Abstract
Purpose
It has been demonstrated that circulating microRNA profiles are affected by physiological conditions. Several studies have demonstrated that microRNAs play important roles in the regulation of adiposity. However, few have investigated the relationship between circulating microRNAs and obesity, which has become a major public health problem worldwide. This study investigated the association between circulating microRNAs and obesity in a Japanese population.
Methods
Obesity parameters, such as subcutaneous and visceral fat adipose tissue, body fat percentage, and body mass index were assessed in a cross-sectional sample of 526 participants who attended health examinations in Yakumo, Japan. In addition, five circulating microRNAs (miR-20a, -21, -27a, -103a, and -320), which are involved in adipocyte proliferation and differentiation, were quantified using real-time polymerase chain reaction amplification.
Results
We compared the circulating microRNA concentrations in a percentile greater than 75th (high) with below the value (low) of subcutaneous adipose tissue, visceral fat adipose tissue, body mass index, and per cent body fat. For visceral fat adipose tissue, significant decrease in miR-320 expression was observed in high group. Also, for body mass index, significant change of miR-20a, -27a, 103a, and 320 expression level was observed in high group. Multiple linear regression analysis demonstrated that circulating levels of some microRNA such as miR-27a were significantly associated with subcutaneous adipose tissue, visceral fat adipose tissue, and body mass index.
Conclusions
Our findings support the need for further studies to determine whether such changes are consistent across different populations and whether the identified microRNAs may represent novel biomarkers to predict the susceptibility and progression of obesity-related disorders.
Keywords
Introduction
The balance between energy intake and expenditure is precisely regulated in vivo. It has been well established that chronic disturbance of this balance leads to metabolic diseases such as obesity. Obesity, which often induces insulin resistance, type 2 diabetes, and cardiovascular disease, 1 has become a major public health problem worldwide. Extensive attempts devoted to the determination of obesity-related genes have been made to understand its pathogenesis. 2 For example, inflammatory adipokines such as tumour necrosis factor alpha and interleukin-6 have been identified as potent inducers of obesity-related disorders. 3 However, the detailed pathogenesis of obesity has not yet been elucidated owing to its complicated mechanisms.
MicroRNAs (miRNAs) comprise small non-coding, single-stranded RNAs that are typically 22 nucleotides in length. They primarily regulate gene expression post-transcriptionally, through the suppression of messenger RNA translation or its degradation. 4 miRNAs are involved in the regulation of various biological processes including cell proliferation, apoptosis, metabolism, stress responses, and angiogenesis. 5 To date, more than 2000 different miRNAs have been described in humans and their number is still increasing (miRBase). 6 Notably, alterations of miRNA patterns frequently occur in various disorders; furthermore, recent studies have demonstrated that miRNAs also play important roles in the pathogenesis of obesity.5,6 The development of obesity appears to depend on adipose tissue hypertrophy and hyperplasia mediated by the differentiation of precursor cells into new adipocytes. 7 In particular, it has been demonstrated that certain miRNAs are involved in adipocyte formation. Among these, miR-21, -27a, -103a, and 320 have been reported as being involved in adipose proliferation and differentiation.8–11
miRNAs can be detected in body fluids (e.g. plasma, serum, urine, high-density lipoprotein (HDL), and saliva) as well as in tissues.12–14 Circulating miRNAs are stable in serum and are conveyed in exosomes and microvisicles.15,16 Therefore, circulating miRNAs may represent relatively non-invasive and specific diagnostic and/or prognostic molecular biomarkers for several diseases; in particular, they are considered as promising novel biomarkers of cancers, 17 cardiovascular diseases, 18 autoimmune disease, 19 and diabetes. 20 Given the prior evidence of adipose-related miRNAs in the blood, physiological conditions of adipose tissue such as its size may be affected by circulating miRNAs. 21 In support of this conjecture, studies involving patients with obesity have demonstrated that the concentrations of certain circulating miRNAs such as miR-140-5p are increased, whereas those of others including miR-15a were observed to decrease in these patients. 2 In addition, Pescador et al. 22 demonstrated that up-regulated serum concentrations of miRNAs including miR-138 are observed in patients with obesity. However, currently published studies lack long-term outcome data regarding disease activity, prognostic expectations, and pathogenesis. As most research related to circulating miRNAs in obesity examined cases of morbid obesity or patients undergoing treatments or with diseases caused by obesity,23,24 limited knowledge is available regarding the association between circulating miRNAs and obesity in healthy individuals. In particular, the association between circulating miRNAs and the health effects of being overweight has not, to our knowledge, yet been examined.
It is difficult to identify and estimate changing circulating miRNA profiles caused by obesity, because circulating miRNAs vary widely according to age and gender, and may also be influenced by environmental factors such as smoking and alcohol intake. 25 The association between circulating miRNAs and obesity in healthy individuals would need to be subjected to detailed adjustment for these influencing factors. Furthermore, the estimation of circulating miRNA concentrations should have adequate sensitivity and be performed with sufficient power to ensure adequate statistical validity of the result.
In consideration of these factors, in this study we examined the association between obesity and circulating miRNA concentrations in a sample of 526 individuals in a middle-aged Japanese population. In particular, we analysed several obesity parameters including visceral and subcutaneous adipose tissue level (VAT and SAT, respectively), body mass index (BMI), and body fat percentage. We focused on five miRNAs (miR-20a, -21, -27a, -103a, and -320), which have been shown to be involved in adipose proliferation and differentiation.9–11,15 We hypothesized that circulating miRNAs would exhibit changes related to biological parameters of obesity and that such miRNAs may represent novel biomarkers to predict the susceptibility and progression of obesity-related disorders.
Experimental
Study subjects
Health examinations of inhabitants aged ≥ 39 years have been performed in an area of Hokkaido, Japan since 1982.26–28 Cross-sectional and longitudinal studies of lifestyle-related diseases have enrolled individuals who participated in these health examinations. The present cross-sectional study is part of the ongoing Yakumo Study. Our study comprised 556 residents who attended health examinations in Yakumo, a town in Hokkaido, Japan, in August 2012. After excluding 30 subjects who declined to participate in the study, data from the remaining 526 subjects (219 men and 307 women aged 39 years and older) were included in our analysis. All participants provided written informed consent. The Ethics Review Committee of Fujita Health University approved the study protocol (approval number 11-101).
Health and lifestyle habits (including smoking, alcohol and drug consumption, and history of major illness) were assessed via a questionnaire administered by trained nursing staff. 4 Anthropometric indices (height, weight, and waist and hip circumferences) and blood pressure were measured during the health examination. Body height and weight were measured, and BMI was calculated as weight divided by the height squared (kg/m2). Estimated glomerular filtration rate (eGFR) was used to assess kidney function, according to the following equation: eGFR (mL/min/1.73 m2) = 194 ×creatinine−1.094 × age−0.287 ( × 0.739 if female).
Assessment of obesity parameters
Body fat percentage was measured using bioelectrical impedance analysis. 29 The bioelectrical impedance analyses were performed using the Tanita MC780 multifrequency segmental body composition analyser (Tokyo, Japan). The thickness of VAT and SAT was assessed by ultrasound using a ProSound α7 with a UST-9130 convex probe (Hitachi Aloka Medical, Ltd, Tokyo, Japan) operated by three registered medical sonographers (Japan Society of Ultrasonics in Medicine). VAT thickness was defined as the distance (cm) from the peritoneum to the vertebral bodies and SAT thickness was defined as the depth (cm) from the skin to the linea alba.30,31 The ultrasonography images were independently reviewed by each sonographer.
Blood biochemistry
Fasting blood samples were taken during the health examination and sera were separated from blood samples by centrifugation within 1 h of collection. Immediately, serum samples were stored in a deep freezer at −80℃ until analysis. Clinical chemistry exams were assayed using an auto-analyser (JCS-BM1650, Nihon Denshi Co., Tokyo, Japan) in the laboratory at Yakumo General Hospital.
The assessment of serum miRNAs utilized a method similar to that in our previous study. 4 We used TRIzol reagents (Invitrogen, Carlsbad, CA, USA) to isolate serum miRNAs following manufacturer instructions. In brief, proteins were denatured in 10 volumes of TRIzol solution to one volume of serum, vortexed, then incubated at room temperature for 5 min. Next, 5 µL of 5 nM Syn-cel-miR-39 miScript miRNA Mimic was added to all samples to provide a normalized control, then each sample was vortexed immediately. Aqueous and organic phases were separated by adding 0.2 volumes of molecular grade chloroform (Wako, Osaka, Japan), vortexed at the maximum setting for 30 s, followed by centrifugation at 12,000 r/min for 15 min at 4℃. The aqueous phase was rapidly transferred to a new tube and the TRIzol protocol continued. Finally, total RNA was dissolved in 15 µL of RNase-free water. Quantitative real-time reverse transcription polymerase chain reaction (qRT-PCR) was performed using a miScript System (Qiagen, Valencia, CA, USA) that included specific primers for miRNAs, following manufacturer instructions. The RT reaction system contained 1 µL of miScript Reverse Transcriptase Mix, 4 µL 5 × miScript RT Buffer, and 15 µL of RNase-free water. Samples were allowed to react for 60 min at 37℃ and 5 min at 95℃ in a Bio Rad T100 System (Bio-Rad Laboratories, Foster City, CA, USA), and then were diluted with 200 µL of RNase-free water. The reaction system of qPCR contained 10µL of SYBR Green PCR Master Mix (Qiagen), 2µL of miScript universal primer, 2 µL of specific primer, 2 µL of cDNA, and 4 µL of RNase-free water; PCR cycles were completed at 95℃ for 15 min, 45 cycles at 94℃ for 15 s, 55℃ for 30 s, and 70℃ for 30 s. Real-time PCR was performed using an ABI PRISM 7900 Sequence Detection System (Applied Biosystems, Foster City, CA, USA). The relative expression of each miRNA was calculated using the comparative cycle threshold method with spiked cyn-39 as the normalized internal control. 4 We also prepared vectors containing miRNA sequences as standard samples. We ensured the quality of miRNA by quantifying standard samples.4,14 The expression levels of miRNA were calibrated relative to pooled serum from subject controls. 4
The prevalence of obesity, defined by the World Health Organization (WHO) as BMI ≥ 30 is no more than 3% in the Japanese population, in contrast to the 10–20% in Europe and the USA. In Japanese adults, BMI cut points of >25 kg/m2 indicating overweight are widely accepted, as is the terminology of overweight for the lesser extreme and obesity for the greater extreme. BMI of 25 as the cut-off point of obesity are defined for the following reasons: obesity-related adverse effects increase for a BMI ≥ 25, the best combination of sensitivity and specificity for detecting subjects with multiple risk factors is a BMI of 25, and using this standard preserves the international coordination of the WHO criteria and the recommendation of the Steering Committee of the Western Pacific Region of WHO. In this study, 75th percentile of study subjects had a BMI greater than 25.1 kg/m. 2 Over 75th percentile of study subjects was defined as obesity. Thus, miRNA concentrations are compared between the ≥ 75th percentile (obesity) and the < 75th percentile (normal) of obesity parameters (SAT, VAT, BMI, and per cent body fat). The top quartile of our subjects had a SAT, VAT, and per cent body weight greater than 16.6 cm, 67.8 cm, and 29.4%, respectively.
Statistical analysis
All statistical analyses were conducted using JMP version 10.0 (SAS Institute, Cary, NC, USA). Because serum concentrations of miRNAs were distributed logarithmically, these data were log-transformed for the analysis. Serum concentrations of aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, γ-GT, triglyceride, and high-density lipoprotein-cholesterol (HDL-c) are represented as geometric means and 25–75th percentile ranges. Other variables are represented as the mean ± SD. Relationships between circulating miRNA concentrations and the obesity index, which comprises VAT, SAT, BMI, and body fat percentage, are given by Pearson's correlation coefficient and multiple linear regression analysis adjusted for sex, age, smoking habit, drinking habit, systolic blood pressure, haemoglobin A1c (HbA1c), eGFR, and drug use for heart disease. A p < 0.05 was considered statistically significant.
Results
Characteristics of study subjects.
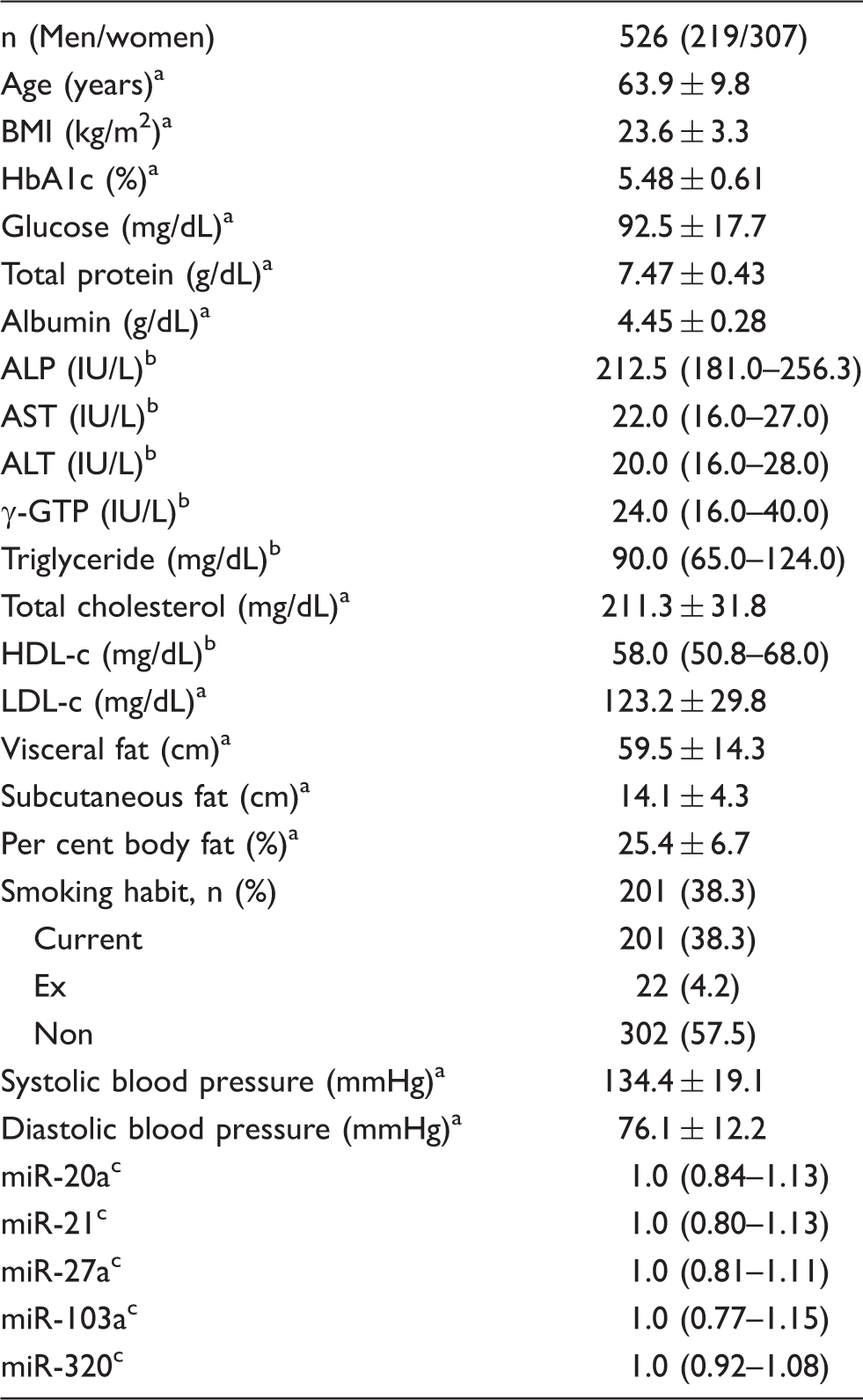
ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; BMI: body mass index; HbA1c; haemoglobin A1c; HDL-c: high-density lipoprotein-cholesterol; γGTP: γglutamyltranspeptidase; LDL-c: low-density lipoprotein-cholesterol.
Data are expressed as mean value ± standard deviation.
Data are expressed as geometric mean values and 25–75th percentheses.
Data are expressed as median values and 25–75th percentheses.
At first, we compared the circulating miRNA concentrations in a percentile greater than 75th (high) with below the value (low) of SAT, VAT, BMI, and per cent body fat. The amount of circulating miR-103a concentrations was significantly increased in high group of SAT (Figure 1(a)). Increasing tendency was also observed in miR-27a (p = 0.075). Significant decreased miR-320 expression level was observed in high group in VAT (Figure 1(b)). Concerning BMI, changed concentrations of miR-20a, -27a, 103a, and 320 were observed in high group (Figure 1(c) to (f)). Increasing tendency of miR-20a was observed in high group of percentage body fat (p = 0.056).
Comparison of miRNA concentrations (miR-20a, -21, -27a, -103a, and 320) between the top 25% (high) and the bottom 75% (low) of obesity parameters (SAT (a), VAT (b), BMI (c), and per cent body fat (d–f)). Circulating miRNA was quantified with real-time PCR method. *p < 0.05, **p < 0.01. BMI: body mass index; miRNAs: microRNAs; SAT: subcutaneous adipose tissue; VAT: visceral adipose tissue.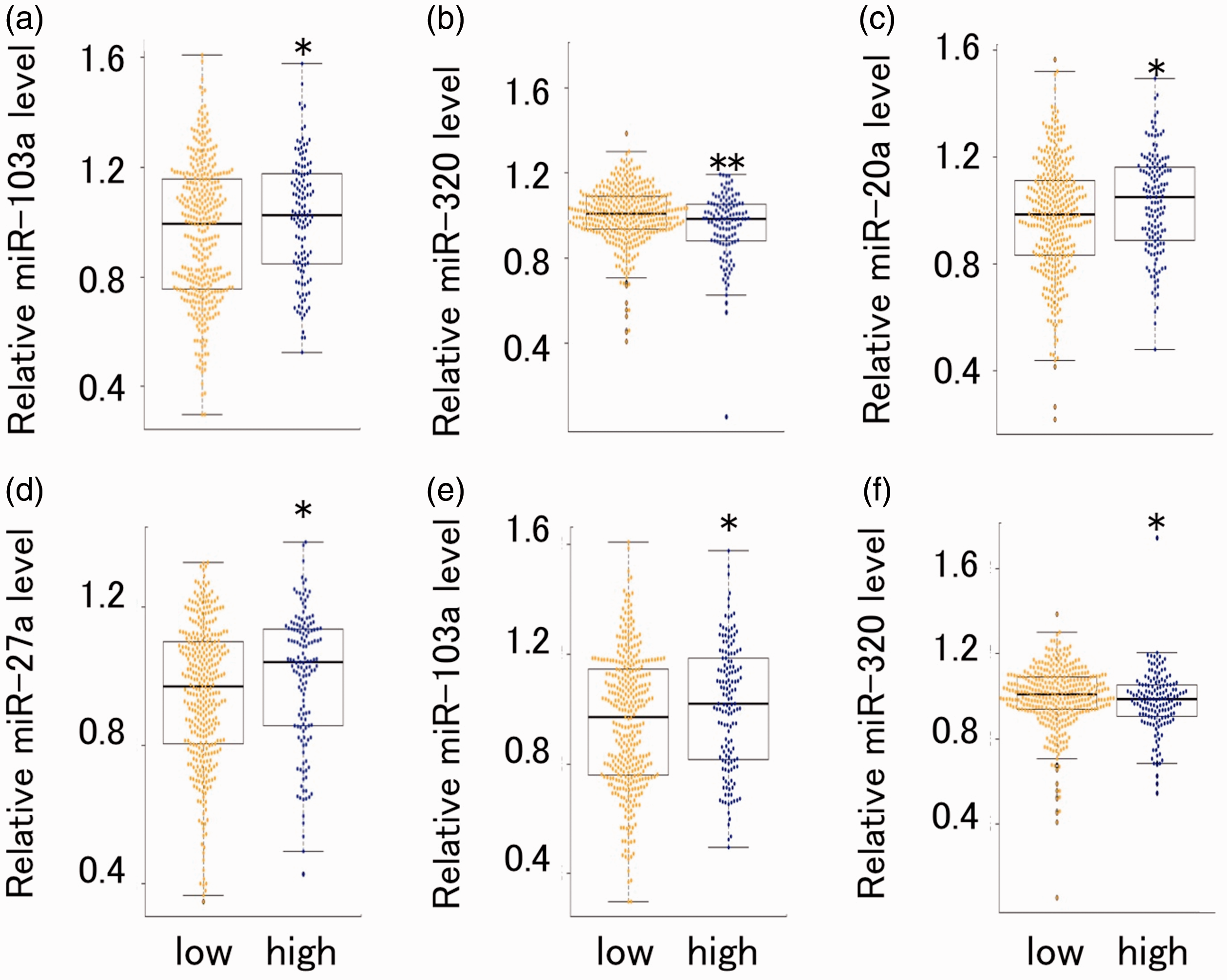
Pearson's correlation analyses of miRNA with SAT, VAT, BMI, and per cent body fat.
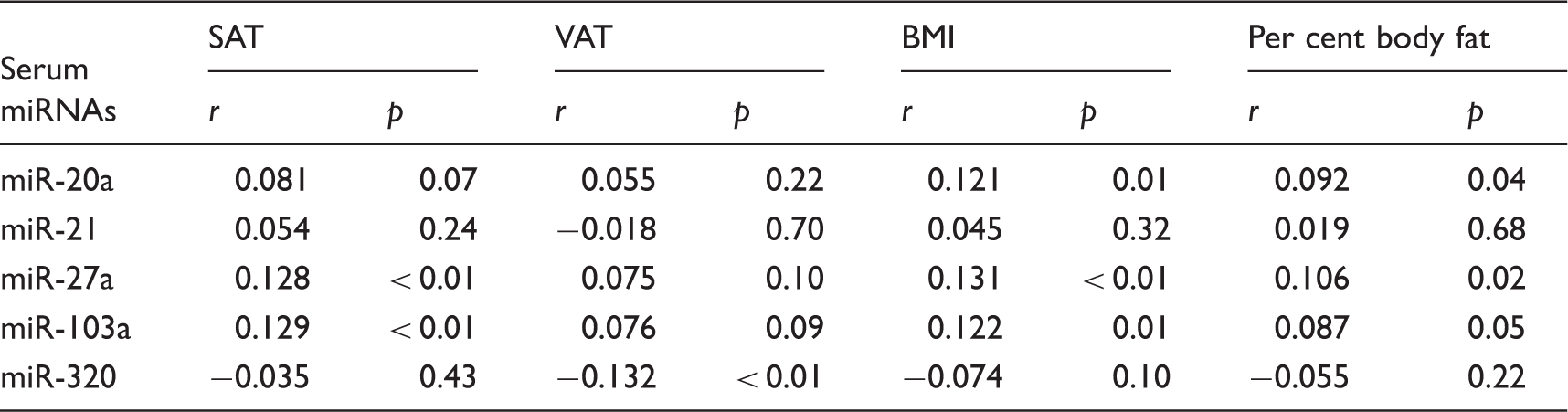
BMI: body mass index; miRNAs: microRNAs; SAT: subcutaneous adipose tissue; VAT: visceral adipose tissue.
r, Pearson's correlation coefficient.
Multiple linear regression analyses of miRNA with SAT, VAT, BMI, and per cent body fat.
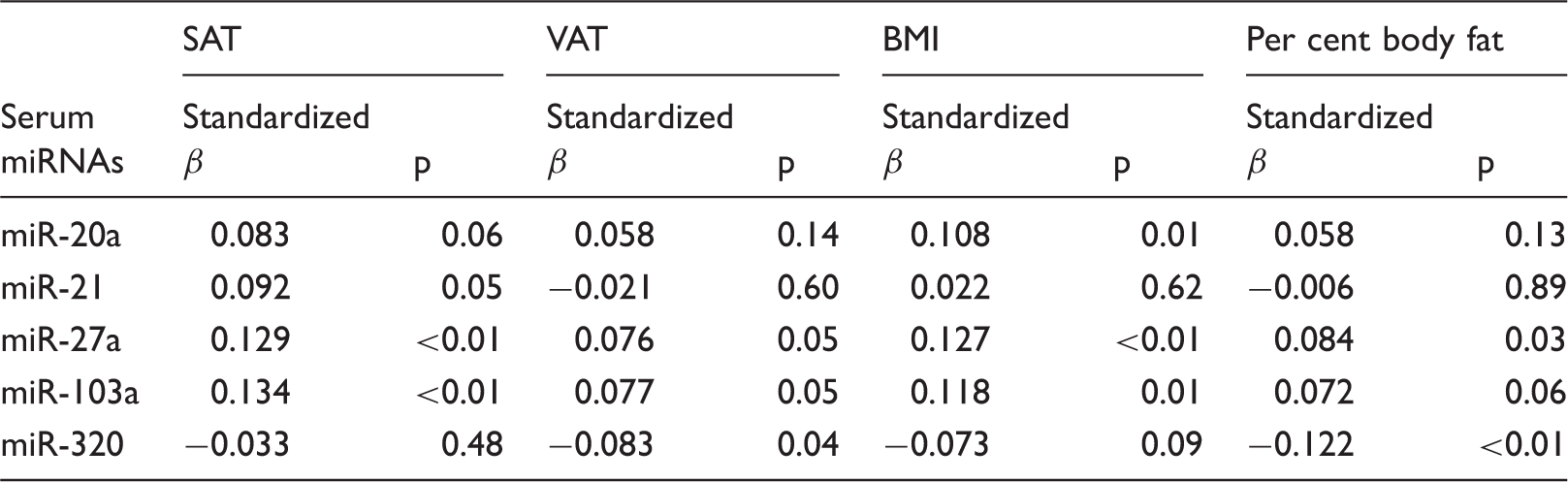
BMI: body mass index; miRNAs: microRNAs; SAT: subcutaneous adipose tissue; VAT: visceral adipose tissue.
Discussion
In the present study, we analysed circulating miRNAs in a sample of 526 individuals who participated in health examinations. We found the altered expression levels of circulating in the high group of obesity index. For example, circulating miR-320 concentrations were significantly decreased in high group. Furthermore, we reveal the association between circulating miRNAs and obesity. In particular, circulating concentrations of miR-21, miR-27a, and miR-103a were significantly associated with SAT level, whereas serum concentrations of miR-27a, miR-103a, and miR-320a were significantly associated with VAT level. These results indicate that circulating miRNAs may be useful as biomarker for obesity and offer new information to better understand the pathogenesis of obesity.
To the best of our knowledge, this is the first report demonstrating that circulating miRNAs differentially correlate with SAT and VAT. We revealed that both miR-27a and miR-103a associate with SAT and VAT; conversely, altered expression levels of miR-21 and miR-320 were specifically associated with SAT and VAT, respectively. The major finding of the present study was that VAT, but not SAT, was associated with significant differences in circulating miRNAs in the general population.
The profile of circulating miRNA may vary by various factors. For example, it is well known that physical condition, such as drinking alcohol and smoking cigarettes, can modulate the expression of miRNAs 32 For example, we have previously reported that cigarette smoking is associated with the elevated expression of various circulating miRNAs. Furthermore, ethanol consumption produces a small increase in circulating miR-122 in healthy individuals. 32 In addition, Sawada et al. 33 reported that acute exercise alters some circulating miRNA concentrations such as miR-146 and -221. Collectively, these data suggest that circulating miRNA expression exhibits high sensitivity to physical conditions. Therefore, the identification of circulating miRNAs as biomarkers may be limited unless the effect of physical condition is closely examined. Concerning this point, we obtained several measures related to the study population (SAT, VAT, BMI, and per cent body fat adjusted for sex, age, blood pressure, HbA1c, eGFR, γGTP, smoking habit, and drinking habit), which enabled us to clarify the association between specific miRNAs and the obesity index using multiple regression analyses.
We revealed that serum concentrations of miR-27a, miR-103a, and miR-320a were significantly associated with VAT level. Notably, miR-320 was associated with VAT but not SAT level, independent of BMI and general obesity. A previous study related to the physiological activity of miR-320 using the 3T3-L1 cell line demonstrated that miR-320 expression in insulin-resistant adipocytes was approximately 50 times higher than that in normal cells. 11 Furthermore, the authors demonstrated that an anti-miR-320 oligo improved insulin-induced glucose uptake in insulin-resistant adipocytes and that miR-320 targets phosphoinositide-3-kinase, regulatory subunit 1. 11 In particular, up-regulated circulating miR-320 concentrations might be associated with adverse effects of VAT, as miR-320 has been shown to regulate endothelial function and angiogenesis in diabetes. 34 According to this report, exosomes released from type 2 diabetic rat inhibited endothelial cell proliferation, migration, and tube-like formation through the transfer of miR-320 into endothelial cells. 34 Notably, excess VAT, rather than SAT, has been recognized as a major predictor of cardiometabolic disease, 35 although the mechanism responsible for this phenomenon remains unknown. Our study may therefore lead to a new understanding of the pathogenesis mechanism for obesity-caused cardiometabolic disease. In turn, circulating miR-320 might serve as a useful biomarker for the prediction of all-cause mortality by obesity including cardiometabolic disease.
It has been reported that VAT causes hypertension and diabetes.36,37 Thus, a circulating miRNA that reflects the amount of VAT may represent a promising biomarker. However, to date, no reports have been published concerning biomarkers for VAT, although specific circulating miRNAs have been shown to exhibit enhanced expression in these diseases themselves. For example, higher expression concentrations of circulating miR-103, -27a, and were observed in hypertension and hyperlipidemia, 38 and altered circulating miR-29 and -103 concentrations were found in diabetes. 39 However, cause and effect relationship between miRNA and disease remains unclear. It should be noted that the miRNAs analysed in current study were altered prior to the onset of disease; therefore, the current results may suggest that these miRNAs may have a causal role with respect to disease pathogenesis. Furthermore, it has been demonstrated that VAT and SAT exhibit different physiological roles in view of their unique gene expression patterns. 40 The information provided by the current study related to their differing contributions to circulating miRNA expression may thus also provide new insights into the pathological mechanisms of obesity-related disorders themselves from the point of view of circulating miRNAs.
It is well established that adipose-derived stem cells differentiate into adipocytes, which leads to adipogenesis. 41 In addition to hormones and transcriptional factors, miRNAs may also play important roles in adipocyte differentiation, as suggested by several reports. 42 Notably, the findings related to some of these identified miRNA are consistent with the results of the current study. For example, Lin et al. 43 demonstrated that the overexpression of miR-27 inhibited adipocyte formation. They also clarified that PPARγ and C/EBPα, which act as master regulators of adipogenesis, represent targets of this miRNA. Furthermore, a recent study has demonstrated that exosomes contain miR-27a and may transfer this into adipose tissue. 44 As miRNAs transferred by this means are thereby able to exert their biological activity in exosome-recipient cells, 45 the dys-regulation of miR-27a (as observed in the current study to be associated with increased SAT and VAT) may be involved in adipose tissue dysregulation in the those cells
There is a report about the association between inflammation and obesity. 46 Some miRNA such as miR-27a and miR-103 are up-regulated in obesity. Taking into consideration that identified miRNAs were present in the serum, the up-regulated inflammatory response in obesity might be associated with the alternation of the circulating miRNA profile. To date, it has been reported that circulating miRNAs regulate cytokine production and inflammatory response. For example, miR-27a regulates inflammatory response by targeting IL-10 expression in macrophage. 47 Up-regulated miR-27a expression level observed in this study may enhance inflammatory response. Also, Hartmann et al. 48 demonstrated that miR-103a induces inflammation in endothelial cells. Collectively, obesity-induced inflammation may be explained by alternation of circulating miRNA concentrations. It is well known that communications between cells via circulating miRNAs play dual roles in modulating immunity. 49 The biological activity of circulating miRNA might vary with obesity-related inflammation. This phenomenon is potentially a novel clue for elucidating obesity-related pathological mechanisms.
There are reports indicating the association between circulating miRNA and obesity-related disease such as diabetes and cardiovascular disease.1,25 Identified miRNA in this study may be associated with inflammation and VAT, suggesting that these miRNA may be involved in pathogenesis of obesity-related diseases. Thus, identified circulating miRNAs might be a potential biomarker for obesity-related disease. However, there is a limitation in this study. The limitation of our study is that, similar to previously published studies, it lacks long-term outcome data regarding causal relationships between expression of these miRNAs and risk for obesity-related disease. However, given that the identified circulating miRNAs have been associated with obesity-related diseases, our healthy population-based data suggest that the circulating concentrations of these miRNAs may have been altered prior to disease onset. Therefore, the miRNA concentrations might represent novel biomarker to predict the susceptibility and progression of obesity-related disorders. In the future, circulating miRNA could have important clinical benefits in terms of development therapies for the prevention and/or for the treatment of obesity-related diseases.
Quantitative assessment of VAT is important for the evaluation of obesity. Technically, non-invasive imaging technologies such as computed tomography (CT), magnetic resonance imaging (MRI), dual-energy X-ray absorptiometry, and ultrasound are available to quantify the specific amounts of VAT and SAT in the body. 50 CT and MRI are considered the gold standard methods for assessing different abdominal adipose tissue compartments. Ultrasound is less accurate and precise than CT and MRI and, in particular, is less specific when differentiating between VAT and SAT, which do not cover the whole abdomen, and thus only provides proxy estimates of the total volumes of VAT and SAT. 50 However, the use of gold standard methods is limited in large epidemiological studies. Therefore, we assessed the thickness of VAT and SAT by ultrasound operated by registered medical sonographers. A subsequent study would strengthen the knowledge obtained in our study.
Conclusions
In this study, we determined that circulating miR-21, miR-27a, and miR-103a were associated with SAT level whereas VAT level was associated with miR-27a, miR-103a, and miR-320. Thus, we revealed an association between circulation miRNAs and VAT/SAT in the middle-aged Japanese population. This information may provide a basis to better understand the pathogenesis of obesity and supports the need for further studies to determine whether such changes are consistent across different populations and whether the identified miRNAs may represent novel biomarkers to predict the susceptibility and progression of obesity-related disorders.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by the Ministry of Education, Culture, Sports, Science, and Technology of Japan via a Grant-in-Aid for Scientific Research and Grant-in-Aid for Young Scientists, 2014–2017 (No. 26860375).
Ethical approval
All participants provided written informed consent. The Ethics Review Committee of Fujita Health University approved the study protocol (approval number 11-101).
Guarantor
KS.
Contributorship
EM, HY, and KS analysed and interpreted the data, and were a major contributor in writing the manuscript. NI, KS, KO, YA, YT, MK, MY, and GM acquired the data. NH interpreted the data. HI and KO were major contributors in writing the manuscript and revised it critically for important intellectual content. All authors read and approved the final manuscript.
