Abstract
Background
The α-melanocyte-stimulating hormone (α-MSH), an endogenous neuropeptide derived from proopiomelanocortin (POMC), has been identified to suppress inflammation and prevent osteoblast damage.
Objective
The present study was aimed to investigate the role of serum α-MSH in non-traumatic osteonecrosis of the femoral head (ONFH).
Methods
Seventy-nine patients diagnosed with non-traumatic ONFH and 79 sex- and age-matched healthy controls were enrolled in the study. Serum α-MSH concentrations were examined with a double antibody radioimmunoassay. The radiographic progression of ONFH was assessed by X-ray plain film according to the FICAT grading system. The symptomatic severity was evaluated by visual analogue scale scores, Harris hip scores and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores. The serum concentrations of protective marker adiponectin and bone necrosis inflammation factor IL-33 concentrations were also examined. The receiver operating characteristic (ROC) analysis curve was performed to explore the diagnostic value of α-MSH, adiponectin and IL-33 for radiographic progression.
Results
Serum α-MSH concentrations were significantly lower in ONFH patients than in healthy controls. The case group included 29 non-traumatic ONFH patients with FICAT grade I/II, 27 with grade III and 23 with grade IV. ONFH patients with grade I/II had significantly higher α-MSH concentrations in serum compared with those with FICAT grades III and IV. ONFH patients with FICAT grade III showed significantly elevated concentrations of α-MSH in serum compared with those with FICAT grade IV. Serum α-MSH concentrations were negatively associated with radiographic progression by FICAT grading system, and symptomatic severity defined by visual analogue scale scores, Harris hip scores and WOMAC scores. In addition, serum α-MSH concentrations were positively related to the expression of adiponectin and negatively associated with IL-33. ROC analysis curve demonstrated that α-MSH exhibited the equal value for the diagnosis of ONFH radiographic progression compared with IL-33.
Conclusions
Serum α-MSH may act as a protective biomarker for non-traumatic ONFH. Systematic application of α-MSH serving as an adjunctive therapy for treating non-traumatic ONFH deserves further investigation.
Keywords
Introduction
Non-traumatic osteonecrosis of the femoral head (ONFH) is a progressively debilitating disease that usually results in the damage of the hip joint. According to the statistics, ONFH accounts for 5% to 12% of all total hip replacement surgeries, with an incidence ranging from 10,000 to 20,000 cases per year in the US. 1 Non-traumatic ONFH is believed to be a multifactorial disease that is associated in some cases with both a genetic predilection as well as exposure to certain risk factors. These risk factors include corticosteroid use, alcohol intake, smoking and abnormal lipid metabolism, etc. 2
Until now, the most important evidence for diagnosing non-traumatic ONFH are complains of symptoms, impaired function and radiographic changes. 3 The symptoms of ONFH are hip joint pain and, depending on the severity, inability to use the joint. However, the disease may show no symptoms at all in the early stages, and worsening pain may appear as the disease progresses. In addition, the radiographic progression including osteosclerotic changes, subchondral collapse and joint space narrowing are almost signs of medium or late stage. 4 Therefore, to seek early, brief and more effective methods such as valuable biomarkers in serum may be meaningful for diagnosis, treatment and prognosis of non-traumatic ONFH.
Alpha-melanocyte-stimulating hormone (α-MSH) is a 13-amino acid peptide that stimulates melanocortin receptor (MCR) and is widely expressed in various tissues and organs including bone and cartilage. 4 The α-MSH plays an important role in a variety of biological processes, such as energy metabolism, bodyweight regulation and modulation of inflammation process.5–7 It is now well recognized that α-MSH participates in the regulation of immune responses by impairing important functions of both antigen-presenting cells and T cells. 8
Recently, the role of α-MSH for preventing osteoblasts against damage has been investigated. Several previous studies found that the toll-like receptor 4 (TLR4) signalling pathway involves in the pathogenesis of ONFH, implying systemic immune and inflammation participating the process of ONFH.9,10 And works have also demonstrated that TLR4 stimulation by lipopolysaccharide administration strengthens the incidence of corticosteroid-induced ONFH. 11 In a further study, Taylor 12 found that α-MSH was able to inhibit lipopolysaccharides (LPS)-stimulated TLR4 with IRAK-M in macrophages. In addition, α-MSH alleviates steroid-induced damages to the cultured osteoblasts via activating MC4R-SphK1 signalling, implicating cytoprotective effect performed by α-MSH in osteoblasts. 13
Therefore, based on the previous studies above, we hypothesize that α-MSH may act as a protective factor during the process of non-traumatic ONFH. To our knowledge, there were no works available investigating the circulating α-MSH concentrations in non-traumatic ONFH as well as its expression with disease severity. Our study was aimed at measuring serum α-MSH concentrations in non-traumatic ONFH patients to evaluate its potential utility as a biomarker for disease progression of non-traumatic ONFH.
Methods
Study patients
This study was approved by our hospital and was conducted in accordance with the ethical standards of the Helsinki Declaration. A total of 79 unrelated patients with non-traumatic ONFH were consecutively enrolled from July 2016 to May 2017 in the current study. The patients were excluded if they had cardiovascular diseases, congenital diseases, human immunodeficiency virus infection, diabetes mellitus, renal dysfunction and cancer. ONFH were diagnosed by evidence of osteonecrosis using plain radiographs of the FICAT Classification system. 14 Steroid-induced osteonecrosis was defined by a history of taking prednisolone cumulative 2000 mg or an equivalent over 21 days. Alcohol-induced osteonecrosis was defined by the consumption of >900 mL of pure ethanol per week. Seventy-nine age- and sex-matched volunteers who visited our hospital for normal body check were enrolled as controls. All participants were noticed of the study and signed the consent inform.
Laboratory assessment
Blood samples were obtained in the morning at 7:00, and all individuals were fasting and at rest at the time of blood collection. Serum was collected from the non-traumatic ONFH and healthy controls, centrifuged at 3000 r/min and then stored at −80℃ before evaluation. Serum α-MSH concentrations were measured by a commercial human enzyme-linked immunosorbent assay (ELISA) Kit (MyBioSource, CA, USA, Cat. No: MBS018213). The ELISA analytical biochemical technique of the kit is based on α-MSH antibody-α-MSH antigen interactions (immunosorbency) and a horseradish peroxidase colorimetric detection system to detect α-MSH antigen targets in samples. The ELISA Kit is designed to detect native, non-recombinant, α-MSH. The detection range is 3.12 pg/mL–100 pg/mL. The intra- and inter-assay coefficients of variation (CV) for α-MSH were 5.3% and 5.7% following our in-house control. The commercially available ELISAs were used to examine the serum concentrations of adiponectin (R&D Systems Inc., Minneapolis, MN, USA) and IL-33 (Santa Cruz Biotechnology, Santa Cruz, USA). The experiments for all factors were repeated three times, and the results were averaged.
Radiographic evaluation
All patients had a plain X-ray of the hip in two projections (anterior and lateral) according to the FICAT classification.
14
In detail, on X-ray plain radiographs, FICAT stage I: normal or, at most, minor changes (Figure 1(a)); FICAT stage II: diffuse or localized osteoporosis, sclerosis or cysts of femoral head (Figure 1(b)) FICAT stage III: sequestrum, break in articular cartilage, normal or increased joint space (Figure 1(c)) FICAT stage IV: decreased joint space, collapse of femoral head, acetabular osteoarthrotic changes (Figure 1(d)). The grade used for analysis was the higher of the two hips. The grade of radiographs was assessed by two experienced radiologists in our hospital who were blinded to the selection of patients.
Radiographic assessment of FICAT grading system: (a) FICAT grade I, (b) FICAT grade II, (c) FICAT grade III and (d) FICAT grade IV.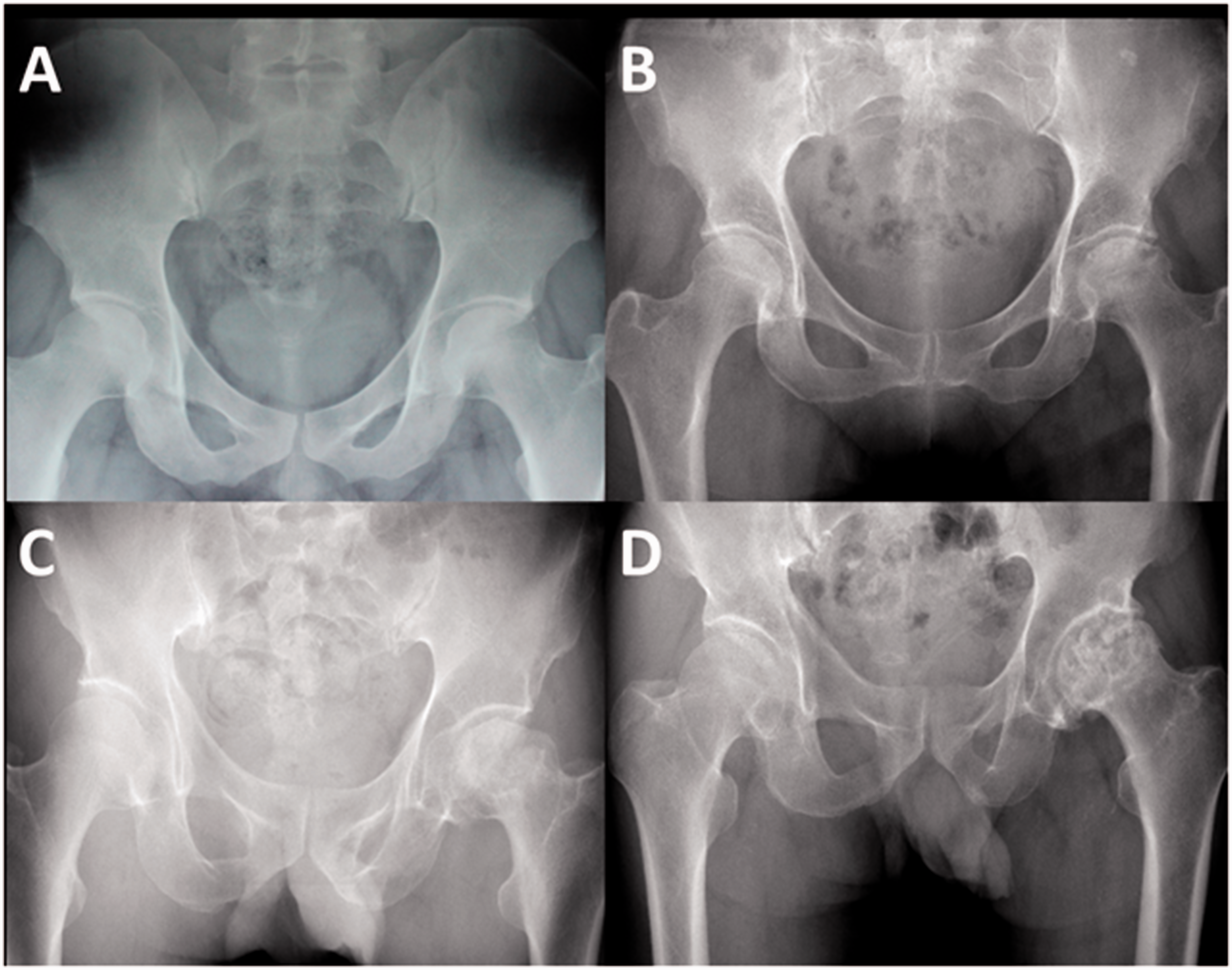
Evaluation of clinical severity
We used visual analogue scale (VAS) scores, Harris Hip Scores (HHS) and Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores to evaluate the clinical severity of non-traumatic ONFH patients. The VAS is a visual measuring instrument designed to document the degree of pain in individual patients, where 0 suggests no pain and 10 indicates extremely pain. 15 The HHS is a disease-specific health status scale that is frequently used to measure the function of hip as well as outcome of total hip arthroplasty. 16 The evaluation of HHS includes pain, function, absence of deformity and range of motion, with the total score of 100. Higher scores represent less pain and better function. The WOMAC system is a validated instrument designed specifically for the assessment of lower extremity pain and function in osteoarthritis (OA) of the knee or hip. 17 The VAS, HHS and WOMAC are all widely used based on clinical evidence and experience.
Statistical analysis
All data were analysed by a qualified statistician using Prism software for Mac, version 6.0c (GraphPad Software, San Diego, CA). Obtained results were presented as means ± SD or median. The Shapiro–Wilk test was evaluated to ensure normal distribution and homogeneity of variance of our data. Comparisons between two groups of individuals were done using Student’s independent t-test or Mann–Whitney U test for unpaired data. The significance of differences in the measured values among groups was performed using one-way analysis of variance (ANOVA). Bartlett’s test was used to test the homogeneity of group variances, followed with Tukey or Tamhane post hoc tests. Correlation among the concentration of α-MSH in the serum and disease severity was analysed by Pearson’s or Spearman’s correlation coefficient (r). Kappa values were generated by setting the observed proportion of agreement in relation to the proportion of agreement expected by chance with regard to the radiographic assessment. Kappa value>0.65 was considered significant. The area under curve (AUC) of serum α-MSH, adiponectin and IL-33 were calculated by ROC curve analysis to decide their values in radiographic progression. Decision to combine α-MSH, adiponectin and IL-33 to diagnose ONFH progression was performed by the decision tree method. The value of P < 0 .05 was considered statistically significant.
Results
Basic data of patients
Basic data of the patients.

Note: All data are given as the mean value ± SD or median. ONFH: osteonecrosis of the femoral head; BMI: body mass index; VAS: visual analogue scale; HHS: Harris Hip Scores; WOMAC: Western Ontario and McMaster Universities Osteoarthritis Index; a-MSH: a-melanocyte-stimulating hormone.
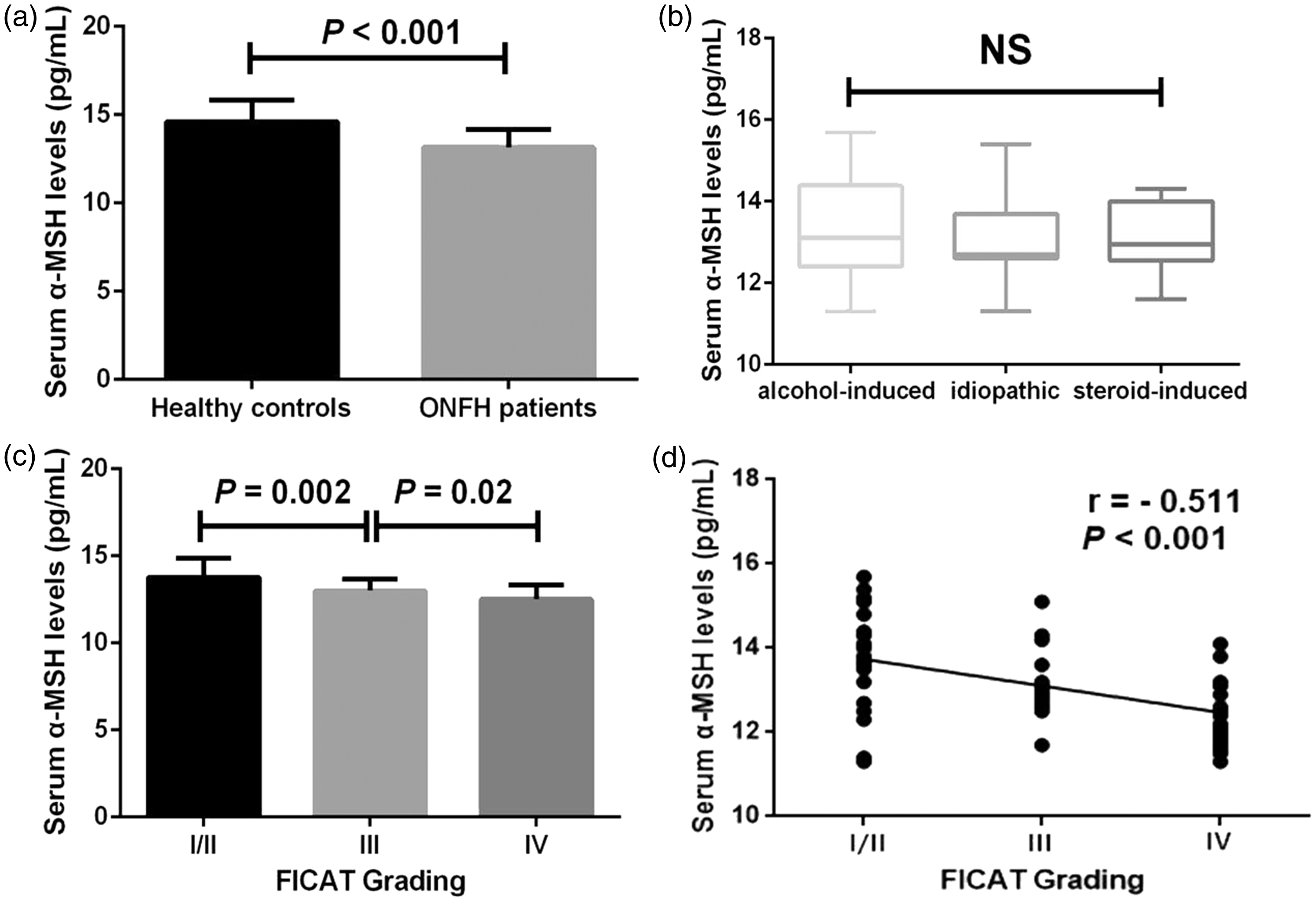
(a) Comparison of serum α-MSH concentrations between healthy controls and ONFH patients, (b) comparison of serum α-MSH concentrations among alcohol-induced, idiopathic and steroid-induced groups (c) comparison of serum α-MSH concentrations among different stages of FICAT grading (d) correlation of serum α-MSH concentrations with FICAT grading (*P < 0.05).
Serum α-MSH concentrations in non-traumatic ONFH patients with radiographic severity
The Kappa value was 0.72 for radiographic progression evaluation after calculation. In accordance with the FICAT classification, 29 patients were FICAT grade I/II, whereas 27 patients were FICAT grade III and 23 patients were FICAT grade IV. Serum concentrations of α-MSH were evaluated and compared in relation to radiological FICAT system. Non-traumatic ONFH patients with FICAT grade I/II showed significantly elevated α-MSH concentrations in serum compared with those with FICAT grade III (13.8 ± 0.2 pg/mL vs. 13.0 ± 0.1 pg/mL, P = 0.002). Non-traumatic ONFH patients with FICAT grade III had significantly higher serum concentrations of α-MSH compared with those with FICAT grade IV (13.0 ± 0.1 pg/mL vs. 12.5 ± 0.2 pg/mL, P = 0.02) (Figure 2(c)). α-MSH concentrations in serum of non-traumatic ONFH patients were negatively correlated with radiographic progression evaluated by FICAT grading (r = −0.511, P < 0.001) (Figure 2(d)).
Serum α-MSH concentrations in non-traumatic ONFH patients with clinical severity and biochemical indices
The correlation of serum α-MSH concentrations with clinical severity determined by VAS, HHS and WOMAC scores was explored. Serum α-MSH concentrations were negatively associated with VAS scores (r = −0.333, P = 0.003) (Figure 3(a)), HHS scores (r = 0.433, P < 0.001) (Figure 3(b)) and WOMAC scores (r = −0.392, P < 0.001) (Figure 3(c)). We further investigated the relationship between serum concentrations of α-MSH and damage marker IL-33 as well as protective marker adiponectin. We found that serum concentrations of α-MSH were negatively associated with IL-33 and were positively correlated with adiponectin (Figure 4). These differences still remain significant after adjusting for age and body mass index (BMI) (Supplementary Table 1).
Correlation of serum α-MSH concentrations with clinical severity determined by VAS (a), HHS (b) and WOMAC scores (c).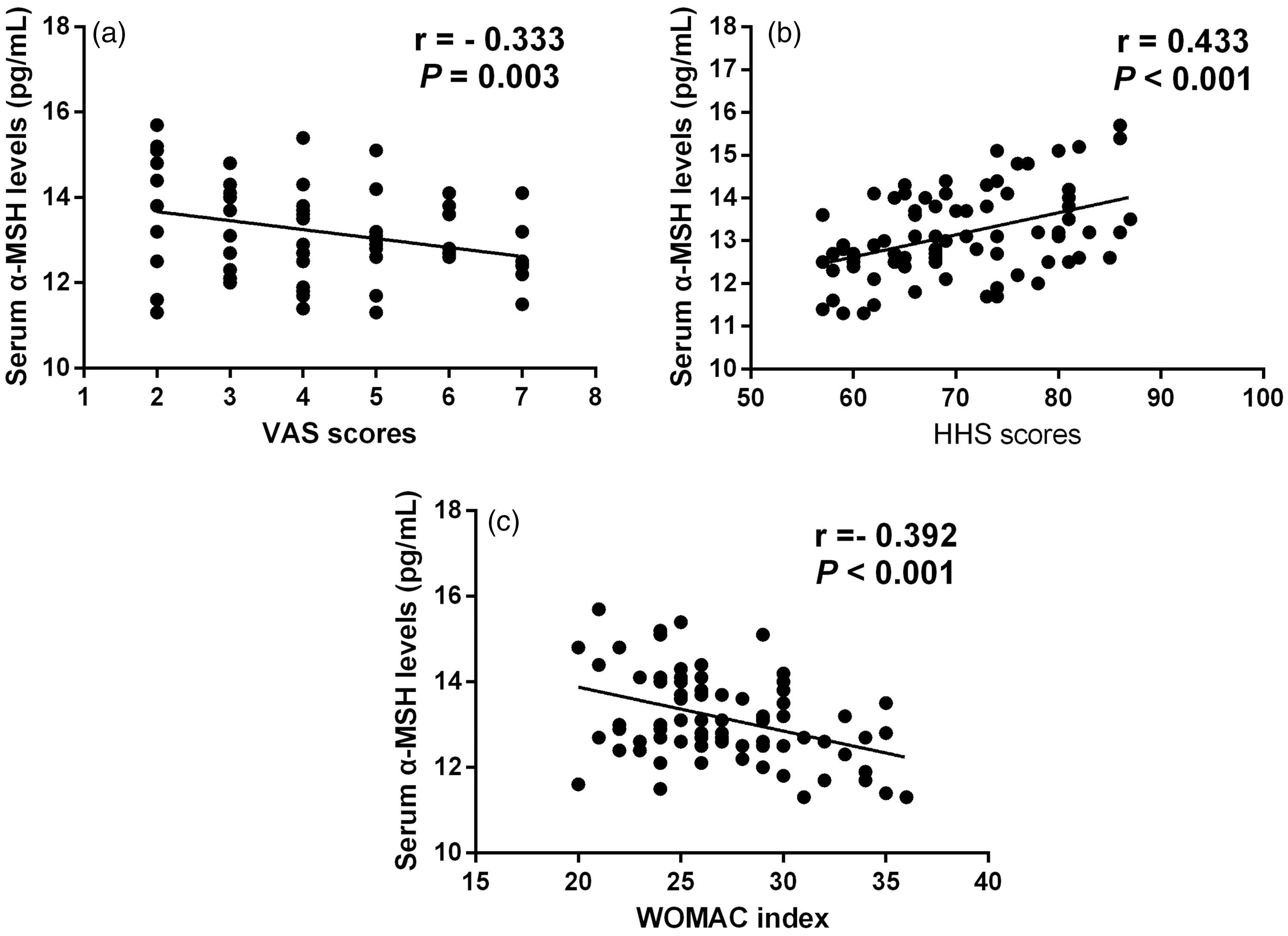
ROC curve analysis and combined analysis
We next performed ROC curve analysis to compare α-MSH with IL-33 and adiponectin to investigate the diagnostic value of these factors for radiographic severity of ONFH.
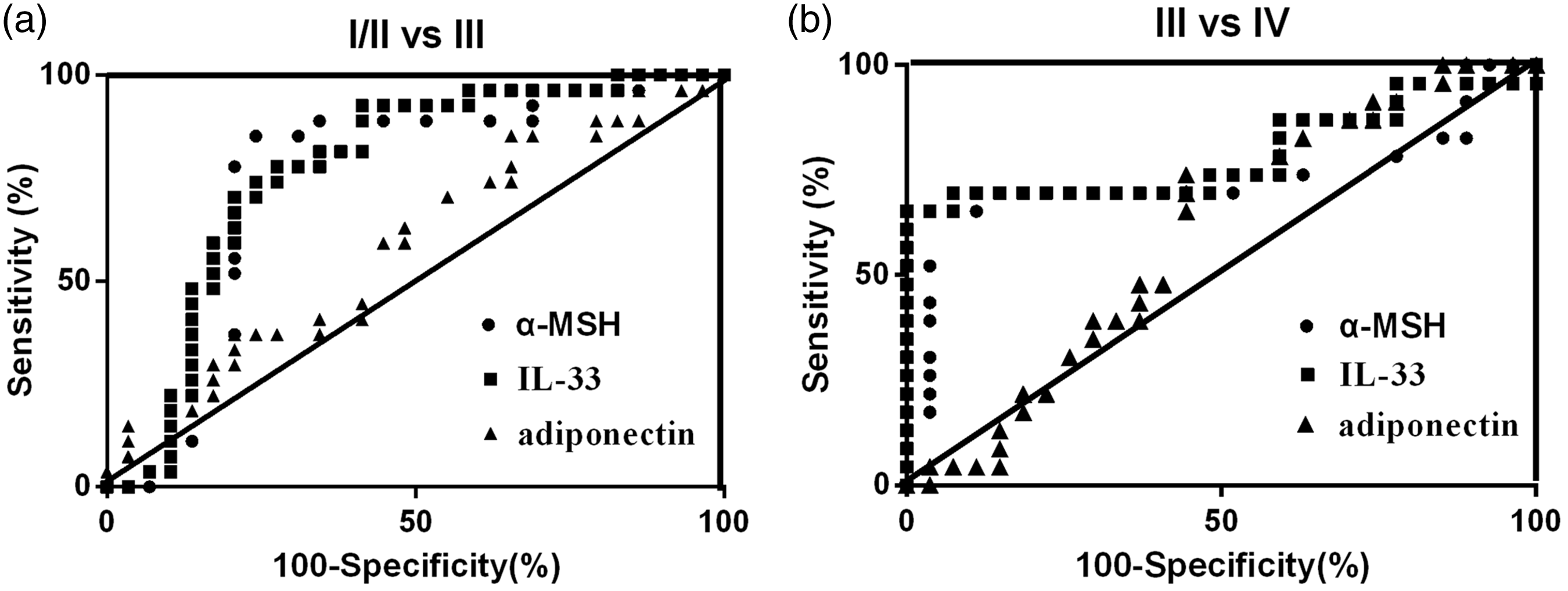
As shown in Supplementary Tables 2 and Table 3 and Figure 5, for both FICAT grade I/II vs. grade III and FICAT grade III vs. grade IV, α-MSH had a similar AUC (I/II vs. III: AUC = 0.750, P = 0.001; III vs. IV: AUC = 0.725, P = 0.006) with IL-33 (I/II vs. III: AUC = 0.771, P < 0.001; III vs. IV: AUC = 0.787, P < 0.001) and adiponectin (AUC = 0.587, P = 0.261) (Figure 3(c), Supplementary Tables 2 and 3). The results demonstrate that α-MSH show similar diagnostic value with IL-33 for assessing radiographic progression at both in early and medium–late stages. Decision tree for the combined analysis of α-MSH, IL-33 and adiponectin in ONFH FICAT Grading are shown in Supplementary Figure 2.
Correlation of serum α-MSH concentrations with IL-33 (a) and adiponectin (b). α-MSH: α-melanocyte-stimulating hormone; IL-3: interleukin-3. Area under curve of α-MSH, IL-33 and adiponectin for diagnosing radiographic progression by ROC curve analysis. (a) FICAT I/II vs. FICAT III (b) FICAT III vs. FICAT IV.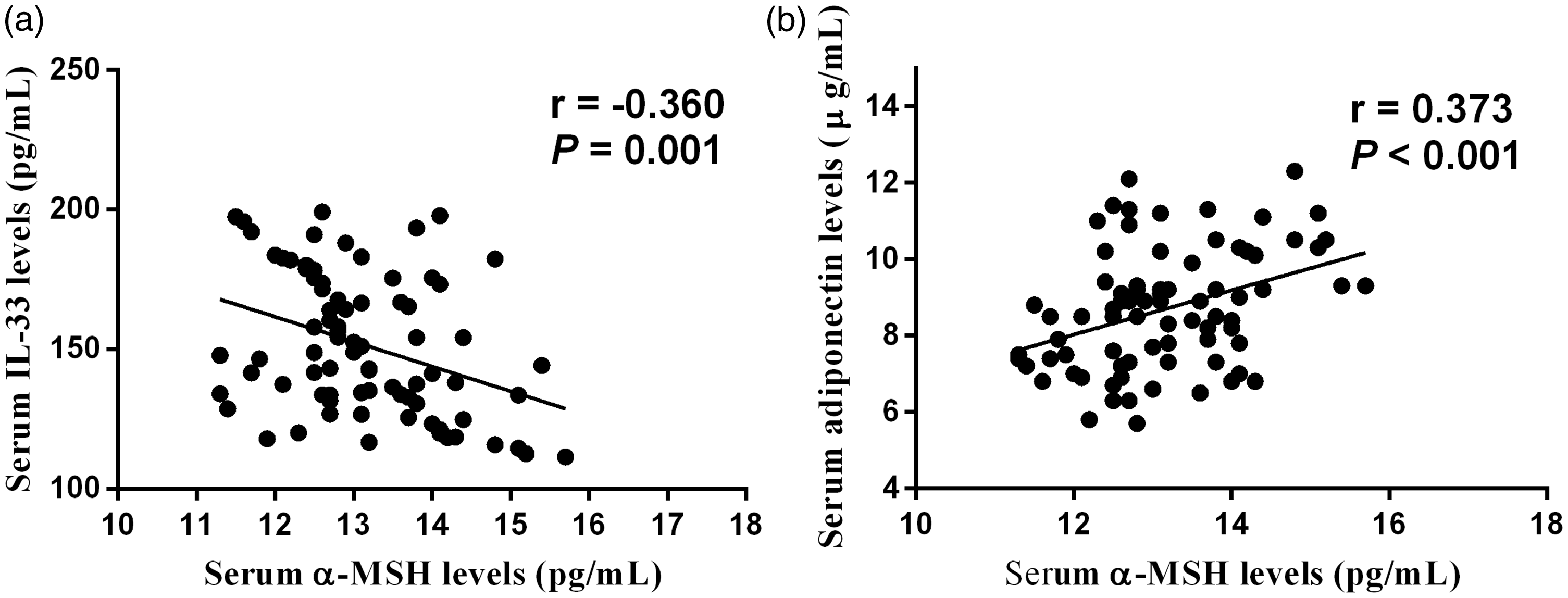
Discussion
The current study investigated the possible involvement of serum α-MSH concentrations in patients with non-traumatic ONFH. To our knowledge, this is the first published work on serum α-MSH concentrations in non-traumatic ONFH. We found that serum α-MSH concentrations were significantly lower in non-traumatic ONFH patients than in sex- and age-matched controls. In addition, serum α-MSH concentrations were negatively associated with radiographic progression and clinical severity. These differences still remain significant after adjusting for age and BMI.
The disease activity of ONFH is often in accordance with the changes of metabolism of bone protective or damage factors. In this respect, investigation of serum biomarkers may serve as a minimally invasive and quick method to reflect time course and changes in disease progression. Therefore, it has raised the hope that non-traumatic ONFH may soon be detected earlier and more accurately with novel and reliable biochemical markers than current possible radiographic changes.
The melanocortin α-MSH is a 13-amino acid peptide, synthesized within the brain and pituitary gland. The biological effects of α-MSH are mediated via MCRs. 18 Until now, at least five MCRs, including (MC1R to MC5R), have been characterized. 18 Recent evidence demonstrated that one of the five receptors, MC4R, could be the receptor mediating α-MSH's actions in osteoblasts. In vitro studies have identified that MC4R is present in an osteoblast-like cell line (UMR 106) and in primary osteoblasts, which is responsible to α-MSH treatment.19,20
One of the important causes of osteonecrosis is reduced blood flow. Under many pathological conditions, the vasculitis is attributed for the blood flow reduction. The possibility of vasculitis as the underlying cause of avascular necrosis of femoral head in systemic lupus erythematosus patients has been identified, implicating the role of vasculitis in the progression of ONFH. 21 In endotoxin-induced vasculitis murine model, single injection of α-MSH significantly suppressed the deleterious vascular damage and haemorrhage by inhibiting the sustained expression of vascular E-selectin and VCAM-1. 22 This phenomenon suggests that α-MSH may prevent the process of avascular necrosis by alleviating vasculitis.
Previous studies have shown that adiponectin and IL-33 involve in the progression the ONFH. 23 IL-33 is specifically released by necrotic cells constitutively expressed in human bone, in particular by osteocytes, osteoblasts. 24 IL-33 also modulates angiogenesis, vascular permeability, osteoclastogenesis and bone resorption, indicating that IL-33 may play a role in ONFH. One recent study by Zheng et al. found that elevated serum IL-33 concentrations were detected in non-traumatic ONFH patients and were positively related to FICAT grading system. 25 One another identified protective marker of ONFH, adiponectin, has been identified lower in ONFH patients than in healthy controls. 26 Moreover, adiponectin could act as an independent predictor for the presence of non-traumatic ONFH. 26 We found that serum α-MSH concentrations were negatively associated with IL-33 and positively related to adiponectin. In the next ROC curve analysis, we found α-MSH exhibiting a similar value of diagnosing radiographic progression compared with IL-33.
We explored serum α-MSH concentrations between males and females in ONFH patients, and we found that although serum α-MSH concentrations seem to be higher in male that in female, the differences did not reach significance (Supplementary File 1). Previous studies investigating the role of peripheral α-MSH in humans had somewhat conflicting results. Two studies have shown a correlation between serum α-MSH and measures of body composition as well as insulin resistance in obese male participants compared with lean controls.27,28 On the other hand, in a large study of young lean participants, no correlation between serum α-MSH and body composition parameters was found. 29 Obese men have been reported to have higher concentrations of α-MSH compared with controls.27,28 In women, no difference in serum α-MSH between obese subjects and controls was found. 30 Therefore, α-MSH concentrations seem to have a tighter correlation with BMI or bodyweight rather than sex, likely as most of our patients enrolled in our study. And also, these findings should be verified further in a larger sample size study in the future.
It should be considered that there are some inherent limitations to this study. First, our study was performed based on a relatively small number of enrolled patients. A further study with a random sample of a larger population will be required to make a more definite conclusion. Second, the study was limited to those patients who attended our hospital. Consequently, the results may not be directly applicable to subjects from other ethnic groups. Finally, this study was cross-sectional in its design and, therefore, no conclusions regarding cause and effect relationships can be drawn. However, prospective longitudinal studies can now be designed to document disease progression and define the precise role of α-MSH in non-traumatic ONFH.
To summarize, we demonstrated a significant decrease in the systemic expression of α-MSH and identified a strong negative association with the degree of radiographic progression and symptomatic severity in patients with non-traumatic ONFH. The results of the present study implicate that serum α-MSH may serve as a useful protective factor to reflect the disease severity of non-traumatic ONFH. And also, α-MSH concentrations in serum positively correlated with serum adiponectin and negatively with IL-33. This is the first study to identify such an association and should be followed up with a prospective study in order to substantiate these findings. Although potential mechanisms of this correlation and their cause-and-effect relationships are not fully illustrated, our findings may contribute to the understanding of the role of α-MSH in the pathogenesis of non-traumatic ONFH.
Footnotes
Supplementary material
Additional supplementary information may be found with the online version of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the ethical committee of Linyi People’s Hospital (LW2017001).
Guarantor
YYB.
Contributorship
YYB, MZ and LG designed and performed the study; YYB obtained ethical approval, analysed the data and wrote the first draft of the manuscript. YHT and CJJ were responsible for sample and data collection. LD and XMP were responsible for biochemical analyses. All authors reviewed, edited and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
