Abstract
Background
Extracorporeal membrane oxygenation procedure (ECMO) has been established in the therapy of respiratory insufficient infants with congenital diaphragmatic hernia. In congenital diaphragmatic hernia newborns, a delay in transfer to an ECMO centre is associated with a sharp increase in mortality. Predictive factors for ECMO support are urgently needed. We evaluated the routine parameters of the first blood withdrawal after birth in congenital diaphragmatic hernia infants, hypothesizing that early signs in bone marrow affecting haematology parameters for early regulation of potentially post birth hypoxia are predictive factors for ECMO support.
Materials and methods
In 44 patients born with congenital diaphragmatic hernia, differential blood cell count from the first blood withdrawal after birth was examined. Descriptive statistics included median, 95% confidence intervals, minimum and maximum differentiating ECMO/early mortality vs. no ECMO. Odds ratios with CI were calculated by binary logistic regression analysis. Best predictive markers were further checked in combination with the liver-up situation in two factorial regression models.
Results
In our cohort, the survival rate was 77.3% (34/44). While 18 neonates received ECMO support, 26 experienced no ECMO during hospital stay. Odds ratio calculations showed that risk for ECMO support increases with augmenting leukocytes, erythrocytes, haemoglobin, haematocrit, mean cell volume and absolute immature granulocytes. Further, the risk advanced in line with the severity of congenital diaphragmatic hernia assessed by prenatal ultrasound.
Conclusions
We conclude that these parameters are associated with disease severity in congenital diaphragmatic hernia newborns and may be considered potentially predictive biomarkers for the necessity of ECMO support.
Introduction
Congenital diaphragmatic hernia (CDH) is a rare disease with an incidence of 1 to 2500–3000 in live births. Its clinical presentation varies widely and is associated with high mortality and long-term morbidity. 1 During development, an orifice remains in the diaphragm, which ultimately leads to organ herniation into the thorax, hypoplasia of the pulmonary vasculature and the airway system and pulmonary hypertension with insufficient oxygenation after birth. Additional malformations are often involved. 2 Although much effort has been spent to improve therapeutic options, mortality rates remain high. 3 For several years, extracorporeal membrane oxygenation procedure (ECMO) has been established in the therapy of respiratory insufficient infants with CDH. 4 Despite being controversially discussed, it is a widely used procedure. 5 Since a delayed transfer (i.e. > 24 h after birth) to an ECMO centre is associated with a sharp increase in mortality in CDH newborns, predictive factors for ECMO support are urgently needed. 6 In infants with CDH, a lower observed-to-expected lung-to-head ratio (o/e LHR) and an intrathoracic liver position (liver-up) are associated with worse outcome, and risk stratification is possible in line with this information.7,8 Although many studies are investigating the pathomechanisms, little is known regarding changes in haematopoiesis. 9 Following admission to a hospital setting, the complete blood cell count is always determined in CDH newborns. While there are continuous improvements in the analysis technologies, complete blood cell count should be evaluated as a predictive factor for ECMO support in CDH infants. Blood cell count parameters are of outstanding importance in a variety of childhood diseases. Immature platelet fraction (IPF) is a parameter that differentiates causes for thrombocytopenia. 10 Cremer et al. have shown its value in sepsis diagnosis in neonates. 11 Immature granulocytes (IG) seem to be a predictive factor in neonatal sepsis. 12 Elevated nucleated red blood cells (NRBC) are predictive of mortality in very low birth weight infants. 13 Foetal erythropoiesis is regulated by complex mechanisms due to physiologically low oxygen partial pressure in utero in contrast to erythropoiesis after birth. 14 While the role of regulation in bone marrow activity for all cell lines has not yet been evaluated in CDH infants, data from an animal model indicate that erythropoietin (EPO) as a progenitor to red cell differentiation is impaired in CDH. 15
The aim of the present study was to assess blood cell count values early after birth as part of routine analysis for predicting ECMO in these patients.
Patients and methods
Setting
This is a single centre retrospective observational study performed at the central laboratory and the Department of Neonatology and Pediatric Critical Care Medicine of the University Clinics Bonn, Germany. The department of neonatology is a large level III perinatal centre with approximately 600 admissions/year (approx. 80 very low birth weight infants, 20–30 CDH infants, approx. 15 neonatal ECMOs).
Patients
The collective included data from 44 patients born with CDH from February 2014 to April 2016. One child died immediately after birth, and data were not included for further analysis. Data were collected from the central laboratory information system (SWISSLAB II, Roche, Berlin, Germany), from the neonatal information system (Neodat, Tubingen, Germany) and the internal CDH database. Criteria for ECMO were based on current recommendations of the CDH-Euro Consortium: preductal oxygen saturation <85% or postductal saturation <70%, OI ≥40 consistently present, increased PaCO2>70 mmHg with a pH <7.15, a peak inspiratory pressure ≥28 cm H2O or mean airway pressure ≥17 cm H2O or persistent systemic hypotension (mean arterial pressure <40 mmHg). 16
Methods
Venous EDTA whole blood samples were examined. In all samples, analysis was performed within 15 min of arrival at the central laboratory. We analysed values from the first blood withdrawal after birth. Values at the central laboratory were measured with Sysmex XN1000™ (Sysmex, Norderstedt, Germany). Hb concentration was photometrically measured with the sodium lauryl sulphate (SLS) haemoglobin method. Erythrocytes were measured with impedance technique. Thrombocytes and IPF were measured by fluorescent staining. Haematocrit and mean cell volume (MCV) were determined with summarizing impulse heights; mean corpuscular haemoglobin and mean corpuscular haemoglobin concentration were computed parameters. Lymphocytes, monocytes, neutrophil granulocytes, eosinophil granulocytes and IG were measured with fluorescence flow cytometry in the WDF channel, while basophil granulocytes were measured in the WNR channel. All methods are part of routine methods at the central laboratory (accredited according to DIN EN ISO 15189:2014) and are performed in line with legal requirements (RiliBÄK, Guideline of the German Medical Association) with stipulated internal and external quality controls.
Statistics
Data were statistically analysed using IBM SPSS® Statistics, Version 20 (Ehningen, Germany) and MedCalc® (Ostend, Belgium). P < 0.05 was considered statistically significant. Variables were described with median value (95% confidence interval [CI]), minimum and maximum differentiating ECMO/early mortality vs. no ECMO. Values were obtained during the first blood withdrawal. Next, odds ratios with CI and P-values were calculated for a set of potentially predictive markers by binary logistic regression analysis. Best predictive markers were further checked in combination with the liver-up situation in two factorial regression models and odds ratios with CI and P-values were obtained. Box-plots were generated with median, interquartile ranges, minimum and maximum. Receiver operating characteristics (ROC) curves and areas under the curve (AUC) were calculated.
Ethics
In article six of the German Data Protection Act (https://recht.nrw.de/lmi/owa/br_text_anzeigen?v_id= 10000000000000000495#), it is stipulated that the physician may use existing patient data for retrospective analyses without explicitly asking for the consent of patients. The study design is consistent with the Declaration of Helsinki. According to information obtained from the Institutional Review Board (Ethikkommission an der Medizinischen Fakultät der Rheinischen Friedrich-Wilhelms-Universität Bonn, Chairman K. Racké, MD, PhD, Professor, University Bonn), retrospective analysis of data obtained during routine treatment and diagnosis does not require consultation by the ethics committee according to article 15 of the German medical professional code.
Results
In our cohort, the survival rate was 77.3% (34/44), survival-to-discharge was 77.3% and the median age at discharge in survivors was 45 days (range: 13–255 days). No fatalities occurred after discharge and all patients attended regular follow-up visits in our outpatient clinic. The median age at the latest follow-up was 3.4 years (range: 2.1–4.7 years). While 18 neonates received ECMO support, 26 required no ECMO during hospital stay. One patient died immediately after birth; four children with early mortality due to contraindication to ECMO despite fulfilling ECMO criteria were included in the ECMO group. Male to female ratio was 3.2:1. Ten infants in the ECMO/early mortality group died (10/18, 55.5%) and 27 infants (61.4%) suffered from CDH with liver-up condition. In all patients, blood for CBC analysis was withdrawn after insertion of an umbilical catheter 20 and 45 min after birth. The median age of initiating ECMO support was 9.6 h (IQR 7.1–19.4 h). Patient characteristics, including gestational age, birth weight, lung-to-head ratio, duration of ventilation, hospital stay, score for neonatal acute physiology-II (SNAP-II) and analytes for patients with or without ECMO therapy are presented in Table 1. Odds ratios, 95% CI and P-values for potentially predictive markers for ECMO therapy are presented in Table 2.
Descriptive statistics from first blood withdrawal of children born with CDH treated with or without ECMO therapy.
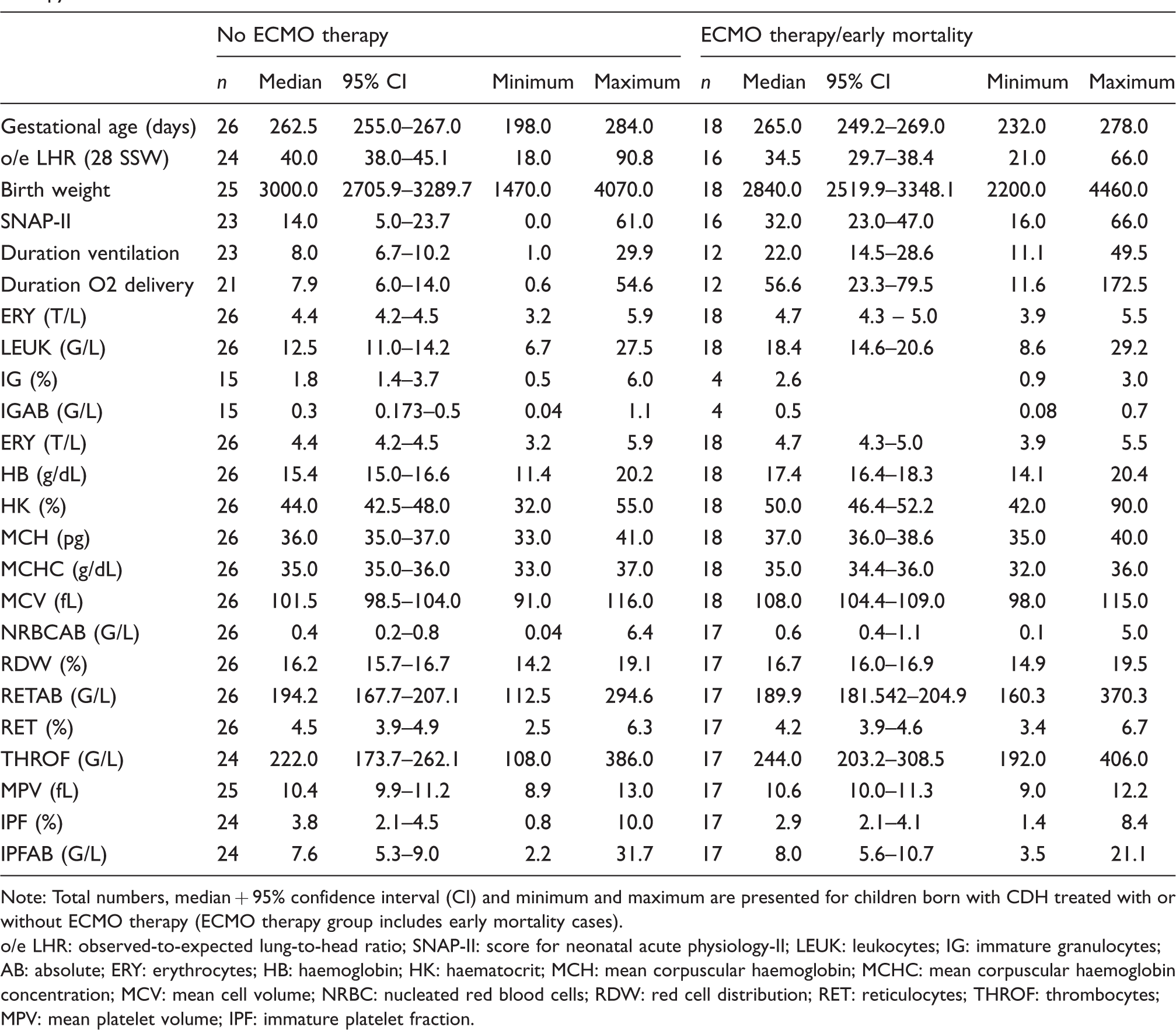
Note: Total numbers, median + 95% confidence interval (CI) and minimum and maximum are presented for children born with CDH treated with or without ECMO therapy (ECMO therapy group includes early mortality cases).
o/e LHR: observed-to-expected lung-to-head ratio; SNAP-II: score for neonatal acute physiology-II; LEUK: leukocytes; IG: immature granulocytes; AB: absolute; ERY: erythrocytes; HB: haemoglobin; HK: haematocrit; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; MCV: mean cell volume; NRBC: nucleated red blood cells; RDW: red cell distribution; RET: reticulocytes; THROF: thrombocytes; MPV: mean platelet volume; IPF: immature platelet fraction.
Odds ratios, 95% confidence intervals (CI) and P-values for predictive markers for ECMO therapy.
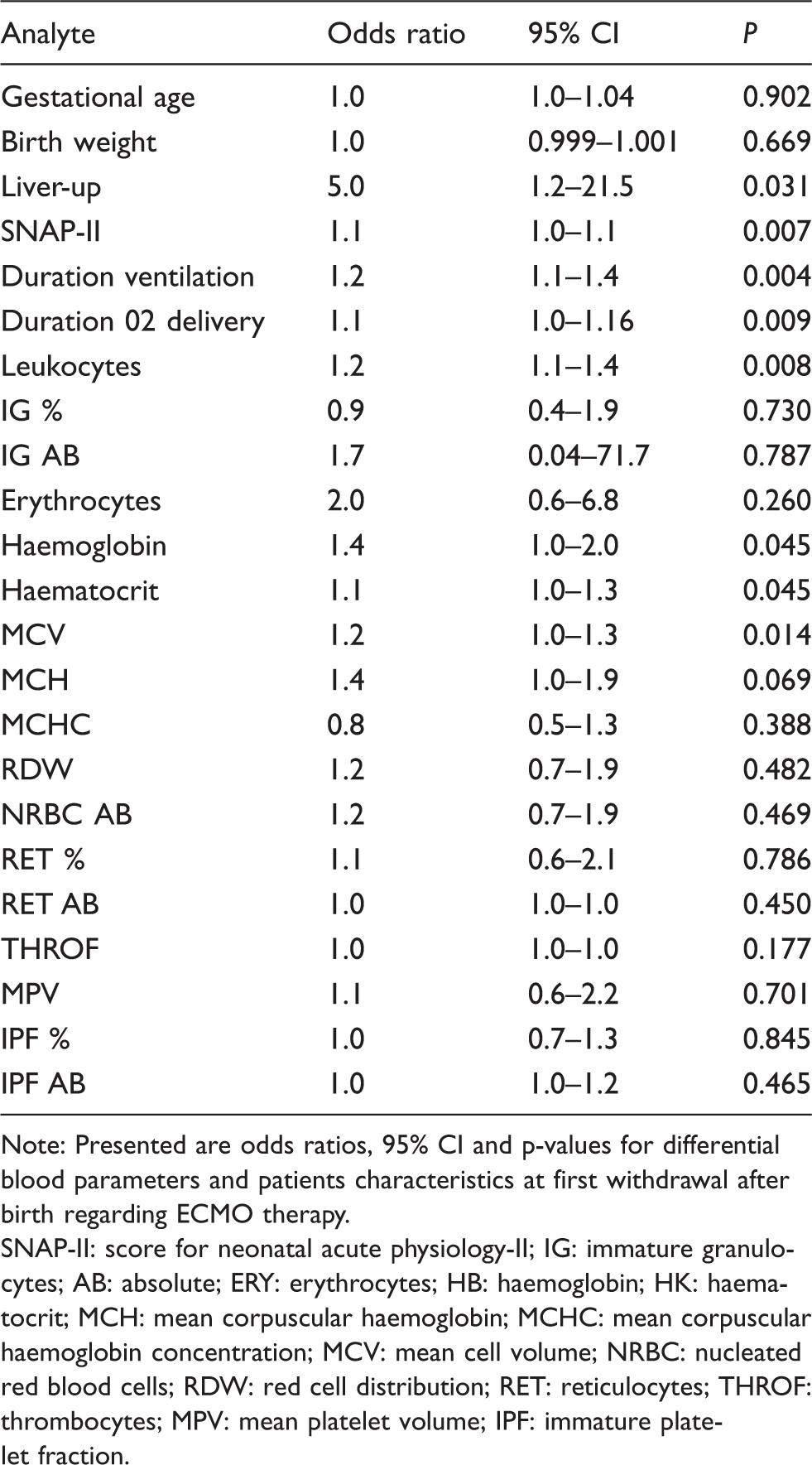
Note: Presented are odds ratios, 95% CI and p-values for differential blood parameters and patients characteristics at first withdrawal after birth regarding ECMO therapy.
SNAP-II: score for neonatal acute physiology-II; IG: immature granulocytes; AB: absolute; ERY: erythrocytes; HB: haemoglobin; HK: haematocrit; MCH: mean corpuscular haemoglobin; MCHC: mean corpuscular haemoglobin concentration; MCV: mean cell volume; NRBC: nucleated red blood cells; RDW: red cell distribution; RET: reticulocytes; THROF: thrombocytes; MPV: mean platelet volume; IPF: immature platelet fraction.
Value distributions of analytes and outcome parameters in ECMO vs. no ECMO groups are displayed in boxplots (see Supplemental Figure 1).
Supplemental Figure 2 shows ROC curves and areas under the curve for the analysed parameters (see Supplemental Figure 2). Best predictive markers were further analysed in combination with the liver-up situation in two factorial regression models. Results for odds ratios with 95% confidence intervals and P-values are presented in Table 3.
Odds ratios, 95% confidence intervals (CI) and P-values assessed in combination with the liver-up situation in two factorial regression models for best markers as predictive factors for ECMO therapy.
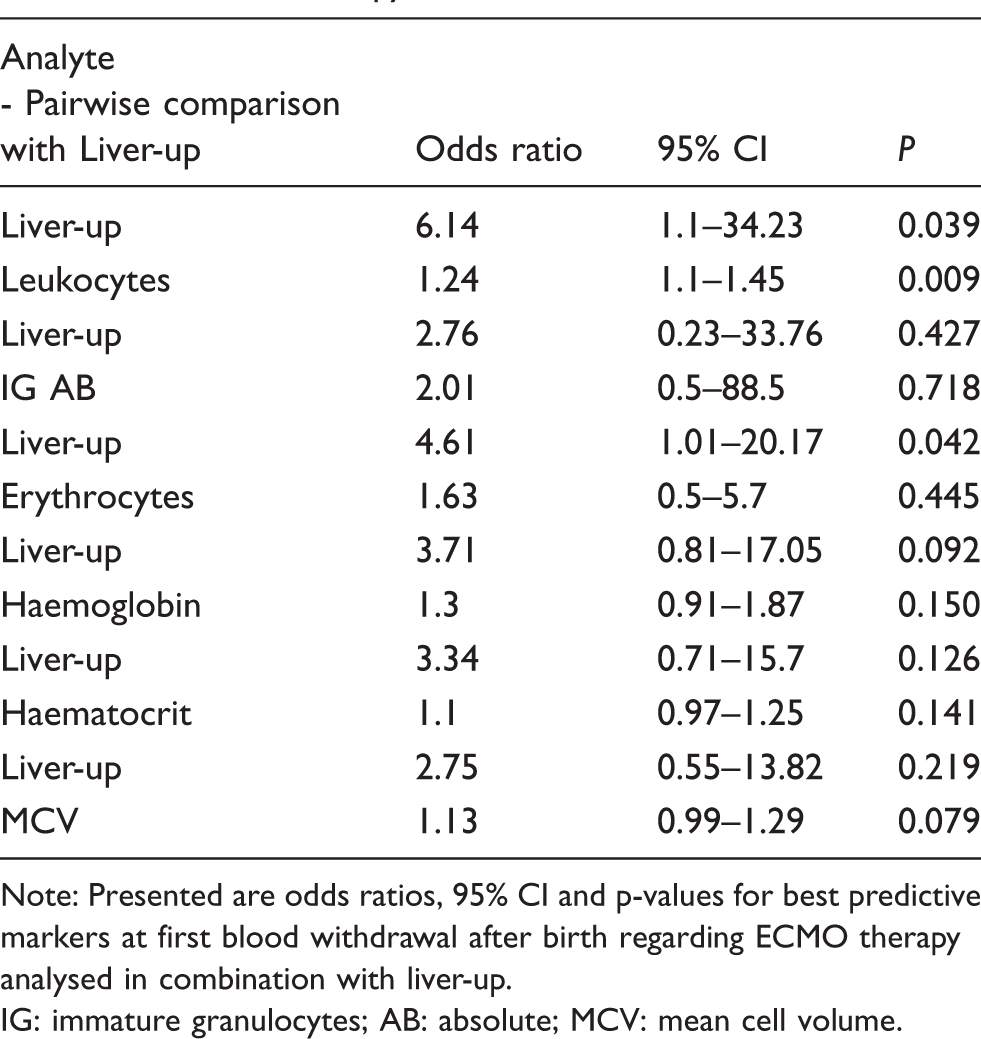
Note: Presented are odds ratios, 95% CI and p-values for best predictive markers at first blood withdrawal after birth regarding ECMO therapy analysed in combination with liver-up.
IG: immature granulocytes; AB: absolute; MCV: mean cell volume.
Discussion
Management of CDH newborns remains a great challenge today with high morbidity and mortality. 17 Schaible et al. reported a sharp increase in mortality in CDH newborns following delayed (i.e. > 24 h after birth) transfer to an ECMO centre. 6 Additionally, newborns surviving after ECMO support might demonstrate neurocognitive impairment as a crucial side-effect. 18 Therefore, factors predicting the need for ECMO support are paramount in these patients. Although ECMO treatment outcome in neonates is generally satisfactory, ECMO support in CDH patients can cause disorders, such as problems in hearing and vision, impaired neurocognitive development, concentration problems, etc. 19 We assessed the values of blood count analytes immediately (ca. 30 min) after birth for predicting ECMO in children born with CDH. In order to establish a new predictor for outcome, it is important that measurement of CBC is not to close to the predicted event. In our study, the median time of approximately 9 h from blood sampling to initiation of ECMO support, represents long enough duration to plan ECMO cannulation. Odds ratio calculations showed in combination with liver-up that risk for ECMO support grows with increased levels of leukocytes. Erythrocytes, haemoglobin, MCV and absolute IG in combination with liver-up situation show an elevated risk for ECMO therapy. Further, the risk increased in line with severity of CDH as assessed by prenatal ultrasound (i.e. o/e LHR, liver-up). We conclude that these parameters are associated with disease severity in CDH newborns and may be considered potentially predictive biomarkers for the necessity of ECMO support. ECMO support is a therapeutic option in critically respiratory insufficient neonates. 5 Our results indicate that these parameters are not only predictive for the necessity of ECMO support but may also predict different outcome parameters during the hospital treatment (e.g. duration of ventilation, oxygen dependency). Haemoglobin is the most important component of the erythrocyte, which is linked to the oxygen transport capacity. Reticulocytes can be seen as an indicator for effectiveness of erythropoiesis. 20 To our knowledge, changes in blood cell lines have not been previously investigated in CDH newborns.
Evidence from a CDH animal model shows that EPO synthesis is suppressed in CDH rats and that it is associated with more severe CDH. 15 However, based on these findings, we would expect to observe a reduced amount of haemoglobin. Erythroid cells mature with the help of EPO, but EPO does not affect differentiation and survival of erythrocytes. 21 Additionally, it is well described that erythropoiesis is stimulated not only by EPO but also by several other factors, including interleukins IL-3, IL-6 and IL-9.22,23 Previous studies suggest an increased expression of several proinflammatory cytokines, including IL-6, in more severely affected CDH newborns. 24 Therefore, increased IL-6 expression during foetal life could be responsible for the significant alterations in the blood cell lines observed in our study. Future studies need to investigate a potential interaction between inflammation and erythropoiesis in CDH newborns. However, EPO measurement is expensive and requires a high sample volume for measurement making it less attractive as a biomarker candidate in CDH newborn research.
Theoretically, the reported changes in haematopoietic cells might result from severe hypoxia due to CDH-associated lung hypoplasia, which may stimulate EPO-mediated erythropoiesis. However, in the majority of patients, blood was drawn for analysis within the first 30 min after delivery, allowing only a limited time period for potential hypoxia to affect blood count parameters. Furthermore, our cohort consisted of patients with prenatally diagnosed CDH, and in this setting, all infants received optimal care within a standardized protocol, including immediate intubation and ventilation with 100% oxygen as recommended. 16 Therefore, a substantial number of patients may not have experienced profound hypoxia during initial resuscitation. Additionally, delayed cord clamping was not performed in any patient during the study period and volume supplementation during early postnatal resuscitation was similar between groups (data not shown).
In a recent study, the commercially widely available high-sensitivity troponin T and N-terminal pro-brain natriuretic peptides were not predictive for subsequent ECMO support in CDH newborns. 25 Beside biomarkers, foetal lung size and intrathoracic liver herniation can be used as prenatal predictive factors for ECMO support. 26 Additionally, approaches using echocardiographic assessment or calculation of SNAP II have been reported.27,28 We focused on haematopoietic cells with their indices for several reasons: our reported analytes can be measured from a very small sample volume, the technique is inexpensive, with a round-the-clock availability in every hospital setting. Since blood sampling from infants could lead to relevant anaemia, a further advantage was that no extra blood had to be taken, and analysis was carried out rapidly as no centrifugation step is necessary for haematology analysis. In contrast to previous studies, IPF measuring was performed with improved fluorescence technique for the haematology analyser.10,11,29 All analytes underlie internal and external quality control schemes in our accredited laboratory. In previous studies, standardization and comparability of the analysed materials were rarely provided. 25 Our results for SNAP-II as a predictive factor for ECMO support are in line with previously reported results. 29
Limitations of the study
Routine diagnostic was carried out in the context of first treatment of our patients and took place on average within 20 min during routine conditions after placement of umbilical vessel catheter. We did not correct the P-value for multiple testing in our study. Therefore, the chosen significance level of P < 0.05 incorporates the likelihood of false-positive associations given the number of significance tests performed and should be considered to be explorative. An additional limitation is the retrospective character of our study, and all findings need to be confirmed with a prospective design.
Conclusion
We evaluated routine parameters of the first blood withdrawal after birth in CDH infants hypothesizing that early signs in bone marrow affecting haematology parameters of children with CDH for early regulation of potentially post birth hypoxia are predictive factors for ECMO support. We found that increased leukocytes, erythrocytes, haemoglobin, HK, MCV, absolute IG and liver-up condition are predictive factors for ECMO support. Our results need confirmation by a larger observational study.
Supplemental Material
Supplemental Figure legends - Supplemental material for Haematopoietic alterations in neonates with congenital diaphragmatic hernia receiving extracorporeal membrane oxygenation support
Supplemental material, Supplemental Figure legends for Haematopoietic alterations in neonates with congenital diaphragmatic hernia receiving extracorporeal membrane oxygenation support by Ramona C Dolscheid-Pommerich, Alexander Kreuzer, Ingo Graeff, Birgit Stoffel-Wagner, Andreas Mueller and Florian Kipfmueller in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Figure1 - Supplemental material for Haematopoietic alterations in neonates with congenital diaphragmatic hernia receiving extracorporeal membrane oxygenation support
Supplemental material, Supplemental Figure1 for Haematopoietic alterations in neonates with congenital diaphragmatic hernia receiving extracorporeal membrane oxygenation support by Ramona C Dolscheid-Pommerich, Alexander Kreuzer, Ingo Graeff, Birgit Stoffel-Wagner, Andreas Mueller and Florian Kipfmueller in Annals of Clinical Biochemistry
Supplemental Material
Supplemental Figure2 - Supplemental material for Haematopoietic alterations in neonates with congenital diaphragmatic hernia receiving extracorporeal membrane oxygenation support
Supplemental material, Supplemental Figure2 for Haematopoietic alterations in neonates with congenital diaphragmatic hernia receiving extracorporeal membrane oxygenation support by Ramona C Dolscheid-Pommerich, Alexander Kreuzer, Ingo Graeff, Birgit Stoffel-Wagner, Andreas Mueller and Florian Kipfmueller in Annals of Clinical Biochemistry
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable. The study design is consistent with the Declaration of Helsinki.
Guarantor
FK.
Contributorship
RCDP, BSW, AM and FK researched literature and conceived the study. RCDP, AK, IG, BSW, AM and FK were involved in protocol development, gaining ethical approval, patient data recruitment and data analysis. RCDP, AK, IG, BSW, AM and FK wrote the article. All authors reviewed and edited the article and approved the final version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
