Abstract
Background
The study was designed to evaluate the analytical performances of two ERM-DA471/IFCC traceable cystatin C (CysC) reagents available on the market for urinary CysC (u-CysC) quantification. In addition, clinical relevance was assessed by measuring u-CysC in healthy controls and in patients with tubular dysfunction.
Methods
CysC in urine was measured by a particle-enhanced nephelometric immunoassay using Siemens reagents and by a particle-enhanced turbidimetric immunoassay using DiaSys reagents. Imprecision, linearity, limit of detection and limit of quantification were evaluated according to CLSI recommendations. The two methods were tested on 150 urinary samples from 50 healthy subjects, 50 HIV patients with tubular dysfunction and 50 patients who developed acute kidney acute injury.
Results
Within-laboratory coefficients of variations were below 4%. The lower limit of quantification of the assay was found at 0.043 and 0.046 mg/L for DiaSys and Siemens, respectively. The following Passing-Bablok regression equations were obtained: DiaSys = 0.99 Siemens + 0.00. Using Bland-Altman analysis, the mean bias was –0.004 mg/L on the analytical range between 0.02 and 1 mg/L. Median u-CysC in 50 HIV patients with tubular dysfunction and in 50 patients with AKI was higher than in control subjects.
Conclusions
Both Siemens and DiaSys reagents demonstrated reliable and reproducible performances allowing easy determination of u-CysC on automated platforms in clinical practice with potential interest for the detection of tubular dysfunction.
Introduction
Urinary Cystatin C (u-CysC) has emerged as a promising biomarker for early detection of tubular dysfunction. 1 Under physiological conditions, urinary concentration of CysC is very low. An abnormal presence of these proteins in the urine is a marker of tubular dysfunction. 2 In 2015, the ‘Société Française de Biologie Clinique’ working group conducted a multicentric evaluation of automated serum CysC assays after standardization using the certified reference material ERM-DA 471. In the latter study, only Siemens reagents on the Siemens systems, and to some lesser extent the DiaSys reagents on the Cobas system, provided results that satisfied the minimum performance criterion. 3 To the best of our knowledge, no measuring system for u-CysC is Communautés Européennes marked; only adaptation of pre-existing CysC application in accordance with the specific instrument’s settings and the performance characteristics provided by the manufacturer are available. The aim of this study was to evaluate analytical performances of the two best ERM-DA471/IFCC traceable CysC reagents available on the market for u-CysC quantification. In addition, clinical relevance was assessed by measuring u-CysC in healthy controls and in kidney disease patients with tubular dysfunction.
Materials and methods
CysC in urine was measured by a particle-enhanced nephelometric immunoassay (PENIA) using Siemens N Latex Cystatin C reagents on BN Prospec (Siemens Healthcare Diagnostics, Marburg, Germany) and by a particle-enhanced turbidimetric immunoassay (PETIA) using DiaSys Cystatin C FS reagents (DiaSys Diagnostic Systems GmbH, Holzheim, Germany) implemented on Mindray BS480 analyser (Mindray Bio-Medical Electronics Co., Ltd, Shenzhen, China). The Siemens CysC assay protocol was designed for serum analysis with automatic dilution of samples at 1:100 with two subsequent dilutions of 1:5 and 1:20. In this study, the 1:20 predilution of specimens was used for urinary determination of CysC which corresponds to a measuring range from approximately 0.06 to 1.96 mg/L. 4 An optimized u-CysC application was developed by Diasys to obtain better analytical sensitivity using higher volume of sample, decrease volume of reagent, decrease volume of buffer and a calibration generated by manual dilution of the calibrator to cover the range 0–1 mg/L. 5
Imprecision studies were based on the Clinical and Laboratory Standards Institute (CLSI) EP15-A3 protocol by repeat analysis of three urine pools with concentrations of 0.14, 0.41 and 0.71 mg/L. 6 A one-way analysis of variance was performed to estimate repeatability (%CVR) and within-lab imprecision (%CVWL). The limit of blank (LoB) and the limit of detection (LoD) were determined using a method based on CLSI EP17-A guideline. 7 In this protocol, 10 replicates of one analyte-free urine sample and two urine samples that approximate three and four times the assay’s sensitivity claimed by the manufacturer were assayed. LOD was calculated as LOD = LOB + 1.645 σ S, where σ S is the standard deviation of the population of the low concentration sample measurements. To determine the lower limit of quantification (LLOQ), a precision profile was established using a panel of five urine samples with target concentrations at approximately 0.5, 0.22, 0.12, 0.05 and 0.03 mg/L. Samples were measured in duplicate each day during five days. LLOQ was determined by plotting the coefficients of variation (CVs) calculated at each concentration against the mean concentration. LLOQ was defined as the lowest value with an imprecision ≤ 10%. Linearity was evaluated following the CLSI EP6-A8 protocol 8 by successive dilutions of patient urine samples at a high CysC concentration (0.8 and 1.8 mg/L for DiaSys and Siemens measuring system, respectively) with CysC-free urinary samples at the following theoretical concentrations of 100%, 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20% and 10% of the initial concentration and analysed in duplicate over three days. Comparison between expected and obtained u-CysC values was made by linear regression analysis. The panel of urine used was kept frozen at –80°C until measurement in an appropriate number of aliquots (single use), and each aliquot was thawed just before analysis.
Clinical relevance was assessed by measuring urinary CysC in three groups of patients. The control group consisted of 50 patients with normal glomerular filtration rate (estimated GFR >60 mL/min/1.73 m2) and no pathological proteinuria (urinary protein to creatinine ratio <23 mg/mmol). The HIV patients group with kidney tubular dysfunction comprised of 50 patients with the presence of at least three abnormalities: urine protein to creatinine ratio >23 mg/mmol, urine albumin to creatinine ratio <0.5 and urinary percentage maximal tubular reabsorption of phosphate <0.77. The acute kidney injury (AKI) group consisted of 50 patients with defined AKI as urine volume <0.5 mL/kg/h for 6 h or/and a serum creatinine increase ≥26.4 μmol/L or a serum creatinine increase of 1.5 times the baseline value (KDIGO ≥1). 9 Samples collected for this study were registered at the Ministère de l’Enseignement Supérieur et de la Recherche with the number DC-2008–417 and DC-2012–1579 after approval by our institution’s ethical committee. Passing-Bablok regression analysis was performed to compare data from the different methods used for urinary CysC determination. The scatter of differences was visualized with the Bland and Altman graphical method. Mean and limits of agreement (mean ± 1.96 SD) have been computed. The Kruskal-Wallis test was used to compare u-CysC between the three groups. When comparisons were statistically significant, two-by-two comparisons were carried out with the Mann-Whitney test.
Results and discussion
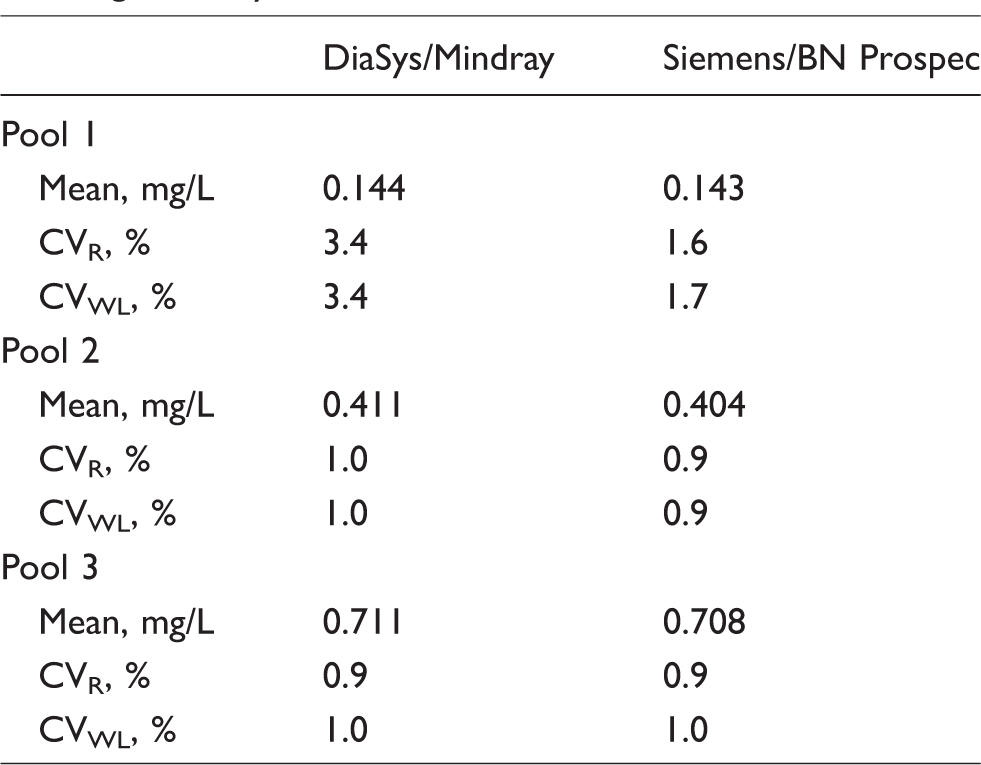
Repeatability and within-laboratory CVs were below 2%, except for DiaSys implemented on the Mindray BS 480 in pool 1 (Table 1). Mean LOD was found at 0.015 and 0.016 mg/L for DiaSys and Siemens, respectively. The LLOQ of the assay was found at 0.043 and 0.046 mg/L for DiaSys and Siemens, respectively. The obtained dilution profiles (y = 1.034 x–0.098, r2 = 0.99 and y = 1.032 x + 0.020, r2 = 0.99 for DiaSys and Siemens, respectively) exhibited a high degree of linearity. The recoveries for the different dilutions ranged from 80% to 102% and from 101% to 110% for DiaSys and Siemens, respectively. Both assays exhibited reliable precision comparable to those previously obtained by PENIA (5%)2,4 and PETIA (2–4%)10–12 methods. Time span for imprecision assessment could explain why the observed analytical performances were slightly better in our study. The slightly higher CV obtained for DiaSys reagent with the urine pool at 0.144 is comparable that previously found using DiaSys reagent on a cobas c502 (Roche Diagnostics GmbH, Germany). 5 Moreover, the results achieved for LOD and LLOQ confirmed the assay precision. They were comparable to those previously published.5,10
Mean values, repeatability (CVR) and within-lab (CVWL) coefficient of variation for the three urinary pools and the reagent–analyser combinations.
The hook effect, which can lead to falsely low results in conditions of antigen excess, represents a potential problem in homogeneous immunoassay methods for analytes with a wide range of concentrations as well described for urinary albumin.13,14 It is also the case for urinary Cyst C values that ranges from undetectable values in healthy volunteers to more than 30 mg/L in patients with tubular injury. During the study, the antigen excess was not determined using spiked samples with varying concentrations of Cyst C5,11,12 but all patients’ results above the highest calibrator value were flagged by the analyser and re-analysed with the appropriate dilution. In addition, no discordant result was found between the two assays. Absorbance values extracted from the instrument software showed no prozone effect for urinary Cys C values up to 30 mg/L since none of the absorbance values fell under that of the highest calibrator for each method. Absorbance of the highest calibrator was about 10,500 Siemens and 1500 for Diasys. Using Siemens reagents, urinary samples’ absorbance above the highest calibrator increases to about 13,000 for Cys C of 3 mg/L and stayed at this value for Cys C up to 30 mg/L. In agreement with previous study, 5 the Diasys assay exhibited a high-dose hook effect from 3 to 4 mg/L urinary Cys C concentration; however, the security range expands up to 30 mg/L. Indeed, using Diasys reagents, urinary samples’ absorbance above the highest calibrator increases to about 3700 for Cys C of 3–4 mg/L and then decreases to reach a minimum value of 2000 for urinary Cys C concentration from 15 up to 30 mg/L.
For comparison study, a total of 150 selected urinary samples were analysed covering the mean range from <0.02 to 36.3 mg/L. There was no significant difference between mean values obtained with DiaSys (1.632 ± 5.291 mg/L) or Siemens (1.764 ± 5.468 mg/L) reagents (P = 0.40). The following Passing-Bablok regression equations were obtained: DiaSys = 0.99 (95% confidence interval, –0.96 to 1.01) Siemens + 0.00 (95% CI, –0.00 to −0.00). The mean and limits of agreement of the difference between DiaSys and Siemens u-CysC were –0.098 (from –1.461 to 1.266) mg/L and were –0.004 (from –0.087 to 0.078) mg/L when focused on the analytical range between 0.02 and 1 mg/L (n = 123 patients). These results highlighted the beneficial effect of CysC standardization in urine as previously demonstrated by our group in serum. 3
To assess the clinical relevance of u-CysC determination, 50 healthy subjects and two groups of patients with renal tubular dysfunction were studied (Table 2). In healthy subjects, u-CysC results ranged from undetectable values to 0.234 and 0.246 mg/L with Siemens and DiaSys reagents, respectively. In this small group, the upper reference limits defined as <95 percentiles were 0.220 and 0.235 for Siemens and DiaSys reagents, respectively. Using the same methodology, the upper reference limit of u-CysC was determined at 0.28 by nephelometry 4 and 0.14 mg/L by turbidimetry. 5 In addition, we observed a significant difference in u-CysC results among the three groups of patients (P < 0.001) with both methods. Significant differences in pairwise comparisons were found between healthy controls and the two groups of patients with kidney injury (P < 0.001) as well as between the group of HIV patients with tubular dysfunction and the group of patients with AKI (P = 0.003 and P = 0.005 for u-CysC measured with Siemens and DiaSys reagents, respectively). These data confirmed that u-CysC could be an interesting biomarker to detect tubular dysfunction. Data from meta-analysis demonstrated that both serum and urinary CysC show a good diagnostic performance for predicting AKI in adults 15 and children. 16 Moreover, the combination of serum CysC as a functional biomarker and urinary NGAL as a damage marker yields a significantly superior discriminative performance for AKI detection. 17 In this context, performing both serum and urine CysC using one reagent on the same automated platform could offer to the physician the possibility to evaluate both renal function and damage.
Urinary Cystatin C values in healthy controls and in patients with HIV proteinuria and in patients with AKI.
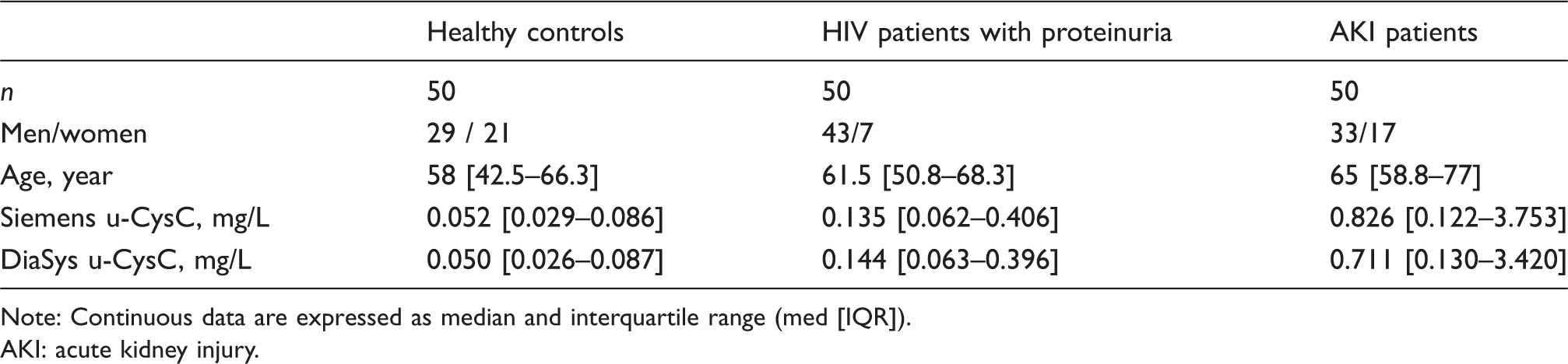
Note: Continuous data are expressed as median and interquartile range (med [IQR]).
AKI: acute kidney injury.
Conclusion
In conclusion, u-CysC determination using Siemens and DiaSys reagents has reliable analytical performances allowing diagnosis of tubular dysfunction in clinical practice. Our study shows that results were comparable between the Siemens PENIA and the DiaSys PETIA methods.
Footnotes
Acknowledgements
Reagents for cystatin C assay used in this study were kindly provided by DiaSys and Siemens.
Contributorship
ASB and JPC researched the literature and conceived the study. ASB, VA, KK, MP, NK, AMD and SB are involved in clinical and biological data collection and analysis. ASB and JPC have written the first draft of the article with major contribution of SB and AMD to the critical reading of the manuscript. All authors reviewed and edited the article and approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Samples were registered at the Ministère de l’Enseignement Supérieur et de la Recherche with the number DC-2008–417 and DC-2012–1579 after approval by our institution’s ethical committee (Lapeyronie University Hospital of Montpellier).
Guarantor
JPC.
