Abstract
Background
Patients with mutations of succinate dehydrogenase B (SDHB) and succinate dehydrogenase D (SDHD) are at high risk of paraganglioma necessitating surveillance. Chromogranin A has been proposed as a biochemical marker of paraganglioma. We sought to determine the diagnostic utility of chromogranin A in a population-based SDHx sample.
Methods
Tasmania is an island state with one tertiary referral centre for endocrine neoplasia. We performed a cross-sectional analysis of all adult SDHB (n = 52) and SDHD (n = 10) patients undergoing paraganglioma surveillance between 2011 and 2017. Chromogranin A was referenced against the outcome of paraganglioma surveillance with a minimum of 18F-fluorodeoxyglucose positron emission tomography/computed tomography (18F-FDG PET/CT) and plasma metanephrines (metanephrine and normetanephrine).
Results
Chromogranin A correctly predicted the result of paraganglioma surveillance more often in patients with SDHB compared with those with SDHD (77% vs. 22%, P = 0.003). In the SDHB group, chromogranin A demonstrated a sensitivity of 67% and specificity of 79% compared with 22% and 0% in the SDHD group. Chromogranin A identified one of three PET/CT-visualized SDHB-related paragangliomas with normal plasma metanephrines at the expense of nine false-positive results. A normal chromogranin A demonstrated a negative predictive value of 92% for SDHB-related paraganglioma. In patients with SDHB, plasma normetanephrine and metanephrine offered superior specificity (100%, P = 0.01 and 100%, P < 0.01, respectively) with comparable sensitivity (67%, P = 1.0 and 11%, P = 0.06, respectively) to chromogranin A.
Conclusion
Chromogranin A does not provide additive benefit to standard surveillance for predicting the presence of SDHB- or SDHD-related paraganglioma, but has a useful negative predictive value when normal in patients with SDHB mutation.
Keywords
Introduction
Paragangliomas (PGLs) are rare neuroendocrine tumours that arise from autonomic ganglia and may either be functional, producing catecholamines, or non-functional. Hereditary conditions predisposing to PGL are increasingly recognized and currently represent 25–30% of all PGL diagnoses. 1 While sporadic PGLs are rare, patients affected by hereditary PGL syndromes are at increased risk and may experience synchronous, metasynchronous or metastatic disease.1,2 Loss of functional mutations involving succinate dehydrogenase B (SDHB) and succinate dehydrogenase D (SDHD) results in an autosomal dominant increased risk of PGL.1–3
SDHB and SDHD have a highly penetrant phenotype, up to 50% and 80% lifetime risk, respectively.4–8 SDHB-related PGLs are predominately derived from sympathetic ganglia of the thoracoabdominal region and are characterized by an aggressive disease course with malignant PGL over-represented.9–11 Due to maternal imprinting, SDHD-related disease only manifests when the mutation is inherited paternally and is characterized by non-malignant multifocal disease predominately involving parasympathetic ganglia of the head and neck.3,4,8 Diagnosis of SDHx-related PGL can be delayed due to an increased frequency of biochemically silent and clinically asymptomatic phenotypes, potentially increasing the risk of malignant transformation. 12
The potential for early onset, aggressive and highly penetrant phenotypes necessitates lifelong surveillance as the standard of care for patients with SDHx mutation.13,14 Surveillance protocols for SDHx-related disease continue to be refined. 15 Current surveillance algorithms emphasize specialized imaging, such as magnetic resonance imaging/computed tomography, fluorodeoxyglucose (18F) positron emission tomography/computed tomography (18F-FDG PET/CT) or 68 Ga DOTATATE PET/computed tomography and biochemistry, including plasma metanephrines.15–21 Challenges to this approach include the need for specialized testing and technical expertise, sometimes mandating significant travel time for patients, cost and radiation exposure across the lifespan. The relevance of interval biochemistry is uncertain.
Chromogranin A (CgA) is a major soluble protein in the secretory dense-core granules of neuroendocrine cells and cosecreted into serum with other stored peptides. 22 Consequently, CgA is a widely available diagnostic biomarker for neuroendocrine tumours and is commonly used for the surveillance of patients with SDHx mutations.23,24 Recently, CgA was demonstrated to have comparable and complementary diagnostic performance to plasma metanephrines, with a sensitivity of 73.2% and specificity of 95.9%, in an SDHB and SDHD referral population with high prevalence of metastatic and multifocal disease under ideal diagnostic conditions. 22 However, the utility of CgA for PGL surveillance in unselected SDHx populations is controversial, with false-positive results being a particular challenge.23,25 False-positive CgA results may occur due to proton pump inhibitor (PPI) use, chronic kidney disease (CKD), heart failure or atrophic gastritis. 23 We examined the diagnostic value of CgA results in a population-based sample of adults with SDHB and SDHD.
Materials and methods
Tasmania is an island state in Australia with a single referral centre (the Royal Hobart Hospital) for patients with or at risk of endocrine neoplasia, including those with SDHB and SDHD mutations. As previously described, 16 all adult patients with SDHB and SDHD mutations undergoing PGL surveillance with 18F-FDG PET/CT and plasma metanephrines at the Royal Hobart Hospital between 1 July 2011 and 30 September 2017 who had contemporary (within six months) assessment of CgA concentration were considered eligible for inclusion. Surveillance for PGL in asymptomatic adult SDHB and SDHD carriers at the Royal Hobart Hospital consists of annual biochemistry (plasma metanephrines and CgA) and second yearly imaging (four yearly neck and renal ultrasounds alternating with four yearly 18F-FDG PET/CT). The research programme was approved by the Southern Tasmanian Health and Medical Human Research Ethics Committee (reference number H0014866).
Patients were considered positive for a PGL-related abnormality if either 18F-FDG PET/CT or plasma metanephrines were positive. PGLs were considered functional when plasma metanephrines (metanephrines or normetanephrine) were elevated. CgA concentration was referenced against the result of PGL surveillance to determine the diagnostic value of CgA. CgA concentration was considered abnormal if it was greater than the upper limit of normal. Patient data were assessed in a cross-sectional manner to determine the ability of CgA concentration to predict the result of PGL surveillance.
Measurement of plasma metanephrines
Plasma metanephrines considered in this study included metanephrine and normetanephrine, but not 3-methoxytyramine. Plasma metanephrines were analysed using liquid chromatography–tandem mass spectrometry at the Clinical Pharmacology and Therapeutics Laboratory, Austin Health. Briefly, following the addition of deuterated internal standard, solid phase extraction, dry down and reconstitution, samples were derivatized using cyanoborohydride and acetaldehyde. Chromatography was performed using Agilent 1200 Infinity high performance liquid chromatography (Agilent Technologies, Mulgrave, Australia) and a reversed phase column (Atlantis T3 150 mm× 2.1 mm; 3 μm packing, Waters Australia) using a 0.2 mL/min flow of mobile phase delivering a linear acetonitrile gradient (4–24% over 5 min with 3 min re-equilibration) in 0.2% formic acid. Tandem mass spectrometric detection was performed using an Agilent 6460 series instrument (Agilent Technologies, Australia). Electrospray ionization was used in positive ion mode at unit mass resolution and optimized detector settings for voltages, gas temperatures and flows.
CgA measurement
Patients were requested to fast prior to CgA assessment. All samples for CgA measurement were collected in a serum tube and placed in ice–water mixture to prevent breakdown of endogenous CgA. CgA was quantified using the DAKO (DAKO, Glostrup, Denmark) assay until 2014 (inter-assay coefficients of variation 7% at a CgA concentration of 53 U/L, derived from 40 estimations). Subsequent testing was done using the Cisbio (Cisbio Assay, Codolet, France) assay (inter-assay coefficients of variation were 5.8% and 6.7% at a CgA concentration of 20 μg/L and 200 μg/L, respectively, both derived from 30 estimations). Both assays used an ELISA format but different antibody specificity. The DAKO assay used two polyclonal antibodies with epitope specificity towards a 23 kD ‘C’ terminal fragment of CgA. The Cisbio assay used two monoclonal antibodies that had specificity directed to amino acid sequence 145–197 and 198–245. The reference range (<21.8 U/L) for the DAKO assay was derived from the analysis of samples from 40 healthy volunteers. The reference range (27–94 μg/L) for the Cisbio assay was derived from the analysis of samples from 60 healthy volunteers. Statistical correlation between the assays was high (R2=0.99). Consistency of clinical interpretation of results between the two methods was also high (94%) with only two samples, both close to the assay cut-offs, yielding different results between the assays. As the reference range for these assays differed, CgA concentration was expressed as multiples of the upper limit of normal (xULN, 21.8 U/L for the DAKO assay and 94 μg/L for the Cisbio assay).
Medical records and biochemistry were reviewed to determine PPI use, the presence of CKD (defined as an estimated glomerular filtration rate ≤ 60 mL/min/1.73 m2), 26 atrophic gastritis or heart failure. Patients were not excluded from the analysis if these conditions were present, but their presence was noted and impact assessed.
Data were collated and statistical analysis was performed using GRAPHPAD PRISM Version 7.03 (GRAPHPAD Software Inc., La Jolla, CA, USA) and SigmaPlot Version 13 (Systat Software, San Jose, CA, USA). CgA was analysed as multiples of the upper limited of normal (xULN). Student’s t-tests and Fisher exact tests were used to compare the differences in means and proportions, respectively. Where data were not normally distributed, log transformations were performed. Where data failed to meet the equal variance test or Shapiro-Wilk normality test post log transformation, Mann-Whitney Rank Sum t-tests were used. McNemar’s t-test was used to compare the diagnostic performance of CgA and plasma metanephrines. Statistical significance was defined as a two-tailed P value ≤0.05.
Results
There was no significant difference between patients with SDHB and those with SDHD with regard to age, sex or mean CgA concentration (Table 1). Patients with SDHB were significantly (P < 0.001) less likely to have a PGL manifest at baseline compared with those with SDHD (17% vs. 90%). When PGL was present, CgA was significantly higher (P = 0.02) in patients with SDHB compared with those with SDHD (3.00 ± 3.81 vs. 0.80 ± 0.55). Patients with SDHB were more likely to have functional PGLs (P = 0.03) and for these PGLs to affect the thoracoabdominal region (P = 0.002). There was no significant difference between SDHB and SDHD patients with regard to the prevalence of malignant PGLs (33% vs. 22%). Three patients with SDHB and two patients with SDHD had malignant PGLs with metastatic disease above and below the diaphragm in all cases.
Characteristics of patients.
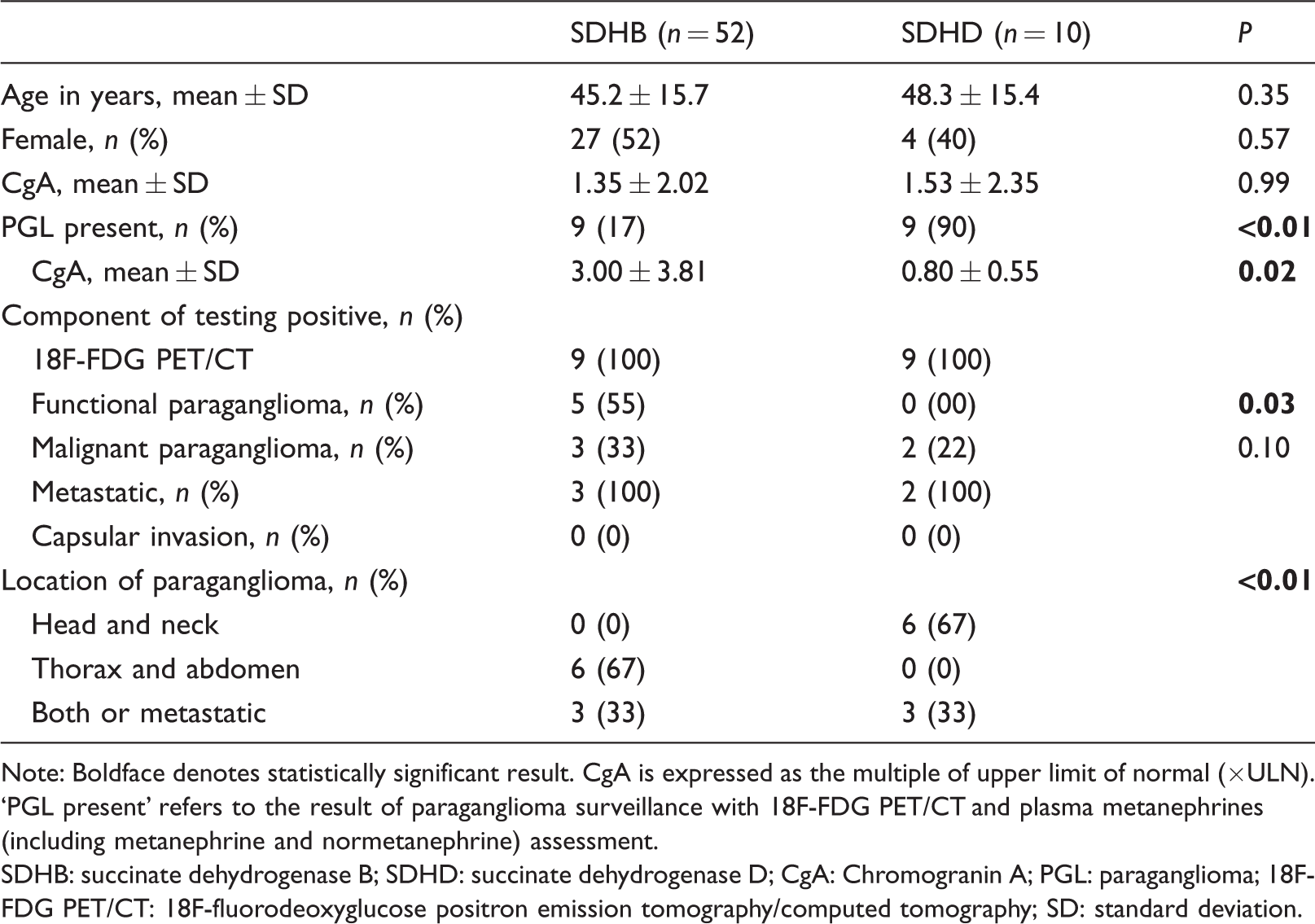
Note: Boldface denotes statistically significant result. CgA is expressed as the multiple of upper limit of normal (×ULN). ‘PGL present’ refers to the result of paraganglioma surveillance with 18F-FDG PET/CT and plasma metanephrines (including metanephrine and normetanephrine) assessment.
SDHB: succinate dehydrogenase B; SDHD: succinate dehydrogenase D; CgA: Chromogranin A; PGL: paraganglioma; 18F-FDG PET/CT: 18F-fluorodeoxyglucose positron emission tomography/computed tomography; SD: standard deviation.
Table 2 summarizes the diagnostic performance of CgA concentration referenced against the outcome of PGL surveillance. CgA was significantly more likely to yield a concordant result for patients with SDHB compared with those with SDHD (77% vs. 22%, P = 0.003). This was primarily due to normal CgA better predicting the absence of PGL in patients with SDHB vs. SDHD (92% vs. 0%, P < 0.001), rather than a positive result predicting the presence of a PGL (40% vs. 67%, P = 0.56). In patients with SDHB, the sensitivity and specificity of CgA for abnormal surveillance results were 67% and 79%, respectively, compared with 22% and 0% in patients with SDHD. The negative predictive value of a normal CgA concentration was 92% in patients with SDHB. Excluding all SDHB patients on PPIs (n = 6), with significant CKD (n = 1), heart failure (n = 0) or atrophic gastritis (n = 0) from analysis regardless of the outcome of PGL surveillance yielded a sensitivity of 75% (35–97%) and specificity of 84% (69–94%) in patients with SDHB. If plasma metanephrines were included in analysis and biochemistry defined as positive if either CgA or plasma metanephrines were elevated, then sensitivity increased to 78% (40–97%) and specificity to 80% (65–91%). In the SDHB cohort, plasma normetanephrine had similar sensitivity of 67% (30%–93%, P = 1.0) but superior specificity of 100% (91%–100%, P = 0.01) compared with CgA for SDHB-related PGL (Figure 1(b)). Plasma metanephrine also had superior specificity of 100% (92–100%, P < 0.01) with comparable sensitivity of 11% (0–48%, P = 0.06) to CgA for SDHB-related PGL (Figure 1(c)).
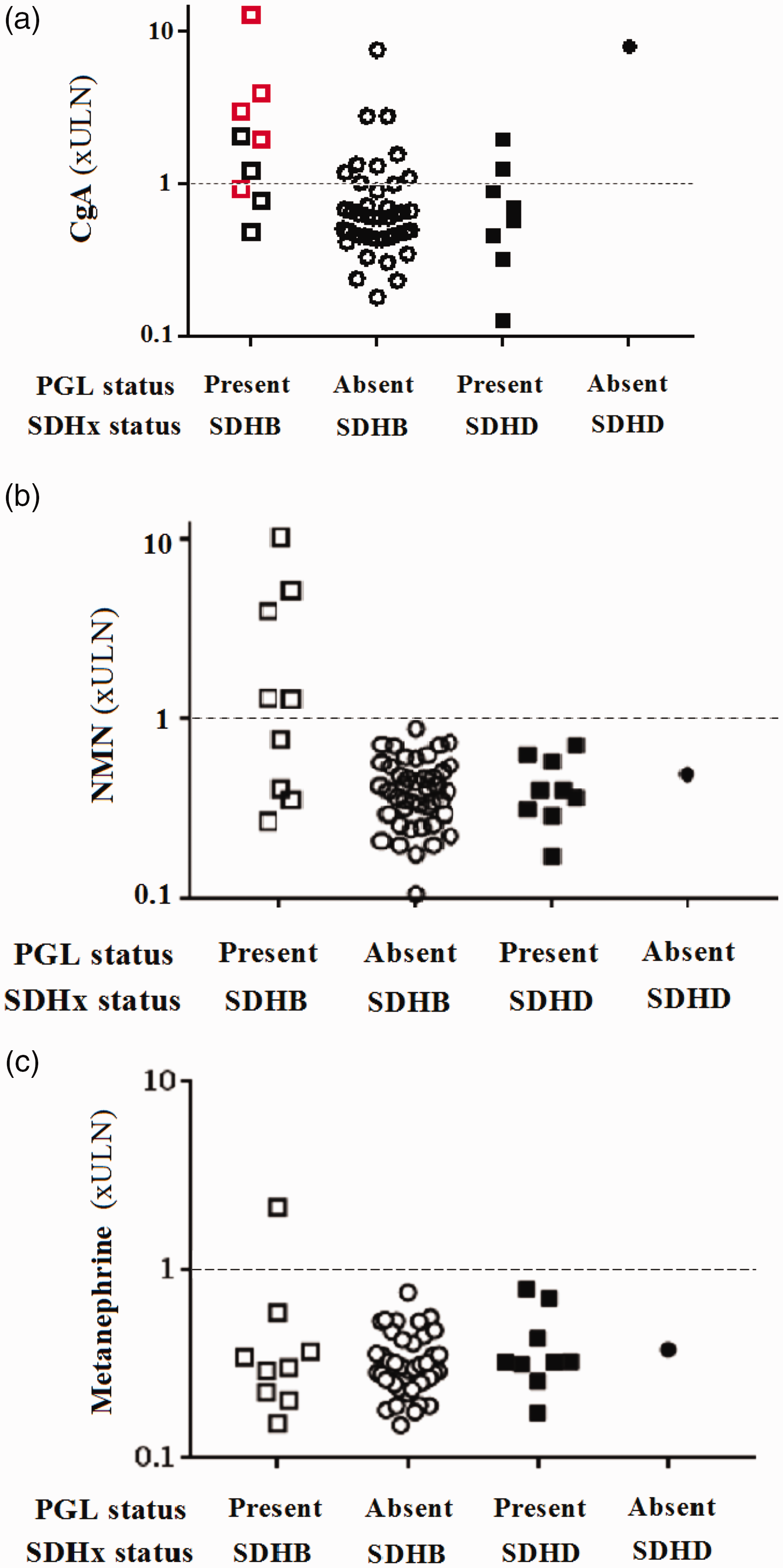
Spectrum of chromogranin A (CgA, a), normetanephrine (NMN, b) and metanephrine (c) results on a per patient basis stratified by SDHx and PGL status. Red symbols in (a) denote functional PGLs (defined by elevated normetanephrine or metanephrine concentration). The dashed line represents the upper limit of normal. Patients with SDHB and a PGL were more likely to have an elevated CgA. CgA was more likely to be elevated in patients with a functional PGL compared with those with a non-functional PGL (a). Plasma normetanephrine and metanephrine had similar sensitivity but superior specificity compared with CgA for SDHB-related PGL (b and c).
Diagnostic performance of CgA for predicting outcome of PGL surveillance.
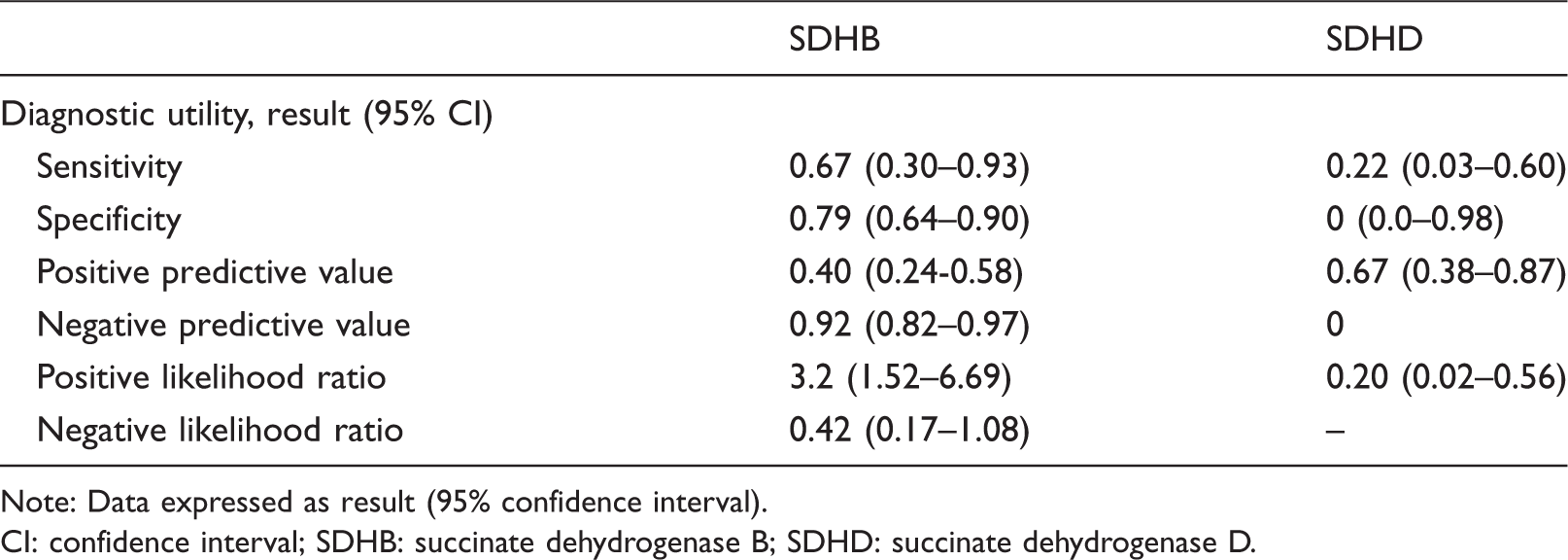
Note: Data expressed as result (95% confidence interval).
CI: confidence interval; SDHB: succinate dehydrogenase B; SDHD: succinate dehydrogenase D.
Patients with SDHB and a PGL were more likely to have an elevated CgA than SDHB patients without PGL (P = 0.01, Figure 1(a)). CgA was also significantly higher (3.00 ± 3.81 × ULN vs. 0.89 ± 1.17 × ULN, P = 0.001) in patients with SDHB and PGL compared with SDHB patients without PGL. In patients with SDHD, CgA was not significantly different (P = 0.33) between patients with and without PGL. CgA was more likely to be elevated in patients with a functional PGL compared with those with a non-functional PGL (86% vs. 18%, P = 0.045). One patient with SDHB-related PGL had elevated CgA without concurrent elevation of plasma metanephrines; however, this PGL was also confirmed on concurrent 18F-FDG PET/CT imaging. One patient had elevated plasma metanephrines without elevation of CgA and two patients with normal CgA and plasma metanephrines had imaging-detected PGLs.
Discussion
In this population-based study, CgA had limited additive diagnostic value for predicting the presence of SDHx-related PGL, but offered potentially useful negative predictive value for patients with SDHB mutation. CgA demonstrated better diagnostic performance in SDHB-related compared with SDHD-related PGL surveillance. Corroborating existing literature, 22 we found that CgA was (1) better able to predict the outcome of PGL surveillance in patients with SDHB compared with those with SDHD and (2) more likely to be elevated and significantly higher when PGL was present in patients with SDHB, but not in patients with SDHD. A normal CgA better predicted the absence of SDHB-related PGL with infrequent false-negative results and a potentially useful negative predictive value. However, in our population-based SDHB cohort, CgA did not identify any PGLs that would not have been detected by routine surveillance, limiting its positive predictive utility in both SDHB- and SDHD-related PGL surveillance.
PGL related to SDHB and SDHD differs phenotypically. SDHD-related PGLs preferentially arise from parasympathetic ganglia in the head and neck region, whereas SDHB-related PGLs more frequently arise from sympathetic ganglia and involve the thoracoabdominal region with a higher incidence of malignant disease.4,7,8 Our data reflect this anatomical divergence and support the growing body of literature suggesting that SDHB- and SDHD-related PGLs are biochemically divergent with CgA lacking diagnostic efficacy in patients with head and neck PGLs (exclusively SDHD patients in our cohort).24,27 CgA was higher in patients with SDHB-related PGL compared with those with SDHD-related PGL. CgA was higher in patients with functional PGLs, possibly reflecting the necessary role of CgA in dense-core secretory granule biogenesis 28 or cosecretion with catecholamines. 29 The biochemical divergence between SDHB- and SDHD-related PGL potentially reflects the parasympathetic origin of head and neck predominant SDHD-related PGLs or may be attributable to other genotype-specific differences.27,30 We were unable to differentiate these possibilities due to the pronounced anatomical segregation by genotype in our cohort.
Compared with a recent, highly controlled study in an SDHB referral population, 27 the sensitivity, specificity and ability of CgA to complement plasma metanephrine testing in our SDHB cohort were lower. This partially reflects the impact of PPI use and CKD in our cohort as exclusion of these participants increased sensitivity and specificity to the extent that the sensitivity of CgA was comparable, although specificity remained less than previously observed. 27 The residual difference in specificity may be due to the lower prevalence of metastatic disease in our cohort. The complementary nature of CgA and plasma metanephrines for PGL diagnosis was also less apparent in our cohort. CgA was elevated in only one of three (33%) SDHB-related PGLs with normal plasma metanephrines, was less specific than plasma metanephrines alone and the sensitivity of CgA and plasma metanephrines together was only slightly higher than CgA alone. Thus, in our cohort, CgA was able to detect one additional PGL, which was concurrently visualized on 18F-FDG PET/CT, at the expense of nine false-positive results.
False-positive results are a major limitation of CgA testing 23 but do not detract from the negative predictive value of a normal CgA result. In our SDHB cohort, the negative predictive value of a normal CgA was 92%. Therefore, while a raised CgA is unlikely to meaningfully add to routine PGL surveillance, a normal CgA in a patient with SDHB mutation offers potentially informative negative predictive value as an ancillary test. These data should aid decision-making in daily clinical practice but are not sufficient to recommend CgA displace existing biochemical or imaging approaches to PGL surveillance.
3-Methoxytyramine is the O-methylated metabolite of dopamine and has emerged as a valuable biomarker of prevalent and metastatic SDHx-related PGL.31–33 Assessment of 3-methoxytyramine is particular useful in head and neck PGLs, 34 which are less likely than abdominothoracic PGLs to be detected using metanephrine or normetanephrine. 35 Given our data suggesting that CgA offers limited positive predictive value to diagnostic algorithms including 18F-FDG PET/CT and plasma metanephrines, it is conceivable that the inclusion of 3-methoxytyramine in these algorithms will further supplant the role of CgA as a second line test. 21
This study has potential limitations. Patients on PPIs and with mild renal dysfunction were included. This potentially underestimated the diagnostic performance of CgA due to false-positive results, which we accounted by further analysis excluding relevant participants. The major benefit of this inclusive study design, combined with a population-based sample from the only referral centre in an island state, is strong external validity and relevance to clinical practice. Our sample size was moderate, potentially limiting statistical analysis. However, in the context of an exceedingly rare disease, our sample size compares favourably and allowed comparisons between patients with SDHB and SDHD. Finally, two different assays were used to quantify CgA during the study period, potentially introducing additional variability. However, both the statistical correlation and clinical consistency of assay results were high, suggesting that the overall impact of this change was limited.
In conclusion, CgA does not meaningfully add to standard surveillance for predicting the presence of SDHB- or SDHD-related PGL; however, it provides a potentially informative negative predictive value in patients with SDHB mutation.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The research programme was approved by the Southern Tasmanian Health and Medical Human Research Ethics Committee (reference number H0014866).
Guarantor
MJWT.
Contributorship
MJWT: study design, data collection, analysis, article drafting and revisions.VP: data collection, article drafting and revisions.JRB: study conception and design, data collection and analysis, article drafting and revisions.
