Abstract
Background
It has been reported that both of the miR-132/212 (micro-RNA) cluster members, miR-132 and miR-212, are downregulated in hepatocellular carcinoma. Nevertheless, the expression pattern and clinical utility of serum miR-132/212 in hepatocellular carcinoma are still unknown.
Methods
In this study, serum concentrations of miR-132 and miR-212 were measured in 80 hepatocellular carcinoma patients, 51 controls with chronic liver diseases and 42 healthy volunteers by using quantitative real-time polymerase chain reaction.
Results
In hepatocellular carcinoma patients, serum concentrations of miR-132 and miR-212 were significantly reduced and strongly correlated (r = 0.603, p < 0.001). Receiver operator characteristic analyses showed that serum miR-132 and miR-212 might have a potential role in the diagnosis of hepatocellular carcinoma. Moreover, the combination of serum miR-132, miR-212 and alpha-fetoprotein improved the diagnostic efficiency for hepatocellular carcinoma, especially in sensitivity and negative predictive value. Serum miR-132 was associated with tumour differentiation degree (p = 0.021) and tumour–node–metastasis stage (p = 0.002); serum miR-212 correlated with tumour size (p = 0.023) and tumour–node–metastasis stage (p = 0.007). Kaplan–Meier analyses indicated poorer overall survival in hepatocellular carcinoma patients with lower serum concentrations of miR-132 (p < 0.001) and miR-212 (p = 0.005).
Conclusions
Our results suggest that both components of the miR-132/212 cluster have potential roles as non-invasive serum biomarkers for diagnosis and prognosis of hepatocellular carcinoma.
Introduction
Hepatocellular carcinoma (HCC) is one of the commonest human malignancies worldwide, accounting for more than 600,000 deaths annually. Current treatment strategies, including hepatectomy, liver transplantation and chemoembolization, have significantly improved the life quality of HCC patients, but five-year survival rate has not changed much over the past decade. Early diagnosis and effective, timely treatment are likely to prolong the survival of HCC patients.1,2 Serum alpha-fetoprotein (AFP) has been routinely used for early detection of HCC for many years. However, its sensitivity and specificity remain suboptimal, underpinning an urgent need to develop novel biomarkers in early diagnosis, prognosis and assessment of treatments for HCC.
MicroRNAs (miRNAs) are conserved non-coding RNA sequences 18–25 nucleotides long, which regulate gene expression by interacting with the 3′-untranslated region of the target messenger RNAs (mRNAs), thereby leading to repression of post-transcriptional translation and/or degradation of mRNAs. There is mounting evidence that deregulation of miRNAs contributes to the occurrence and development of many human malignancies, including HCC. Additionally, miRNAs are involved in cancer cell proliferation, differentiation, apoptosis, migration and invasion, angiogenesis, etc.3,4 Therefore, better knowledge of the roles of miRNAs during hepatocarcinogenesis might provide new avenues for HCC diagnosis and treatment. Importantly, miRNAs are detected not only in tissues but also in cell-free body fluids, including serum, plasma, urine, etc. which are much more readily obtainable. Their resistance to endogenous RNase activity and stability under different conditions makes miRNAs potentially promising as diagnostic or prognostic markers and attractive therapeutic targets for human cancers.5,6
MiR-132 and miR-212, collectively termed as the miR-132/212 cluster, are highly conserved among vertebrates. Both are located on chromosome 17p13.3 and share an identical 5′ seed sequence. MiR-132 and miR-212 genes are arrayed in tandem; miR-212 is about 260 bp from miR-132. The miR-132/miR-212 genes are co-transcribed to primary (pri)-miRNA-212/132 in the form of a gene cluster. This is processed and cleaved to the mature miR-132 and miR-212.7,8 Previous studies have indicated that miR-132 and miR-212 are dysregulated in many human malignancies and play important roles in carcinogenesis, including HCC.9–13 However, the expression pattern and clinical utility of concentrations of the serum miR-132/212 cluster in human cancers remain to be established. In this study, we measured serum concentrations of miR-132 and miR-212 in HCC patients and examined their potential clinical utility in the diagnosis and prognosis of HCC.
Materials and methods
Patients and serum samples
A total of 173 subjects were enrolled, including 80 patients with primary HCC, 51 patients with chronic liver diseases (CLD) and 42 healthy volunteers. All cases of HCC were diagnosed on the basis of clinicopathological findings. The grade of tumour cell differentiation was estimated according to the World Health Organisation classification system, and the clinical pathological stage was established according to the tumour–node–metastasis (TNM) staging system of the International Union against Cancer Society. HCC patients were followed up for three years after surgical resection or hepatectomy combined with trans-arterial chemoembolization. The CLD group was comprised of 31 patients with chronic hepatitis, 16 patients with liver cirrhosis, and four patients with non-alcoholic fatty liver disease. Three millilitres (mL) of peripheral blood was taken in the morning from all individuals. The samples were centrifuged at 4°C, 3000 g for 20 min, and serum was separated and stored at −80°C for further assay. All subjects enrolled in this study had given written informed consent, and this study was approved by the Ethics Committee of the Affiliated Hospital of Nantong University.
Detection of serum miRNAs by quantitative real-time PCR (qPCR)
Total RNA from serum samples was isolated with miRNeasy Serum/Plasma Kit (Qiagen, Germany) according to the manufacturer’s instructions. Purity and concentration of RNA were evaluated by the absorbance ratio at 260/280 nm, measured with NanoDrop, 2000 spectrophotometer (Thermo Fisher Scientific, USA). The integrity of extracted RNA was evaluated by formaldehyde denatured agarose gel electrophoresis. 14 The reverse transcription reactions were carried out with the miScript II RT Kit (Qiagen, Germany) according to the manufacturer’s protocol. Serum concentrations of miR-132 and miR-212 were measured with the miScript SYBR Green PCR Kit (Qiagen, Germany) on the ABI7500 qPCR detection instrument (Applied Biosystems, Foster, USA). The qPCR cycling conditions were as follows: initial denaturation at 95°C for 15 min, followed by 40 cycles of denaturation at 95°C for 15 s, annealing at 56°C for 30 s and extension at 70°C for 30 s. The cel-miR-39 was used as an internal control. All the primers were designed and purchased from RiboBio (Guangzhou, China). The relative serum miR-132 and miR-212 concentrations were calculated after being normalized against cel-miR-39 by using the equation 2–ΔΔCT, and a mixed serum sample from healthy volunteers was used as a control, ΔΔCT=(CtmiR-test−CtmiR-39-test) −(CtmiR-control− CtmiR-39-control).
Detection of serum AFP
Concentrations of serum AFP were measured by chemiluminescence immunoassay with ARCHITECT i2000SR analyser (Abbott, USA).
Statistical methods
Statistical analyses were performed using Statistical Product and Service Solutions Statistics 18.0 software. Differences in serum miR-132/212 concentrations were determined by non-parametric Mann–Whitney U test or Kruskal–Wallis H-test. The association between serum miR-132/212 and clinicopathological parameters was analysed by Fisher’s exact test. In addition, a receiver operator characteristic (ROC) curve was drawn to evaluate the diagnostic value of serum miR-132/212 for HCC. Kaplan–Meier survival curve was constructed and then log-rank test was used to compare the survival time in different subgroups. Statistical significance of differences between groups was set at p values of less than 0.05.
Results
Serum concentrations of miR-132/212 in HCC patients
Serum concentrations of miR-132 and miR-212 were analysed in patients and healthy controls, by qPCR. In accordance with the convention of using cel-miR-39 as an internal control,15,16 we did likewise; we detected the expression of cel-miR-39 in serum samples from the subjects and found that there was no obvious difference between groups (HCC, CLD, healthy volunteers) (all p > 0.05, data not shown). As shown in Figure 1(a), the concentrations of serum miR-132 (Median, Maximum–Minimum, 0.5, 0.04–1.3) were reduced in HCC patients, as compared with CLD (0.73, 0.06–1.52) and healthy control (0.87, 0.09–1.58) groups, p = 0.003 and p < 0.001, respectively. On the other hand, the concentrations of serum miR-212 (0.51, 0.04–1.1) were also decreased in HCC patients, as compared with CLD (0.67, 0.10–1.15) and healthy control (0.7, 0.08–1.13) groups, p = 0.023 and p = 0.003, respectively (Figure 1(b)). No significant difference was observed in concentrations of serum miR-132 or miR-212 between the CLD and healthy control group, p = 0.092 and p = 0.449, respectively. Moreover, Spearman correlation analysis showed that concentrations of serum miR-132 and miR-212 were strongly correlated in the HCC group (r = 0.603, p < 0.001) (Figure 2).
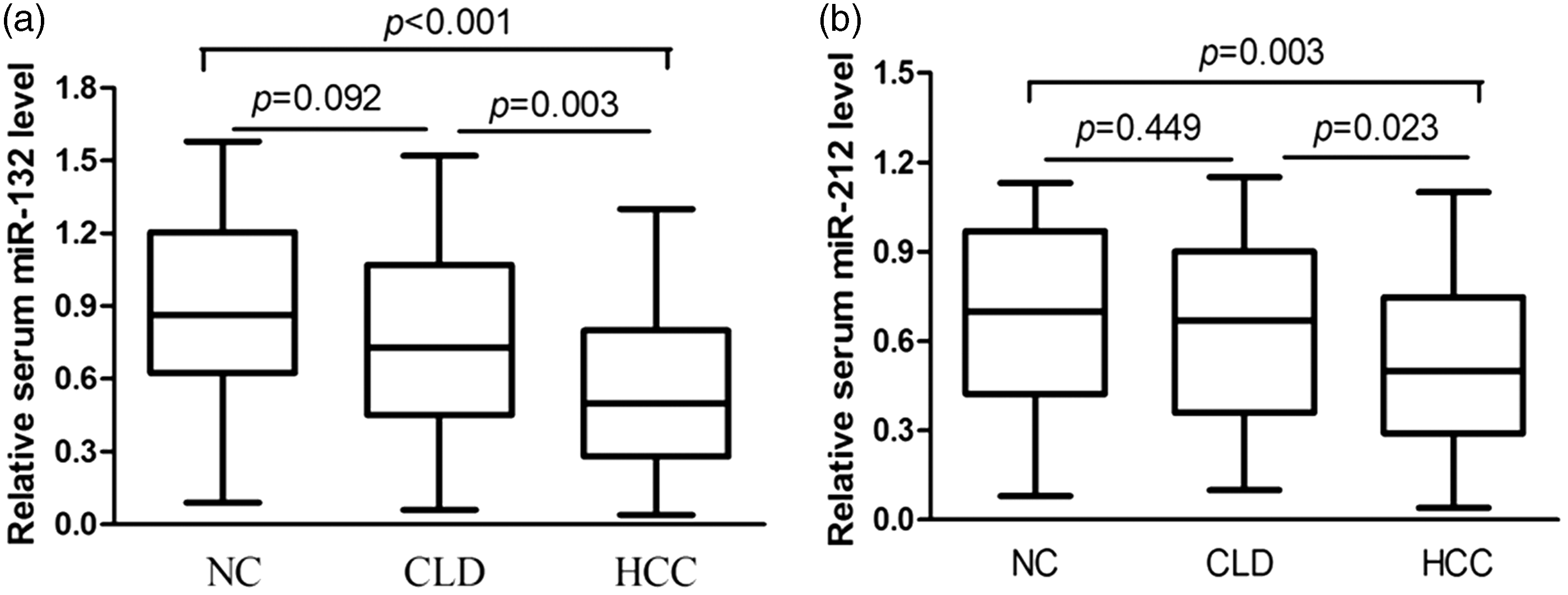
Detection of serum miR-132 and miR-212 expression levels by qPCR. The relative concentrations of serum miR-132 (a) and miR-212 (b) in healthy controls (NC, n = 42); patients with CLD (n = 51) and HCC (n = 80) were analysed and normalized to the internal control cel-miR-39. CLD: chronic liver diseases; HCC: hepatocellular carcinoma; NC: healthy controls.
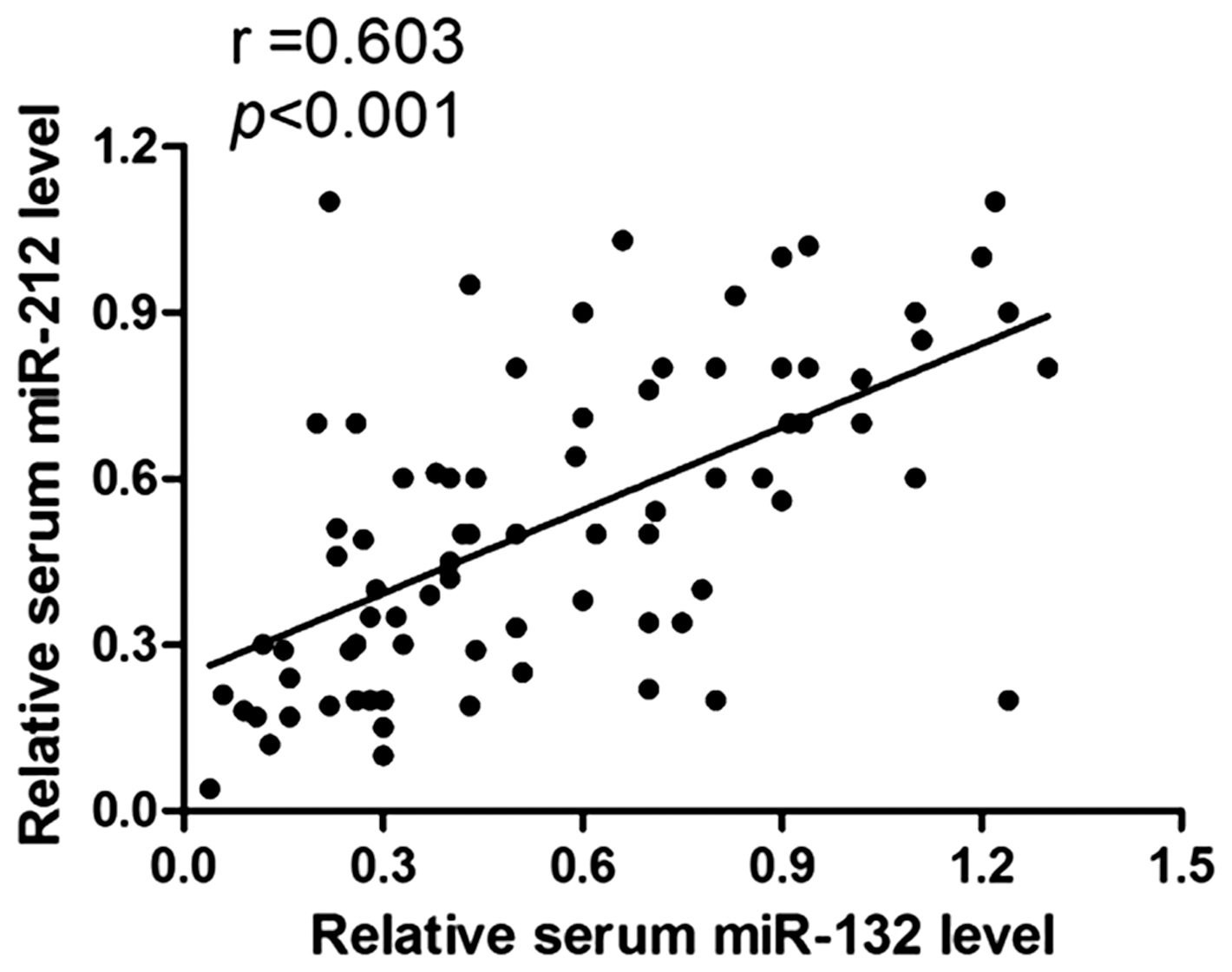
Spearman correlation analyses between serum miR-132 and miR-212 in HCC patients. Scatter plots were demonstrated that there was a positive correlation between the two members of serum miR-132/212 cluster, r = 0.603, p < 0.001.
Diagnosis values of serum miR-132/212 for HCC
Next, a ROC curve was drawn to evaluate the diagnostic performance of the serum miR-132/212 cluster in differentiating HCC from CLD and healthy controls (Figure 3). The area under the curve (AUC) (±95% confidence interval (CI)) of serum miR-132 was 0.747 (0.675–0.809), p < 0.001. When the cut-off value was defined at 0.51, sensitivity was 57.50% (95% CI, 45.9–68.5), specificity was 81.72% (95% CI, 72.4–89.0) and the Youden index was 0.3922. The AUC of serum miR-212 was 0.706 (0.632–0.733, p < 0.001). At a cut-off value of 0.64, sensitivity was 68.75% (57.4–78.7), specificity was 66.67% (56.1–76.1) and the Youden index was 0.3542.
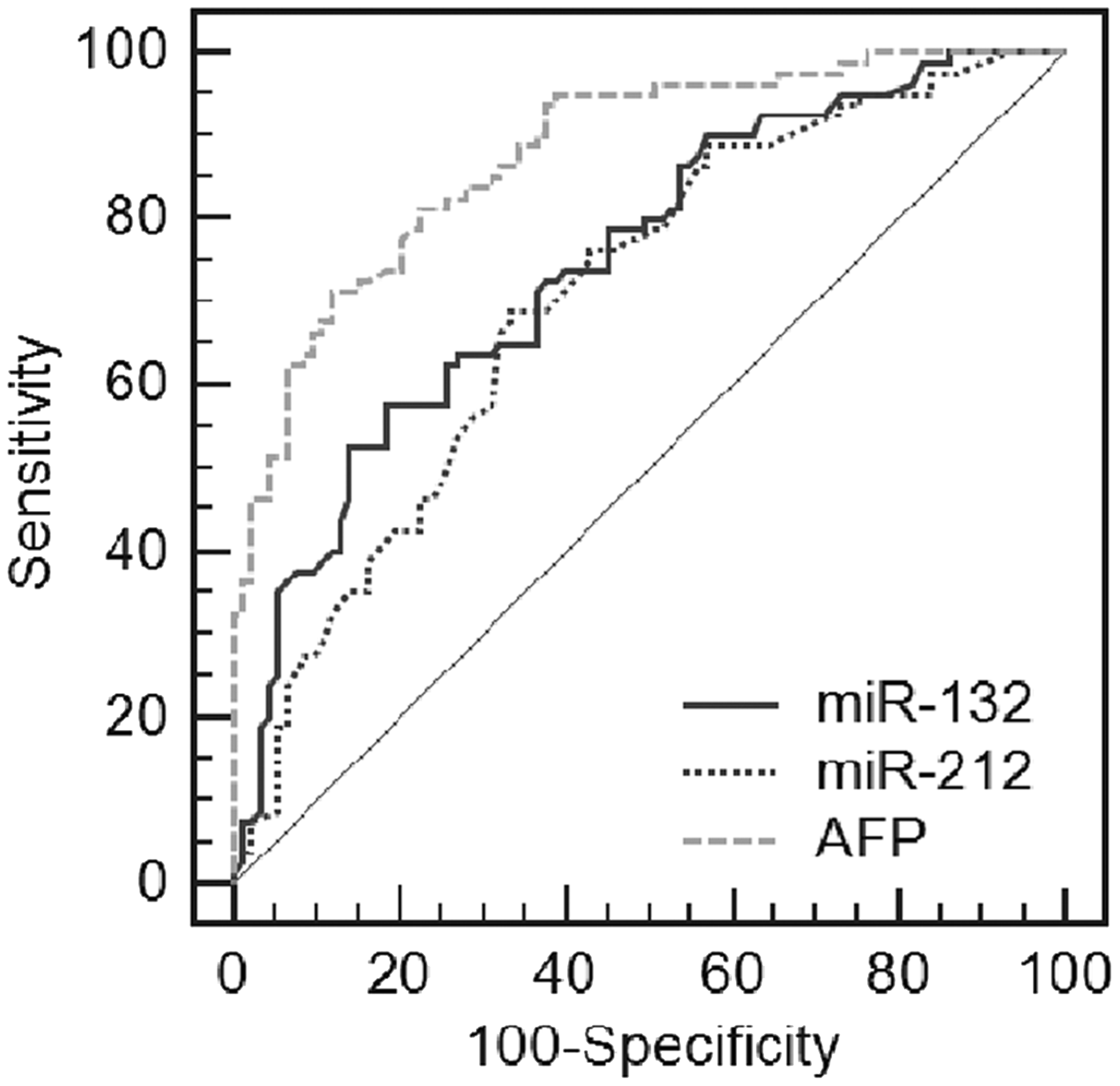
Evaluation of the diagnosis value of serum miR-132/212 for HCC. ROC curve analyses of the diagnostic value of serum miR-132, miR-212 and AFP in differentiating HCC from CLD and healthy controls.
We also investigated the diagnostic performance of these markers by a combination of serum miR-132, miR-212 and AFP. Compared with serum miR-132 or miR-212 alone, the sensitivity, negative predictive value and total efficiency were improved by a combination of serum miR-132 and AFP (all p < 0.05). Moreover, the sensitivity and negative predictive value were improved by a combination of serum miR-212 and AFP, compared with serum miR-132 or miR-212 alone (all p < 0.05). Importantly, the combination of serum miR-132, miR-212 and AFP could substantially increase the sensitivity and negative predictive value, compared with serum miR-132, miR-212 or AFP alone, all p < 0.05. Our results indicate that although the diagnostic value of serum miR-132 or miR-212 alone is not better than AFP, the combination of serum miR-132, miR-212 and AFP is associated with improved diagnostic efficiency for HCC (Table 1).
Diagnosis of HCC by serum miR-132, miR-212 and AFP.
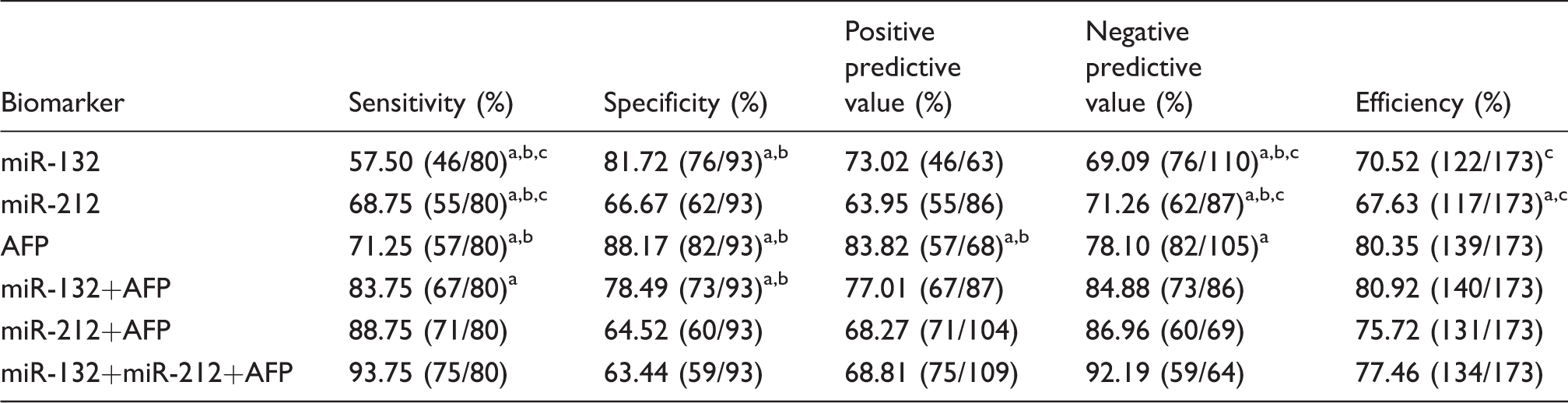
AFP: alpha-fetoprotein; HCC: hepatocellular carcinoma.
Compared with miR-132 + miR-212 + AFP, aP < 0.05; compared with miR-212 + AFP, bP < 0.05; compared with miR-132 + AFP, cP < 0.05.
Association between serum miR-132/212 and clinicopathological features of HCC
To analyse whether serum concentrations of the miR-132/212 cluster were associated with clinicopathological features of HCC, HCC patients were stratified by age, sex, hepatitis B virus (HBV) infection status, liver cirrhosis status, serum AFP, tumour size, degree of tumour differentiation and TNM stage. On the basis of the median concentrations of serum miR-132 and miR-212, HCC patients were classified into two groups: low-expression group and high-expression group. The statistical analyses revealed that low expression of serum miR-132 was associated with tumour differentiation degree (p = 0.021) and TNM stage (p = 0.002), but not with other clinicopathological features (all p > 0.05). Low expression of serum miR-212 positively correlated with tumour size (p = 0.023) and TNM stage (p = 0.007), but no other clinical parameter was associated with serum miR-212 (all p > 0.05, Table 2). The results suggest that serum miR-132/212 cluster may help to predict the degree of malignancy and assess the progression of HCC.
Association of serum miR-132 and miR-212 with clinicopathologic characteristics of HCC patients.
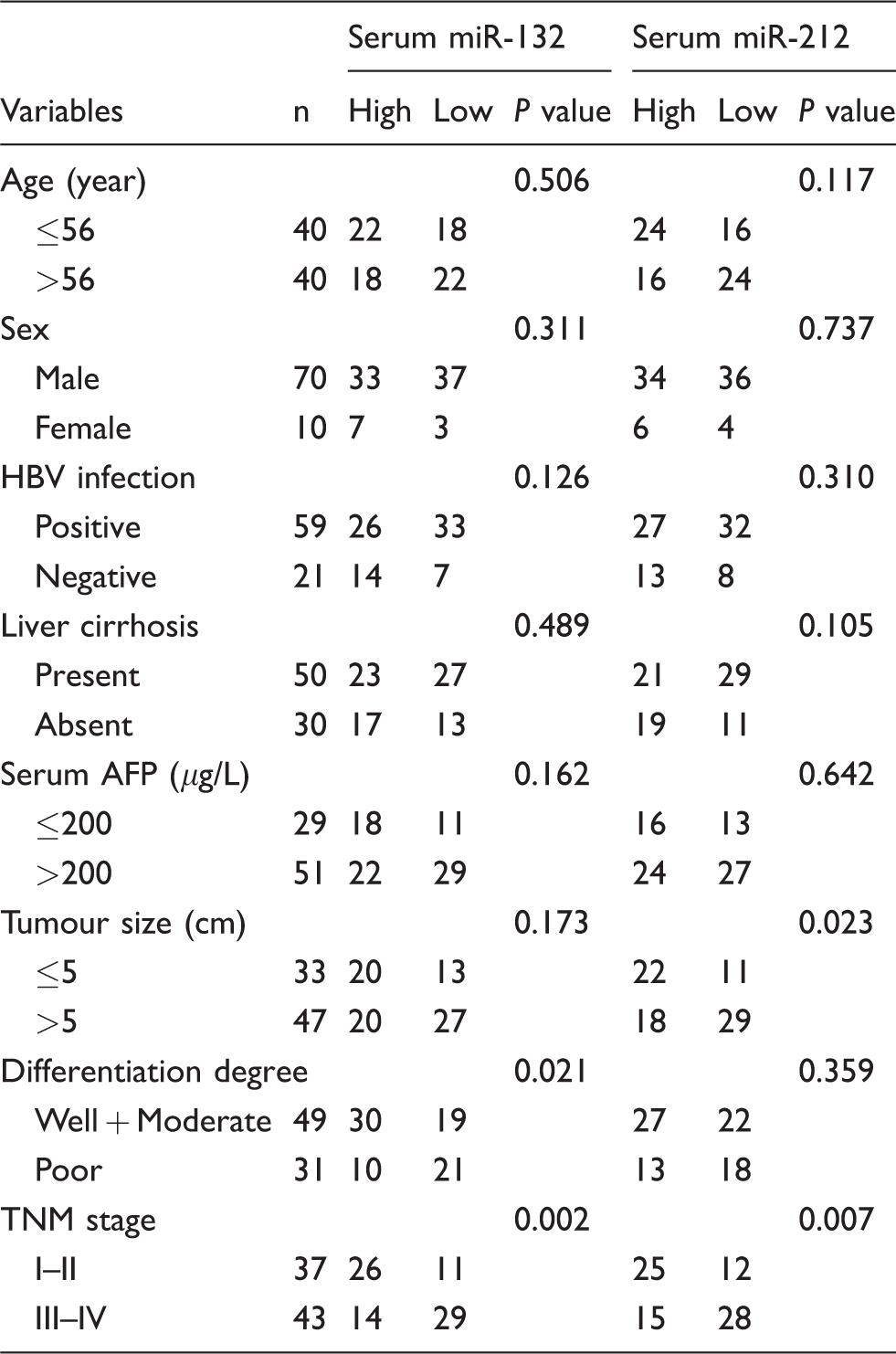
AFP: alpha-fetoprotein; HBV: hepatitis B virus; HCC: hepatocellular carcinoma; TNM: tumour–node–metastasis.
Correlation of serum miR-132/212 with overall survival of HCC patients
In order to investigate the prognostic value of the serum miR-132/212 cluster in HCC patients further, Kaplan–Meier survival analyses were conducted using post-treatment follow-up data. According to median concentrations of serum miR-132 and miR-212, HCC patients were classified into low-expression and high-expression groups. As shown in Figure 4(a) and (b), patients with lower concentrations of serum miR-132 and miR-212 had significantly poorer overall survival rates than those with higher concentrations of serum miR-132 and miR-212, p < 0.001 and p = 0.005, respectively, showing their potential as prognostic markers for HCC.
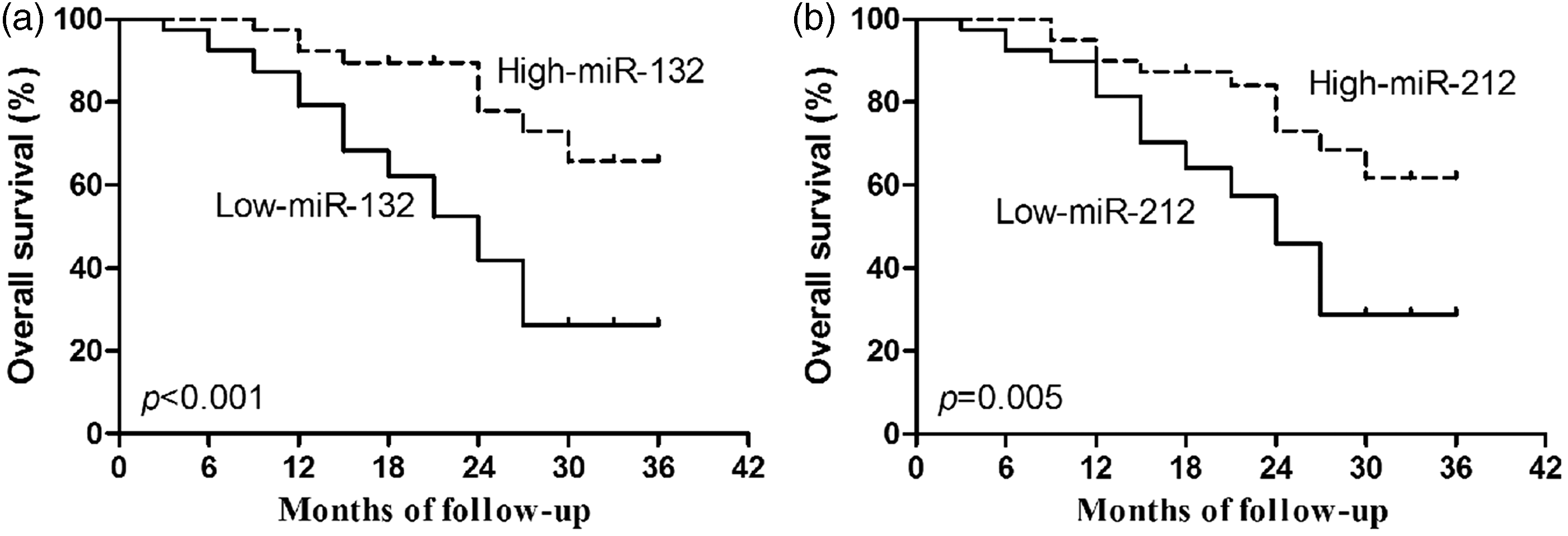
Association of serum miR-132/212 with overall survival (OS). OS rate of HCC patients was analysed by Kaplan–Meier curves and log-rank test according to the serum concentrations of miR-132 (a) and miR-212 (b).
Discussion
HCC is a complicated, multifactorial disease, resulting from the mutation of multiple susceptibility genes and the impact of various environmental factors. Diagnosis of HCC is often made at an advanced or terminal stage; early diagnosis is fundamental for optimal HCC management. Serum AFP has been used in the diagnosis of HCC over the past few decades. Its use underlines the paucity of good diagnostic markers, since its sensitivity and specificity are relatively unsatisfactory.17,18 Hence, there is a real need to identify novel HCC diagnostic biomarkers that are non-invasive, easily operable and highly accurate. Evidence is accumulating that miRNAs can be detected in blood, are stable, non-invasive and easily quantified in serum, and show potential as diagnostic or prognostic values. Moreover, these findings are consistent with their biological roles, which are mechanistically linked to tumour progression. Due to these characteristics of circulating miRNAs, they are promising biomarkers in many human malignancies, including HCC.19–21
MiR-132 and miR-212 are downregulated in HCC and cell lines and play important roles in hepatocarcinogenesis. Zhang et al. 12 found that miR-132 expression levels were significantly reduced in HCC tissues and associated with malignant progression of HCC. Further studies showed that miR-132 could impede cell growth and boost apoptosis in HCC cells via vascular endothelial growth factor, neurotrophin and mitogen-activated protein kinase signalling pathways. 22 Moreover, miR-132 can inhibit cell proliferation, invasion and migration of HCC by targeting phosphoinositide-3-kinase regulatory subunit 3 by inactivating the AKT/mTOR signalling pathway, showing that miR-132 might act as a tumour suppressor and serve as a potential target in the treatment of HCC. 23 Wei et al. reported that Hepatitis B virus x protein could induce DNA hypermethylation of the promoter of miR-132 gene, thereby epigenetically repressing miR-132 expression. This novel mechanistic insight into HBV-mediated hepatocarcinogenesis suggested that miR-132 might have a potential role as a marker and have therapeutic applications in HBV-related HCC. 24 Dou et al. 13 found that miR-212 was downregulated in HCC and could inhibit HCC cell viability and proliferation, inducing apoptosis by targeting FOXA1, thus exerting a tumour suppressive role. 25 Additionally, it has been found that miR-212 can negatively regulate expression of the histone H3 lysine 4 demethylase retinoblastoma binding protein 2 (RBP2) (overexpressed in HCC). 26 This novel pathway, involving miR-212–RBP2–cyclin-dependent kinase inhibitors, may be important in the pathogenesis of HCC.
Notwithstanding evidence that the miR-132/212 cluster may have important roles in the occurrence and progression of HCC, the expression pattern and clinical performance of serum miR-132/212 cluster in HCC remain to be established. In the current study, we have found that the expression of serum miR-132 and miR-212 is reduced in HCC patients compared with CLD patients and healthy controls. Moreover, our finding that serum miR-132 and serum miR-212 are strongly correlated in HCC patients, r = 0.603, p < 0.001, are in keeping with previous studies of the expression of miR-132 and miR-212 in HCC tissues,12,13,23,25 indicating that the genes of miR-132 and miR-212 are transcribed synchronously, with both serving as tumour suppressors in hepatocarcinogenesis. In addition, ROC analyses showed that the AUC of serum miR-132 was 0.747 (95% CI 0.675–0.809), the AUC of serum miR-212 was 0.706 (95% CI, 0.632–0.733), both p < 0.001, suggesting a potential diagnostic role in HCC. We also found that although the diagnostic value of serum miR-132 or miR-212 alone was not better than AFP, the combination of serum miR-132, miR-212 and AFP markedly improved sensitivity and negative predictive value, compared with AFP alone. Thus, the combination of all of three biomarkers may improve the diagnosis and differential diagnosis of HCC.
Our stratified analyses revealed that low concentrations of serum miR-132 were closely associated with tumour differentiation degree and TNM stage. Furthermore, low concentrations of serum miR-212 positively correlated with tumour size and TNM stage. It has been reported that low concentrations of miR-132 in HCC tissues correlate with HBV infection status, tumour differentiation degree, advanced TNM stage, lymph node metastasis and tumour infiltration.12,23 Low concentrations of miR-212 in HCC tissues correlate with high AFP, large tumour size, high Edmondson–Steiner grading and advanced TNM stage.13,25 Together, all of these data suggest that expression levels of miR-132 and miR-212 in serum samples and in tissues may help to predict the degree of malignancy and assess the progression of HCC. Additionally, the expression levels of miR-132 and miR-212 in HCC tissues correlate closely with the prognosis of HCC patients.12,13,25 In the current study, we have confirmed that lower concentrations of serum miR-132 and miR-212 significantly correlated with shorter overall survival of HCC patients, showing their potential prognostic value for HCC.
In conclusion, this study has for the first time described in detail the clinical utility of the serum miR-132/212 cluster in HCC patients. We have shown that downregulated serum miR-132/212 cluster may improve clinical diagnosis, prediction of progression and assessment of prognosis for HCC. The combination of serum miR-132, miR-212 and AFP improves the diagnostic efficiency for HCC. Our study is a preliminary evaluation of the clinical utility of the serum miR-132/212 cluster in HCC. Our sample size was relatively small, and all cases were recruited from a single institution. Other factors which may have introduced bias include compliance during treatment, lack of long-term follow-up and standardized detection of miRNA. Further studies are required to confirm the present findings, in particular to confirm the early diagnostic value of the serum miR-132/212 cluster in HCC and especially in the diagnosis of AFP-negative HCC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (81601842), Science and Technology Development Project of Nantong City (HS2014061, MS22016057), Jiangsu Provincial Medical Youth Talent Project (QNRC2016689, QNRC2016401).
Ethical approval
The study was approved by the Ethics Committee of the Affiliated Hospital of Nantong University, China (Protocol number 2012–004).
Guarantor
LC.
Contributorship
LC and WC contributed to conceive and design of the study and to the interpretation of the data. FW, JW and LJ performed the experimental work, data analysis and wrote the manuscript. All authors approved the final version of the manuscript.
