Abstract
Dear Editor,
We have occasionally observed significantly elevated serum total testosterone concentrations in female patient samples using the Abbott Architect 2nd generation total testosterone assay which have not been confirmed by mass spectrometry, suggesting the possibility of interference.
17α-Ethynyl-19-nortestosterone, commonly known as norethisterone (also as 19-norethisterone, norethindrone and 19-norethindrone), and a structurally similar compound, norethisterone acetate, are steroidal compounds of the progestogen drug class found in the combined oral contraceptive pill and other forms of contraceptive, e.g. progestogen-only pill. These compounds share the same steroid backbone as testosterone and have the potential to cross-react in testosterone assays.
Interference from norethisterone has been reported previously in other immunoassays, 1 but not to our knowledge for the Abbott Architect assay. In order to investigate this possible interference, we undertook a spiking experiment using norethisterone.
19-Norethindrone (⩾98% purity) was purchased from Sigma-Aldrich (now Merk) in powder form. Powder (5 mg) was dissolved in 16.75 mL of high performance liquid chromatography (HPLC) grade methanol to give a solution with a concentration of 1mmol/L. This solution was diluted 1000-fold with HPLC-grade methanol to produce 1 μmol/L stock solution.
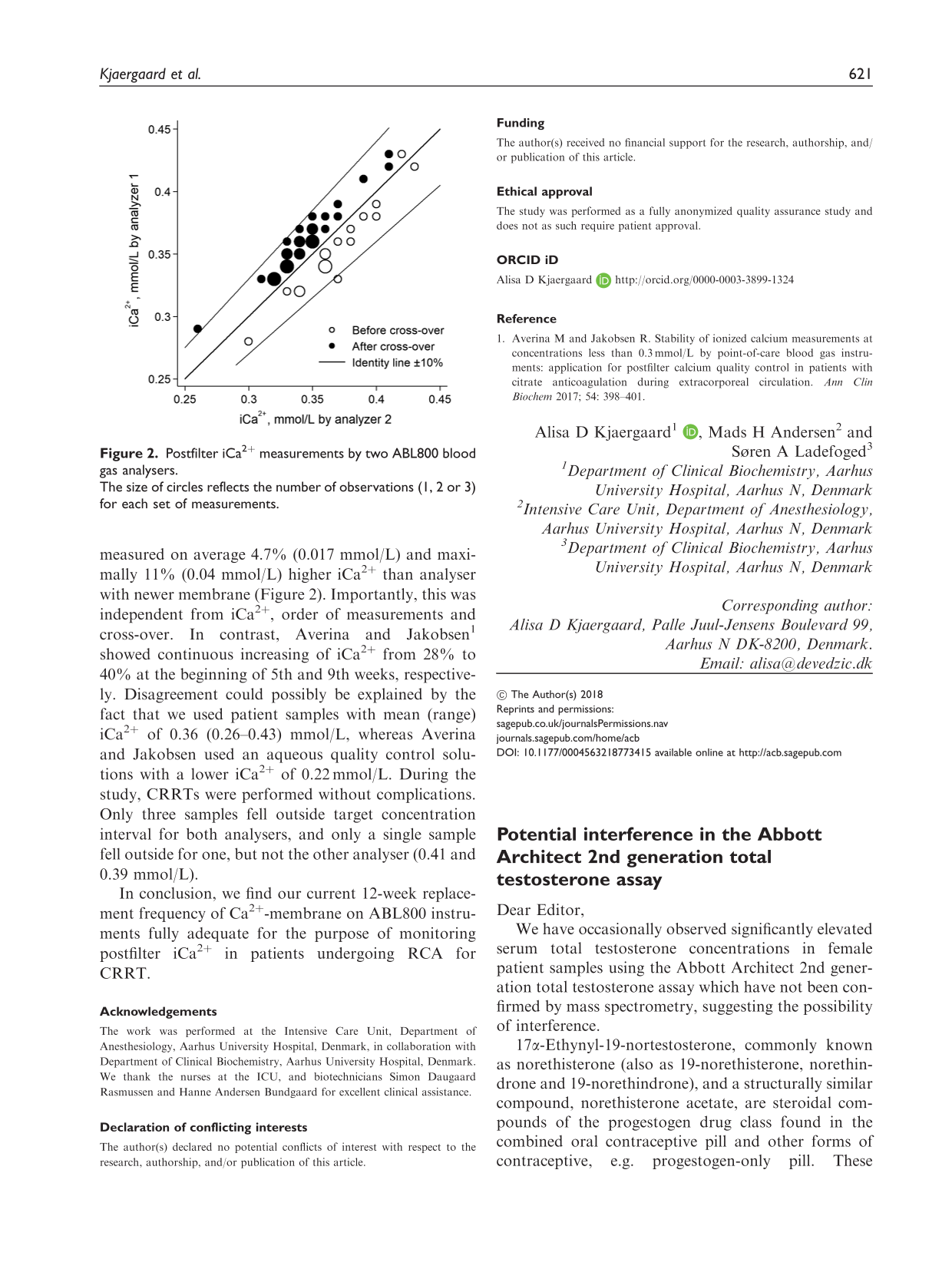
Total testosterone was measured on nine female patient samples and on a pooled female patient serum sample (baseline concentration). The stock solution was then added to each of these samples to produce 19-norethindrone concentrations of 100nmol/L, 50 nmol/L and 10 nmol/L, and total testosterone concentration re-measured (Table 1). Increasing addition of 19-norethindrone was associated with an increase in total testosterone concentration from baseline in all samples. It is not known if these patients were on any form of contraception.
Effect of increasing concentrations of spiked 19-norethindrone on serum total testosterone.
Users should be aware of the potential interference from norethisterone in the Abbott Architect 2nd generation total testosterone assay when interpreting increased serum testosterone concentrations in female patients.
Footnotes
Acknowledgements
The authors would like to thank the Biochemistry Department at East Kent Hospitals for purchasing the 19-norethindrone used in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
CR.
Contributorship
CR researched the literature and conceived the study. CR was involved in protocol development. CR and SR performed the sample analysis and data analysis. CR drafted the first manuscript and SR critically revised the manuscript. All authors approved the final version of the manuscript.
