Abstract
Background
Abundant evidence indicate the increased levels of oxidative stress in patients with autism. Advanced glycation end products and advanced lipoxidation end products and their precursors play a major role in increased oxidative stress in numerous metabolic and neurologic diseases. Carnosine is a natural dipeptide with antiglycation effects. The aim of this trial was to examine the effects of carnosine supplementation on the advanced glycation end products and the precursors of advanced lipoxidation end products in patients with autism.
Method
This randomized double-blind, placebo-controlled clinical trial was conducted on 36 autistic children, 18 in the carnosine group and 18 in the placebo group. The groups received a daily supplement of 500 mg carnosine or placebo for two months, respectively. Plasma concentrations of glycation and precursors of lipoxidation markers were evaluated by enzyme-linked immunosorbent assay method.
Results
In all, 63.9% of the autistic children had normal nutritional status. Carnosine supplementation did not significantly alter plasma concentrations of advanced glycation end products and precursors of advanced lipoxidation end products in autistic children.
Conclusion
The findings indicate that supplementation of carnosine could not change advanced glycation end products and precursor of advanced lipoxidation end products in autistic children.
Keywords
Introduction
Autism spectrum disorder (ASD) is a disorder with impaired verbal and non-verbal communication and interactions, repetitive patterns of behaviours and limited interests. 1 The prevalence rate of ASD is four times more in boys than girls. 2 The Centers for Disease Control and Prevention (2014) has reported that one of 68 children in the United States is suffering from ASD. 3 The findings of a systematic review of epidemiological surveys showed that worldwide prevalence of autistic disorders is 1 in 160 children. 4 Apart from their main challenges, autistic children might have other problems, including sleep disturbances, self-injury, metabolic defects, 5 attention-deficit hyperactivity disorder, anxiety, epilepsy, depression 6 and obesity. There is no specific treatment for ASD, and the actual treatment aims to relieve the symptoms.7–9
According to the literature, aside from genetic issues, other factors such as prenatal problems, 10 air pollution, 11 neurotoxic compounds 12 and toxicants, 6 nutritional risk factors 13 and oxidative stress14,15 could contribute to the pathogenesis of ASD. In a study on autistic patients, Tang et al. found increased levels of oxidative stress in the brains of autistic children compared with healthy children. 16 Another study on 90 autistic and healthy people in 2011 found that as the severity of autism increased, the levels of urinary oxidative stress, such as 4-hydroxynonenal and thiobarbituric acid increased and levels of antioxidants decreased. 17 Advanced glycation end products (AGEs) and advanced lipoxidation end products (ALEs) are compounds that can be produced under oxidative stress conditions or can themselves aggravate oxidative stress condition.18,19
AGEs and ALEs are compounds that are formed by different non-enzymatic mechanisms through reaction with heterogeneous precursors like DNA and proteins. 20 The pathological roles of AGEs and ALEs are specified in different oxidative-based diseases, including neurological disorders. 21 Several evidence indicated the increased levels of AGEs and ALEs in autistic patients.15,22,23 Furthermore, it has been established that agents with antioxidant properties such as zinc, 24 curcumin 25 and carnosine26,27 are potentially able to prevent AGE and ALE generation in the body.
Carnosine is a natural dipeptide found in many animal and human tissues, including muscles and the brain. 28 Various properties have been identified for carnosine, including antioxidant, antitoxic, metal ion chelating and intracellular buffering.29,30 Recently, the ability of carnosine to prevent the production of oxidative stress markers, in particular non-enzymatic forms (AGEs and ALEs and their precursors), has been recognized. Pepper et al. demonstrated that carnosine can overcome carboxymethyl lysine and other AGEs in a prokaryotic model. 31 In another study on dogs receiving carnosine, carnosine reduced levels of AGEs and ALEs such as pentosidine in synovial fluid. 32
In autistic children, Chez et al. showed that supplementation of 800 mg carnosine for two weeks resulted in improved social behaviour, better speech and neuroprotective effects. 33 Also, in our previous study, supplementation of carnosine significantly decreased parasomnias, sleep duration and total sleep disorders in autistic patients compared with the placebo group. 34
Emerging reports indicate a link between mitochondrial dysfunction and ASD.35,36 Abnormalities in mitochondrial function lead to developmental regression, learning disability and several behavioural disorders.35,37 The findings of a meta-analysis study showed that the prevalence of mitochondrial dysfunction in the ASD population is much higher than that in the general population. 35 Mitochondrial dysfunction can contribute to the pathophysiology of neurological disorders through overproduction of reactive oxygen species (ROS). Impaired homeostasis of endogenous metals such as copper, iron and zinc is another factor reported to contribute to the initiation and progression of autism. 36 Carnosine may possibly be useful in the treatment of autism due to its prevention of mitochondrial abnormalities,38,39 its free-radical scavenger and metal chelating properties,40,41 or increasing the activity of antioxidant enzymes such as glutathione peroxidase and superoxide dismutase,42,43 thereby subsequently attenuating the AGE and ALE concentrations.
Due to the increase in oxidative stress, AGEs and ALEs in autistic patients and given that carnosine has antioxidant and antiglycation properties, we aimed to investigate the beneficial effects of carnosine on reducing AGEs and precursors of ALEs in autistic children.
Materials and method
Study design
This study is a double-blind, placebo-controlled clinical trial to evaluate the effect of carnosine supplementation on plasma concentrations of glycation and precursors of lipoxidation markers in children with autism, from June 2016 to December 2016. The study was approved by the Ethics Committee of Tabriz University of Medical Sciences, and each patient’s parental consent was obtained.
Participants
According to the American Psychological Association, autism is defined by the presence of difficulties in social interaction, impaired communication, restricted interests and repetitive behaviours. Autism was diagnosed by a child psychiatrist according to the Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM V). Individuals who met the three items of sociability criteria and at least two items of restrictive interest and/or repetitive behaviour criteria are identified as having ASD. 44 Autistic children aged 4–14 years with invariant medication at least from two months ago were included in the study. Participants with particular diets or with chelators or other supplements, with a neurological (cerebral palsy, epilepsy, etc.) or metabolic (phenylketonuria, maple syrup urine disease, etc.) disease and those who received less than 75% of carnosine supplements were excluded from the study.
Using a standard equation (Pocock) and based on mean ± SD of pentosidine from a previously published study, 26 with the 5% significance level test (α = 0.05) and power of 80% (β = 0.2), yielded a minimum sample size of 18 individuals to each group, which was increased to 22 people in anticipation of 25% expected dropouts. According to the inclusion and exclusion criteria, 44 autistic children were recruited to the trial using convenience sampling and then randomly assigned into carnosine (n = 22) or placebo groups (n = 22) by blocking (blocks with a minimum volume of 4 to prevent blindness unfolding) using RAS software. Two groups were matched for age and sex.
Intervention procedures
The children in the carnosine group received a daily supplement of 500 mg carnosine capsule (Nova Nutritions, Scotch Plains, New Jersey) and children in the placebo group received 500 mg of corn starch capsule with a lunch for two months.
Data collection and blood sampling
Height, weight and body mass index (BMI) were measured at the beginning of the study. The short form of the international physical activity questionnaire (IPAQ) was used to evaluate the physical activity of the participants. Food intake was assessed using a three-day 24-h recall survey (two working days and one weekend day) for three times at the beginning, middle and end of the study. The adjusted nutritionist IV (N4.1) software for Iranian foods was used to analyse the data from dietary intake.
Determination of plasma levels of AGEs and ALEs
At the beginning and end of the study, 5 mL of venous blood was drawn from each child. Plasma concentrations of methylglyoxal, carboxymethyl lysine, pentosidine, malondialdehyde and 4-hydroxynonenal were determined using the BT Bioassay Technology Laboratory (Shanghai Crystal Day BiotechCo, Ltd, Shanghai, China) enzyme-linked immunosorbent assay kits. Briefly, 50 μL of standards and 40 μL of the samples were added to the appropriate wells of a precoated plate together with the secondary antibody labelled with biotin. After an hour of incubation at 37°C, the wells were washed five times with wash buffer, chromogen solution was added and the mixture was incubated for 10 min at 37°C. Colour development was then stopped, and the absorbance of the wells was measured at 450 nm.
Statistical analysis
All analyses were performed using SPSS version 22 (SPSS, Chicago, IL, USA). Qualitative data were presented as frequency (per cent), and normal quantitative data were reported as mean ± SD and abnormal quantitative data as median (minimum and maximum). Mann-Whitney test was used to detect the differences between the two independent groups. Wilcoxon test was performed to determine differences within each group (before and after the intervention), and analysis of covariance was used to adjust baseline values, dietary intakes and confounders after taking logarithms of the variables. P-value less than 0.05 was found to be significant.
Results
Four individuals from carnosine group were excluded from the study due to resistance to blood sampling, thyroid hormone disorders and no desire to continue receiving supplements. Also, in the placebo group, four participants were excluded due to lack of referral for the second stage of blood sampling and reluctance to continue the study. Finally, 18 in the carnosine group and 18 in the placebo group completed the study (Figure 1).
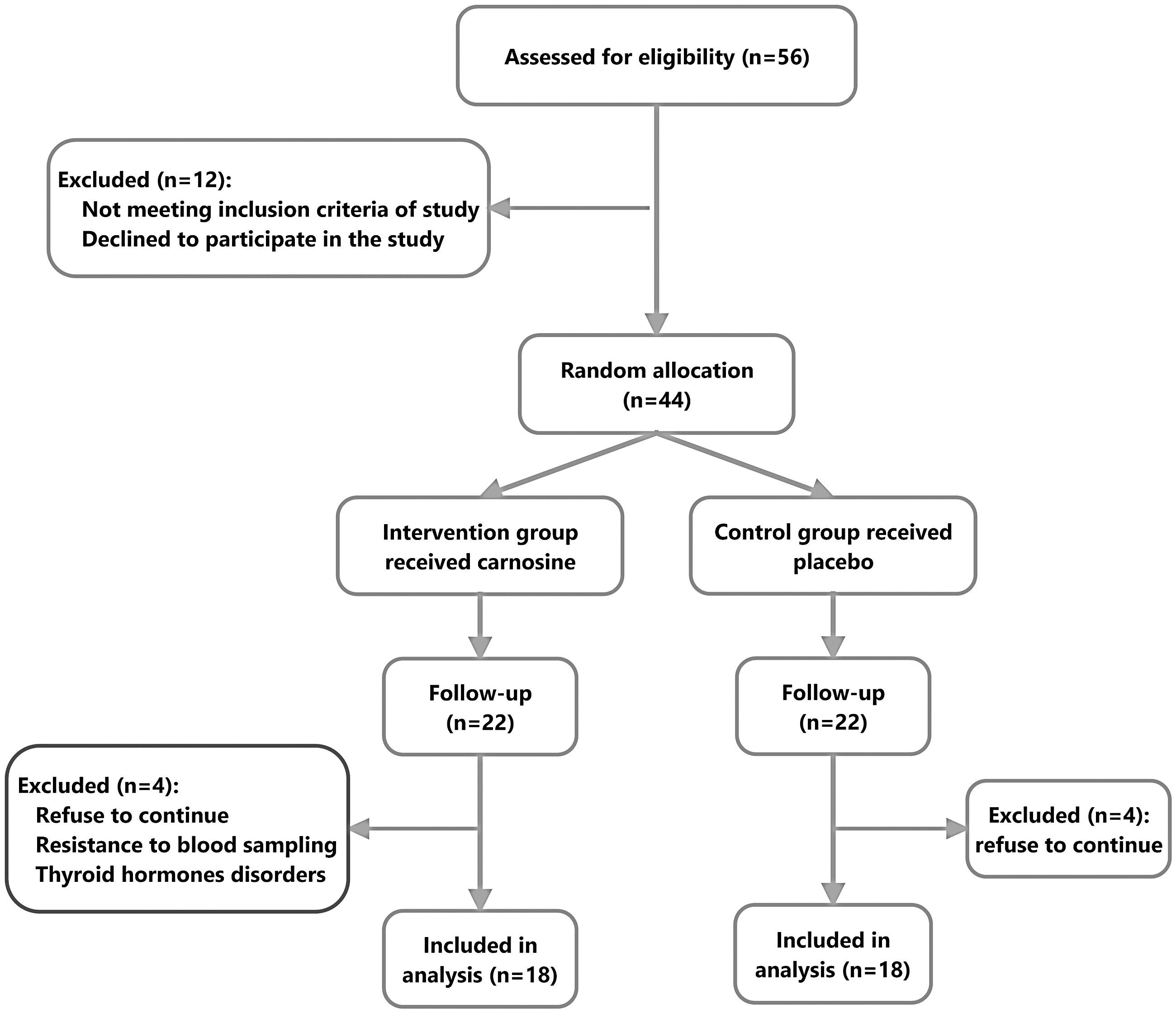
Flowchart of sample selection.
The demographic data of the participants are presented in Table 1. Thirteen children of the carnosine group and 14 children of the placebo group were male, and the rest were female. In terms of nutritional status, 63.9% of the children were at normal levels. Almost half of the children (44.4%) had low physical activity. There was no significant difference in age, gender, physical activity, height, weight or BMI of children between the two groups (P > 0.05). Also, there were no significant differences in dietary intakes between the two groups at the beginning and end of the study (P > 0.05).
Demographic characteristics of the studied groups at baseline (n = 36).
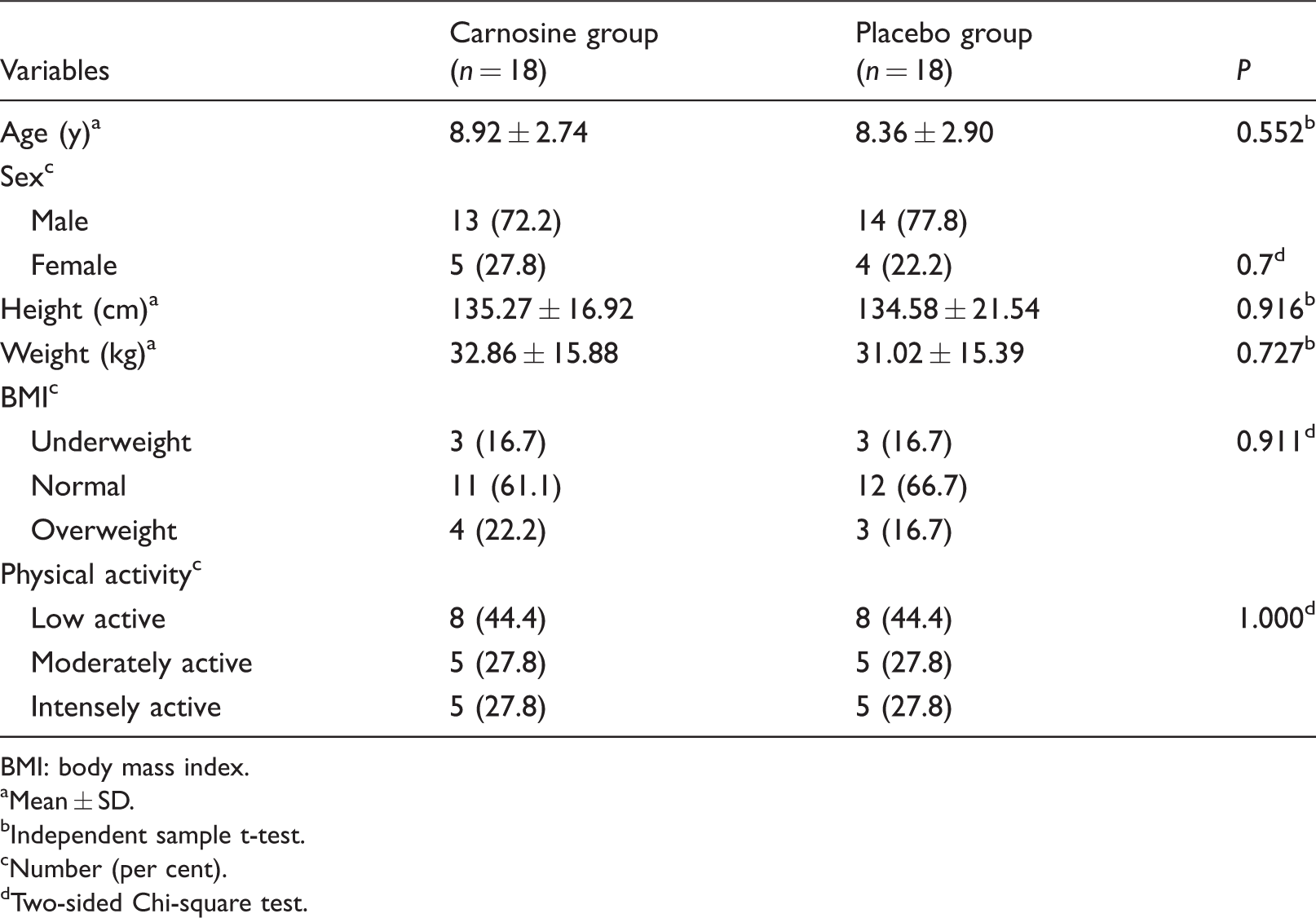
BMI: body mass index.
Mean ± SD.
Independent sample t-test.
Number (per cent).
Two-sided Chi-square test.
Both at the beginning and end of the study, plasma concentrations of AGEs (methylglyoxal (MG), carboxymethyl lysine (CML) and pentosidine) and precursor of ALEs (malondialdehyde (MDA) and 4, hydroxynonenal (4-HNE)) did not differ between the two groups (P > 0.05), and carnosine supplementation could not change plasma concentrations of AGEs and the precursor of ALEs. At the end of the study, plasma concentrations of AGEs and precursor of ALEs did not significantly differ from the baseline values in the either carnosine or placebo groups (P > 0.05) (Table 2), except pentosidine, which significantly declined in placebo group (P = 0.022).
Effect of carnosine supplementation on AGEs and precursor of ALEs.
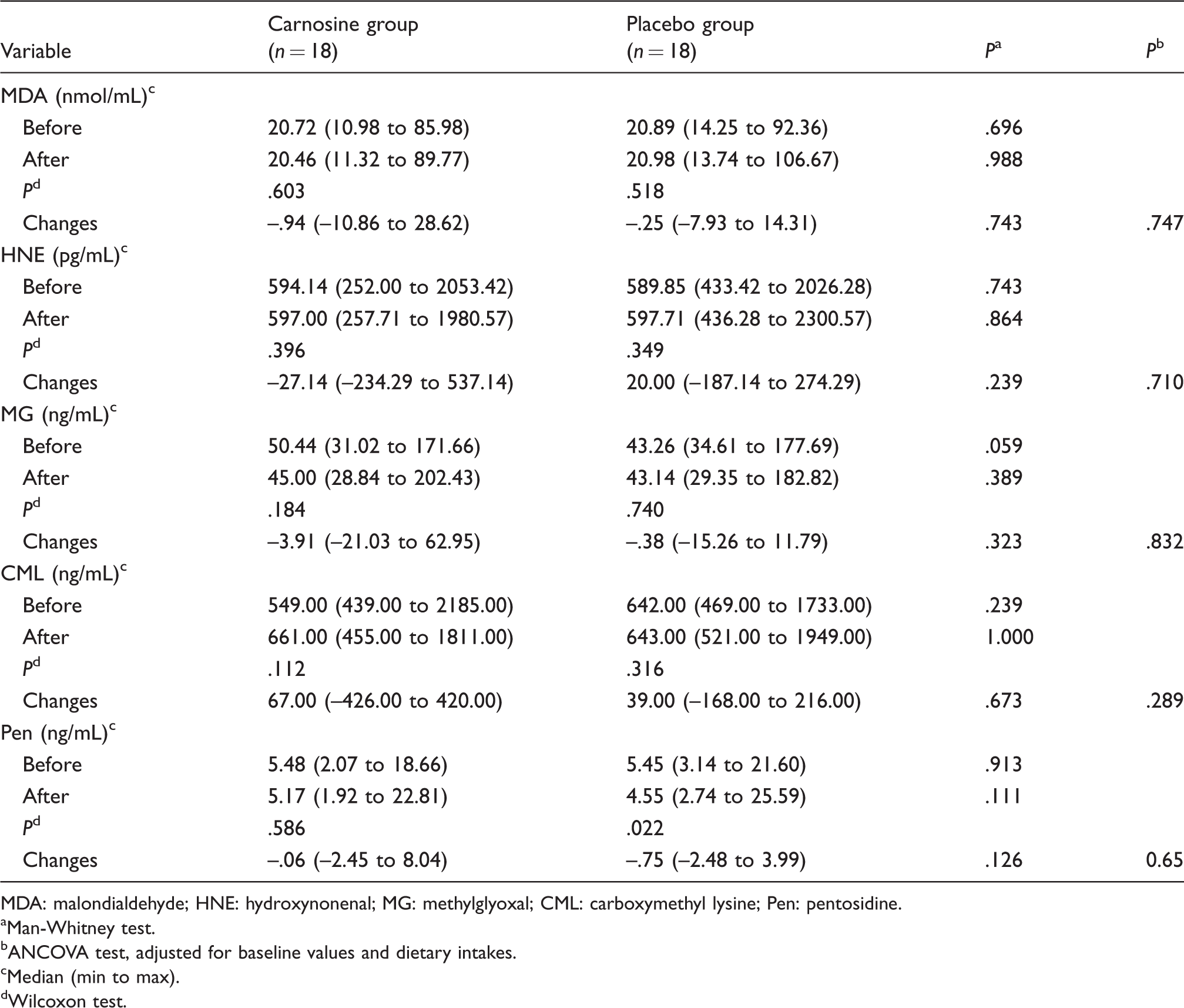
MDA: malondialdehyde; HNE: hydroxynonenal; MG: methylglyoxal; CML: carboxymethyl lysine; Pen: pentosidine.
Man-Whitney test.
ANCOVA test, adjusted for baseline values and dietary intakes.
Median (min to max).
Wilcoxon test.
As Table 2 shows, percentage changes of AGEs (MG, CML and pentosidine) and precursors of ALEs (MDA and 4-HNE) also did not differ significantly between the two groups (P > 0.05), both before and after adjusting for age, gender and dietary variables (P > 0.05).
Discussion
As noted, the increase in ALEs and AGEs can have irreparable effects on human health. In this study, supplementation of carnosine once daily at 500 mg showed no effect on reducing the concentration of AGEs and the precursor of ALEs. Human studies on this topic are rare. A study by Alhamdani et al. assessed the effect of carnosine peptide on the rate of AGE formation in peritoneal fluid and observed reduced formation of AGEs by carnosine peptide. 45 In this study, carnosine was studied on a small number of samples (eight) and in injection form to the peritoneal fluid of peritoneal dialysis patients. 45 Also, in our previous investigation, carnosine supplementation with a dose of 1000 mg per day for three months attenuated AGEs serum concentration in patients with type 2 diabetes. 26 Therefore, differences in the age, dose and duration of supplementation, variety in sites of AGEs studied (serum, plasma and peritoneal fluid), the nature of disease and the number of participants in the studies may explain the contradictory findings in human studies.
The findings of animal model studies in this area are also inconsistent. In agreement with our findings, the results of several studies showed that carnosine supplementation did not inhibit AGEs formation in animals. Pfister et al. found that oral carnosine treatment did not prevent AGEs formation after oral supplementation of carnosine on retinopathic rats. 46 Riedl et al. have also shown that oral supplementation of carnosine for three months did not significantly change MGO and CML levels in diabetic rats. 47 In their study on rats with myocardial infarction, Evran et al. found that supplementation of carnosine led to an increase in superoxide dismutase and glutathione peroxidase, but it was not able to reduce malondialdehyde. 48 In contrast, several other studies have reported the decremental effect of carnosine on AGEs and precursors of ALEs. Shi et al. showed that oral doses of carnosine decreased concentrations of pentosidine, 4-hydroxynonenal–protein adducts in dogs with osteoarthritis. 32 Stegen et al. reported that supplementation of carnosine for eight weeks reduced CML and total AGEs concentrations in plasma and urine of rats. 49 Xie et al. demonstrated that intraperitoneal administration of carnosine decreased MDA and oxidative stress in rat models. 50
In in vitro studies, Pepper et al. showed that adding different amounts of carnosine to the mixture of AGEs and Escherichia coli bacterium leads to more survival of bacteria and that carnosine could prevent glycation. They concluded that since the chemical procedures of glycation in eukaryotes and prokaryotes are similar, carnosine may show the same effect in eukaryotes. 31 Also, Hipkiss et al. found that carnosine can prevent the formation of AGEs, ROS and malondialdehyde in cultured cells. 51
But what we overlook is that the entire pharmacokinetic stages of chemical agents in humans can be different from other organisms and may be due to the different absorption rates in humans and various other factors. An enzyme called carnosinase 52 in the human body can also convert the amount of additional carnosine into its constructive amino acids, which may be considered one of the most important other causes in the study.
It is well known that oxidative stress is a key player in the pathophysiology of numerous neurological disorders. However, notably, a variety of factors including diabetes mellitus, 53 high blood pressure and atherosclerotic disease54,55 and air pollution 56 can synergistically contribute to the pathophysiology of neurological disorders by increasing the oxidative damage and stress.57–59 Similarly, the inflammatory response that follows acute stroke can lead to a dramatic increase in the production of ROS that ultimately increases the secondary induced brain damage and impairs functional recovery.60,61 Therefore, despite the observation that carnosine supplementation had no remarkable effect on AGEs and ALEs, it may have beneficial applications in the field of other oxidative stress-based neurological diseases, including cognitive impairment and stroke.
A relatively small sample size remains a notable limitation of this study. Furthermore, we were unable to cut off medications to avoid any effect of various drugs on oxidative factors due to ethical considerations. However, the medications of the study participants were kept constant throughout the study. Furthermore, measurement of enzymes such as glyoxalase I and carnosinase, as well as superoxide dismutase and glutathione peroxidase, could be more effective in confirming the results of the study.
Conclusion
The findings of the study indicate that carnosine supplementation could not reduce the concentration of AGEs and precursors of ALEs in autistic children. It is suggested that future studies be performed with larger samples and control of other confounders so the results can be more decisive.
Supplemental Material
Supplemental material for Carnosine supplementation does not affect serum concentrations of advanced glycation and precursors of lipoxidation end products in autism: a randomized controlled clinical trial
Supplemental material for Carnosine supplementation does not affect serum concentrations of advanced glycation and precursors of lipoxidation end products in autism: a randomized controlled clinical trial by Ramin Ghodsi, Sorayya Kheirouri and Rahmat Nosrati in Annals of Clinical Biochemistry
Footnotes
Acknowledgements
The results of this study are provided from the master's thesis of Ramin Ghodsi, approved at the Tabriz University of Medical Sciences, Tabriz, Iran. The authors would like to thank all the children and their families for their participation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This trial is supported by the faculty of Nutrition and Food Sciences of Tabriz University of Medical Sciences, Iran.
Ethical approval
The Research Ethics Committee of the Tabriz University of Medical Science approved the study (Ethical approval: IR.TBZMED.REC.1396.617).
Guarantor
SK.
Contributorship
SK and RG were involved in conceptualizing the research proposal and data acquisition. SK, RG and RN participated in data analysis and interpretation and manuscript drafting. All authors read and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
