Abstract
We report a 49-year-old woman with an acute swollen left knee due to acute pseudogout with chondrocalcinosis as a presenting feature of Gitelman syndrome due a novel homozygous mutation of the SLC12A3 gene. This report highlights the under-recognized importance of excluding metabolic disease, including Gitelman syndrome, in younger patients whose sole presenting feature may be chondrocalcinosis with or without pseudogout, as this may impact on management and risk of further episodes. We also suggest that chondrocalcinosis and hypomagnesaemia with or without hypokalaemia are diagnostic of Gitelman syndrome.
Keywords
Introduction
Gitelman syndrome (GS), also known as familial hypokalaemia-hypomagnesaemia, is an autosomal recessive renal tubulopathy characterized by hypokalaemic metabolic alkalosis with hypomagnesaemia and hypocalciuria.1–3 It is typified by high phenotypic variability from asymptomatic to severe clinical manifestations secondary to the electrolyte abnormalities.1–3 We report a 49-year-old woman with GS presenting solely with acute pseudogout and chondrocalcinosis and discuss the pathophysiology, differential diagnosis and management of this under-appreciated association.
Case report
A 49-year-old woman presented to the emergency department (ED) with a three-day history of a non-traumatic, painful and swollen left knee with difficulty in weight-bearing. Her medical history included partial thyroidectomy for hyperthyroidism, subclinical autoimmune primary hypothyroidism and alpha-thalassaemia trait.
On examination, she was afebrile with a blood pressure of 94/55 mmHg and a regular heart rate of 78 beats per minute. Her left knee was tender with an effusion, restricting active and passive range of movements.
Radiography of her left knee showed chondrocalcinosis (articular cartilage calcification). Aspirated synovial fluid revealed rhomboid-shaped positively birefringent crystals under polarized light microscopy typical of calcium pyrophosphate dehydrate (CPPD) crystals diagnostic of pseudogout. Culture of synovial fluid excluded sepsis. Her white cell count, C-reactive protein, uric acid and routine biochemistry were normal.
She was treated with oral non-steroidal anti-inflammatory agents (NSAIDs) and discharged from the ED with planned follow-up in rheumatology outpatients and the option of rapid access to the rheumatology service should she not improve.
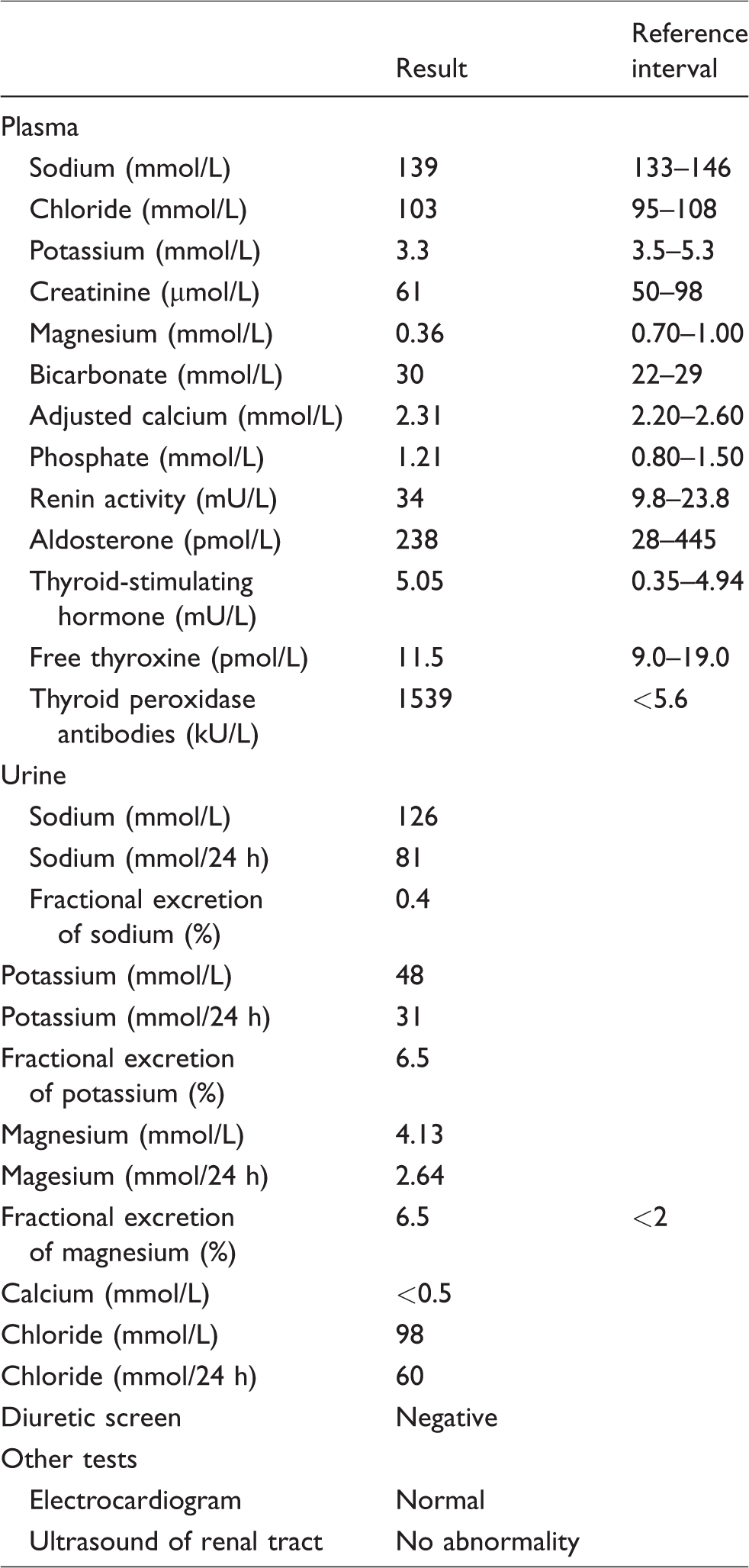
At her rheumatology outpatient assessment, she was found to be hypokalaemic (plasma potassium 3.0 mmol/L) and review of previous results showed intermittent mild hypokalaemia (plasma potassium 3.2–3.7 mmol/L). She was, therefore, referred for metabolic assessment. At review, having recovered from acute pseudogout, she was asymptomatic without polyuria, nocturia or myopathic symptoms. Biochemical investigations (Table 1) were typical of GS showing hypokalaemic metabolic alkalosis with hypomagnesaemia, hypocalciuria and hyperchloriduria, raised plasma renin (in the presence of hypotension) and a negative diuretic screen.1–3 Subsequent sequencing analysis of the SLC12A3 gene detected a novel homozygous mutation c.2687G>A in exon 23, consistent with GS.
Investigations.
The patient was counselled about GS, symptoms of hypokalaemia and hypomagnesaemia and advised when to seek medical help. A liberal salt diet high in potassium and magnesium was recommended. Magnesium supplements were initiated, but magnesium L-aspartate and magnesium glycerophosphate caused diarrhoea. She had two further acute attacks of pseudogout before commencing magnesium L-lactate SR tablets, but diarrhoea limited treatment to one tablet twice daily (7 mmol/day) and subsequently amiloride was added. These corrected her hypokalaemia but only partially corrected her hypomagnesaemia. She has had one further attack of acute pseudogout. The patient’s siblings did not have electrolyte abnormalities.
Discussion
The patient presented with acute monoarthritis, defined as a single joint arthritis of less than two weeks’ duration. 4 Crystals, infection and trauma are the most common causes of acute adult monoarthritis. 4 In the absence of trauma and infection, the cause of the acute monoarthritis in this patient based on X-ray and synovial fluid findings was chondrocalcinosis and acute pseudogout due to CPPD crystal deposition disease (CPPD-CDD). Gout, an important differential diagnosis, is caused by monosodium urate monohydrate (MSU) crystals. Under a polarizing filter and red compensator filter, MSU crystals appear needle-shaped and negatively birefringent whereas CPPD crystals are smaller, more difficult to detect, weakly positively birefringent or not birefringent and polymorphic with predominantly rhomboids but also rods and cubes. However, CPPD and MSU crystals may co-exist. 5 While the first metatarsophalangeal joint is most frequently affected in acute gout, large joints are more commonly affected in acute pseudogout – the knee most commonly, followed by the wrist, ankle, elbow, toe, shoulder and hip. 6 Unlike gout, pseudogout is not associated with tophi or renal stones. Rarely, calcium oxalate, apatite and lipid crystals may cause arthropathies. 4
Acute pseudogout may occur spontaneously or be provoked by trauma, surgery, severe medical illness and pamidronate therapy. 6 Acute pseudogout (acute CPPD crystal arthritis) occurs in 25% of patients with CPPD-CDD. The other distinct presentations of CPPD-CDD are asymptomatic CPPD, pseudo-osteoarthritis (OA with CPPD), pseudo-rheumatoid arthritis and pseudo-neuropathic arthropathy. 6
Synovial fluid culture and blood cultures (if systemically toxic) are mandatory to exclude superimposed septic arthritis. 1 Treatment interventions for acute aseptic pseudogout depend on the number of involved joints and feasibility of joint injections. If one or two joints are acutely inflamed, arthrocentesis and intra-articular glucocorticoids are the preferred option unless the likelihood of a septic joint cannot be ruled out on clinical grounds (e.g. fever, rash, foci of infection and purulent appearing synovial fluid). In such cases, intra-articular glucocorticoids should be deferred until the results of synovial fluid and blood cultures are available. A failure to achieve a response within 48–72 h or multiple involved joints necessitates the use of an oral anti-inflammatory medication (NSAID, colchicine or glucocorticoid). 6 In patients with three or more attacks of acute pseudogout per annum, prophylactic treatment with colchicine is an option. 6
Chondrocalcinosis, radiographic calcification in hyaline and fibrocartilage, is typical of pseudogout. Chondrocalcinosis is present on radiographs in 4.5% of those over the age of 40 years and increases to 50% in those over 85 years. Chondrocalcinosis in the elderly is largely idiopathic, but its occurrence with or without acute pseudogout in individuals less than 60 years should prompt consideration of autosomal-dominant familial chondrocalcinosis and other associated diseases. 6 Epidemiological studies have reported CPPD-CDD to be associated with hyperparathyroidism, hypomagnesaemia, haemochromatosis, rheumatoid arthritis, osteoporosis and gout (Table 2).5–7 The relationships between acromegaly, Wilson’s disease, hypophosphatasia, diabetes mellitus and CPPD-CDD are less clear and largely based on case reports (Table 2).5–7
Learning points.

CPPD-CDD: calcium pyrophosphate dehydrate-crystal deposition disease.
This patient’s age of presentation, hypotension, biochemical findings and genetic studies confirmed GS. GS is the most common inherited tubulopathy with a prevalence between 1 and 10 per 40,000.1–3 GS is due to mutations in the SLC12A3 gene, which encodes the thiazide-sensitive sodium chloride (NaCl) co-transporter (NCC).1–3 NCC mutations lead to reduced NaCl reabsorption in the distal collecting duct (DCT) resulting in hyperchloriduria and mild volume contraction. Volume contraction, manifesting as low or low normal blood pressure, activates the renin-angiotensin-aldosterone system increasing renin and aldosterone. Secondary hyperaldosteronism results in hypokalaemic metabolic alkalosis with an inappropriate kaliuresis. The mechanisms of hypocalciuria and hypomagnesaemia are unclear. It has been proposed that in GS, increased passive calcium reabsorption in the proximal tubule and downregulation of TRPM6 magnesium channels in the DCT explain the hypocalciuria and renal wasting hypomagnesaemia, respectively.1–3
Type III Bartter syndrome, caused by mutations in the CLCNKB gene (encoding the chloride channel ClC-Kb), clinically and biochemically overlaps with GS but may be differentiated by the response to thiazide and loop diuretics. 3 In GS, there is a blunted response to thiazides, whereas in Bartter syndrome, there is a blunted response to loop diuretics. These tests are rarely performed because the occasional marked diuresis may produce adverse effects and have been replaced by more definitive genetic testing. 3 Other types of Bartter syndrome present earlier with a more severe phenotype. 3 Surreptitious causes of hypokalaemia including diuretic abuse, laxative abuse and chronic vomiting (as in anorexia/bulimia) should be excluded. 8 Laxative abuse causes a hyperchloraemic normal anion-gap metabolic acidosis and hypokaliuric hypokalaemia and therefore is readily distinguished from GS. Vomiting is characterized by a hyperkaliuric hypokalaemic metabolic alkalosis, but the associated low urine chloride differentiates it from GS, in which urine chloride is invariably high (typically above 40 mmol/L). Thiazide diuretic abuse may produce identical biochemical features to those of GS. Urinary chloride is variable in diuretic abuse; high after ingestion but falls as diuretic effect wears off. A high urine chloride may, therefore, only be attributed to GS after a negative diuretic screen. Although biochemical findings may be similar, we suggest that the presence of chondrocalcinosis and pseudogout is indicative of GS, as they are not associated with thiazide diuretics, 5 but this remains to be fully clarified. 9
Chronic hypomagnesaemia has been implicated in the pathogenesis of CPPD-CDD. 9 Magnesium is a cofactor for alkaline phosphatase (ALP). Decreased ALP activity due to hypomagnesaemia increases extracellular ionic inorganic pyrophosphate (PPi). Since magnesium is also unavailable to chelate PPi, the increased ionic product of calcium and PPi predisposes to CPPD crystal deposition. 10 Supporting data are from reports of increased synovial fluid PPi in patients with hypomagnesaemia. 10 In addition, observational studies have reported that magnesium supplements reduce the recurrence of acute attacks of CPPD-CDD in patients with and without hypomagnesaemia-related CPPD-CDD. 10
To prevent further attacks of pseudogout, treatment is aimed at correcting hypomagnesaemia with oral magnesium supplements, 10 but their use may be limited by magnesium-induced diarrhoea as described in this case. Drugs which block distal renal tubular sodium–potassium exchange (spironolactone, amiloride, eplerenone) may also help increase plasma magnesium and potassium concentrations.
Genetic counselling is recommended. As GS is an autosomal recessive condition, the risk of having children with GS is low (about 1:400) unless in a consanguineous partnership. 2 Patients must be counselled about the symptoms of hypokalaemia and hypomagnesaemia and factors, such as diarrhoea and vomiting, which may precipitate severe hypokalaemia and hypomagnesaemia. Severe electrolyte abnormalities may be associated with neuromuscular and cardiac manifestations requiring treatment with intravenous magnesium with or without potassium.1–3,8 The long-term prognosis, however, of GS is excellent.
In summary, chondrocalcinosis and acute pseudogout may be the sole presenting features of GS, of which we suggest the hallmark is hypomagnesaemia since this case indicates that hypokalaemia may not always be present. This report underlies the importance of excluding secondary associations of chondrocalcinosis and acute pseudogout, such as GS, in younger individuals as treatment of these may prevent the recurrence of acute pseudogout.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Informed written consent obtained from the patient.
Guarantor
IB.
Contributorship
RMG and IB researched the literature and wrote the manuscript. IB, JW, NB and RG contributed to the data. RG and NB are responsible for the clinical care of the patient. IB conceived the case report. All authors critically reviewed the manuscript and approved the final version of the manuscript.
