Abstract
Background
The contribution of tear protein expression in patients with presumed diagnosis of Sjögren syndrome is underestimated. We aimed to evaluate the role of tear proteins in the Sjögren syndrome early diagnosis.
Methods
Charts from 110 patients suspected of Sjögren syndrome were analysed and the subsequent diagnosis retrieved. Subjective symptoms (ocular surface disease index, OSDI), tear film break-up time (TFBUT), Schirmer test, Jones test, tear clearance (TC), corneal (NEI score) and conjunctival staining (van Bjerstelveldt score), esthesiometry, cytology, tear protein analysis (total protein [TP] content, lysozyme-C [LYS-C], lactoferrin [LACTO], lipocalin-1 [LIPOC-1] and albumin [ALB]) were analysed. The diagnostic performance with area under the curve (AUC) and odds ratio (OR) for each parameter were calculated.
Results
Thirty-five patients (31.8%) had been diagnosed as affected by Sjögren syndrome. Clinical tests showed lower diagnostic performance (OSDI > 44 [AUC 0.57], Schirmer ≤ 5 mm [0.59], TFBUT ≤ 3 s [0.72], TC > 1/16 [0.68], Jones ≤ 4 mm [0.68], corneal staining > 2 [0.51], conjunctival staining > 2 [0.78]) compared with tear proteins (LYS-C ≤ 1.5 mg/mL [0.79], LACTO ≤ 20% [0.94], LIPOC-1 ≤ 10% [0.89], ALB ≥ 15% [0.79]). LYS-C, LACTO, LIPOC-1 and ALB showed a significant association in predicting Sjögren syndrome vs. not-Sjögren syndrome dry eye (OR, respectively, 4.9, 5.5, 7.2, 6.7).
Conclusions
Tear proteins’ concentrations showed a significant higher accuracy compared with the traditional ocular clinical tests for reaching Sjögren syndrome’s diagnosis. In particular, LACTO and LIPOC-1 provided an excellent diagnostic performance and thus could likely be considered promising biomarkers of Sjögren syndrome.
Introduction
Sjögren’s syndrome (SS) is a multisystem autoimmune disease characterized by T-cell infiltration and B-cell hyperactivity in lachrymal and salivary glands, determining fibrosis and progressive destruction of these tissues. It is considered one of the most prevalent autoimmune diseases, with a female to male ratio varying from as high as 20:1 to 9:1. 1 The involvement of lachrymal and salivary glands results in the typical features of dry eye (DE) and xerostomia, the symptoms are often highly variable in presentation and can progress slowly, making timely diagnosis a challenging issue. 2 It is estimated that the disease remains undiagnosed in more than half of affected adults, and that patients experience symptoms for an average of more than three years before the diagnosis. 3 A delayed diagnosis compromises early treatment, leading to potentially serious consequences for patient’s quality of life, with the increase of socioeconomic burden and of life-threatening sequelae. The clinical work-up typically involves a variety of tests, including tear and salivary function tests, serological autoantibody biomarkers, salivary gland biopsy and systemic endocrine findings. However, some of these criteria, such as salivary scintigraphy or salivary gland biopsy, are cumbersome and not always performed in the routine practice, due to high invasiveness and low patients’ compliance.
The search for biomarkers is a convenient and non-invasive tool for the diagnosis of SS. Traditional serum biomarkers include SS-A/Ro, SS-B/La, antinuclear antibody (ANA) and rheumatoid factor (RF). Although being considered important for the diagnosis of SS, they are not always positive in patients with SS, especially in early cases. In addition, SS-A/Ro and SS-B/La are positive only in half of patients with SS who refer DE symptoms. 4
Tear proteomic is a potential tool for the non-invasive diagnostic detection of DE syndrome, and multimarker models with high specificity and sensitivity in the DE diagnosis were established.5–8 Although DE is a typical diagnostic feature of SS, it is a very common entity with a wide range of underlying pathologies, thereby making it quite non-specific for SS by itself.9–11
Tear protein alterations have previously been detected in patients suffering from SS-related DE, but only after that SS diagnosis was already reached. The aim of this study was to characterize the different patterns of tear protein expression in patients with initial symptoms of DE and still suspected of SS (but not yet diagnosed) and to estimate the accuracy of these parameters in the subsequent diagnosis of SS.
Materials and methods
This is a retrospective analysis of prospective collected data retrieved from clinical charts of 110 patients (107 females and 3 males) suspected of SS and referred to the Ophthalmology Unit (S.Orsola-Malpighi Teaching Hospital, Bologna, Italy) for the recent onset of ocular symptoms of DE over the period September 2009–April 2014. The study was conducted according to the Declaration of Helsinki. When returning for subsequent check-ups, all patients were informed on the purpose of the research and signed the consent.
The protocol established in the current practice of our rheumatology and ophthalmology units for the management of patients suspected of SS is hereby briefly explained.
First rheumatologic access
At this time, patients had a face-to-face interview with a single expert Rheumatologist using a standardized form that included question about demographic data and medication used. Subjective ocular and oral symptoms had been evaluated according to the American-European Consensus Group (AECG) sicca questionnaire, 12 with the following questions: (1) Have you had daily, persistent, troublesome DEs for more than three months? (2) Do you have a recurrent sensation of sand or gravel in the eyes? (3) Do you use tear substitutes more than three times a day? (4) Have you had a daily feeling of dry mouth for more than three months? (5) Do you frequently drink liquids to aid in swallowing dry food? (6) Have you had recurrent or persistent swollen salivary glands as an adult?
Patients who had given affirmative response to at least one of the above-listed screening questions underwent further examinations, including ocular surface and tear protein analyses, blood sample analysis, salivary scintigraphy, as below detailed.
Ocular surface analysis
Subjective discomfort symptoms were graded on the basis of the DE discomfort symptoms questionnaire (ocular surface disease index, OSDI) score which ranges from 0–12 (no disability), to 13–22 (light DE), to 23–32 (moderate DE), to 33–100 (severe DE). 13 Schirmer test was carried out as described in DEWS methodology 14 by using sterile Schirmer strips (ContaCare Ophthalmic and Diagnostics, Gujarat, India), placed at the outer lateral inferior canthus with open eyes without (Schirmer test I, performed twice) or with (Jones test) anaesthesia (oxybuprocain 0.5%). Pathological value was regarded as ≤5 mm/wetting after 5 min for Schirmer I and ≤ 3 mm/wetting after 1 min for Jones test. The tear film break-up time (TFBUT) was measured and recorded (average of three measurements) after instillation of 2 μL unpreserved 2% sodium fluorescein (galenic preparation from Fluoralfa 0.25%, Alfa Intes, Italy). A TFBUT higher than 10 s was considered as a normal value. Corneal and conjunctival surface damage were assessed, respectively, by instillation of 2% sodium fluorescein and lissamine green. Fluorescein staining details were enhanced by the aid of a 7503 Boston yellow filter (equivalent to Kodak Wratten 12) and graded according to the National Eye Institute (NEI) score 15 and van Bjesterveldt score, 16 respectively. Corneal sensitivity was measured using a Cochet-Bonnet esthesiometer (Luneau, Chartres, France) and was recorded in millimetres nylon filament length. A measurement < 50 mm was considered low corneal sensitivity. 17 Tear clearance rate was carried out as prevoiusly described, 18 graded onto the colour of the fluorescein dye fading compared with a control staining scale dilution (pathological values ≤1/8 dilution). For the statistical analysis, the tear clearance rate results were converted into numerical values: dilution 1:2 = 5; 1:4 = 4; 1:8 = 3; 1:16 = 2; 1:32 = 1; 1:64 = 0. Scrapings were performed in both eyes at the lower and upper tarsal conjunctiva to evaluate inflammation, and smears were read following the Scraping Cytology Score System described elsewhere, with pathological values >4 out of 12. 19 Imprint cytology was used to evaluate conjunctival epithelial metaplasia and goblet cell density in the nasal bulbar area. Epithelial metaplasia was scored grading from 0 to 5, as described by Tseng, with values >1 being considered pathological. 20
Tear protein analysis
A minimum amount of 5 μL of tears was collected with a laboratory micropipette (Pipetman P, Gilson Int.l B.V., Den Haag, Netherlands) using sterile disposable tips. Chip-based analysis was performed with the Agilent 2100 Bioanalyzer system (Agilent, Waldbronn, Germany) as validated and extensively described in other papers from our group.7,21 This proteomic technology works on just 2 μL sample, with the advantage to obtain data individually and not on pooled samples, an option mostly applied in the proteomic current practice. 22 In addition, this ‘lab-on-a-chip’ device offers the advantage to quantitatively estimate several protein species with diagnostic potential without dividing the original patient sample and when the amount of tear available is limited, as it often occurs in DE patients. 21 The system consists of a miniaturized capillary gel electrophoresis device integrating all steps of a ‘conventional’ electrophoretic run, from sample preparation, labelling, gel loading, separation up to detection by a laser-induced fluorescence detector. The Agilent Protein P230 Kit was utilized for tear protein detection, the analytical specification can be downloaded at (https://www.agilent.com/cs/library/usermanuals/Public /_G2938–90054_Protein 230 Assay_KG.pdf). The lower limit of detection has been estimated as 30 ng/μL BSA in 0.5 M NaCl. Two representative electropherograms are shown in Figure 1.
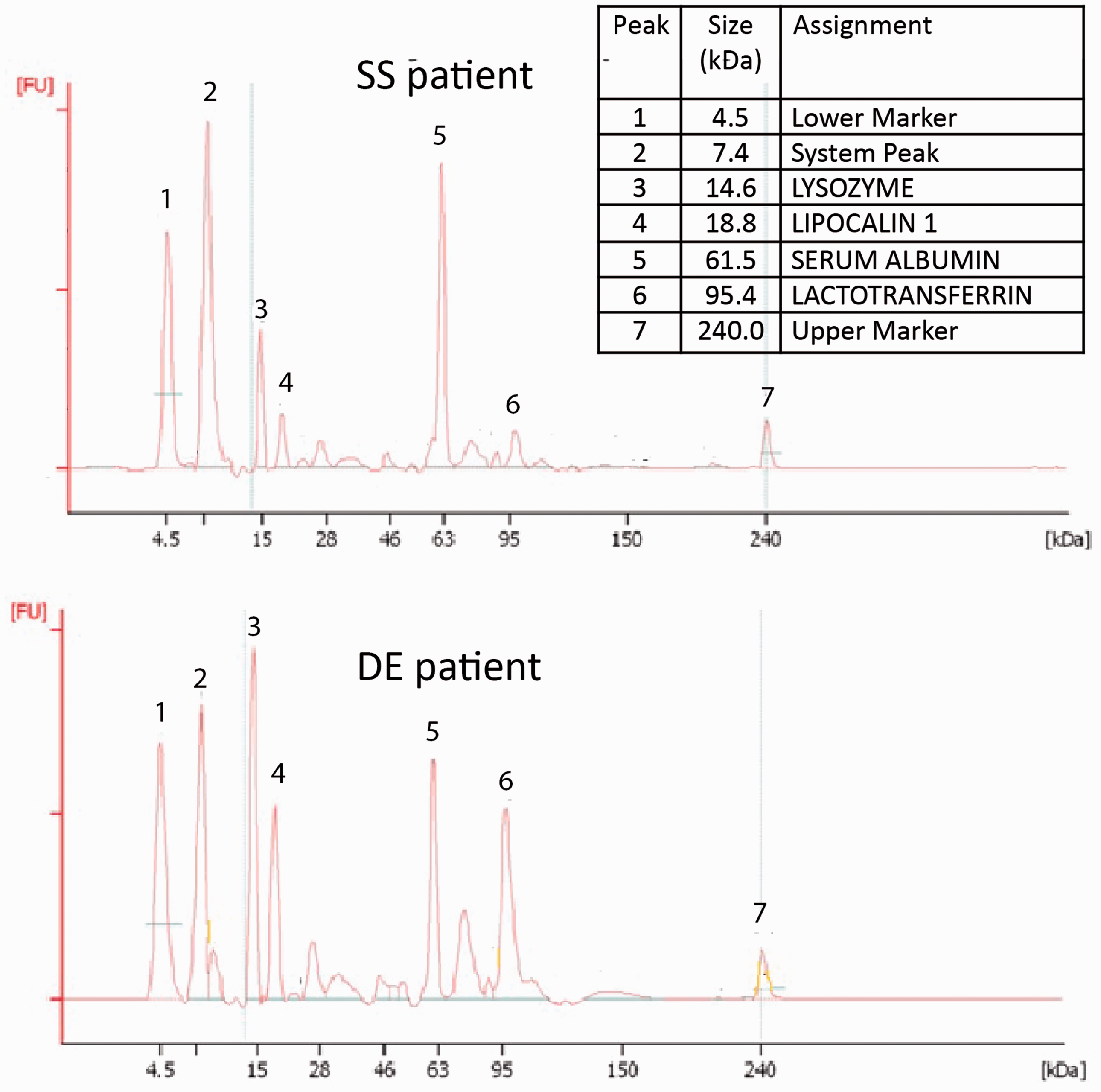
Representative electropherograms of tear specimens from a dry eye (DE) patient and from a Sjogren's syndrome (SS) patient. Figures were selected from those showing more clearly the differential expression among the tear proteins of interest.
Total protein (TP) content and peaks for lysozyme C (LYS-C), lipocalin-1 (LIPOC-1), lactoferrin (LACTO) and exudated serum albumin (ALB) were recognized at given molecular weights. Proteins were expressed either as % vs. TP content and as mg/mL tear sample.
Blood analysis
Blood sample was drawn for γGlobulins concentrations and autoantibody testing, including anti-Ro/SSA and anti-La/SSB (named hereinafter as ENA– extractable nuclear antigen) determined by enzyme-linked immunosorbent assay (ELISA), ANA (antinuclear Antibodies) by indirect immune-fluorescence using Hep-2 cells, and RF determined by ELISA. The normal or abnormal value for each test was determined by the local laboratory standard reference value.
Salivary scintigraphy
Standardized stimulated dynamic salivary gland scintigraphy tests were performed in all patients 60 min after the intravenous injection of 370 MBq of technetium-99m-pertechnetate and after per os stimulation with a 0.5 g tablet of ascorbic acid administered 40 min after the injection. The presence of chronic sialoadenitis was determined as the progressive decrease of uptake of radioactive tracer, and a semi-quantitative score grading (mild, moderate, severe) was performed.
Lip biopsy
Lip biopsy (LB) was proposed in all patients but only 19% of patients gave their consent to be performed. In this case, LB was performed after local anaesthetic infiltration to harvest 4–10 glands, 23 processed and evaluated by a pathologist expert in the diagnosis of focal lymphocytic sialoadenitis, who was blinded to the patients’ demographic, clinical or serological characteristics. The specimens were considered as positive if they showed lymphocytic foci ≥ 50 cells in a glandular area of at least 4 mm2. The focus score was determined by using the Chisholm and Mason grading. 24
Patients were diagnosed as suffering from DE according to the DEWS Criteria. 25 Diagnosis of primary SS was made by an expert Rheumatologist according to the AECG Criteria. 12
Statistical analysis
Data from both eyes were collected from charts, but only the value from the worst eye was taken into consideration for statistical purposes. Statistical analysis was performed with computer software (IBM SPSS Statistics for Windows, Version 20.0; Armonk, NY, USA) and MedCalc 5.0 (MedCalc Inc., Ostend, Belgium). All data were expressed as mean ± SD and median (min value; max value) (95% confidence interval for the median).
Differences between groups were evaluated by Mann-Whitney test for independent samples. Each parameter was analysed for sensitivity, specificity, area under the receiver-operating characteristics (ROC) curves (AUC, ranging 0–1), likelihood ratio, calculated comparing SS vs. not-SS DE patients. The prevalence of the SS-I was calculated having as a reference the population included in the present study. The relationship between AUC and the diagnostic accuracy was estimated as follows: 0.9–1.0 excellent, 0.8–0.9 very good, 0.7–0.8 good, 0.6–0.7 sufficient, 0.5–0.6 poor and < 0.5 test not useful.
Chi-square and multivariate logistic forward regression analysis were used to assess the association between demographic, ocular and blood-related variables and SS vs. not-SS DE diagnosis. Risk for primary Sjogren's syndrome (pSS) diagnosis was also estimated by odds ratio (OR) with 95% confidence intervals that independently predicted the disease. Data were considered to be statistically significant if P < 0.05.
Results
The analysis of data retrieved from clinical charts was conducted on April 2017, allowing at least a 36-month window from the latest patient included in the study and the present analysis. This time interval can be considered similar to the length of the median time necessary to reach SS diagnosis in daily clinical practice. Thirty-five out of 110 patients (31.8% of the total) had been diagnosed as primary SS. Median interval between symptoms onset and SS diagnosis was 19 months (range: 6–80 months). The remaining 75 patients were diagnosed as DE not-SS related. Patients’ characteristics are summarized in Table 1.
Demographic characteristics of the population under study, divided into the two groups of patients diagnosed as SS or as not-SS related DE sufferers.
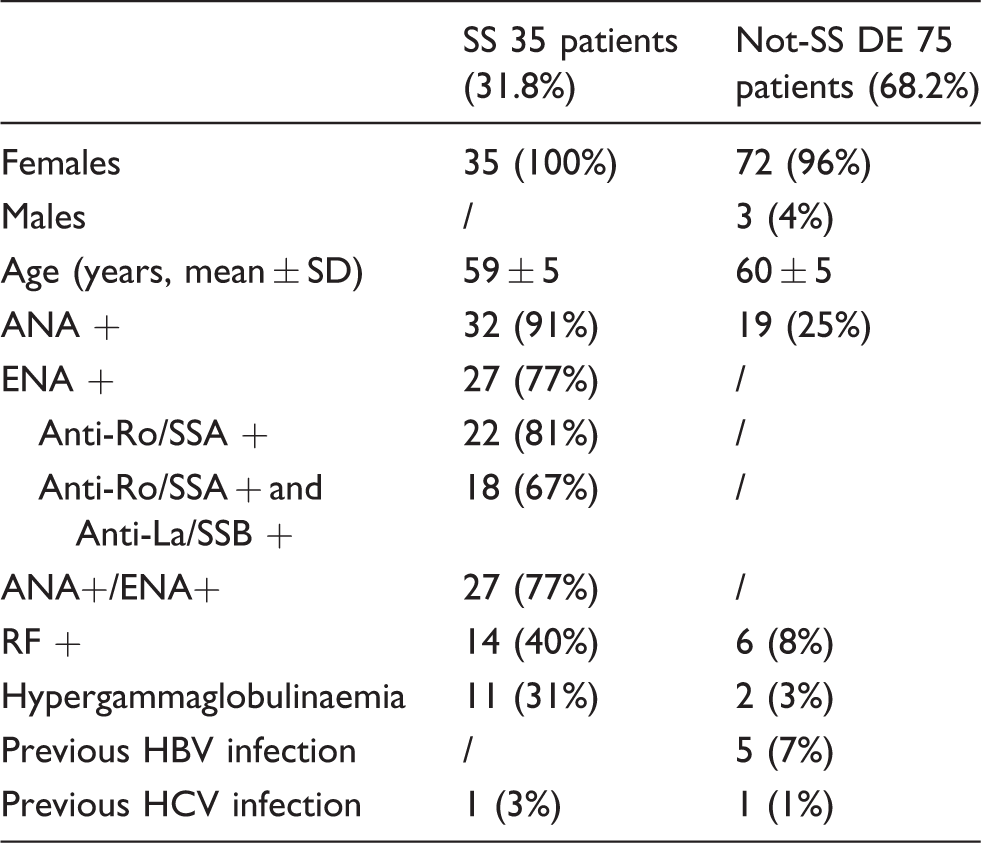
SS: Sjogren’s syndrome; DE: dry eye; ANA: antinuclear antibodies; ENA: extractable nuclear antigen; RF: rheumatoid factor; HCV: hepatitis C virus; HBV: hepatitis B virus.
Laboratory data from both SS and DE patients are summarized in Table 2, with the different parameters grouped by blood, ocular surface, cytology and tears. Patients diagnosed as SS showed significant lower values for TFBUT, Jones test and tear clearance compared with not-SS DE patients, whereas conjunctival staining score was significantly higher in SS group compared with not-SS DE group. Values from OSDI score, Schirmer test, corneal staining score, corneal esthesiometry and scraping/imprint cytology did not change significantly between the two groups (P > 0.05).
Laboratory data from both SS and DE patients are summarized, with the different parameters grouped by blood, ocular surface, cytology and tears.
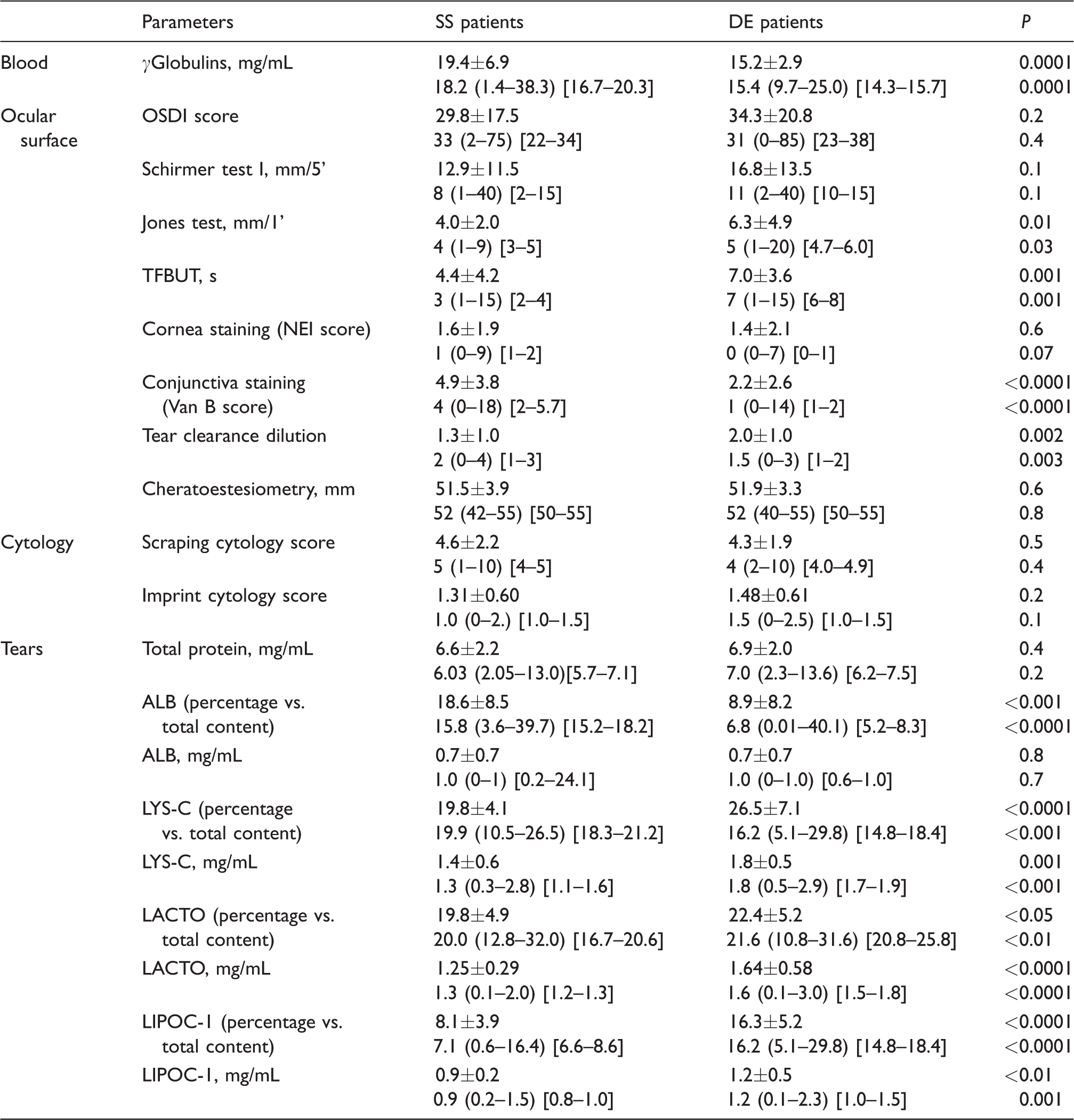
Note: Values are expressed as mean ±SD (first line) and median (minimum value−maximum value) (95% CI) (second line).
TFBUT: tear film break-up time; NEI: National Eye Institute; OSDI: ocular surface disease index; LYS_C: lysozyme-C; LACTO: lactoferrin; LIPO-1: lipocalin-1; ALB: albumin; SS: Sjögren’s syndrome; DE: dry eye.
All tear samples were analysed, and the protein bands of interest were always visible total TP amount did not significantly differ between groups, whereas the tear protein profiles were different between the SS and the not-SS DE group. In particular, significant lower content was demonstrated for LYS-C, LACTO and LIPOC-1 in the SS group compared with the non-SS DE either as the % vs. TP content or as mg/mL (P ranging from <0.01 to <0.0001). On the contrary, SS patients showed higher values of exudated ALB compared with not-SS DE patients as percentage vs. total content (Table 2). The statistically significant difference between groups found in blood parameters is biased from obvious inclusion criteria.
The performance of parameters for SS vs. not-SS DE diagnosis is summarized in Table 3. Among blood parameters, only ANA and ENA showed a very good diagnostic accuracy, but the result suffers again from an inclusion bias. Both imprint and scraping cytology scores were shown to be poor in discriminating between the two conditions under comparison (AUC, respectively, 0.58 and 0.54). The diagnostic accuracy of corneal esthesiometry, subjective symptom score, corneal staining and the Schirmer test (even at a threshold 5 mm/5’) was also poor (AUC, respectively, 0.51, 0.57, 0.51, 0.59). A sufficient diagnostic accuracy was shown for the Jones test and tear clearance (AUC 0.68 for both), and only conjunctival vital staining along with TFBUT (at a very low cut-off of 3 s) exhibited good characteristics as discriminative tests (AUC, respectively, 0.78 and 0.72). Despite the TP concentration failed to be a useful discriminative test, a higher diagnostic accuracy was found for single tear proteins. In particular, LACTO and LIPOC-1 showed an excellent to a very good diagnostic performance (AUC, respectively, 0.94 and 0.89), and a good diagnostic accuracy was shown for LYS-C and ALB (AUC 0.79 for both).
Performance of parameters for SS vs. not-SS DE diagnosis is summarized, with the different parameters grouped by blood analytes (including the scintigraphic score, as this is performed by rheumatologists), ocular surface cytology, ocular surface parameters and tear proteomics.
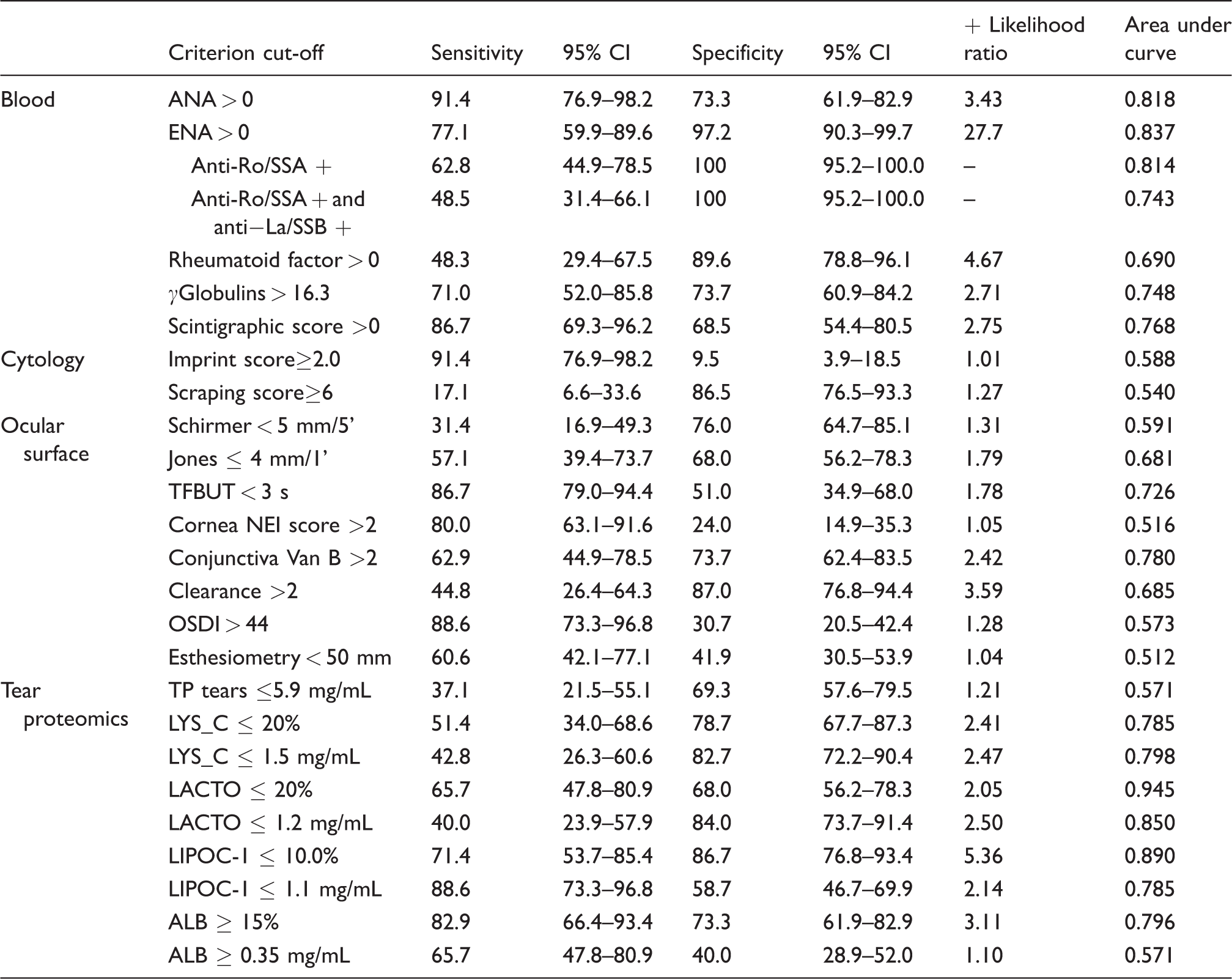
ANA: antinuclear antibody; ENA: extractable nuclear antigen; TFBUT: tear film break-up time; NEI: National Eye Institute; OSDI: ocular surface disease index; TP: total proteins; LYS_C: lysozyme-C; LACTO: lactoferrin; LIPOC-1: lipocalin-1; ALB: albumin.
Table 4 summarizes data from univariate and multivariate logistic regression, showing variables with positive association in predicting SS vs. not-SS DE. Blood autoantibodies ANA and ENA along with scintigraphic score were significantly associated to SS diagnosis in both types of models, again as the result of inclusion bias. No association was found for cytologic parameters, and among ocular surface variables only the Jones test, conjunctival vital staining and tear clearance exhibited positive OR but in the univariate only (OR, respectively, 2.5, 3.1 and 4.9). All tear proteins at the given thresholds showed a significant positive association with SS diagnosis in the univariate model, and only LACTO, LIPOC-1 and ALB also in the multivariate analysis (OR, respectively, 5.5, 7.2 and 6.7).
The different variables are grouped by blood analytes (including the scintigraphic score, as this is performed by Rheumatologists), cytology, ocular surface and tear proteomics.
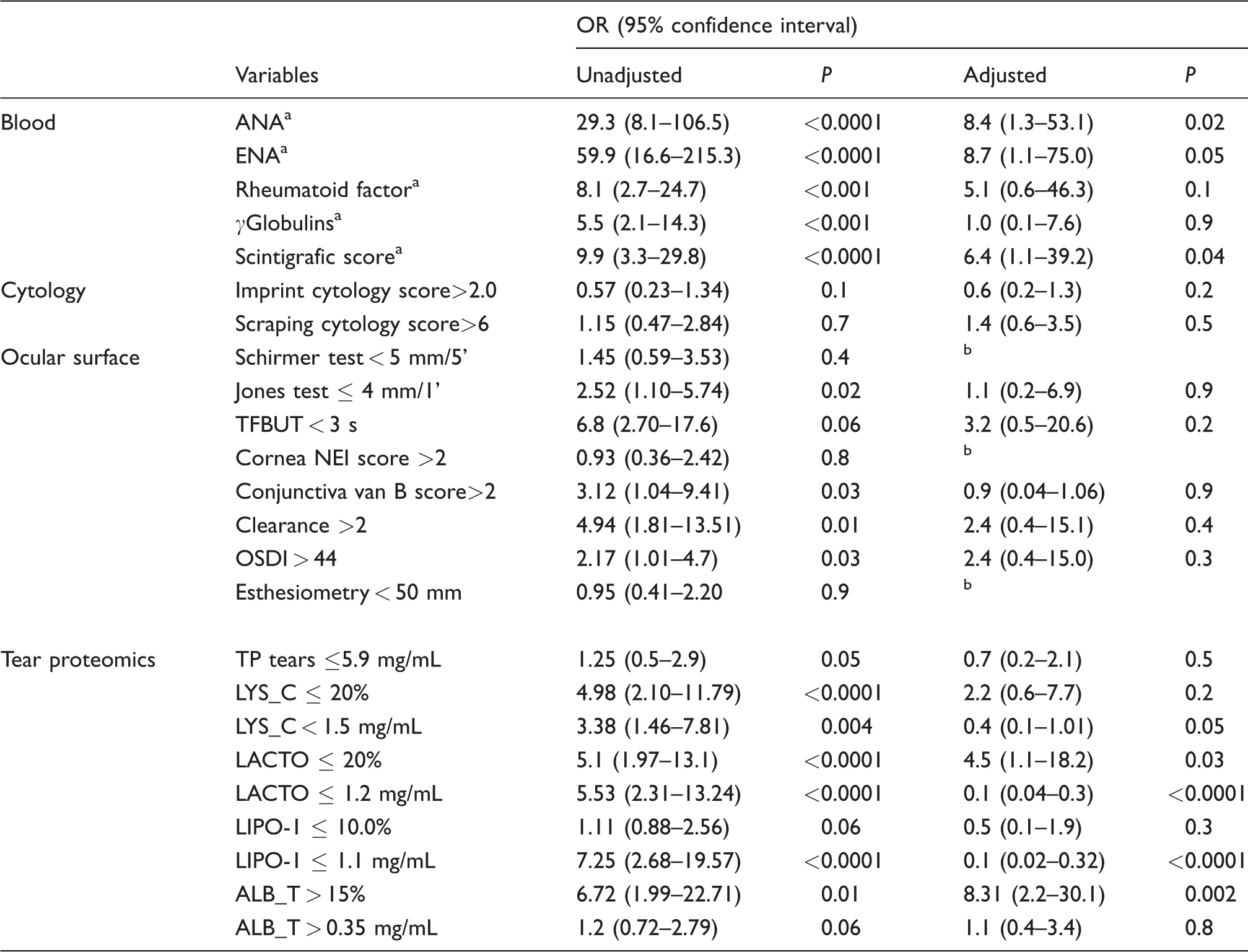
Unadjusted and adjusted (univariate and multivariate logistic regression, adjusting for the other factors shown in the table) odds ratios – OR – and 95% confidence intervals of risk factors in parentheses.
ANA: antinuclear antibody; ENA: extractable nuclear antigen; TFBUT: tear film break-up time; NEI: National Eye Institute; OSDI: ocular surface disease index; TP: total protein; LYS_C: lysozyme-C; LACTO: lactoferrin; LIPO-1: lipocalin-1; ALB: albumin.
aFactors which showed a positive association in predicting SS vs. not-SS DE, in the univariate analysis. The multivariate analysis was run for each group of variables, and included in the model only those variables resulted to be significant in the univariate analysis.
bVariables not included in the multivariate analysis.
Discussion
Data from this study highlight the unique nature of tear chemistry of SS-related DE already in the early stages of the disease, when ocular symptoms arose, and that tear LACTO and LIPOC-1 appear strongly predictive for SS diagnosis. To the best of our knowledge, this work is the first attempt to predict the chance to develop SS already at the time of initial onset of ocular symptoms, by analysing ocular surface parameters and tear proteins at this specific time. In fact, all the previous studies characterized and distinguished SS-related from not SS-related DE when the diagnosis of SS had already been reached, and therefore did not allow any predictive analysis.
Early symptoms of SS, such as DE and dry mouth, are often present in patients without SS, and patients complaining of ocular discomfort symptoms are often given a generic diagnosis of ‘idiopathic DE’. Although DE is the typical diagnostic feature of SS, it is one of the most commonly encountered conditions in the ophthalmic practice with a wide range of underlying diseases, thereby making it quite non-specific for SS by itself.25,26
Different criteria have been proposed, with the aim of improving classification performance. All these systems (namely from the oldest to the most recent, AECG, 12 American College of Rheumatology [ACR] 27 and ACR/European League Against Rheumatism classification criteria 28 ) include reduced tear production and ocular surface staining as main ocular criteria for the diagnosis of SS.
Human body fluid other than blood (e.g. tears or saliva) analysis appears attractive as they provide several key advantages compared with traditional work-up, including low invasiveness, minimum cost and easy sample collection and processing. 29 The ability to probe the protein content of human tear fluid has enormous potential for deepening the knowledge of the pathology of ocular and systemic diseases such as SS and enabling novel non-invasive tear-based diagnostic tools. The healthy lachrymal gland secretes a complex fluid that contains proteins, nutrients, hormones, growth factors and immunoglobulins in an isotonic electrolyte solution. Lachrymal gland function impairment in SS is, in part, caused by lymphocytic infiltration. It was believed in the past that this change was the only cause of reduced secretion; it is now clearer that multiple mechanisms, including neurological influences, participate in SS acinar cell dysfunction. 30 DE syndrome in SS patients is associated with an altered proteomic profile with dysregulated expression of proteins involved in a variety of cellular processes: in particular, proteins involved in antibacterial action were found to be down-regulated, those involved in the inflammation, immunity and oxidative stress were up-regulated, and in addition, proteins uniquely present in the tear fluid of SS patients with DE were identified. 31 Other studies differentiated SS from DE and controls on the basis of changes in tear film-specific protein expression,5,32–35 leading to the suggestion that biomarker profiling may significantly contribute to patient characterization.8,36
Both clinical tests and tear proteome analysis showed different trends in the two populations (SS and not-SS DE), at least for some of the analysed parameters in the present study. In particular, SS patients exhibited lower TFBUT values as compared with DE patients, in agreement with some authors31,37 but not with others,38,39 while Schirmer test was not significantly different in the two groups in agreement with our previous work 40 but in disagreement with others.31,37,38,41 A more general agreement was reached for ocular surface staining, Jones test and clearance test, that were found to be worse in the SS group as compared with the not-SS DE, in agreement with others.38–43 Similarly, a significant lower content was demonstrated for LYS-C, LACTO and LIPOC-1 in SS group compared with not-SS DE, while on the contrary, SS patients showed higher values of exudated ALB compared with not-SS DE patients, in agreement with previous studies.36,40,44,45
It must be noticed that it is difficult to compare the results obtained in the present study with those available in the literature as they have been performed in different stages of SS natural course, and this could explain the discrepancy of our data with those available in the literature. We analysed patients soon after the initial onset of ocular symptoms when the suspicion of SS had just arisen, while the other studies analysed ocular parameters later, when the diagnosis of SS had already been reached. In addition, the proteomic technology we had utilized overcomes the challenge of too little tear sample, which generally leads to the pooling of samples from several patients, a practical approach that, however, lacks to provide association with individual clinical history and biological variability. 46
The reduction of LACTO and LIPOC-1 content showed an excellent diagnostic performance to distinguish patients affected by SS from patients with ‘idiopathic’ DE, comparable with that from autoantibody profile, and also LYS-C and ALB showed a good diagnostic accuracy.
It is remarkable in this context that the tear proteins LACTO along with LYS-C are co-localized with LIPOC-1 in the lachrymal gland secretory granules, and it is believed that they are most likely secreted together, suggesting a role as indicators for the lachrymal gland function. 47 LACTO and LYS-C are multifunction chain polypeptides with properties such as anti-inflammatory, bacteriostatic and antioxidant secreted by the acini of the main lachrymal gland. LIPOC-1 is the main lipid carrier in human tears and is crucial in the ocular surface protection. They could either have similar functions or influence the activity of each other.44,45,47 ALB can be considered as an indirect sign of subclinical inflammation as it occurs when there is an increase of protein leakage from inflamed conjunctival vessels.18,48
The diagnostic accuracy of all ocular clinical tests including Schirmer test was poor, except for Jones, clearance, conjunctival vital staining along with TFBUT (at a very low cut-off of 3 s) that showed a sufficient diagnostic performance. Surprisingly, none of the ocular parameters showing high diagnostic accuracy in this study are considered in the current diagnostic criteria, at the expense of Schirmer test that on the contrary showed poor accuracy in this study. This finding could explain, at least in part, the often delayed and challenging diagnosis of SS in daily clinical practice.
Our analysis of retrieved data from charts was conducted after at least 36 months from the latest patient included: this window period allowed to follow the natural history of the disease, given the – often long – time interval from symptoms onset and SS diagnosis. However, it might be possible that some patients included in the not-SS DE group because of the lack of sufficient criteria to reach the diagnosis at the time of this analysis could likely develop SS later, and this represents the major limitation of the study.
In conclusion, data from this study show that tear LACTO and LIPOC-1 could likely be considered promising biomarkers, providing high accuracy in reaching SS diagnosis, in the hope to reduce or eliminate the need for more invasive diagnostic procedures. Future efforts should further explore and support the usefulness of incorporating tear proteomic analysis both in the work-up and in the diagnostic criteria of SS.
Footnotes
Acknowledgement
We gratefully acknowledge the Epidemiology and Biostatistics Unit, Research and Innovation Department Sant'Orsola-Malpighi Hospital and University of Bologna for the support in the statistical analyses of data.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The ethics committee of S.Orsola-Malpighi Hospital approved this study (REC number: Reuma_Ofta_2016).
Guarantor
NM.
Contributorship
PV, NM, ECC – substantial contributions to the conception and design of the work; interpretation of data for the work – revising critically the work for important intellectual content.
GG, GV, RM – acquisition, analysis and interpretation of data for the work.
PV – correspondent author, final approval of the version to be published.
