 97.9–104.6% and 99.7–105.1%, 96.6–105.7% and 98.5–104.4%, 98.3–105.4% and 97.9–102.1%, 98.7–103.3% and 98.9–104.8%, and 91.6–101.3% and 94.1–104.6%, respectively (n = 6). The intra- and inter-day imprecisions of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were 0.9–2.4% and 6.0–10.9%, 1.3–2.1% and 6.2–8.4%, 1.0–2.1% and 6.6–9.5%, 1.1–2.4% and 7.0–8.4%, 1.5–3.7% and 6.2–9.4%, and 1.0–2.5% and 6.9–8.1%, respectively.
Intra- and inter-day accuracies and imprecisions of tolvaptan and its five major metabolites in human plasma.
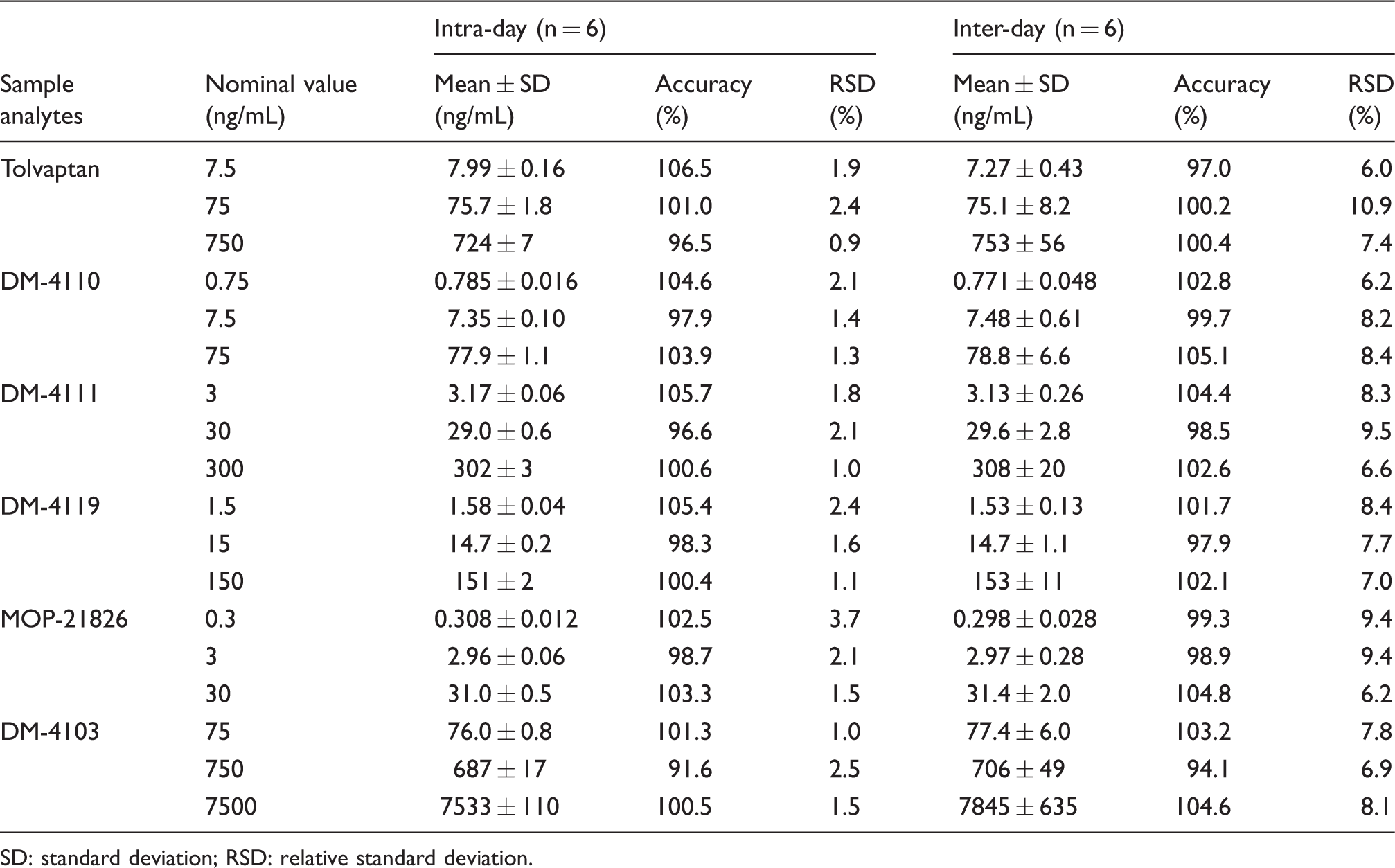
SD: standard deviation; RSD: relative standard deviation.
Stability
Tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826, DM-4103 and IS in plasma specimens were stable at 4°C (% of initial value, 95.7–107.9%) for up to 24 h and also stable at room temperature (% of initial value, 90.2–113.2%) for up to 24 h. The analytes and IS in injection solutions were stable at 4°C (% of initial value, 89.3–110.7%) for up to 24 h. The analytes and IS in plasma specimens were stable at –80°C (% of initial value, 85.1–110.5%) for up to one month. The stock solutions of the analytes and IS were stable at 4°C (% of initial value, 86.2–107.7%) for up to three months. The analytes and IS in plasma specimens were stable through twice freeze–thawed cycles (% of initial value, 86.9–103.0%).
Plasma concentrations of tolvaptan and its metabolites
Figure 4 shows the plasma concentration ranges of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 in 20 heart failure patients treated with 3.75–15 mg, which were 9.37–280, 1.91–16.3, 3.43–88.9, 1.43–10.4, 0.160–1.01 and 40.2–1471 ng/mL, respectively. The concentration ranges of all the analytes in the plasma samples actually collected from 20 heart failure patients were measurable within their calibration curves.
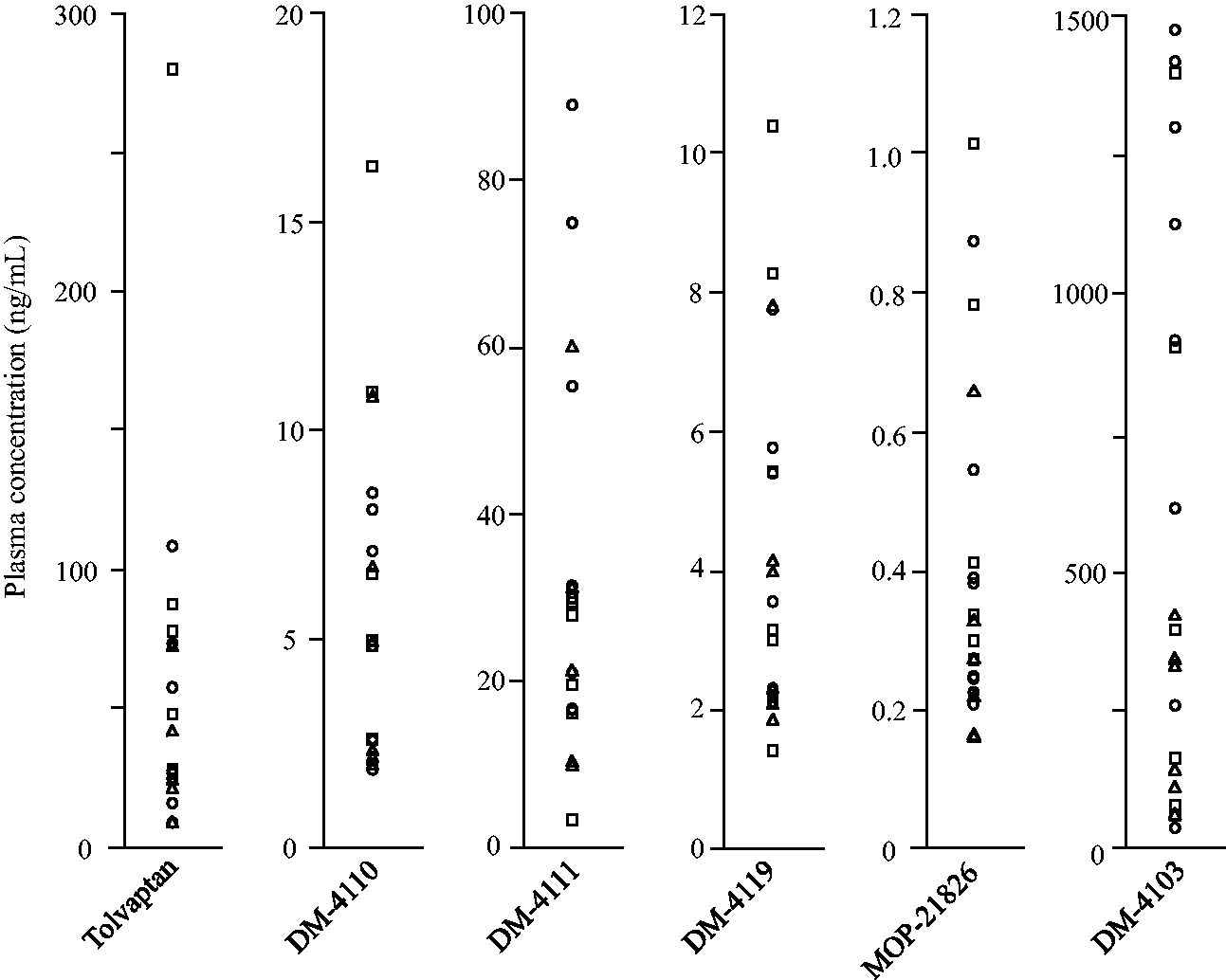
Plasma concentrations of tolvaptan and its five major metabolites obtained from 20 heart failure patients just before dosing on the seventh day after initiation of therapy or later. Tolvaptan dose, 3.75 mg: triangle, 7.5 mg: circle and 15 mg: square.
Variation in plasma exposure and metabolism of tolvaptan
The median and interquartile range (IQR) of dose and body weight adjusted plasma concentrations of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were 224 and 151–427, 35.5 and 18.3–52.8, 157 and 118–302, 30.7 and 14.8–44.2, 2.64 and 1.34–4.31 and 4303 and 820–6440 ng/mL per mg/kg, respectively (Figure 5). The median metabolic ratios to DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 were 0.123 (IQR, 0.090–0.180), 0.715 (0.326–1.17), 0.103 (0.058–0.160), 0.00876 (0.00607–0.0182) and 8.67 (3.93–22.0), respectively.
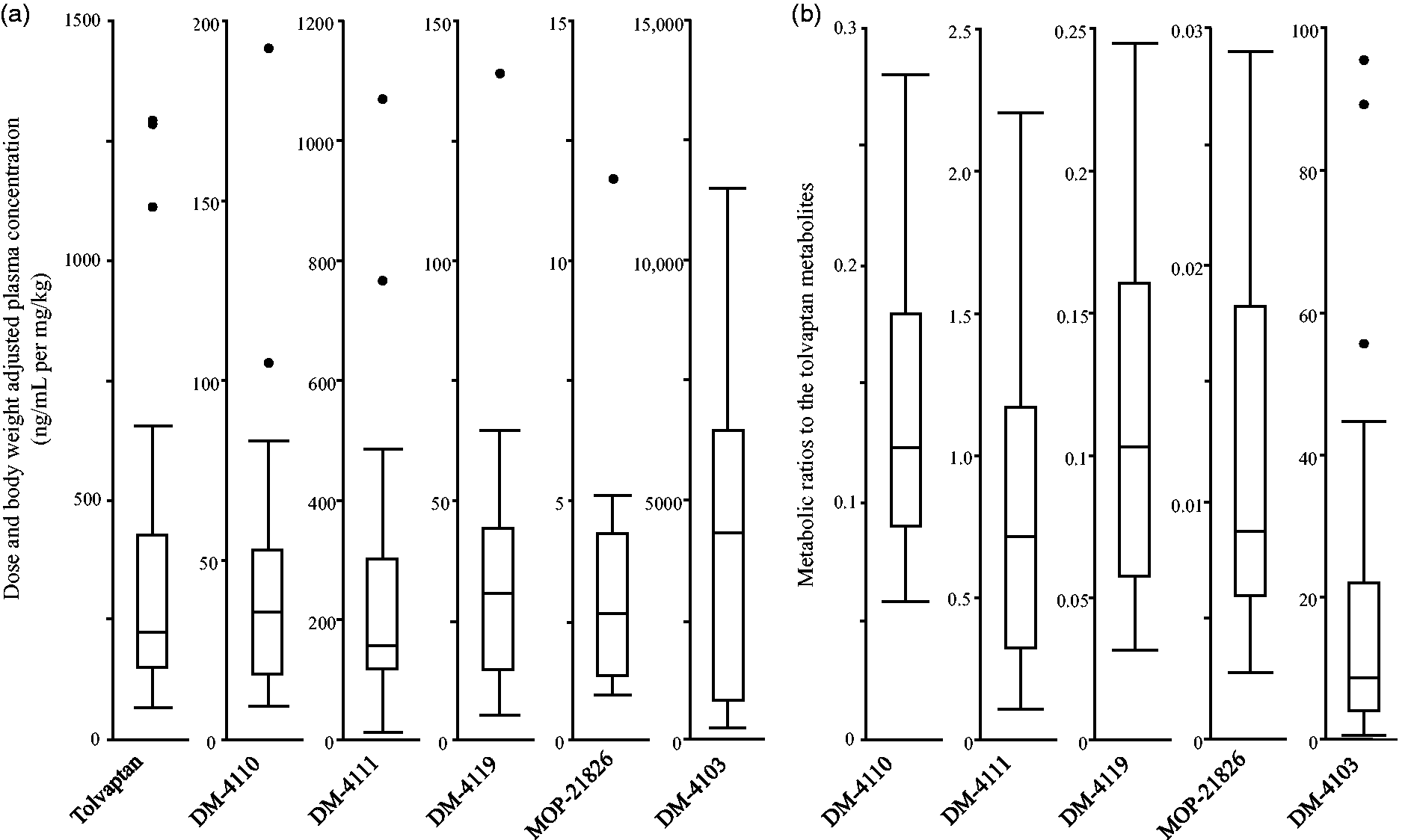
The variations in plasma exposure (a) and metabolic ratios (b) of tolvaptan and its metabolites in heart failure patients. Box plot explanation: upper horizontal line of box, 75th percentile; lower horizontal line of box, 25th percentile; horizontal bar within box, median. The length of the box plot whiskers is specified as 1.5 times the interquartile range (25th to 75th quartiles) of the data. Circles represent outliers.
Discussion
Tolvaptan is converted to several major metabolites including three monohydroxylates (DM-4110, DM-4111 and DM-4119), an oxidate (MOP-21826) and a carboxylate (DM-4103) in humans. This study established a simultaneous quantitative method for tolvaptan and its five major metabolites in human plasma using a simple gradient LC-MS/MS and assessed its clinical suitability in heart failure patients with repeated dosing of tolvaptan. Acetonitrile precipitation was employed for sample pretreatment without extraction. A simple linear LC gradient program using a 3-μm particle size octadecylsilyl column with 250 mm length achieved the separation of the three monohydroxylates and the separation between tolvaptan and MOP-21826 within 15 min. This method possessed adequate sensitivity for the quantitation of all the peaks in the plasma of 20 heart failure patients. Additionally, it is validated in the US FDA bioanalytical method validation guidance, 18 to be clinically practical for the quantitation of tolvaptan and its five major metabolites. To the best of our knowledge, this is the first practical quantitative method for tolvaptan monohydroxylates and oxidates in addition to the parent drug simultaneously in human plasma.
Acetonitrile was used for the deproteinization of plasma specimens in the present study. The sample pretreatment recovery rates and matrix factors of all six analytes met the guidance. Jiang et al. also reported a simultaneous quantitative method for tolvaptan and its carboxylates in human plasma using an LC-MS/MS. 11 Our method can simultaneously quantify the six analytes, including monohydroxylates and oxidates, with smaller variations in pretreatment recovery rates of tolvaptan and DM-4103. Furukawa et al. 9 used solid-phase extraction in the sample pretreatment for determining tolvaptan and its metabolites in rat serum. In human plasma, the method of Jiang et al. employed large amounts of acetonitrile and methanol through relatively complicated processes including solid-phase extraction. 11 In the present study, small amounts of organic solvents were used for sample pretreatment, leading to a simple procedure with a low-cost and low-occupational exposure and better applicability to clinical use.
No interfering peaks were found in drug-free plasma specimens. The LLOQs of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 in human plasma were 3.125, 0.3125, 1.25, 0.625, 0.125 and 31.25 ng/mL, respectively. Pei et al. 16 and Derangula et al. 17 reported quantitative methods of tolvaptan with LLOQs of 0.457 and 0.05 ng/mL in human plasma, respectively. Jiang et al. also reported that both LLOQs of tolvaptan and DM-4103 were concentrations of 1 ng/mL, 11 which are not needed for clinical use. Furukawa et al. reported the simultaneous quantitation of tolvaptan and its nine metabolites, including MOP-21826, in rat serum using an LC-MS/MS method. 9 MOP-21826, which exists at a remarkably low concentration in human plasma, was successfully quantified in our method with optimization of the collision energy and mobile phase. The LLOQs of tolvaptan metabolites in our method were found to be low enough for their quantitation in plasma samples collected from 20 heart failure patients receiving a clinical dose of tolvaptan.
The chromatographic separation of tolvaptan and MOP-21826 was not completely accomplished in earlier reports.11,16,17 The two analytes have close retention times due to their similar chemical structures and polarities (Figure 1). They also have product ions with almost the same mass-to-charges, which originates in a chlorine with stable isotopes. In our method, their chromatographic separation was performed using a mobile phase that consisted of 80% acetonitrile. The LC separation of the three monohydroxylates, which have theoretically identical molecular masses (Figure 1), was achieved using a mobile phase that consisted of 41% acetonitrile. A 3-μm particle size column with 250-mm length was needed to separate the three monohydroxylates from a peak observed in the patients’ plasma, which has a mass-to-charge that is identical to the monohydroxylates. The peak area of DM-4110 was well quantified by splitting areas on the vertical line bordered with a slightly overlapping peak (Figure 3(d)). In a supplementary experiment using additional 20 plasma samples collected from heart failure patients, no significant difference was observed between the plasma concentrations estimated by the validated method and an ad hoc method that only detects DM-4110 (Figure S1). Our LC method can separate tolvaptan, its five major metabolites and unidentified compounds including non-targeted metabolites without noise signals, in plasma samples from patients.
All the validation criteria of the US FDA guidance were satisfied with the data obtained using this method. The intra- and inter-day accuracies and imprecisions were 91.6–106.5% and 0.9–10.9% for tolvaptan and its five major metabolites. DM-4107, which was quantified in the method of Jiang et al., 11 was not validated in the present method. Its matrix factor was strongly fluctuating for every measurement with its short retention time at 2.6 min. All the analytes in plasma specimens can be stored stable at room temperature for up to 24 h and at –80°C for up to one month. Ninety-six plasma samples theoretically can be determined in succession using this method due to their stability in mobile phase for up to 24 h after preparation. Our validated method can be utilized for the pharmacokinetic evaluation of tolvaptan and its five major metabolites in patients.
The plasma concentration ranges of all the analytes were measurable within their calibration curves. The trough plasma concentrations of tolvaptan, DM-4110, DM-4111, DM-4119, MOP-21826 and DM-4103 have wide ranges, which were 9.4–279.8, 1.9–16.3, 3.4–88.9, 1.4–10.4, 0.2–1.0 and 40.2–1471 ng/mL, respectively, in 20 heart failure patients treated with 3.75–15 mg of tolvaptan. Jiang et al. used samples taken from volunteers who received a single dose of 15 mg tolvaptan, resulting in the calibration ranges of 1–500 ng/mL for both tolvaptan and DM-4103. 11 The plasma concentration of DM-4103 exceeded 500 ng/mL in some of our data, likely due to its long half-life. In the present study, the calibration range was determined through consideration of the patients’ trough plasma concentrations on the seventh day after starting medication or later. The peak plasma concentrations of all the analytes including DM-4103 would be measurable within the present calibration ranges because the plasma concentration ratio of maximum to minimum of tolvaptan is relatively small. 3
Although the relationship between clinical effects and plasma concentrations of tolvaptan and its metabolites remains to be clarified in clinical settings, their simultaneous quantitation in human plasma in early stage of treatment may be helpful for the prediction of tolvaptan efficacy and adverse effects. Dose and body weight adjusted plasma concentrations of tolvaptan and its metabolites had a large individual variation, which can be a result of individual differences in the absorption and elimination process. Tolvaptan is mainly metabolized by CYP3A4 and CYP3A5 and is recognized by ABCB1.8,19,20 The metabolic ratios of tolvaptan also had a large individual variation, although the patients who had concomitant drugs with strong interactions were excluded in the study. Individual differences in CYP3A4 activity and genetic polymorphisms of CYP3A5 and ABCB1 can modify the plasma exposure and metabolic ratios of tolvaptan. Our method can be clinically applied to further investigation into factors of individual variation in plasma tolvaptan exposure, leading to better treatment based on each patient’s characteristics.
The current method has several limitations. First, application of the method is limited to heart failure patients. Patients with liver cirrhosis or autosomal dominant polycystic kidney disease are expected to have higher plasma concentrations because of less hepatic CYP activity or higher dose of tolvaptan. 21 The method needs to be evaluated with respect to its suitability in patients with these diseases. Second, the method required a total run time of 15 min using a conventional 3-μm particle size octadecylsilyl column of 250 mm in length. If a high-pressure LC system using an octadecylsilyl column with sub 2-μm particles is employed, the total run time could be shortened. In our method, the maximum pressure was approximately 18 MPa throughout the gradient analysis. The peaks of tolvaptan and its five metabolites in human plasma would be well separated even using a conventional LC system. Third, a few unidentified peaks were found in plasma samples, under the condition of the detection of DM-4110, DM-4111 and DM-4119. Those peaks, including the one positioned to the immediate left of DM-4110 in Figure 3(d), are expected to originate from non-targeted monohydroxylates, probably including the diastereomer of DM-4119. Tolvaptan is racemic with a chiral quaternary carbon centre; therefore, its monohydroxylates inherently exhibit chirality. Although the separation of diastereomers of the monohydroxylates was achieved in the method, that of enantiomers was not implemented. Further separation with a chiral column is needed to separate and quantify enantiomers of tolvaptan and its monohydroxylates.
In conclusion, the present validated method with acceptable analytical performance can be utilized for evaluating the pharmacokinetics of oral tolvaptan, including the determination of its major metabolites, in heart failure patients.
Supplemental Material
Supplemental Material1 - Supplemental material for Validated liquid chromatography coupled to tandem mass spectrometry method for simultaneous quantitation of tolvaptan and its five major metabolites in human plasma
Supplemental material, Supplemental Material1 for Validated liquid chromatography coupled to tandem mass spectrometry method for simultaneous quantitation of tolvaptan and its five major metabolites in human plasma by Kohei Hoshikawa, Takafumi Naito, Masao Saotome, Yuichiro Maekawa and Junichi Kawakami in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Material2 - Supplemental material for Validated liquid chromatography coupled to tandem mass spectrometry method for simultaneous quantitation of tolvaptan and its five major metabolites in human plasma
Supplemental material, Supplemental Material2 for Validated liquid chromatography coupled to tandem mass spectrometry method for simultaneous quantitation of tolvaptan and its five major metabolites in human plasma by Kohei Hoshikawa, Takafumi Naito, Masao Saotome, Yuichiro Maekawa and Junichi Kawakami in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Material3 - Supplemental material for Validated liquid chromatography coupled to tandem mass spectrometry method for simultaneous quantitation of tolvaptan and its five major metabolites in human plasma
Supplemental material, Supplemental Material3 for Validated liquid chromatography coupled to tandem mass spectrometry method for simultaneous quantitation of tolvaptan and its five major metabolites in human plasma by Kohei Hoshikawa, Takafumi Naito, Masao Saotome, Yuichiro Maekawa and Junichi Kawakami in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
We thank Otsuka Pharmaceutical Co., Ltd for their donation of several tolvaptan metabolites.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: DM-4110, DM-4111, DM-4119 and DM-4103 were donated by Otsuka Pharmaceutical Co., Ltd.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This study was approved by the Ethics Committee of Hamamatsu University School of Medicine (17–012). All patients signed an informed consent form.
Guarantor
TN.
Contributorship
KH and TN planned and designed this study. Acquisition of data was carried out by KH. KH, TN and JK contributed to the analysis and interpretation of data. All authors contributed to drafting and revision of the manuscript for important intellectual content and provided final approval for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
