Abstract
Background
Because autotaxin reportedly has a better performance than hyaluronic acid as a marker for liver fibrosis for the prediction of cirrhosis caused by hepatitis C, we aimed to further evaluate the role of autotaxin in liver fibrosis of other aetiologies.
Methods
Autotaxin antigen was measured in serum samples from 108 patients with chronic hepatitis B and 128 patients with non-alcoholic fatty liver disease who had undergone a liver biopsy as well as healthy subjects and patients with chronic kidney disease, diabetes mellitus, rheumatoid arthritis and cardiac dysfunction.
Results
When evaluated using receiver operator characteristics curves, the performance of autotaxin for the prediction of significant fibrosis (F2–F4) in chronic hepatitis B patients was better than that of hyaluronic acid or type IV collagen 7S. In non-alcoholic fatty liver disease patients, however, the performance of autotaxin for the prediction of significant fibrosis was poorer than that of hyaluronic acid or type IV collagen 7S. The increase in the serum autotaxin concentrations was less notable than that of hyaluronic acid or type IV collagen in patients with chronic kidney disease, diabetes mellitus, rheumatoid arthritis or cardiac dysfunction. Food intake did not affect the serum autotaxin concentrations.
Conclusions
Autotaxin is useful as a serum marker for liver fibrosis caused by not only chronic viral hepatitis C but also by hepatitis B, although it was less useful in patients with non-alcoholic fatty liver disease. The increase in serum autotaxin concentrations is fairly specific for liver fibrosis, and the serum autotaxin concentrations can be analysed without consideration of food intake before blood collection.
Introduction
We have been working to clarify the roles of autotaxin (ATX), which possesses the enzymatic activity required to generate the lipid mediator lysophosphatidic acid (LPA), in liver pathophysiology. We previously reported that LPA stimulates the proliferation of hepatic stellate cells, which are key players in liver fibrosis in vitro. 1 During the course of experiments to unveil the in vivo roles of LPA in liver fibrosis, we subsequently found that both the serum ATX concentrations and the plasma LPA concentrations increased in correlation with the stage of liver fibrosis in rats 2 and humans.3,4 The correlation between an increase in the serum ATX concentrations and liver fibrosis can be explained by the reduced uptake of ATX from the bloodstream by sinusoidal endothelial cells, 5 reflecting a phenotypic change that occurs during the process of liver fibrosis. 6 In addition, an increase in the mRNA expression of ATX in fibrotic liver has been observed in humans 7 but not in rats with chronic carbon tetrachloride treatment. 2 Nonetheless, these lines of evidence suggest that ATX and LPA might be useful for predicting the stage of liver fibrosis.
From the viewpoint of clinical laboratory medicine, ATX has many advantages over LPA as a blood marker; for example, ATX is temperature stable after sample preparation, 8 and a high-throughput test for ATX mass measurement has been developed. 9 Using this method, we found that the serum ATX concentrations were the best parameter for predicting cirrhosis when compared with serum hyaluronic acid (HA) and the aminotransferase/platelet ratio index (APRI) in patients with chronic liver disease caused by hepatitis C virus (HCV; C-CLD), 4 suggesting that ATX could be used as a serum marker for liver fibrosis in clinical settings. Very recently, in line with our previous finding, ATX was shown to perform well as a serum marker for liver fibrosis in C-CLD patients, with ATX being superior to the FIB-4 index and Forn’s index. 10
When considering the practical use of ATX as a marker for liver fibrosis, one must first determine its performance in patients with hepatitis B virus (HBV), another hepatitis virus prevalent worldwide, and in patients with non-alcoholic fatty liver disease (NAFLD), another common cause of chronic liver disease around the world, 11 as both of these conditions can ultimately lead to cirrhosis and hepatocellular carcinoma. 12 Furthermore, we sought to clarify whether the serum ATX concentrations might be altered in patients with common diseases that do not involve liver fibrosis, such as chronic kidney disease (CKD), diabetes mellitus (DM), rheumatoid arthritis (RA) or cardiac dysfunction (CD). In addition, we wondered whether the serum ATX concentrations might be affected by food intake.
Methods
Participants
One hundred and eight patients with chronic liver disease caused by HBV infection (B-CLD) were diagnosed based on the presence of positive HBV markers in blood samples and histopathological findings from liver biopsies between 1987 and 2011 at Toranomon Hospital. Forty subjects with normal liver function tests and no history of liver diseases and 128 patients with NAFLD diagnosed based on histopathological findings from liver biopsies (steatosis in more than 5% of the hepatocytes) and the exclusion of other liver diseases were enrolled between 2008 and 2014 at the University of Tokyo Hospital. Serum samples from patients with CKD (n = 31, men/women = 16/15), DM (n = 47, men/women = 26/21), RA (n = 54, men/women = 14/40) or CD (n = 54, men/women = 32/22) but without liver diseases and from healthy subjects (n = 56, men/women = 23/33) were obtained from Tsukuba Medical Laboratory of Education and Research (TMER) between 2011 and 2012. CKD was diagnosed based on the presence of kidney injury and/or a reduced glomerular filtration rate (<60 mL/min/1.73 m2); DM was diagnosed based on a fasting plasma glucose concentration ≥126 mg/dL, a 75-g oral glucose tolerance test at 2 h ≥200 mg/dL, or a casual plasma glucose concentration ≥200 mg/dL; RA was diagnosed based on the presence of synovitis in multiple joints 13 and CD was diagnosed based on the presence of a plasma brain natriuretic peptide concentration ≥100 pg/mL. Whether food intake might affect the serum ATX concentrations was examined in healthy subjects (n = 5) enrolled in the study in 2011 at the University of Tokyo Hospital.
In all the patients with liver diseases, blood samples were taken on the day of liver biopsy at each hospital; the serum was separated from all the samples and stored at −20℃ or lower until the ATX antigen measurement.
This study was performed in accordance with the ethical guidelines of the Declaration of Helsinki and was approved by the Research Ethics Committee of Toranomon Hospital, TMER, and the University of Tokyo Hospital. Informed consent was obtained from each of the patients and subjects at each hospital.
Measurement of ATX
The serum ATX antigen concentrations were determined using a specific two-site enzyme immunoassay, as previously described. 4
Measurement of HA and type IV collagen 7S
HA was measured using Hyaluronic Acid LT assay from WAKO Pure Chemical Ind. Ltd, Tokyo, Japan, and type IV collagen 7S was measured using Type IV collagen 7S kit from Sceti Medical LABO, Tokyo, Japan.
Statistical analysis
The trends in the serum ATX concentrations in accordance with the fibrosis stage were assessed using the Jonckheere-Terpstra test. A two-sided P-value less than 0.05 was considered statistically significant. The predictive accuracy of the parameters for the stage of liver fibrosis was assessed using a receiver-operator characteristics (ROC) curve analysis. The area under the curve (AUC) was evaluated as the ability to predict the stage of liver fibrosis, and comparisons were analysed using a non-directional two-tailed significance level. The sensitivity and specificity of the ROC analysis were decided by the cut-off value determined by the Youden index. The statistical procedures were performed using StatFlex software (Version 6.0; Artec Inc., Osaka, Japan).
Results
Performance of ATX for predicting the liver fibrosis stage in patients with B-CLD
Characteristics of healthy subjects and patients with B-CLD and NAFLD.
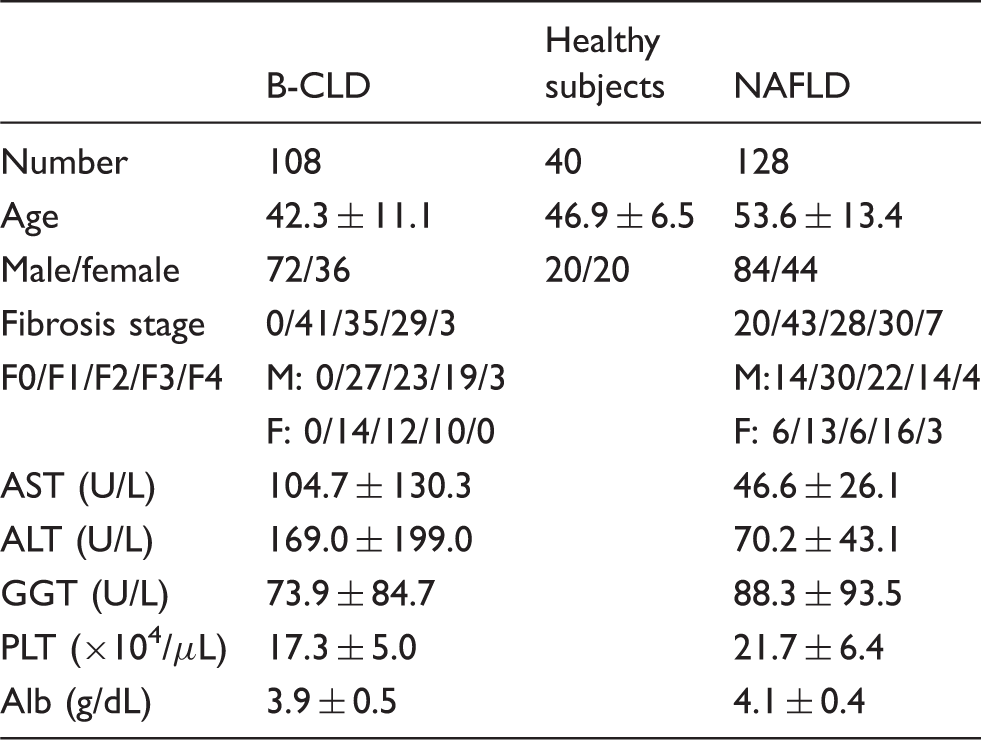
AST: aspartate aminotransferase; ALT: alanine aminotransferase; GGT: gamma-glutamyl transferase; PLT: platelet; Alb: albumin; NAFLD: non-alcoholic fatty liver disease; B-CLD: chronic liver disease caused by HBV infection.
Performance of ATX, HA or type IV collagen 7S to predict significant fibrosis (≥F2) in patients with B-CLD.
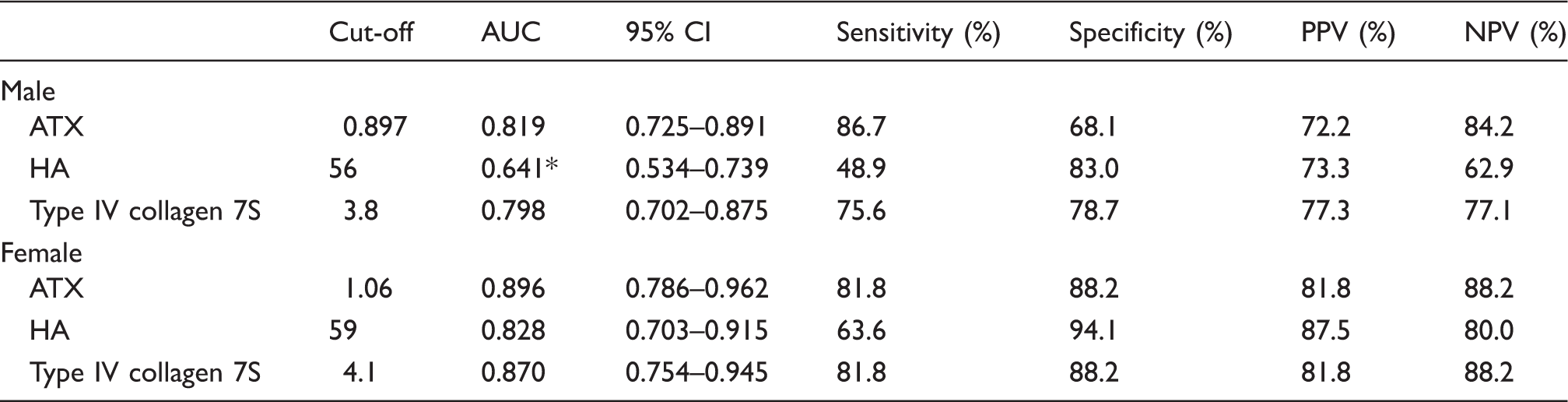
AUC: area under the curve; PPV: positive predictive value; NPV: negative predictive value; ATX: autotaxin; HA: hyaluronic acid; CI: confidence interval; B-CLD: chronic liver disease caused by HBV infection.
P < 0.01 (non-directional two-tailed significance level).
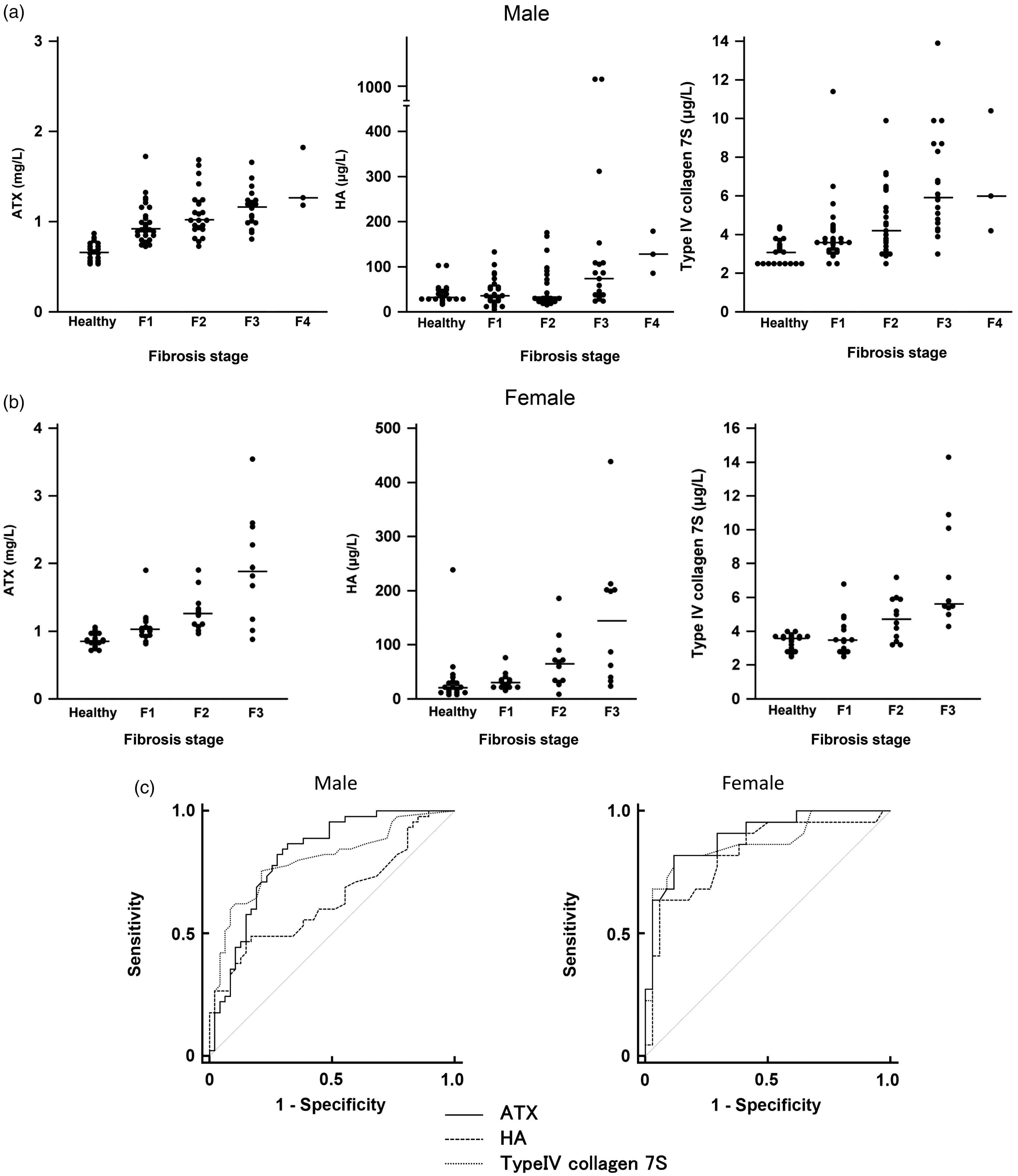
Relationships between serum concentrations of ATX, HA or type IV collagen 7S and the liver fibrosis stage in men (a) and women (b) with B-CLD and healthy subjects and in men (d) and women (e) with NAFLD. Data from 72 men and 36 women with B-CLD and 20 men and 20 women without liver diseases, who were assumed not to have liver fibrosis, were used to analyse the relationship between the serum concentrations of ATX, HA, or type IV collagen 7S and the liver fibrosis stage. Data from 84 men and 44 women with NAFLD were used to analyse the relationship between the serum concentrations of ATX, HA or type IV collagen 7S and the liver fibrosis stage. The vertical bars denote the median. ROC curves illustrating the relationship between sensitivity and 1 – specificity for the serum concentrations of ATX, HA or type IV collagen 7S for discriminating significant fibrosis (F2−F4) in men and women with B-CLD (c), and in men and women with NAFLD (f). Data from the above-described patients were used in the ROC statistical analyses to compare the accuracy of ATX, HA and type IV collagen 7S for the discrimination of significant fibrosis (F2−F4).
Performance of ATX for predicting the liver fibrosis stage in patients with NAFLD
Performance of ATX, HA or type IV collagen 7S to predict significant fibrosis (≥F2) in patients with NAFLD.
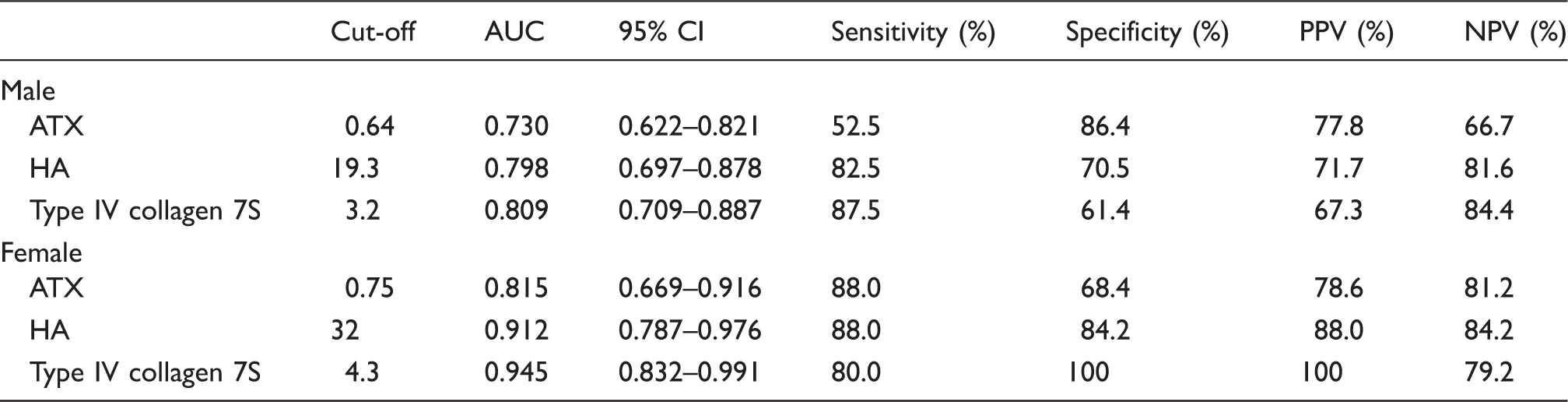
AUC: area under the curve; PPV: positive predictive value; NPV: negative predictive value; ATX: autotaxin; HA: hyaluronic acid; CI: confidence interval; NAFLD: non-alcoholic fatty liver disease.
Serum concentrations of ATX in patients with non-liver diseases
For the use of ATX as a marker for liver fibrosis in clinical settings, it is important to know whether the serum ATX concentrations might be elevated in patients with non-liver diseases without fibrosis, such as CKD, DM, RA or CD, as well as in healthy subjects. As shown in Figure 2(a) and (b), the serum concentrations of HA were higher in patients with CKD, DM, RA or CD than in healthy subjects. The serum concentrations of type IV collagen 7S were also increased in men with CKD, DM, RA or CD, compared with the concentrations in healthy subjects. In contrast, the serum concentrations of ATX were slightly increased in patients with RA and in men with CD, compared with the concentrations in healthy subjects. Thus, ATX is a more specific marker for liver fibrosis than HA or type IV collagen 7S, suggesting its usefulness for the prediction of liver fibrosis even in patients with CKD, DM, RA or CD.
Serum concentrations of ATX, HA or type IV collagen 7S in male (a) and female (b) healthy subjects and CKD, DM, RA or CD patients. Serum samples from patients with CKD (n = 31), DM (n = 47), RA (n = 54), or CD (n = 54) without liver diseases and from healthy subjects (n = 56) were used to examine the ATX, HA, and type IV collagen 7S concentrations. The vertical bars denote the median and an asterisk indicates a significant difference from healthy subjects. (c) Circadian serum ATX concentrations before and after food intake. Data from five subjects with 6 points before and after food intake were used to examine the circadian serum ATX concentrations. The measured values were divided by the mean of six values from each subjects and expressed as % of mean. The line represents the mean ATX values of five subjects at each point, and the error bars show the standard deviations.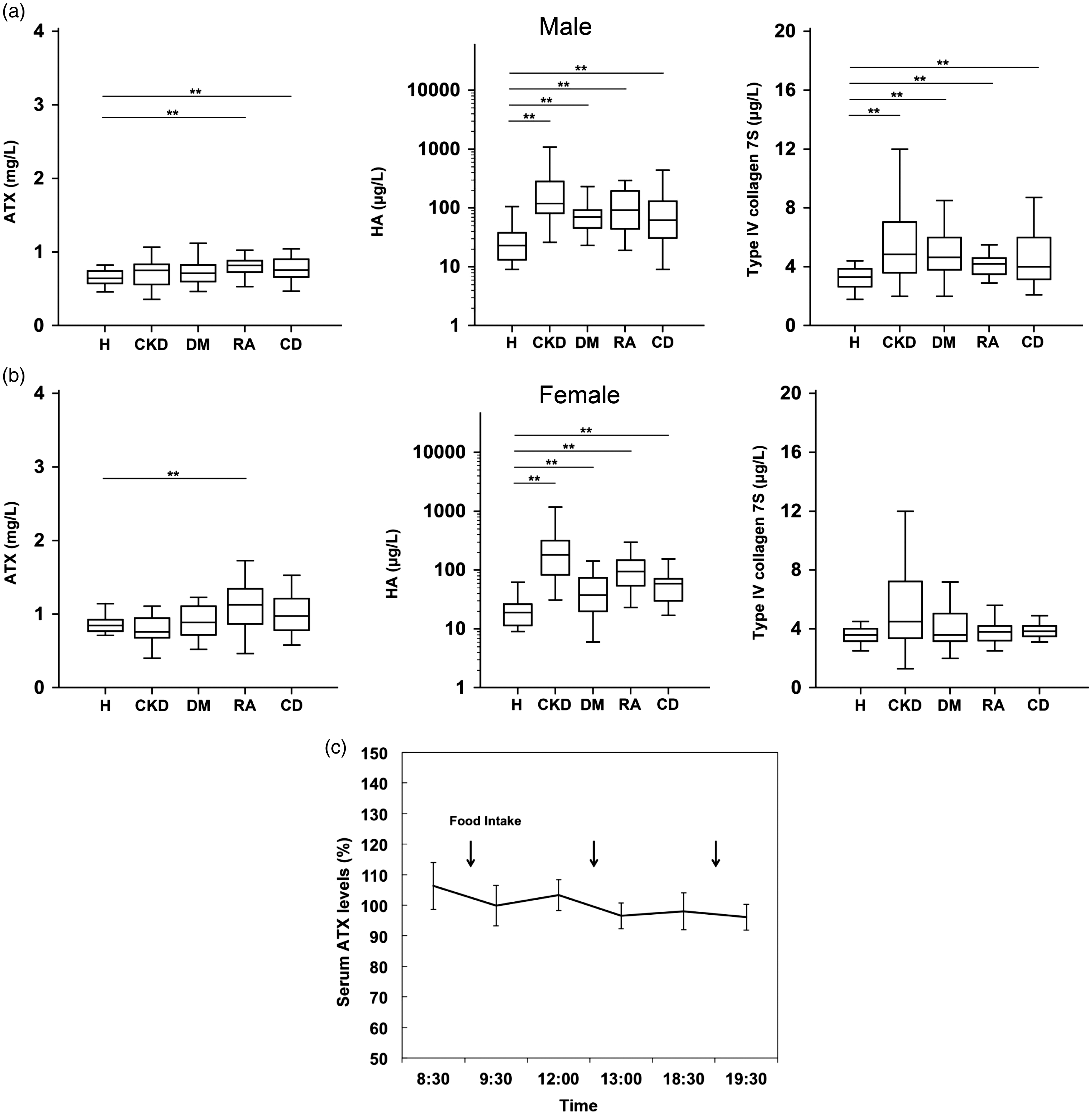
Serum ATX concentrations after food intake
We examined whether food intake before blood drawing might affect the serum ATX concentrations. As shown in Figure 2(c), significant circadian variations in food intake were not observed in healthy subjects (n = 5). This result suggests that serum ATX concentrations can be analysed without any consideration of the timing of serum sample collection in relation to meals.
Discussion
Our previous report indicated that ATX has a superior performance for predicting cirrhosis than HA or APRI, two conventional markers for liver fibrosis, in C-CLD patients. 4 In line with our finding, the usefulness of ATX as a marker for liver fibrosis was recently reported by Yamazaki et al. 10 in C-CLD patients. In this report, the diagnostic accuracy of ATX was compared with that of a novel marker for liver fibrosis Mac-2 binding protein glycosylation isomer (M2BPGi). The AUC to diagnose fibrosis of ≥F2 was 0.861 for ATX and 0.829 for M2BPGi in male patients and 0.801 for ATX and 0.794 for M2BPGi in female patients. The AUC to diagnose fibrosis of ≥F4 was 0.862 for ATX and 0.824 for M2BPGi in male patients and 0.739 for ATX and 0.807 for M2BPGi in female patients. Based on these findings, we wondered whether ATX could be used as a serum marker for liver fibrosis. To address this question, the performance of ATX as a serum marker for liver fibrosis was examined in patients with B-CLD and in patients with NAFLD. Regarding B-CLD, because we could not enrol enough patients with cirrhosis, the performance of ATX to predict significant fibrosis (F2−F4) was examined in comparison with that of HA or type IV collagen, with ATX exhibiting the best performance. On the other hand, in patients with NAFLD, the performance of ATX as a liver fibrosis marker was poorer than the performances of HA and type IV collagen 7S. In addition to uptake by sinusoidal endothelial cells in the liver, 5 serum ATX concentrations are known to be regulated by its production in adipose tissue. 14 In this context, serum ATX concentrations are reportedly reduced in obese subjects. 14 Because NAFLD patients are frequently obese, the serum ATX concentrations in NAFLD patients may reflect not only the presence of liver fibrosis but also obesity, suggesting that ATX may be a less reliable marker for liver fibrosis in patients with NAFLD.
Because we wondered whether the serum ATX concentrations might be increased in patients with diseases that do not involve liver fibrosis, serum samples from patients with CKD, DM, RA or CD were also analysed. As a result, among ATX, HA and type IV collagen 7S, the serum ATX concentrations had the least likelihood of being elevated in patients with CKD, DM, RA or CD, suggesting that ATX is a more specific marker for liver fibrosis than HA or type IV collagen 7S. Of note, the serum ATX concentrations were only increased in a few pathological or specific physiological states, such as the presence of follicular lymphoma 15 or pregnancy. 16 In addition, food intake did not affect the serum ATX concentrations. Furthermore, ATX is temperature stable after sample preparation, 8 and a high-throughput test for measuring ATX concentrations has been developed. 9 Collectively, these features suggest that ATX could be useful as a marker for liver fibrosis.
The difference in the performances of each marker for the prediction of the liver fibrosis stage can be explained by their distinct origins and metabolic pathways in the bloodstream. Although both ATX and HA may be taken up by liver sinusoidal endothelial cells,5,17 only ATX is produced by hepatocytes, 18 and HA is instead produced by hepatic stellate cells, 19 causing the distinct kinetics of ATX and HA in the blood. In fact, the serum ATX concentrations, but not the HA concentrations, exhibit a gender difference. 9 Impaired renal function causes an increase in the serum HA concentrations20,21 but not the ATX concentrations. 22 Furthermore, an individual’s diet affects the serum HA concentration 23 but not the ATX concentration. On the other hand, an increase in serum type IV collagen 7S concentrations may be associated with active fibrogenesis in the liver. 24
Regarding ATX and the liver, some novel findings other than an increase in serum concentrations during liver fibrosis are accumulating. According to a study by Kaffe et al., the expression of ATX in hepatocytes promotes liver fibrosis, and hepatocyte ATX ablation attenuates hepatocellular carcinoma development in mice, suggesting a causative link between ATX and not only cirrhosis but also hepatocellular carcinoma. 18 An organ transcriptome analysis in humans has suggested that the LPA pathway is a central chemoprevention target for hepatocellular carcinoma arising from areas of cirrhosis. 25 On the other hand, we previously reported that higher serum ATX concentrations with enhanced LPA receptor 2 or LPA receptor 6 mRNA expression in hepatocellular carcinoma were associated with poorer differentiation, microvascular invasion and earlier recurrence in humans. The causative role of ATX/LPA in hepatocarcinogenesis should be further studied.7,26
In conclusion, ATX performed well as a serum marker for liver fibrosis in patients with chronic viral hepatitis B as well as those with hepatitis C, but its performance was limited in patients with NAFLD. An increase in serum ATX concentrations was fairly specific for liver fibrosis. Serum ATX concentrations can be analysed without consideration of meal times. Collectively, these features suggest that ATX might be useful as a marker for liver fibrosis in clinical settings.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SS and KI are employees of TOSOH Corporation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by CREST from JST/AMED, a Grant-in-Aid for Scientific Research on Innovative Areas 15H05906 and the Translational Research Network Program, MEXT.
Ethical approval
This study was performed in accordance with the ethical guidelines of the Declaration of Helsinki and was approved by the Research Ethics Committee of the University of Tokyo Hospital (1143).
Guarantor
HI.
Contributorship
HI: study concept and design, analysis and interpretation of data, and drafting of the manuscript.
MK, HK, KE, KK, RO: protocol development, gaining ethical approval, patient recruitment and data analysis.
TN, TK, SS, KI, JA: establishment of ATX measurement system and analysis of ATX data.
MK, MS: statistical analysis and drafting of the manuscript.
YY: literature research and study concept.
