Abstract
Background
Several approaches exist to screen neonates for congenital cytomegalovirus infection. We here describe a new method using cellulose pads for urine collection and its evaluation in an experimental and a clinical setting.
Methods
We systematically tested the effect of storage duration of the pads after exposure to cytomegalovirus-positive urine, meconium contamination and specimen handling on the cytomegalovirus load and the detection rate. Further, the method was tested in clinical practice in a cohort of 500 neonates.
Results
Following exposure of urine pads with cytomegalovirus-positive urine, the viral load decreased after 15 min, 12 h, 24 h, and 7 days to 63.2%, 42.1%, 31.6%, and 9.3% of the baseline value. Cytomegalovirus detection rate after seven days was 100%. Contamination with meconium resulted in a comparable reduction of the viral load. The detection rate for dried urine pads after seven days was 93.3%. In clinical practice, urine collection from pads was successful in 73.6% by the first attempt and in 26.4% by the second attempt.
Conclusions
Urine collection using cellulose pads seems feasible regardless of a reduction of the cytomegalovirus load due to exposure to the pad itself or to meconium. Drying of the exposed urine pad should be avoided.
Introduction
Congenital cytomegalovirus (CMV) infection is the leading non-genetic cause of sensorineural hearing loss (SNHL) in the paediatric population.1–3 The incidence of congenital CMV infection varies in different countries and is proportional to the CMV seroprevalence of the women at childbearing age. Multiple studies investigating the incidence of congenital CMV have been conducted and reported an incidence of 0.2% to 2.2%.4–6 With a presumed incidence of 0.5 to 1.0%, congenital CMV infection affects approximately 25,000 to 50,000 neonates annually in the European Union, which underlines a large socioeconomic impact.
Approximately 10% to 15% of the infected neonates are born with symptomatic CMV infection (e.g. petechia, microcephaly, small for gestational age, hepatomegaly, thrombocytopenia). 7 The remaining 85% to 90% can be considered as asymptomatically infected.8,9 The morbidity in these infants is lower than in symptomatically infected infants, but still 10% develop complications in early childhood. In congenitally CMV-infected infants, SNHL can become significant with a delay of months or years, and therefore, these infants may not be recognized by newborn hearing screenings.10–12
The high socioeconomic impact of the disease may legitimate a nation-wide screening programme for congenital CMV infection. 1 Several approaches have been investigated for CMV screening in newborns, using urine, saliva and dried blood spots (DBS).4,13–17 Realtime polymerase chain reaction (PCR) assays of liquid and dried saliva detect CMV DNA with a high sensitivity and specificity, but bear the risk of false-positive results due to contamination of saliva with CMV-positive breast milk.14,18
Urine is the most commonly used specimen to establish the diagnosis of congenital CMV and has been also used for screening purposes. However, urine collection in neonates and infants can be time consuming and sometimes frustrating, especially in female infants. Paixao et al.19,20 have described the technique of pooling urine samples for CMV screening in an attempt to reduce labour and costs. Other authors used filter discs to collect urine with promising results. 17
The aim of this study was to evaluate a new method of CMV detection using a cellulose pad for urine collection. In the first part of the study, we investigated the influence of storage duration and meconium contamination on CMV detection from exposed urine pads. The second part of the study was designed to test the method in a cohort of 500 infants.
Materials and methods
The Bonn University Hospital Institutional Review Board approved the study. Written informed consent was obtained from parents or legal representatives before enrolment in the study. The study was performed in accordance with the ethical standards described in the Declaration of Helsinki.
Realtime PCR
CMV DNA amplification and quantification were performed using the RealStar® CMV PCR Kit 1.0 (Altona Diagnostics, Hamburg, Germany) according to the instructions of the manufacturer using a LightCycler® 480 II instrument (Roche Diagnostics, Mannheim, Germany). The lower limit for detection (LOD95) of the assay was 91 IU/mL (probit analysis), and the lower limit of quantification was set at 1000 IU/mL. Nucleic acid preparation was performed by using the NucliSENS® easyMag® instrument and chemistry (bioMeriéux, Nürtingen, Germany; urine pools, elution volume 100
Preclinical experiments
During an initial validation phase, we identified three major problems that we then evaluated by an experimental approach: first, the virus detection rate could be reduced due to a delay in processing the urine pads. Second, urine pads may be stained with meconium. Furthermore, we observed that wet urine pads had to be stored in plastic bags to avoid drying. Thus, third, we tested the possibility of CMV detection from dried urine-exposed pads. All urine used in the preclinical experiments was collected from two siblings with symptomatic breast milk-associated CMV infection. In both infants, the same virus genotype (gB2) was detected. Urine was longitudinally collected (over a period of three weeks), which might explain the broad distribution of the CMV DNA concentration. Cellulose-based incontinence pads for maternity care with a size of 6.5 × 22 cm were used for all experiments (Samu Woechnerinnen Vorlagen Classic Mini, Hartmann AG, Germany).
Influence of storage duration
To investigate a possible reduction of the viral load by pad storage, the viral concentration of the CMV-positive urine was determined and in parallel, cellulose pads were soaked separately with 10 mL urine. Cellulose pads were individually stored in lockable plastic bags to reduce evaporation and were manually pressed to collect urine at 15 min, 12 h, 24 h and 7 days after inoculation. CMV DNA concentration was quantified in the respective urine samples by PCR as described above. To investigate the relative decrease in CMV DNA concentration over time, we compared median changes in experiments with a low DNA concentration (i.e. < 5 × 105 IU/mL, group 1) at baseline to changes in experiments using a high DNA concentration (i.e. > 5 × 105 IU/mL, group 2) at baseline. This experiment was performed 30 times.
Influence of meconium
We collected meconium from healthy term infants within the first days of life. One gram meconium and 9 mL normal saline were thoroughly mixed using a vortex mixer. For all experiments, we used CMV-positive urine, collected from preterm infants with postnatally acquired CMV infection. The baseline CMV DNA concentration in the urine was determined, and the urine was mixed with the meconium suspension in a ratio of 1:1 and 4:1 (5 mL urine with 5 mL suspension and 8 mL urine with 2 mL suspension, respectively). For a better understanding, the mixture with a ratio of 1:1 was termed ‘highly contaminated specimen’ and the 4:1 mixture as ‘low contaminated specimen’. The mixture was poured on urine pads that were stored for 24 h or 72 h in a lockable plastic bag at +4°C. The respective urine pads were manually pressed after 24 or 72 h, and the virus concentration was determined in the collected specimen. To account for a reduction of CMV DNA concentration by dilution of the CMV-positive urine with normal saline, the viral load was corrected for the volume of normal saline used in the respective experiments. This experiment was performed 15 times.
Detection rate from dried urine pads
Furthermore, we tested the effect of drying of the urine pads on the detection rate of CMV. Therefore, the CMV load was determined in the CMV-positive urine, and 10 mL urine was put on a urinary pad. The pad was stored at room temperature not sealed in a plastic bag. After seven days of storage, the dry urine pad was soaked with 2 mL of normal saline and centrifuged for 5 min at 4000 ×
Clinical study
Patients
A total of 500 term and preterm infants were prospectively enrolled in the study. On the second day of life, pads were inserted into the diapers of the infants and removed whenever the diaper was changed again (Figure 1). After removing, the pads were stored in lockable plastic bags at +4°C until processing. Within 72 h, the urine-stained pads were manually pressed to collect the urine. Study personnel documented whether the pads were meconium-stained or dry. A volume of 200
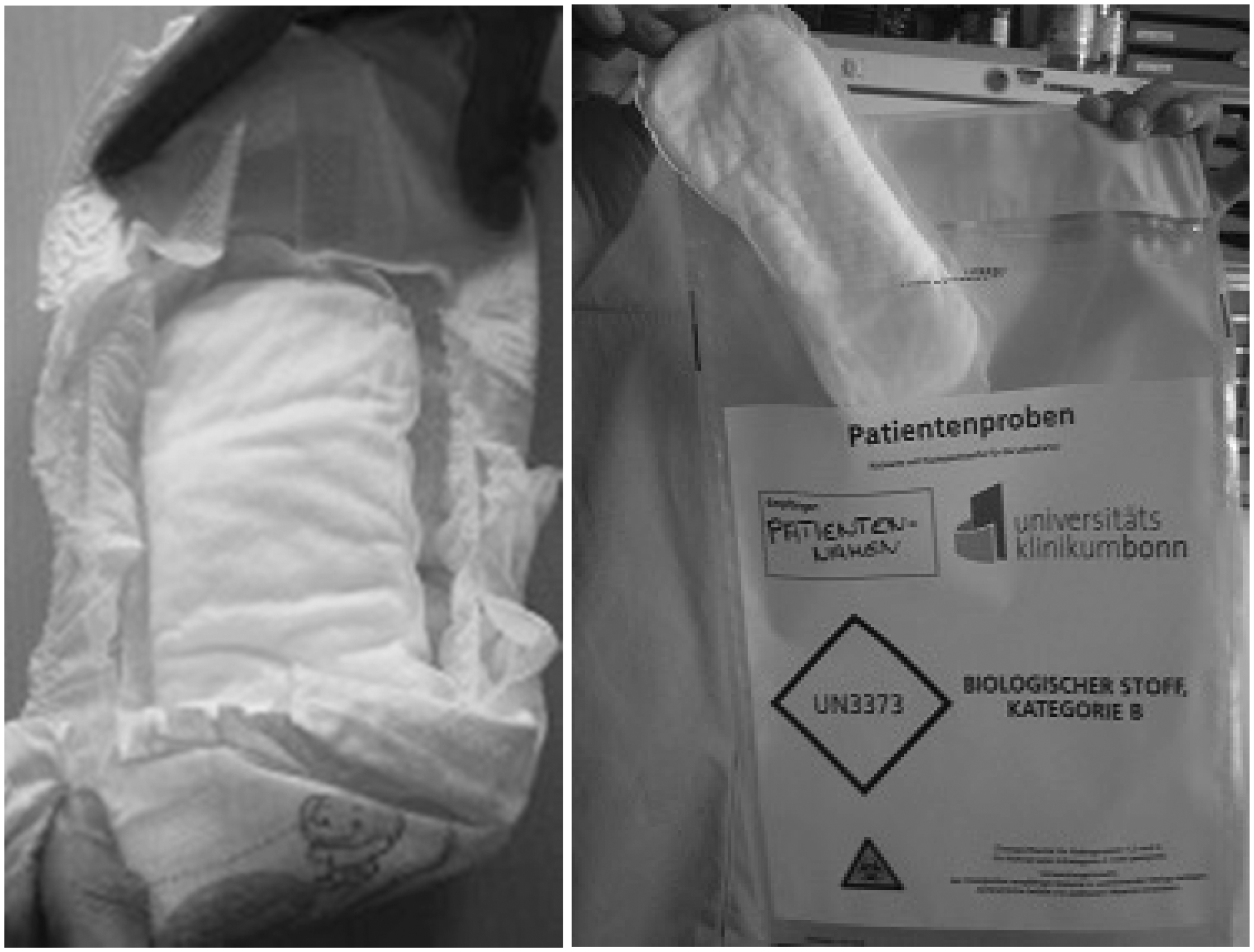
Clinical setting.
For information on maternal CMV seroprevalence, CMV-IgG was determined in a cohort of 100 consecutive mothers using the Abbott CMV IgG Architect assay (Abbott, Wiesbaden, Germany).
Statistical analysis
All statistical tests were performed with a dedicated software package (SPSS release 22.0, SPSS Inc., Chicago, IL, USA). Wilcoxon signed rank test for paired samples was used to compare temporal changes in viral load concentration within the respective groups. A
Results
Preclinical experiments
Influence of storage duration
The median viral load of the urine at baseline was 9.5 × 105 IU/mL. After 15 min, 12 h, 24 h and 7 days of storing, the median CMV concentration decreased to 6.0 × 105 IU/mL, 4.0 × 105 IU/mL, 3.0 × 105 IU/mL, and 8.8 × 104 IU/mL, respectively (Table 1). Considering the baseline viral load as 100%, we observed a median reduction to 63.2%, 42.1%, 31.6% and 9.3% of the baseline value for the respective durations. Individual longitudinal curves (percentage change) of CMV DNA concentration over time are demonstrated in Figure 2. In 12 experiments, the CMV DNA concentration at baseline was < 5 × 105 IU/mL (group 1) and in 18 experiments it was > 5 × 105 IU/mL (group 2). Median baseline CMV DNA concentration was 2.4 × 105 IU/mL in group 1 and 3.9 × 106 IU/mL in group 2. The median change in DNA concentration compared with baseline values after 15 min, 12 h, 24 h and 7 days was 97.4%, 82.3%, 65.9% and 32.5% in group 1 and 51.2%, 37.9%, 28.3% and 8.5% in group 2, respectively. Reduction of the viral load was significant at 12 h (
Influence of storage duration.
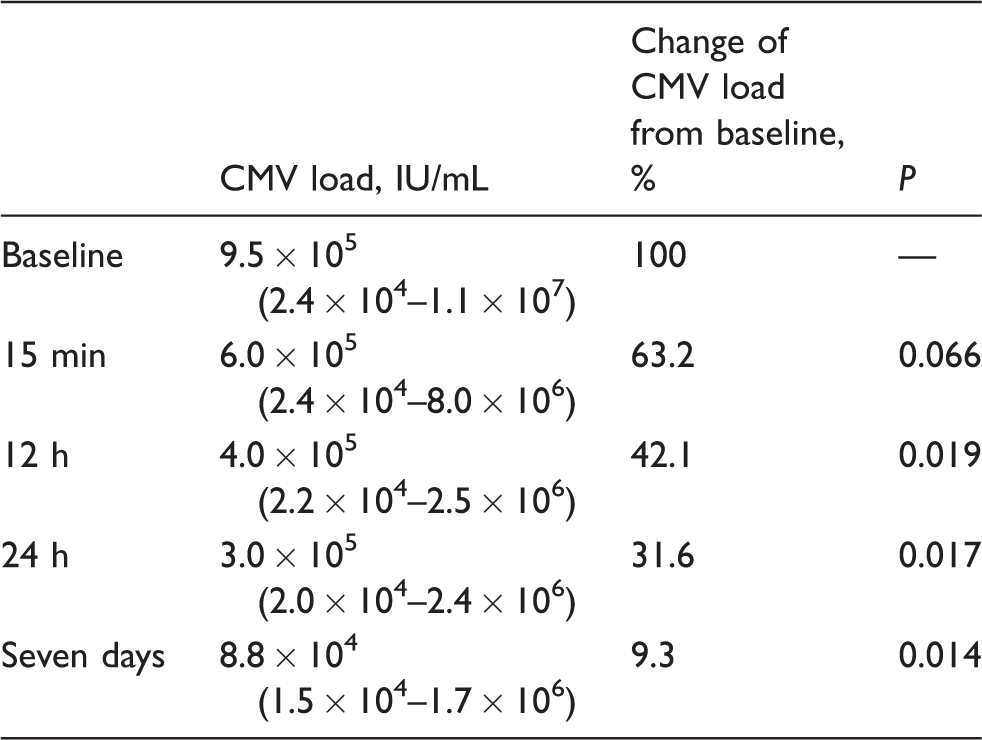
Note: Kinetics of CMV load of 30 samples of CMV-positive urine during exposure to urine pads. Values for IU/mL are reported as median with range in parentheses.
CMV: cytomegalovirus.
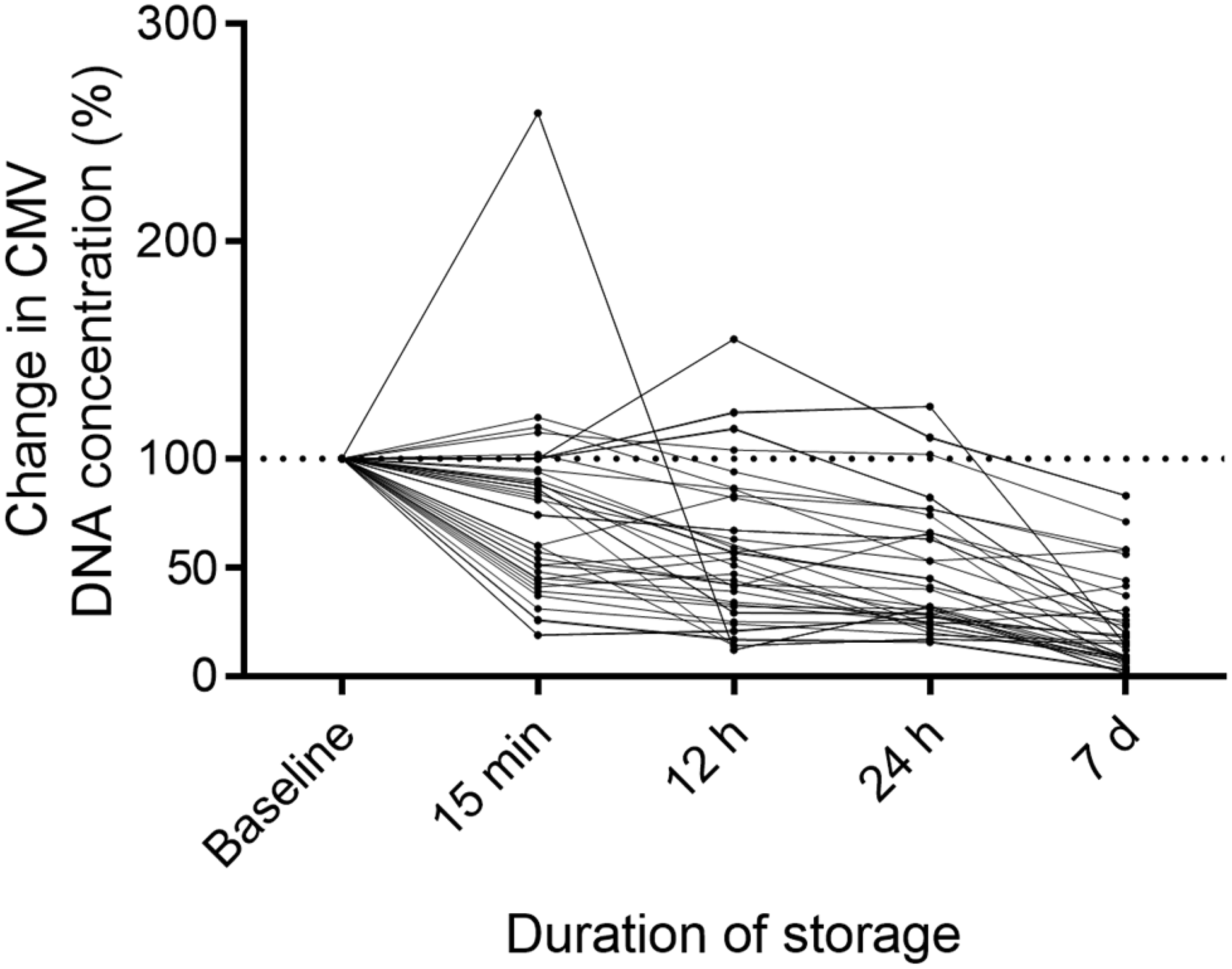
Influence of storage duration.
Influence of meconium
Viral load measurements are summarized in Table 2. The median viral DNA load at baseline was 3.8 × 105 IU/mL. After 24 h of storage, the median viral load decreased to 7.8 × 104 IU/mL (20.5%,
Influence of meconium contamination of urine sample.
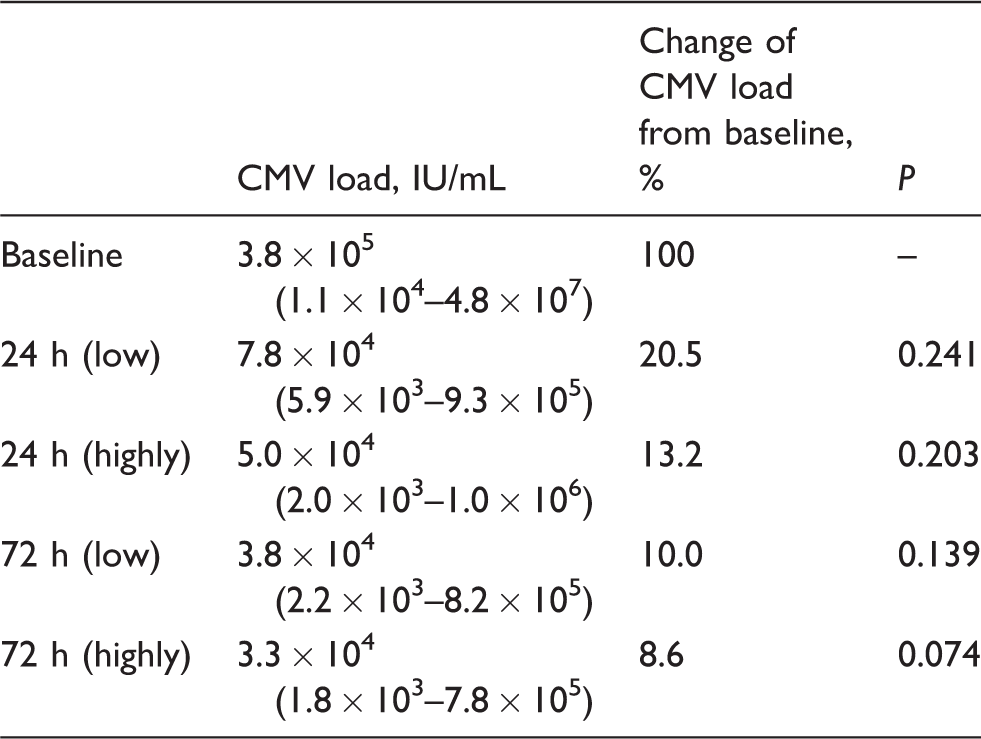
Note: Kinetics of CMV load of 15 samples of CMV-positive urine mixed with two different amounts of meconium to simulate two degrees of contamination (highly/low) of the urine specimen with meconium. The CMV-positive urine was mixed with the meconium suspension in a ratio of 1:1 (highly) and 4:1 (low). Values for IU/mL are reported as median with range in parentheses.
CMV: cytomegalovirus.
Detection rate from dried urine pads
The baseline CMV load ranged between 1.9 × 103 IU/mL and 1.2 × 106 IU/mL. Following seven days of storing at room temperature, CMV DNA was detected in 28/30 dried urine pads (93.3%). Two urine pads (6.7%), with a baseline viral load of 1.9 × 103 and 2.2 × 103 IU/mL, tested negative for CMV DNA after seven days. In all, there were 12/28 positive specimens (42.9%) that were below the lower limit of quantification (i.e. <1000 IU/mL). Viral load of the remaining 16 specimens was significantly decreased and ranged between 1.2 × 103 and 1.0 × 105 IU/mL (
Clinical study
Urine specimens were collected from 500 infants. Overall, there were 237 male infants (47.4%). Mean gestational age was 37.2 ± 2.2 weeks and mean birth weight was 3260 g ± 624 g. Mean maternal age at delivery was 31.2 years. In our cohort, the prevalence of CMV-IgG in mothers was 44%.
A sufficient amount of urine was collected from 73.6% (368/500) of the urine pads in the first attempt. In the remaining 26.4% (132/500), a second attempt was necessary to collect a sufficient amount of urine by inserting a second pad into the diaper. A total of 110 (22%) of the 500 urine pads were partially contaminated with meconium.
All 500 samples were individually tested for CMV DNA as well as in pools of 10 samples each. Two of 50 pools tested positive, and in each pool, one sample was positive. The single sample analysis of all 500 urine samples revealed positive results for CMV DNA in the same two infants. In total, the CMV infection rate in our cohort was 0.4% (2/500).
Discussion
In the present study, we were able to demonstrate the practicability of cellulose pads for urine collection in infants enrolled in a screening programme for congenital CMV infection. In the experimental prestudy carried out to evaluate the reliability of the method, we observed a decrease of CMV DNA concentration over time. However, the concentration remained sufficiently high for CMV DNA detection, even after seven days of storing. On the other hand, the brief exposure (i.e. 15 min) of CMV-positive urine to a urine pad resulted in a reduction of more than one-third of the CMV DNA concentration. This might be attributable to potential virus-binding properties of the cellulose structure within the pad. In the first 12 h, we observed a median reduction of the CMV DNA concentration to 42.1% of the baseline value. After seven days of storage, the median viral load finally decreased to 9.3% (median 8.8 × 104 IU/mL) of the baseline value. We observed a stronger decrease in CMV DNA in experiments with a high baseline concentration compared with those with a low baseline concentration. In fact, in experiments with a CMV DNA concentration < 5 × 105 IU/mL, the median decrease after 12 h of storage was 82.3% compared with 37.9% in experiments with a baseline concentration > 5 × 105 IU/mL. These results demonstrate that infants with congenital CMV infection might not be at risk to get missed by our screening method due to an unreasonable loss of CMV DNA. Although the storage duration of seven days is experimental and does not reflect a realistic clinical scenario, the detection rate was still 100%.
Meconium contamination of the urine was associated with a further reduction of the CMV DNA load, but this reduction was comparable to the decrease in DNA concentration observed in the experiments to investigate storage duration. The results on CMV DNA detection from dried urine pads should be considered with caution, given the simplified technique that we used in our study, as the detection rate after seven days of pad storage was only 93.3%, and in almost half of the analysed specimens, the viral load decreased below 1.0 × 103 IU/mL. Notwithstanding, all specimens with an initial CMV DNA concentration of ≥4.3 × 103 IU/mL tested positive after seven days, which is much lower than median viral load of congenitally infected infants. 21
For practical reasons, we only used one brand of cellulose pads in our study. However, these pads are distributed in numerous countries throughout the world and are widely used in clinical practice. Although the physical and chemical structure of different cellulose pads may influence the effect of the pad on the recovered viral load level, it was beyond the scope of our pilot study to compare absorbing properties of cellulose pads from different companies.
Urine is a commonly used specimen to identify infants with suspected congenital CMV infection.22–26 In general, the collection of urine is performed with urine collection bags. However, this technique can be time consuming, because the adhesive tape may lose contact with the moist skin. Additionally, removal of these bags can be painful, and in female infants, urine collection is more difficult for anatomical reasons. Among parents, this might hamper the unequivocal acceptance of urine bags for a CMV screening programme. Urine pads placed in the diapers could contribute to a higher acceptance among parents and health-care workers, particularly as urine can be collected within one or at most two attempts. Additionally, manually expressing urine pads to collect urine generates approximately the same workload as collecting urine from a urine bag.
In the clinical setting, urine pads were removed from the diapers within 2 to 6 h after insertion. We tested that an amount of approximately 10 mL urine is necessary to moisture the urine pad in a degree that manually pressing reveals a sufficient amount of urine for CMV-PCR testing (i.e. 200
Ross et al. 14 described urine specimen collection using a cotton ball. In their experience, this technique was associated with a dramatic decrease in the detection rate of CMV DNA (56.1%) compared with conventional urine bags. 14 In a follow-up study performing CMV testing by PCR, similar detection rates were observed (cotton ball, 64/67 = 95.5%; bag, 267/279 = 95.7%) urine. 27 Unfortunately, the preanalytical handling of the cotton balls is not described in detail in the published protocol.
In a recent study, Nijman et al. 21 published their data on urinary CMV DNA concentration in congenitally and postnatally infected infants. 21 Congenitally infected infants presented with a median CMV concentration of 8.5 × 106 genome copies/mL, which is roughly equivalent to 1.3 × 107 IU/mL. This was only slightly higher than the median baseline value in our study. Several studies demonstrated that a low CMV DNA concentration is associated with a lower risk for developing SNHL in asymptomatically infected infants.28–30 The use of urine pads would therefore be a practicable method for clinical scenarios where the exact CMV DNA concentration is less important, such as a screening programme for congenital CMV infection. The observed reduction of the CMV DNA concentration after exposure to the urine pad is most likely not of clinical relevance in a screening setting. However, larger studies including more newborns with CMV shedding than in our pilot study presented are necessary to investigate this issue.
Saliva has been successfully tested as specimen in broad national screening programmes for congenital CMV. 18 Studies comparing saliva and urine for CMV screening advocated saliva as the preferred specimen to detect CMV DNA.14,18 This is mostly attributable to a more convenient collection of saliva when compared with urine. In fact, as stated above, urine collection by urine bags can be time consuming, since it requires undressing the infant, opening the diapers, placing the urine bag, closing the diapers and dressing the baby. Additionally, several attempts might be necessary to collect a sufficient amount of urine for analysis. However, when urine collection can be incorporated into the routine care of the baby, it might not be very time consuming. Furthermore, a fast specimen collection technique is more important in a research setting with limited availability of study personnel than in a regular clinical setting with round-the-clock-availability of nursing staff. To our impression, there are two reasons to favour urine as screening specimen: first, urine can be screened for CMV DNA by pooling 10–20 urine samples, which has been demonstrated to be feasible and cost-effective.14,23 Second, using saliva to detect CMV DNA bears the risk of contamination of saliva by CMV-containing breast milk. 18 Although the reported rate of false-positive results is low, this approach might lead to unnecessary parental anxiety and additional testing to rule out or confirm CMV infection. 18
Recently, a targeted screening strategy, testing only infants for CMV infection who do not pass newborn hearing screening (NHS), has been investigated.31,32 The rationale for this approach is a potentially dramatic reduction of overall costs. However, one study including more than 100,000 infants revealed that 43% of all CMV-positive infants and 53% of asymptomatically infected infants passed NHS but were confirmed to have CMV-related SNHL during infancy. 31 Therefore, restriction of CMV testing to infants who failed their NHS cannot be recommended because it will miss a significant proportion of babies who develop late onset hearing loss.
Pooling of urine specimens could result in reduced costs and labour and should therefore be considered as an option for large screening programmes.19,20 With a presumed incidence of 0.4% for congenital CMV, it could be expected that 24 of 25 pools (each with 10 specimens) would be negative when tested for CMV DNA. One positive neonate among 250 neonates could lead to an 86% reduction of PCR runs to detect one infant compared with an approach without pooling (25 pools + 10 individual specimens in case of one positive pool instead of testing of 250 individual specimens). Another aspect to be considered is that the decrease of CMV DNA concentration in urine caused by meconium could be less than indicated by our experimental data, since CMV-infected infants shed CMV DNA in meconium. 33
The high variability of baseline CMV DNA concentrations might raise doubts about the reproducibility of our study design. We discussed the option of using the first International NIBSC/WHO CMV DNA Standard based on a whole virus preparation of the HCMV Merlin strain cultured
As addressed by previous studies, the results of quantitative detection of CMV DNA in clinical specimens can vary and do not perfectly correlate, especially when different assays are used and compared with each other.34,35 Furthermore, there are interlab variations. Thus, an allowed deviation of −0.8 to +0.8 in respect to the target value expressed as log10 is reasonable. Our evaluated method is not feasible to reveal exact true numbers of the virus concentration in the urine but allows the identification of CMV DNA in neonatal urine. Infants with a positive test result need to be referred to a paediatric infectious disease clinic for further evaluation and close follow-up.
In conclusion, the use of cellulose pads for urine specimen collection to detect congenital CMV infection is feasible and especially useful for a screening programme with limited resources. Although we observed a reduction of the viral load as early as after 15 min of exposure to the pad, even after a storage period of seven days CMV DNA was detectable in 100% of the tested samples. Meconium contamination of urine did not result in an unacceptable high rate of loss of the CMV DNA. To reduce the reduction in viral load and to avoid drying of the urine pads, we recommend removing urine from urine pads early after exposure.
Footnotes
Acknowledgements
We would like to thank the nurses in the newborn nursery and the neonatal intensive care unit for assistance in the distribution and collection of urine pads. Furthermore, we would like to express our thanks to the parents of the babies included in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The Bonn University Hospital Institutional Review Board approved the study. Written informed consent was obtained from parents or legal representatives before enrolment in the study. The study was performed in accordance with the ethical standards described in the Declaration of Helsinki.
Guarantor
FK.
Contributorship
FK and KJ contributed equally to development of the study design, patient enrolment and specimen collection, conducting preclinical experiments, data analysis and writing of the manuscript. AMEH and UR contributed to study design, specimen processing, PCR testing, evaluation of the study protocol, data analysis, and contributed to writing of the manuscript. SH provided laboratory infrastructure and contributed development of the protocol, to data analysis and writing of the manuscript. UG contributed to study design, patient enrolment, data analysis and writing of the manuscript. PB and AM contributed to development of the study design, final data analysis and writing the manuscript.
