Abstract
Background
Faecal immunochemical testing is increasingly being used to triage symptomatic patients for suspected colorectal cancer. However, there are limited data on the effect of preanalytical factors on faecal haemoglobin when measured by faecal immunochemical testing. The aim of this work was to evaluate the stability of faecal haemoglobin in faeces and to compare two methods of faecal haemoglobin sampling for faecal immunochemical testing.
Methods
Six patients provided faeces for faecal haemoglobin measurement which were transferred into specialized collection devices at baseline and at 1, 2, 3 and 7 days after storage at either room temperature or 4°C. A total of 137 patients returned both faeces transferred into the specialized collection device and faeces in a standard collection pot. A quantitative immunoturbidometric method was used to measure faecal haemoglobin and results were compared categorically. Discrepant results were assessed against diagnosis.
Results
Faecal haemoglobin concentration declined rapidly within a day of storage at room temperature but results remained ≥10 μg Hb/g faeces in 5/6 patients after two days. A faecal haemoglobin result ≥10 μg Hb/g faeces was obtained in 4/6 patients after storage for seven days at 4°C. Results obtained when patients used specialized collection devices were significantly different from results obtained when faeces was transferred into the specialized collection device in the laboratory.
Conclusion
There is considerable heterogeneity in the sample stability of faecal haemoglobin; therefore, samples should be transferred rapidly into specialized collection devices to prevent false-negative results. Use of collection devices by patients can lead to false-positive results compared with their use in a laboratory.
Introduction
Colorectal cancer is a major global health burden in terms of morbidity and mortality with approximately 700,000 deaths worldwide annually and is the third most commonly diagnosed cancer. 1 Screening programmes for colorectal cancer have historically used guaiac-based methods for faecal haemoglobin (f-Hb) testing (gFOBT), but these are now being superseded by immunochemical methods. 2 Faecal immunochemical testing (FIT) for f-Hb uses either monoclonal or polyclonal antibodies against the globin chain, and quantitative FIT methods typically utilize immunoturbidimetry on an automated analyser platform. 3 In addition to their use in screening programmes in asymptomatic individuals, FIT has excellent negative predictive value for colorectal cancer in symptomatic patients. 4 A positive laboratory result for f-Hb was included as a criterion for urgent referral for suspected cancer in NICE guideline NG12 in 2015. 5 This has since been supplemented by NICE diagnostic guideline DG30 published in July 2017 which recommended FIT using a cut-off value of 10 μg Hb/g faeces as part of a diagnostic pathway for suspected colorectal cancer. 6
Despite the huge potential for introduction of FIT into diagnostic pathways, there are still issues to be addressed especially with regard to analytical comparability of different methods and sample collection.7,8 There are published papers which demonstrated poor stability for f-Hb when analysed by traditional guaiac methodology or by FIT.9,10 For FIT methods, this can be ameliorated by using specialized sample collection devices. 3 Prior to f-Hb testing, a small amount of faecal material must be transferred to the collection device which contains buffer to stabilize the f-Hb. 3 Most publications on f-Hb sample stability have focused on the stability of f-Hb after sample collection into specialized collection devices.11–13 Previous work on stability of f-Hb in faeces using FIT methodology utilized normal faeces spiked with haemoglobin and showed rapid instability at room temperature. 10 Most diagnostic accuracy studies using FIT have provided patients with a specialized collection device for sample collection. 4 However there have been publications where faecal samples are sent to a central laboratory in a standard faeces collection pot for f-Hb transfer to collection device. 14
The aim of this work was to examine the stability of f-Hb in samples from patients at two different storage temperatures for seven days using FIT methodology. In addition, we compared f-Hb results obtained by two different sample collection procedures; primary collection of faeces by patients using a specialized collection device or transport of faeces to a central laboratory in a standard faeces collection pot prior to secondary transfer of the faeces to the specialized collection device by laboratory staff.
Methods
Patient recruitment
Patients were recruited as part of a larger diagnostic accuracy. Briefly, patients presenting with symptoms suggestive of cancer on a ‘two-week wait’ pathway were consented to provide both a faecal sample in standard faeces collection pot and to transfer a portion of the faeces into a specialized collection device. Patients then went on to have ‘gold standard’ investigations (imaging and/or colonoscopy) and these were used to assign a diagnosis after gastroenterology review. Assignment of diagnosis was performed blinded to f-Hb results. Of the 150 patients approached in this part of the wider study, 137 supplied faeces samples in both a standard faeces collection pot and in the specialized f-Hb collection device. These patients also underwent gold standard investigations to assign their diagnosis. Of the 13 patients excluded, nine failed to provide any faeces sample, one provided faeces in a standard faeces collection pot but not a sample in the specialized collection device, two patient samples arrived without a collection date/time and one patient withdrew consent for participation in the wider study.
Ethical approval was granted by East Midlands – Leicester South Research Ethics Committee, REC reference number: 14/EM/0217.
Sample collection and storage
Faeces were collected into standard faeces collection pots (Sarstedt, Nürnbrecht, Germany). Faeces were transported to the laboratory at room temperature on the day of collection and stored at 4°C until a small portion could be transferred into the specialized collection devices by laboratory staff. Following transfer, collection devices were stored at 4°C for up to one week prior to analysis. The collection devices were provided by Kyowa-Medex Co., Ltd (Tokyo, Japan) and consisted of a stick with two small grooves to ensure standard quantity of faecal collection. Transfer of faeces into the collection devices was performed in accordance with the manufacturer’s instructions; these instructions were also provided to patients. Faeces samples transferred by patients into the specialized collection devices were returned to the laboratory at room temperature on day of collection and then stored at 4°C for up to one week prior to analysis.
FIT analysis
FIT was performed using an automated analyser HM-JACKarc (Kyowa-Medex Co., Ltd, Tokyo, Japan). Calibration was performed as per manufacturer’s instructions, and internal quality control samples provided by the manufacturer were analysed in each batch. The analytical co-efficient of variation (%CV) between batch was 4.6% at a concentration of 27 μg Hb/g faeces and 3.6% at a concentration of 102 μg Hb/g faeces. Two distributions of External Quality Assessment samples from NEQAS were analysed and gave results acceptable for targets. A cut-off concentration of 10 μg Hb/g faeces was used to define a positive result. The manufacturer’s quoted limit of quantification (LoQ) of 7 μg Hb/g faeces was used in this study; the imprecision was <15% CV at this concentration. Limit of detection (LoD) was determined as 2 μg Hb/g faeces based on replicate analysis of low concentration f-Hb samples and defined as 1.65 SD above the limit of blank (LoB).
Statistical comparison
f-Hb results were categorized as either positive (≥10 μg Hb/g faeces) or negative (<10 μg Hb/g faeces) and results from patient sampled and laboratory sampled faeces were compared using the McNemar test performed with an online calculator. (https://www.graphpad.com/quickcalcs/McNemar1.cfm)
Results
Stability of f-hb
Faeces samples were provided by six patients in a specialized collection device to give a baseline f-Hb result and also in a standard faecal collection pot transported to the laboratory at room temperature. Following storage of faeces in the standard faecal collection pot at room temperature for 1, 2, 3 and 7 days, faeces was transferred to the specialized collection device and analysed for f-Hb together with the baseline sample (Figure 1). In most patients, there was a rapid decline in f-Hb concentration after one day storage at room temperature, although a result ≥10 μg Hb/g faeces was still obtained in five of six patients after two days storage at room temperature and in three of six patients after seven days storage at room temperature.
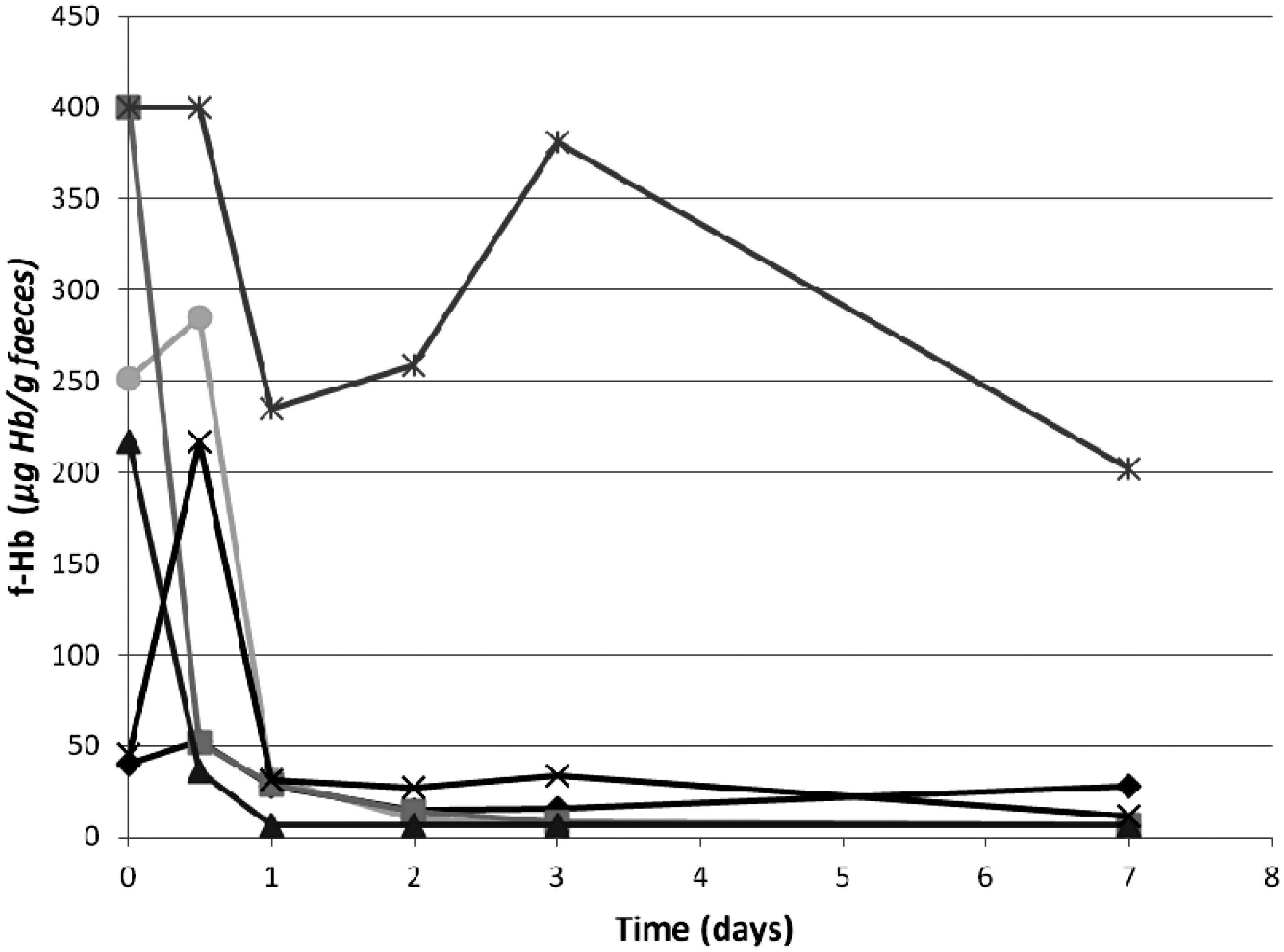
Stability of f-Hb at room temperature. Baseline f-Hb results (μg Hb/g faeces) in six patient samples were obtained using faeces sampled into collection devices by patients, who also provided faeces transported to the laboratory at room temperature. Aliquots of the faeces were stored at room temperature and re-sampled into collection devices at days 1, 2, 3 and 7 post receipt in the laboratory.
An identical study was undertaken with the same six patients except that faeces samples in collection pots were stored at 4°C (rather than room temperature) for 1, 2, 3 and 7 days, prior to transfer of faeces to the specialized collection devices (Figure 2). A result ≥10 μg Hb/g faeces was obtained in five of six patients after storage at 4°C for three days and in four of six patients after storage for seven days at 4°C.
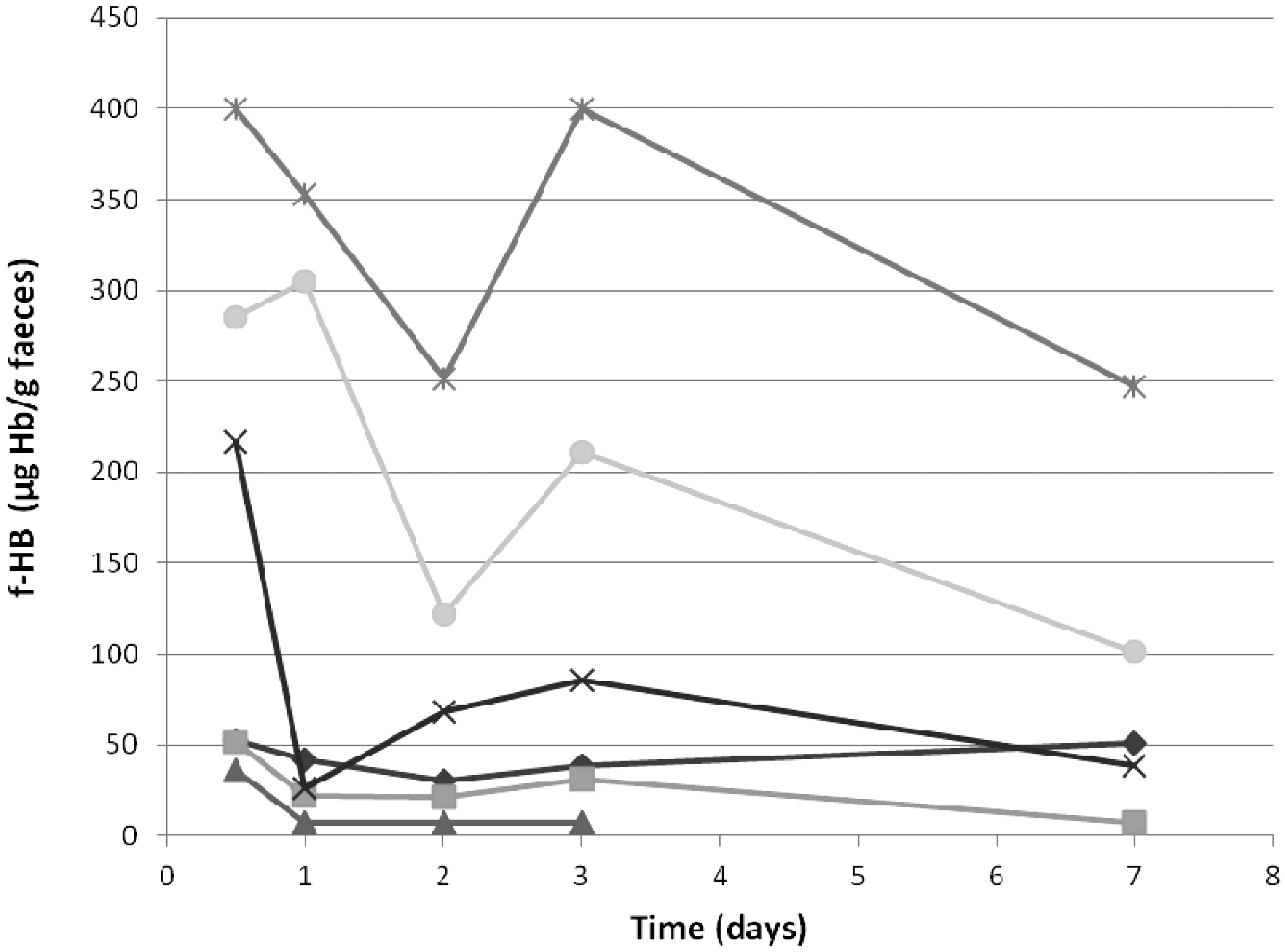
Stability of f-Hb at 4°C. Baseline f-Hb concentrations on six patient samples were obtained using faeces sampled onto specialized collection devices in laboratory on day of collection. Aliquots were stored at 4°C and faeces re-sampled onto collection devices at days 1, 2, 3 and 7 post receipt in the laboratory.
Comparing f-Hb results in these six patients for faeces transferred into the specialized collection device by the patient and faeces collected in standard faeces collection pots and subsequently transferred into the specialized collection device by the laboratory on the day of collection shows considerable heterogeneity (Table 1).
Initial f-Hb results (μg Hb/g faeces) of stability study samples.
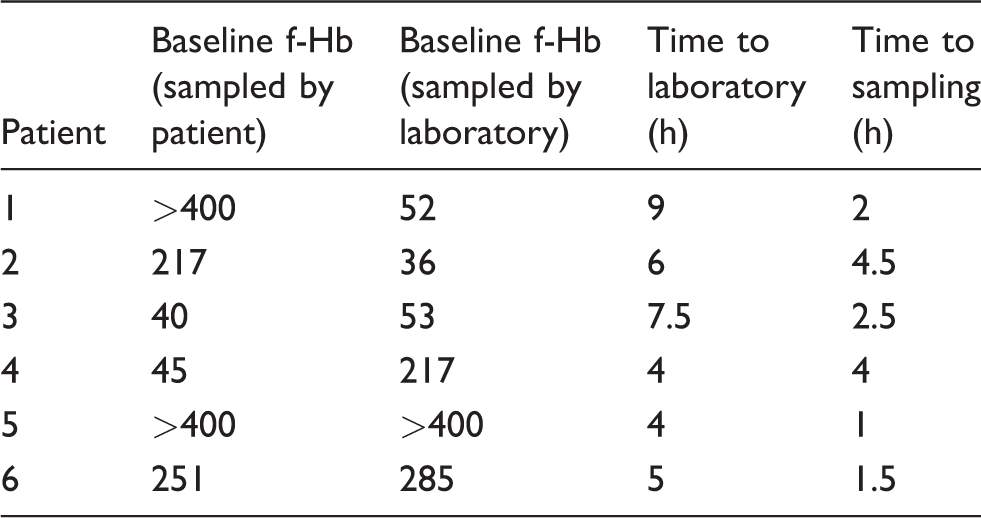
Note: Summary of the f-Hb results and time prior to laboratory transfer into collection devices for the six patient samples used for stability testing.
Comparison of f-Hb sampling by patient and laboratory
As part of a wider diagnostic accuracy study, a total of 137 patients provided both faeces transferred into the specialized collection device and a faeces sample in a standard faeces collection pot. The latter were transferred to a specialized collection device on the same day following receipt by the laboratory. Results were characterized as either undetectable f-Hb (below LoD of 2 μg Hb/g faeces), f-Hb above the LoD but below 10 μg Hb/g faeces, f-Hb between 10 μg Hb/g faeces and 400 μg Hb/g faeces and f-Hb >400 μg Hb/g faeces for comparative purposes (Figure 3(a)). A value of 10 μg Hb/g faeces was chosen as this is the cut-off value for FIT recommended in recent NICE diagnostic guidelines 6 and 400 μg Hb/g faeces represents the upper limit of the analytical working range quoted by the manufacturer for the method used. The data show that more patients fell into the ‘no f-Hb detected’ category when the samples were sent to the laboratory in standard faecal collection pots (with subsequent transfer to the specialized collection device on receipt) than when faeces samples were transferred by the patient into the specialized collection device. There was a corresponding increase in patient numbers in all other categories when transfer into the collection device was performed by patients (Figure 3(a)). The f-Hb results obtained by the two different sample collection methods were not equivalent when compared using McNemar test (P < 0.05). Samples from 13 patients generated numerical f-Hb results above the LoQ and below the upper limit of the analytical working range of the assay by both sampling methods, allowing comparison by Bland Altman plot (Figure 3(b)). This demonstrates poor numerical agreement for the two sampling methods and a wide scatter.
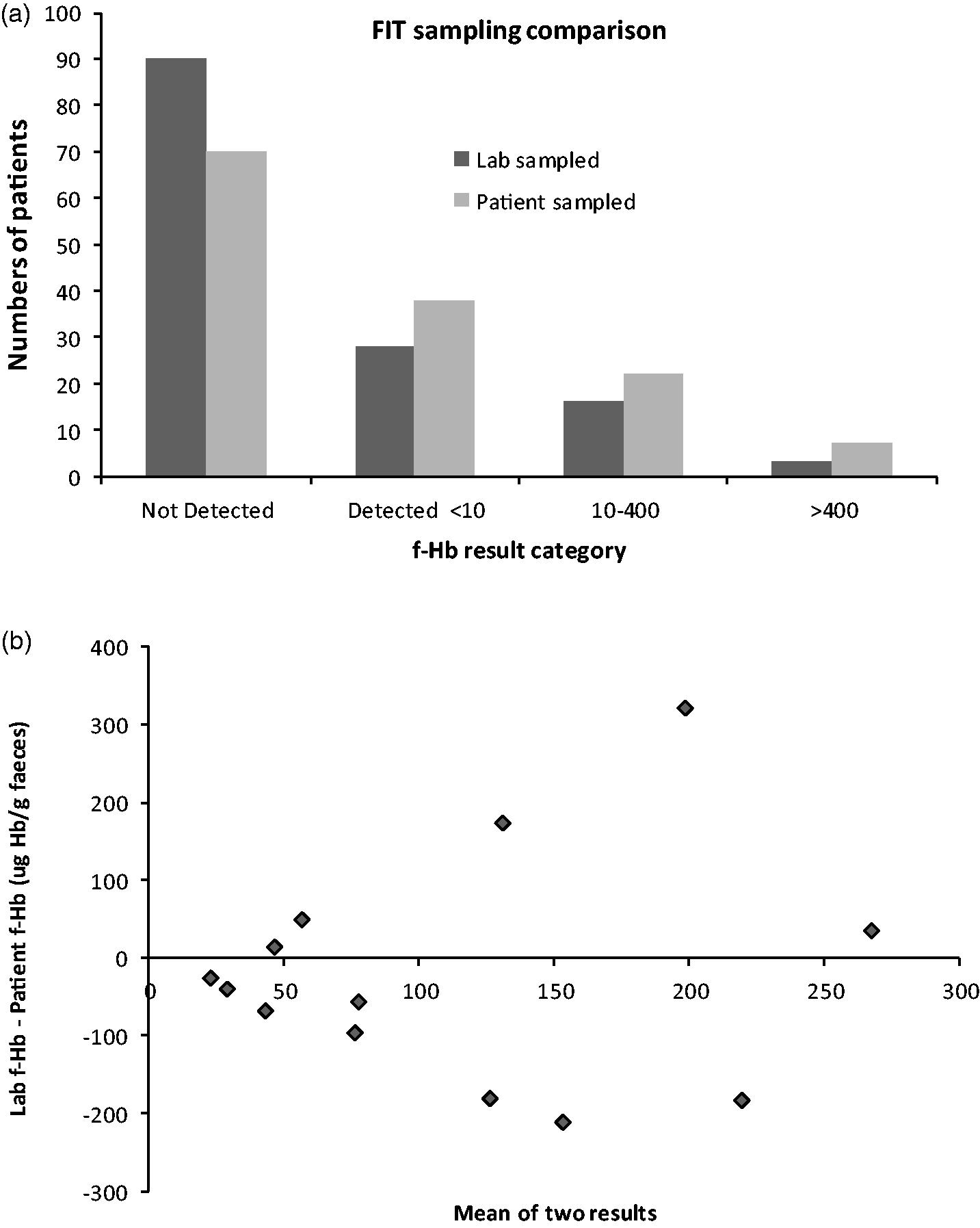
Comparison of patient and laboratory sampled f-Hb results. (a) Comparison of f-Hb results by category between faeces sampling performed by laboratory (dark grey) and sampling performed by patient (light grey) onto collection devices. (b) Bland Altman plot of numerical f-Hb results comparing faeces sampling onto collection devices performed by laboratory (Lab f-Hb) and sampling onto collection devices performed by patient (Patient f-Hb).
Using a cut-off value of 10 μg Hb/g faeces to define a positive f-Hb result, there were 11/137 (8%) patients with a different interpretation (positive or negative) when comparing the two methods of sample collection. In 10 of these 11 patients, a lower f-Hb result was obtained in samples sent to the laboratory in standard faecal collection pots. The relationship between time delay in transferring into the collection device by the laboratory and clinical diagnosis was examined in these patients (Table 2). Discrepant results were seen with a wide range of different time delays in laboratory transfer of faeces into collection devices. Patient diagnosis was assigned by a consultant gastroenterologist and based on colonoscopy and imaging studies. Four patients who had f-Hb results >10 μg Hb/g faeces with primary collection by the patient into specialized collection devices but negative results on samples collected into standardized faeces collection pots were found to have colorectal polyps. Polyps in two patients were found to be high risk, low-grade tubular adenomas (TA) and one patient’s polyps were classified as adenocarcinoma.
Discrepant f-Hb results (μg Hb/g faeces) and diagnosis.
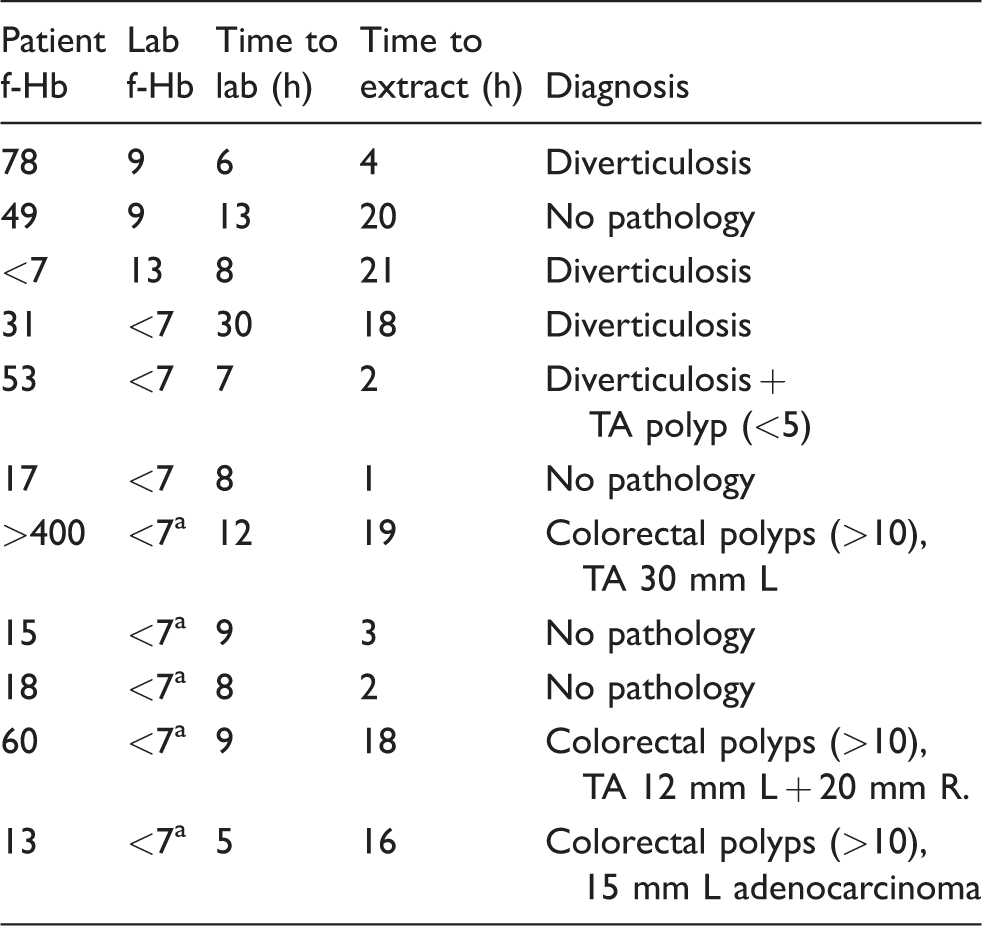
Note: Clinical outcome for patients with discrepant f-Hb results when comparing f-Hb sampling performed in the laboratory to sampling performed by the patient. Positive f-Hb was defined as ≥10 μg Hb/g faeces.
TA: tubular adenoma; L: left colon; R: right colon.
af-Hb above LoD (>2 μg Hb/g faeces) but below the manufacturers quoted LoQ for the assay.
Discussion
The stability of faecal samples for f-Hb testing and the approach used for sample collection will be a key consideration for laboratories as pathways including FIT are rolled out in response to recent NICE guidelines. 6 Colorectal screening programmes have demonstrated increased participation by patients when using FIT collection devices rather than traditional gFOBT, suggesting that patient transfer of faeces into specialized collection devices will be effective. 2 The f-Hb stability results presented here are consistent with the previous work on faeces spiked with haemoglobin which showed rapid decrease in f-Hb concentration after room temperature storage. 10 In the absence of biological variation data, the point at which a decline in results becomes statistically significant can be defined as 2.3 times the analytical standard deviation of the assay. This is based on a one-sided 95% probability factor (Z) of 1.65 multiplied by √2 to account for variation in two measurements. Applying this criterion to our study, the decline in f-Hb concentration was significant by day 1 after storage at room temperature for all six patients. When samples were stored at 4°C, significantly lower f-Hb results were seen in four of six patients by day 1. More patients retained a positive result ≥10 μg Hb/g faeces when samples were stored at 4°C compared with room temperature at both three and seven days post storage. Together this is weak evidence that storage of faeces at 4°C may reduce the rate of f-Hb degradation. Further experiments on fresh faeces and with greater control of faeces storage temperature prior to transfer into collection devices in the laboratory are required to confirm this observation.
Our data show considerable heterogeneity in stability across the six patient samples stored for seven days at 4°C. Of the six patients, two patients maintained a similar level of f-Hb from initial result to day 7 (4°C) result, a further two showed ∼50% reduction in f-Hb concentration by day 7 (4°C) and the remaining two patients had f-Hb <7 μg Hb/g faeces by day 7 (4°C). Heterogeneity in f-Hb stability most likely reflects the heterogeneity of faeces as a sample matrix and may be less apparent in previously published work using faeces spiked with haemoglobin. Since f-Hb stability is variable and cannot be known for an individual sample, all samples tested using FIT must be treated as if they are equally unstable. Rapid transfer of faeces into specialized collection devices appears to be essential to minimize the risk of false-negative FIT results. Improvements to collection device technology mean that current data on f-Hb stability may not be applicable in the future. 15
Comparison of f-Hb results in the 13 patients with results above the LoQ and below the upper limit of the analytical working range of the assay on both sampling methods (Figure 3(b)) shows a slight negative bias for f-Hb when faeces is transferred into a collection device in the laboratory. The wide spread of the data also reflects heterogeneity of faeces. The slight negative bias in f-Hb results when faeces are transferred into a collection device in the laboratory is also borne out when analysing the data categorically (Figure 3(a)). Taken together with the other data presented in this paper, the most likely explanation for this difference would be instability of f-Hb in faeces when tested by FIT methodology. Although there was a statistically significant difference in categorization between the two different sampling methods (P < 0.05), the majority of patients 126/137 (92%) had the same classification using a single cut-off value of 10 μg Hb/g faeces to indicate a positive result. This shows that any delay in f-Hb transfer into collection devices in the laboratory does not affect classification in large numbers of patients using the widely accepted cut-off value of 10 μg Hb/g faeces. However, it does demonstrate that the classification of individual patients can be affected by the f-Hb sampling method chosen and this needs to be considered when interpreting results of diagnostic accuracy studies using FIT.
The two alternative possible explanations for the discrepant f-Hb results between patient and laboratory transfer of faeces into collection devices other than f-Hb instability are: (1) poor or variable patient technique when using specialized collection devices or (2) inherent heterogeneity of faeces as a sample matrix. In this work, we have assumed that patients transferred faeces into the collection device while the faeces were fresh as per instructions. Heterogeneity of f-Hb breakdown in faeces may contribute to the results we have observed as solid material within the faeces may hinder proteolytic breakdown of globin chains. Early in the f-Hb degradation process, there may be increased exposure of Hb epitopes causing an initial increase in results before f-Hb degrades further. Different FIT methods utilize different antibodies recognizing different epitopes on the globin chain; therefore, it is possible stability of f-Hb when measured by FIT methods may be assay specific. If stable peptide degradation fragments of globin exist, then antibodies which cross-react with these may provide improved detection of f-Hb by FIT assays. Patient pathways involving repeat testing of a single faeces sample or multiple faeces samples may minimize the impact of heterogeneity and facilitate correct patient classification. However, a published study looking at collection of two consecutive faeces samples for f-Hb testing suggested that use of a second sample did not improve diagnostic yield. 16
Access to the diagnosis in the 11 patients who gave discrepant positive or negative classification with the different FIT sampling methods provides an indication as to the clinical significance of these discrepancies. The one patient in this group with adenocarcinoma had a borderline positive result of 13 μg Hb/g faeces that was detectable but not quantifiable when the faeces was transferred into collection device in the laboratory. Two other patients with tubular adenomas >1 cm had highly discrepant f-Hb results when faeces was transferred into collection devices in the lab >24 h after patient had transferred faeces into collection devices. This illustrates that while f-Hb stability is variable, it is important that faecal samples for FIT are rapidly transferred into collection devices to minimize the chance of missing positive results, especially those close to the cut-off concentration.
While it is vital to prevent false-negative results due to sample instability, it is also important to ensure that incorrect FIT results (false-positive and false-negative) are not produced by poor patient sampling technique. Patient instructions provided for faeces transfer into collection devices will be vital to the success of any FIT pathways that are introduced as a result of NICE DG30. Important limitations of this study include the small number of patient samples and the lack of control of temperature during transport to lab. Adequately powered diagnostic accuracy studies comparing both possible sampling methods with tight control of preanalytical factors assessed against clinical outcome may assist in definitively determining whether the method of faeces transfer into collection devices influences diagnostic performance of FIT.
Footnotes
Acknowledgements
The authors would like to thank Matthew Davis from Alpha Laboratories for supplying additional consumables for this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work formed part of a larger study funded for JT by York Against Cancer. Alpha Laboratories provided some additional consumables and loan of the HM-JACKarc analyser.
Ethical approval
Ethical approval was granted by East Midlands – Leicester South Research Ethics Committee, REC reference number: 14/EM/0217.
Guarantor
DT.
Contributorship
SM, SB, CC and MF performed f-Hb analysis, JT assigned patient outcome. CC, MF and JT assisted in preparation of manuscript. DT conceived the study and prepared the manuscript. All authors approved the final version.
