Abstract
Background
Faecal immunochemical tests (FIT) are routinely used in colorectal cancer (CRC) screening programmes around the world. More recently, quantitative FIT has been recommended to help triage patients presenting to primary care with symptoms suggestive of CRC. Participants collect faecal samples using sampling probes which are inserted into sample collection devices (SCDs) containing preservative buffer. The SCDs have an internal collar designed to remove excess sample. The aim of this study was to investigate the impact of multiple loading on faecal haemoglobin concentration (f-Hb) using SCDs of four FIT systems.
Methods
Pools of f-Hb negative samples were spiked with blood, homogenised and loaded into SCDs 1, 3 and 5 times, with insertion of the sampling probes into the SCDs with and without mixing between loads. The f-Hb was measured using the relevant FIT system. The percentage change in f-Hb for multiple loads was compared with a single load for each system for the mixed and unmixed groups.
Results
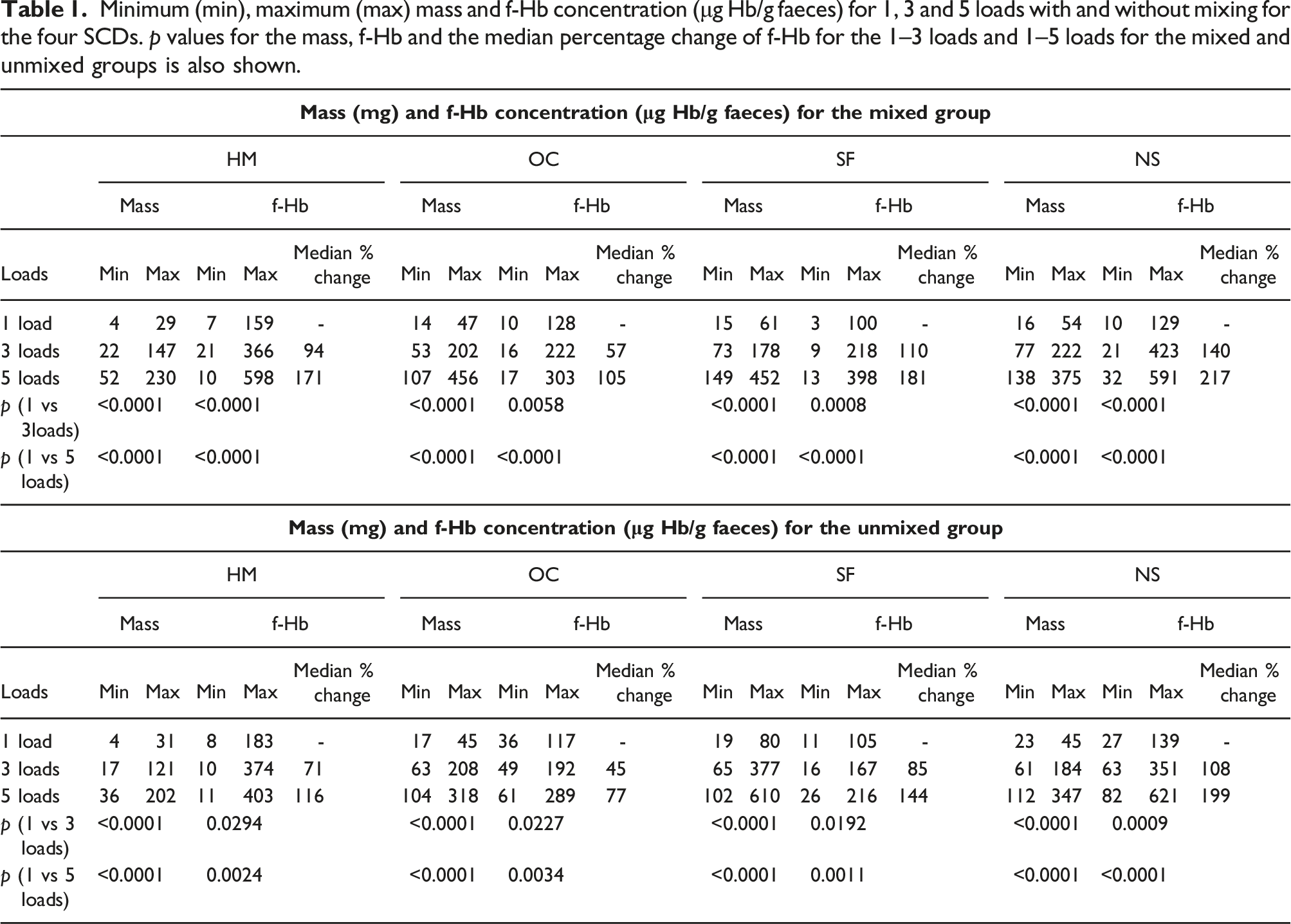
The p values show a significant difference (p < 0.05) in the mass and f-Hb for the mixed and unmixed group, for 1–3 and 1–5 loads for all systems. The median percentage change in f-Hb for the mixed is higher than the unmixed group.
Conclusion
This study showed that multiple loading does significantly increase the f-Hb in the SCDs.
Keywords
Introduction
Colorectal cancer (CRC) is the third most common cancer worldwide. 1 The faecal immunochemical test (FIT) for haemoglobin (Hb) is used as a screening tool for asymptomatic populations and was introduced into the English screening programme in 2019. Quantitative FIT is also now established for triaging to exclude CRC in patients presenting to primary care with symptoms suggestive of CRC.2,3
Following simple instructions, a participant collects a small faecal sample using a sampling probe and inserts this into a bottle containing a preservative buffer. The bottle has an internal collar that is designed to remove excess sample. The sample collection devices (SCDs) are transported to laboratories for analysis.
The collection method means variation in the sample mass is possible. Previous evaluations4,5 found SCDs delivered more sample into the buffer than manufacturers quote. One study 6 found small increases in sample mass loaded onto the probe did not impact the faecal Hb concentration (f-Hb) significantly using four FIT systems. Gross overloading with sample has been observed in the screening programme and, though instructions clearly state to load once, it is obvious that some participants load the SCDs several times. By this, we mean they dip the sampling probe in the faeces, place probe in buffer, remove it, re-dip and re-load. We refer to this as multiple loading.
Aim
To investigate the impact of multiple loading on f-Hb concentration using SCDs of four FIT systems.
Method
Anonymised portions of faecal samples, sent to the routine biochemistry laboratory for calprotectin measurement, were collected into plastic bottles and homogenised for 2 minutes. The initial f-Hb was measured using a quantitative FIT. 177 samples with <10 μg Hb/g faeces (μg/g) were selected and frozen (−20°C).
Hb-lysate was prepared using anonymised blood collected into EDTA bottles with Hb within the reference range, measured using the ADVIA 2120 (Siemens Healthcare Ltd). Aliquots of the lysate were frozen.
The Hb-negative samples were thawed in batches, pooled (5–8 samples). Each batch was homogenised, spiked with Hb-lysate (n = 20, 25–145 μg/g) and homogenised for 3 minutes then divided into four lots (one for each SCD type) and refrigerated; lots were brought to room temperature 10 min before loading, one at a time, and loaded immediately into the FIT SCDs: EXTEL HEMO AUTO-MC Collection Picker (Minaris Medical Co. Ltd), OC-Auto Sampling Bottle 3 (Eiken Chemical Co. Ltd), SENTiFIT pierceTube (Sentinel Diagnostics), or Specimen Collection Container A (Alfresa Pharma Corp). The order of SCDs was varied for each sample to remove bias. 1, 3 and 5 loads were set-up for each SCD type before starting the next.
Samples were loaded in duplicate; two loading techniques were used. Mixed: 20 samples were loaded 1, 3 and 5 times with insertion of the sampling probe into the SCDs and inversion 5 times between loadings (n = 40). Unmixed: 10 samples loaded in the same way without inverting between loads (n = 20). For 1 load, sample was loaded on the grooves only.
While reloading, care was taken to ensure sample was not present on the sampling probe tip. The SCDs were weighed before and after loading using a Sartorius BP210S balance (SD ≤ ± 0.0001 g) to determine the mass of sample loaded. The SCDs were incubated at 15–24°C up to 72 h, mixed by inversion and f-Hb measured using the corresponding analyser (HM-JACKarc (HM), OC-SENSOR PLEDIA (OC), FOB Gold Wide SENTiFIT-270 (SF) and NS-Prime (NS) respectively). Results were calculated in μg/g.
The percentage change in f-Hb for multiple loads was compared with a single load for each system. The Wilcoxon Mann-Whitney Test was used to test the significance of the difference between 1–3 and 1–5 loads for mixed and unmixed samples.
Results
Minimum (min), maximum (max) mass and f-Hb concentration (μg Hb/g faeces) for 1, 3 and 5 loads with and without mixing for the four SCDs. p values for the mass, f-Hb and the median percentage change of f-Hb for the 1–3 loads and 1–5 loads for the mixed and unmixed groups is also shown.
Discussion
In this small study, we have demonstrated that multiple loading significantly increases the f-Hb concentration in samples. This was expected but this data has not been previously published.
This pre-analytical variable of FIT is something that the laboratory has limited control over, other than ensuring that the instructions for use are very clear. Despite the fact that upon visual inspection it is clear that some samples have been multiple loaded there is currently no way to scientifically confirm this.
The clinical impact is that with multiple dipping a result above a defined threshold is more likely and an individual referred for bowel visualisation such as colonoscopy, when if the sample was loaded with one dip they might not. The clinical risk for participants that have multiple loaded the SCDs is low because they are more likely to have a positive result, however with already strained colonoscopy resources this is not ideal.
The analytical impact is that grossly overloaded SCDs are rejected by the analyser or block the analyser’s sample probe giving no result; overloaded samples might also be spoilt due to the cap not fitting. All of these would lead to the subject being requested to provide another sample.
Differences between the design of the different SCD collars (such as width of collar/channel) means when the sampling probe is reinserted, varying amounts of excess sample is removed. Another observation was that inserting the probe at an angle could remove excess sample on the sides of the SCDs before entering the buffer. Manufacturer instructions state to load the SCD’s once and specify excess sample is not required. However, visual inspection of SCD’s returned by the screening population shows otherwise. This was also stated in the study by Zahoor et al. 7 Therefore, it is crucial that the instructions should be made simpler and clearer.
The f-Hb for some 3 load results are highest and some 5 load results are lower than 1 load. Although samples were homogenised, undigested matter could prevent even distribution of f-Hb. Although sample loading once was done with no excess, the mass range shows the amount of sample in the buffer exceeds the manufacturers’ quoted mass as seen in previous studies.4,5 The f-Hb are calculated and expressed in μg Hb/g faeces based on the mass of faeces collected in the grooves with one dip. With multiple loading, results expressed in μg/g is calculated using the expected mass collected with a single dip, and are therefore not accurate due to the increased mass of faeces.
Conclusion
This study shows that multiple loading does significantly affect the f-Hb for the four SCD’s.
Footnotes
Acknowledgements
We would like to thank the FIT system suppliers (HM-JACKarc: Alpha Laboratories, Eastleigh, UK; NS-Prime: Alfresa Pharma, Osaka, Japan; OC-Sensor PLEDIA: Mast Diagnostics Division, Bootle, UK; SENTiFIT 270: Sentinel Diagnostics, Milano, Italy) for supplying the analysers and consumables. We also thank Berkshire and Surrey Pathology Services (Royal Surrey Foundation Trust, Guildford, UK) for supplying samples used in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
CJ.
Contributorship
CJ conceived the study with input from SCB and CP. CJ processed the samples. CJ analysed the data and prepared the manuscript with input from CP and SCB.
