Abstract
Background
There are limited data regarding the contribution of advanced glycation end products in the presence of rheumatoid arthritis. We investigated whether serum Nɛ-carboxymethyllysine and pentosidine were related to the presence and the severity of rheumatoid arthritis.
Methods
Eighty patients with rheumatoid arthritis and 30 control subjects were included in a cross-sectional study. The severity of rheumatoid arthritis was assessed using the disease activity score for 28 joints. Serum Nɛ-carboxymethyllysine and pentosidine were measured by enzyme-linked immunosorbent assay.
Results
Serum Nɛ-carboxymethyllysine and pentosidine concentrations were significantly higher in patients with rheumatoid arthritis vs. control subjects (P < 0.001). Serum Nɛ-carboxymethyllysine and pentosidine concentrations were significantly higher in rheumatoid arthritis patients with high disease activity vs. rheumatoid arthritis patients with moderate disease activity (P < 0.001, P = 0.019, respectively). A multiple logistic regression analysis demonstrated that Nɛ-carboxymethyllysine was independently associated with the presence of rheumatoid arthritis (OR = 1.21, 95% CI: 1.05–1.39, P = 0.006). Furthermore, in a multivariate stepwise regression analysis, Nɛ-carboxymethyllysine was independently correlated with disease activity score for 28 joints (standardized β = 0.43, P = 0.001).
Conclusion
Serum Nɛ-carboxymethyllysine and pentosidine were increased during rheumatoid arthritis, and Nɛ-carboxymethyllysine was independently associated with the presence and the severity of rheumatoid arthritis.
Keywords
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease characterized by inflammation and joint damage with a heterogeneous course and different pathogenic mechanisms. 1 Chronic inflammation can lead to oxidative stress and consequently the formation of carbonyl, reactive carbonyl compounds, which are partly transformed into advanced glycation end products (AGEs).2,3 Non-enzymatic glycation begins with interaction and link between the carbonyl group of a reducing sugar and an aminoterminal group of protein.4,5 Complex rearrangements result in early AGE forms, called Amadori products (haemoglobin A1C [HbA1c]); progressively, they result in more stable AGE precursors 6 and lately in irreversible long-lasting glycoxidation of the proteins, such as Nɛ-carboxymethyllysine (CML), carboxyethyllysine and pentosidine (an AGE with autofluorescent properties).5,6 The increased concentration of AGEs has been reported in the pathophysiology of diabetes and its vascular complications. Kerkeni et al. 7 showed that serum AGEs and especially pentosidine concentrations are related with the presence of diabetic retinopathy, and pentosidine was independently correlated with the severity of diabetic retinopathy. In other studies, Kerkeni et al. 8 find the pentosidine as a biomarker for microvascular complications in type 2 diabetic patients and it was related to the presence and the severity of coronary artery disease. 9 RA disease was an inflammatory disease, and many factors are implicated in its development and induce the severity of RA. Developing new biomarkers to investigate physiopathology mechanisms can ultimately to new treatments. There are limited data regarding the contribution AGEs in the presence of RA.10,11 The aim of this study is that we, firstly, investigated whether serum CML and pentosidine were related to the presence of RA and, secondly, whether these biomarkers are implicated in the severity of RA.
Subjects and methods
Study population
The study protocol was approved by the regional ethical research committee. Written informed consent was obtained from all patients before the enrolment. This study includes 110 participants. Eighty patients recruited in the Rheumatology Department of University Hospital Fattouma Bourguiba Monastir were invited to participate in this study. This study meets the American College of Rheumatology 1987 (ACR) criteria for the diagnosis and classification of RA. 12 These patients completed a questionnaire detailing risk factors, treatment and details of RA. Each patient was clinically evaluated, with at least one chart review to confirm history. Thirty healthy normal people (without any disease) were enrolled as control; they were matched with the patients for age and sex.
Biochemical investigations
In all subjects, blood was drawn in the morning after overnight fasting, and then the samples were stored at −80℃ until analysis. Serum glucose, serum creatinine, uric acid and lipid concentrations (high-density lipoprotein [HDL], low-density lipoprotein [LDL], cholesterol and triglyceride) were measured using enzymatic method (CX9 Auto-chemical analysis instrument (Beckman CX9, USA). HbA1c was measured using G7 HPLC Analyser (Tososh Europe N.V). High-sensitive C-reactive protein (hs-CRP) was quantified according to the instructions of manufacturers using particles enhanced immunonephelomitric method (BNII, Dade Behring). Pentosidine and CML were quantitatively determined in serum by enzyme-linked immunosorbent assay (ELISA) kits provided by YH Biosearch Laboratory (Shanghai, China) according to the manufacturer's instructions. Pentosidine (or CML) present in the sample was probed with an antipentosidine (or anti-CML) antibody, followed by a horseradish peroxidase-conjugated secondary antibody. Pentosidine or CML content in the sample was determined by comparison with a standard curve prepared from pentosidine standards ranging 0–12 μg/L or from CML standards ranging 0–160 μg/L. The absorbance of the final colour product was read at 450 nm by a Bio Rad multiplate reader (Model 680). Anticyclic citrullinated peptides (anti-CCP) were quantitatively determined by a second generation ELISA (Euroimmun®, Germany). The threshold of this test was 5 IU/mL. IgM rheumatoid factor (IgM-RF) was measured by the Waaler–Rose and the test latex agglutination test (Fumouze®, France). The threshold of the two tests was 12 IU/mL.
Disease activity assessment
Disease activity was used to assess the disease activity score for 28 joints (DAS-28 score). 13 Ranges of DAS-28 correspond with disease activity. DAS-28 score <2.6 indicates remission. DAS-28 score from 2.6 to 3.2 indicates low disease activity. DAS-28 score from 3.2 to 5.1 indicates moderate disease activity. DAS-28 score above 5.1 is considered high disease activity. Traditionally, disability was assessed using the health assessment questionnaire (HAQ), a score by questionnaire that examines the disabilities that RA patients encounter in daily living and activities. 14 The final HAQ index ranges from 0 to 3; HAQ scores <0.3 are considered normal. 15
Statistical analysis
All analyses were performed using SPSS version 17.0 (SPSS Inc., Chicago, IL, USA). Continuous variables were expressed as mean ± SD or median (interquartile range), while percentages were used to express categorical variables. ANOVA test was used to compare the continuous variables between subgroups. An unpaired Student’s t-test was used for normally distributed variable, and the Mann-Whitney U-test was used for skewed variables. To determine the independent parameters correlated with the presence of RA, the parameters that correlated significantly in the univariate analysis were tested using multiple backward stepwise regression analysis. A logistic regression analysis was performed to assess the association between AGEs and the presence of RA. A receiver operating characteristic (ROC) curve analysis was performed to identify the optimal cut-off points of serum AGEs concentrations for predicting RA. The area under the curve (AUC) value was calculated to determine the accuracy of the test. A P < 0.05 was considered as statistically significant.
Results
Clinical characteristic of the studied population
Characteristics of patients with RA and control subjects.
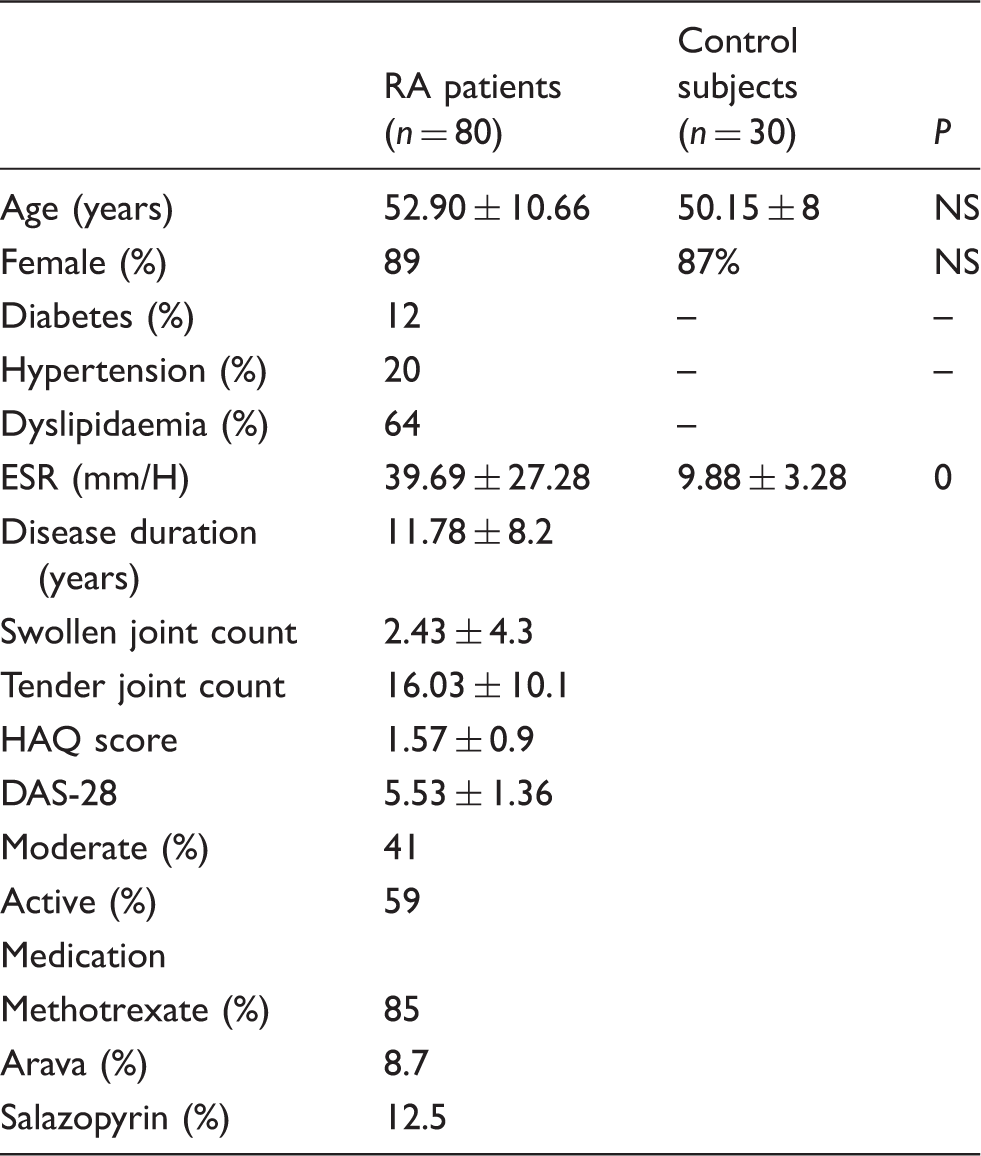
Note: Data represent means ± SD or percentages.
NS: not significant; HAQ: health assessment questionnaire; DAS-28: disease activity score for 28 joints; ESR: erythrocyte sedimentation rate; RA: rheumatoid arthritis.
Biological parameters of patients with RA and control subjects
Biological parameters of patients with RA and control subjects.
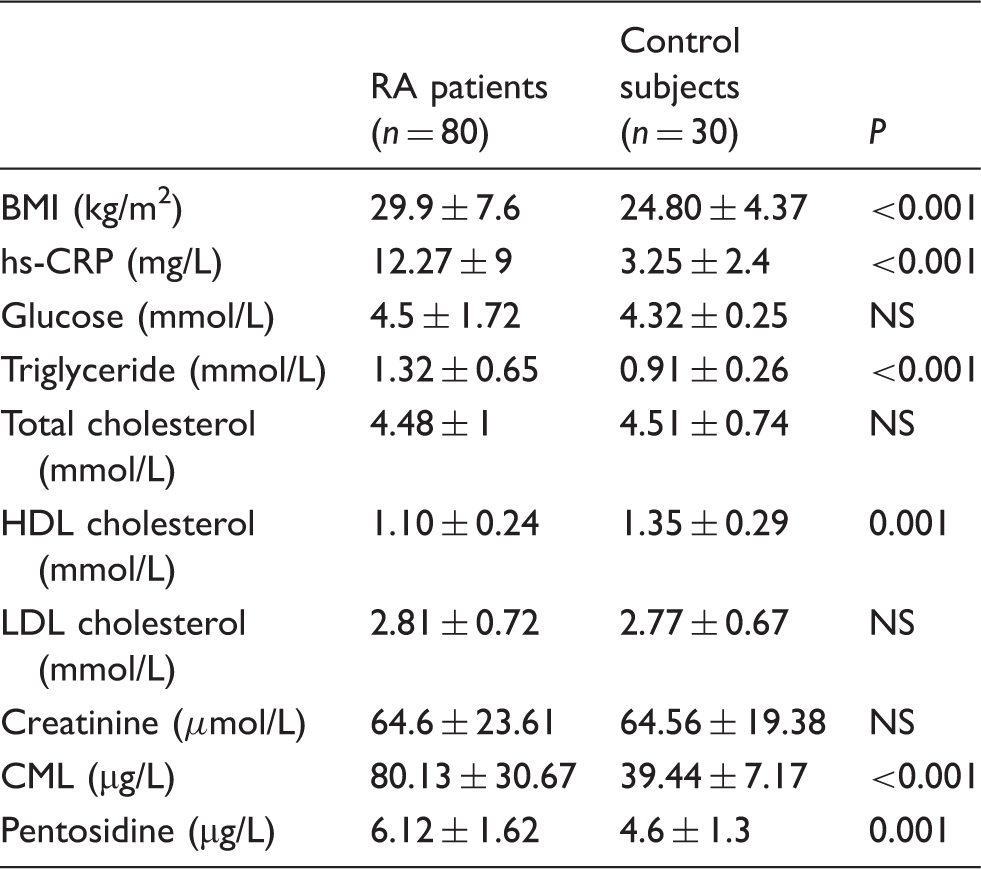
Note: Data represent means ± SD.
NS: not significant; hs-CRP: high-sensitive C-reactive protein; CML: Nɛ-carboxymethyllysine; LDL: low-density lipoprotein; HDL: high-density lipoprotein; BMI: body mass index; RA: rheumatoid arthritis.
Relationship between serum CML, pentosidine concentrations and the presence of RA
Multiple logistic regression analysis was performed using the presence of RA as a dependent variable. The analysis involved age, sex, BMI, glucose, triglyceride, HDL cholesterol, hs-CRP, CML and pentosidine. In result, we find that serum CML (odds ratio [OR] = 1.21, 95% CI: 1.05–1.39; P = 0.006) was an independent predictor of the presence of RA. The ability of serum CML concentrations to distinguish patients with RA from those without RA was assessed using ROC curve analysis. The ROC curves for the presence of RA diagnosis had an AUC of 0.93 (95% CI: 0.88–0.99, P < 0.001). The optimal cut-off value of CML was 45.2 ng/mL, with 88% sensitivity and 82% specificity (Figure 1). We further performed a multivariate stepwise regression analysis, and CML was independently correlated with DAS-28 (standardized β = 0.43, P = 0.001).
Receiver-operating characteristic (ROC) curve analysis for predictive values of serum concentrations in presence of RA. Area under the ROC curve is: 0.93; 95% CI: 0.88–0.99, P < 0.001. The optimal cut-off value of CML was 45.2 μg/L, with 88% sensitivity and 82% specificity.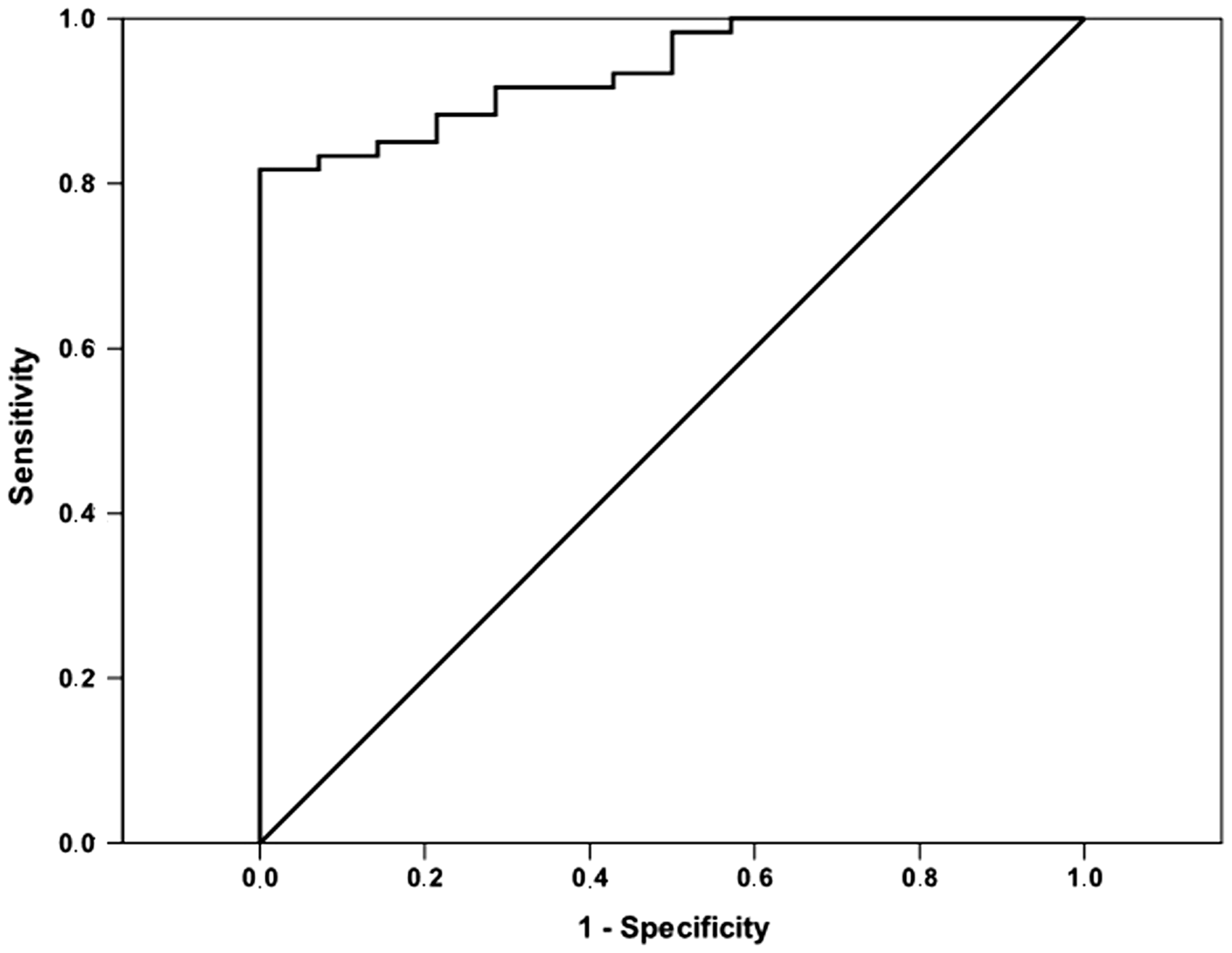
Relationship between serum CML and pentosidine concentrations and the severity of RA
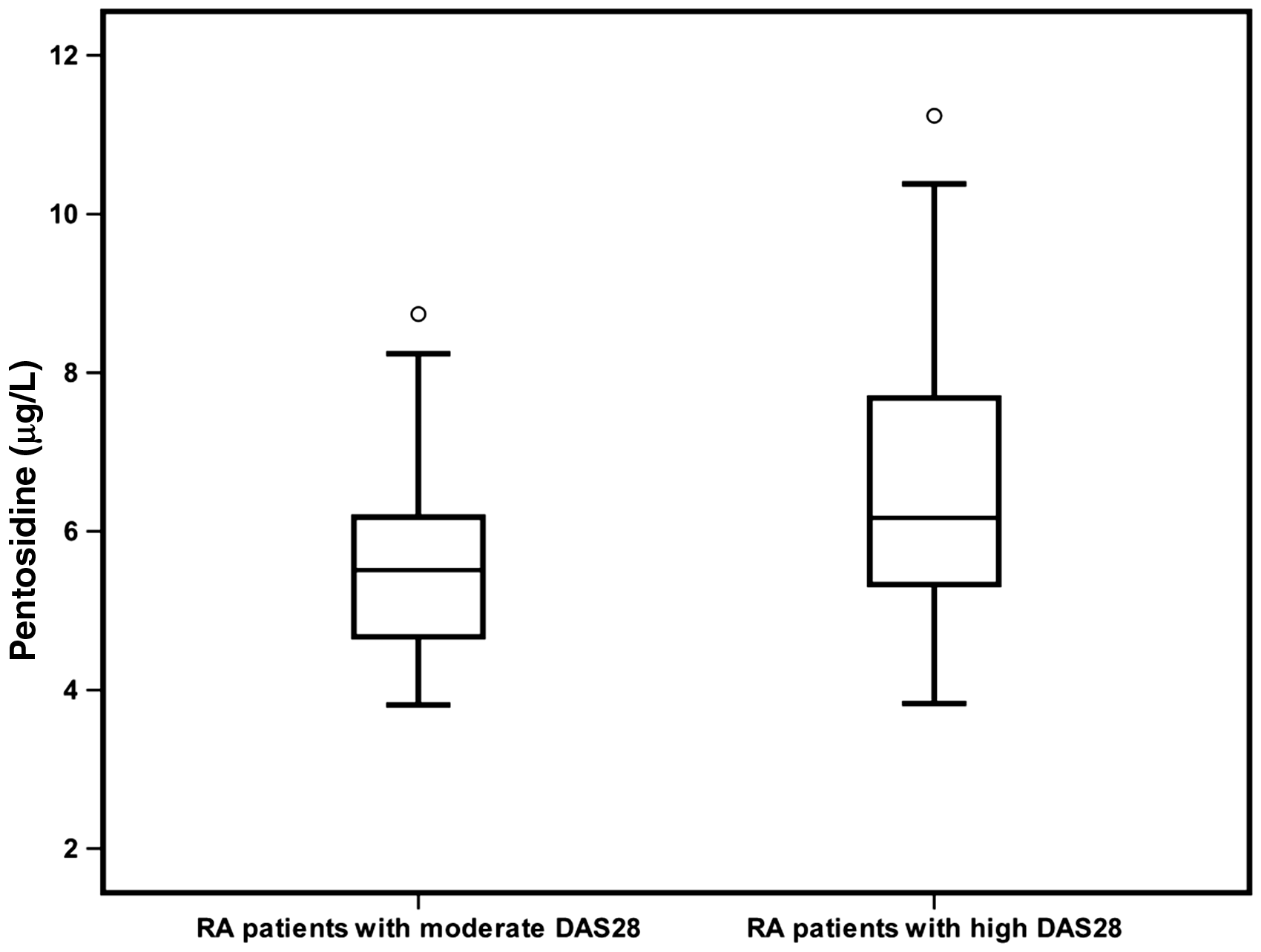
All patients were subclassified into two subgroups according to DAS28. Serum concentrations of CML were increased in patients with high DAS28 compared with the patients with moderate DAS28 (78.73 [72.66–93.27], 53.37 [44.80–67.79]; P < 0.001 respectively), as illustrated in Figure 2. Serum concentrations of pentosidine were increased in patients with high DAS28 compared with the patients with moderate DAS28 (6.19 [5.29–7.75], 5.51 [4.55–6.17]; P < 0.001 respectively), as illustrated in Figure 3.
Serum concentrations of CML in patients with moderate and high DAS28 (P < 0.001). The error bars represent minimum and maximum values, the boxes indicate the interquartile range and the median values. Serum concentrations of pentosidine in patients with moderate and high DAS28 (P = 0.019). The error bars represent minimum and maximum values, the boxes indicate the interquartile range and the median values.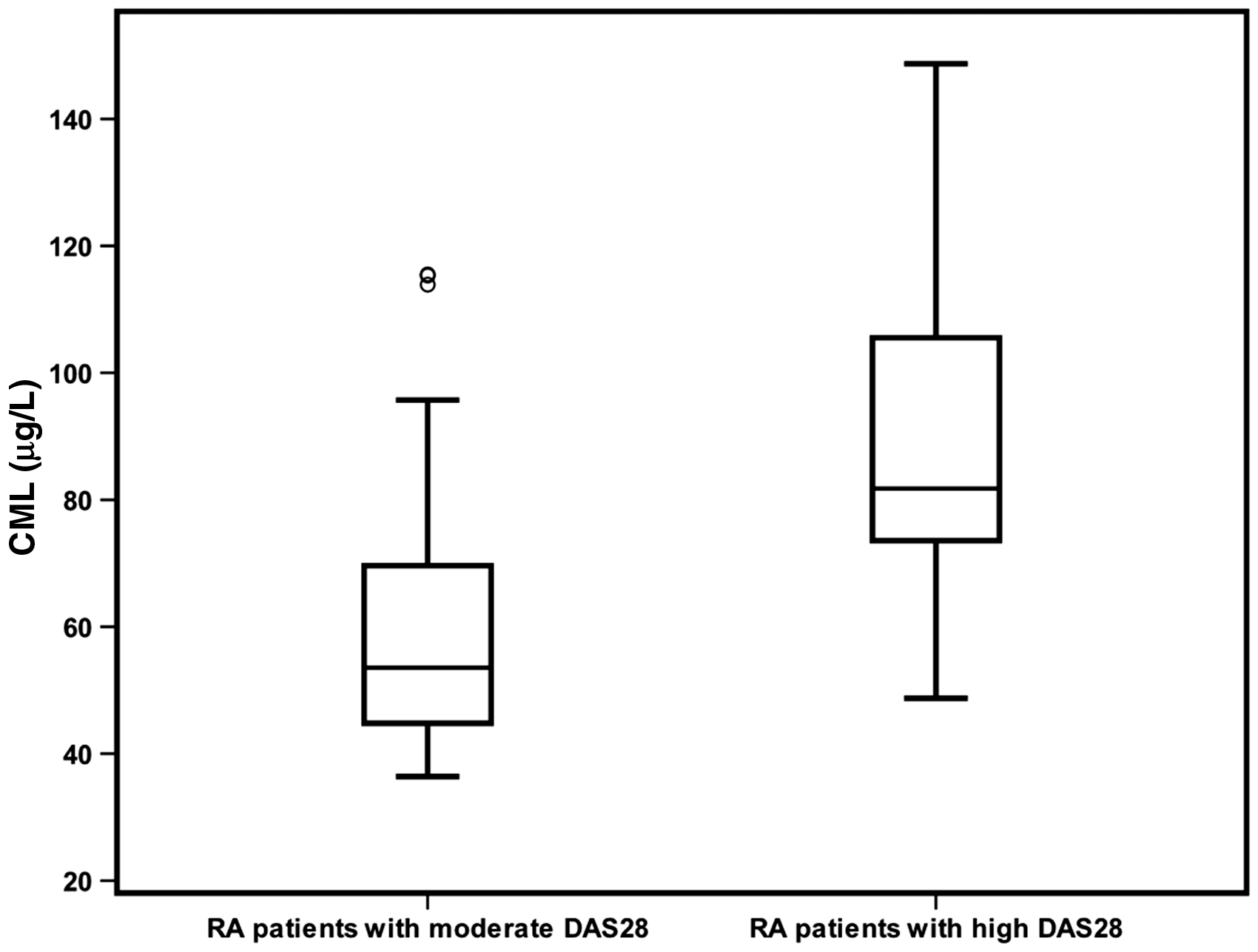
Discussion
RA is a systemic autoimmune disease characterized by chronic inflammation of the synovium, resulting in tenderness and destruction of bone and cartilage in various joints. It affects approximately 1% of the general population, women more often than men, and the inflammatory burden of the disease results in functional disability.16,17 The accumulation of AGEs in circulation and in tissue was observed in the course of ageing18,19 and at higher rates in diabetes20,21 and other inflammatory conditions such as RA 22 and Alzheimer’s disease. 23 Protein glycation has been implicated in the development of arthritis, which may arise through oxidative stress-driven glycation in RA.10,11
The anti-CCP antibody test is now a clinical standard biochemical test for RA, and the diagnosis is often confirmed before damage to the joint occurs. However, the anti-CPP antibody test has relatively low sensitivity 68% sensitivity and 98% specificity in established disease. 24 We hypothesized that damage to proteins of the joint by oxidation and glycation, and with signatures released in plasma as oxidized and glycated proteins may facilitate early-stage diagnosis and typing of arthritis. Hitherto, biomarkers of protein oxidation and glycation have not been developed for clinical diagnostic application in RA, except in studies on plasma. 25 Development of drugs to suppress oxidative damage and glycation damage to collagen in arthritis will be facilitated by the availability of companion diagnostic tests based on these biomarkers. 26
In this study, we examined the serum concentrations of CML and pentosidine in RA patients, and we evaluated their association with activity parameters of this disease. Serum AGEs concentrations were significantly higher in RA patients compared with control subjects. Moreover, these biomarkers were significantly increased in patients with RA with high disease activity. AGEs were generated under the influence of oxidative stress as present in chronic inflammatory diseases. In this way, a positive feedback loop of AGE–receptor for advanced glycation endproducts interaction is established, maintaining an inflammatory status in which AGEs can be formed.27,28 The interest of recent years has focused on the potential of glycated proteins as a source of free radicals and a marker of disease activity.10,29 CML represent a pathological process which appears to associate with RA and its severity. IgG constitutes about 75% of the total immunoglobulin pool in serum. In addition to four N-terminal amino acids, human IgG has ∼80 lysine residues, making IgG a good target for glycation. AGEs alter the three-dimensional integrity of various plasma proteins which could induce functional abnormalities and lead to pathogenesis of several diseases. Newkirk et al. 30 demonstrated that AGE on IgG, as a target for circulating antibodies in North American Indians with RA.
Many studies have shown evidence that the pentosidine is elevated in articular cartilage, serum samples and synovial fluid of patients with RA. 11 However, the mechanisms of these phenomena remain unclear. It is known that chronic hyperglycaemia leads to the accumulation of non-enzymatic products of protein glycosylation, as in diabetes. In the current study, we observed a strong correlation between pentosidine and DAS28 and a significant correlation between pentosidine and ESR (r = 0.319; P = 0.018). Pentosidine concentration rises with age, diabetes 31 and with end-stage kidney disease. However, it is also increased in RA or systemic lupus erythematosus, in the absence of hyperglycaemia or kidney disease. 32 Furthermore, Takahashi et al. 33 concluded that the concentration of pentosidine in serum samples of RA patients reflects the activity of the disease, as the serum concentration of pentosidine correlated with clinical indices of RA activity, and of serum markers of inflammation (CRP, ESR) and the concentration of rheumatoid factor (RF). In addition, the examination of many published studies, correlation between serum/plasma pentosidine and laboratory markers/clinical indices appears equivocal. For example, three11,34,35 of five studies confirmed statistically significant positive correlations between serum pentosidine and CRP but two36,37 did not. Correlation between DAS28 and serum pentosidine was reproduced in two studies3,34 of five.3,35–38
CML is a well-characterized AGE which represents a chemically modified amino acid and is derived in vivo from carbohydrates as well as lipid-derived precursors. CML has been detected in atherosclerosis lesions and is thought to be an endogenous biomarker of local oxidative stress. 10 It is considered a marker of increased oxidative stress. 38 In our results, we demonstrated that the concentration of CML was increased in patients with high DAS28 and in patients with moderate DAS28 compared with control subjects. There have been few studies regarding the implications of CML in RA. It would be of considerable interest to further study the serum concentrations of CML in RA since it is associated with increased cardiovascular risk.39,40 No cut-off value of CML to anticipate the presence of RA has been suggested to date. Our study revealed an optimal cut-off of 45.2 μg/L, with 88% sensitivity and 82% specificity and suggests that serum concentrations of CML are predictors of the presence and the severity of RA. Additionally, multivariate regression analysis demonstrated that CML was an independent marker for the severity of RA. We hypothesize that the synergic effect of increased serum pentosidine and CML may aggravate the severity of RA by increasing local glycoxidation, the related cross-links formation and could amplify an inflammatory phenomenon. Further studies, especially larger, population-based prospective studies are required to confirm the findings suggested by the current study.
Conclusion
The present study is the first to compare the serum concentration of pentosidine and CML in cohorts of RA patients and control subjects. We demonstrated that both high serum concentration of pentosidine and CML were associated with the presence of RA. However, an increase of CML concentrations was markedly associated in the severity of RA.
Footnotes
Acknowledgement
We acknowledge Prof. Anis Belhaj (Hematology Department, CHU Tahar Sfar Madia) for his help in providing the ELISA multiplate reader.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funds from the Biochemistry Laboratory, LR12ES05, Faculty of Medicine, University of Monastir.
Ethical approval
Monastir Hospital public Fattouma Bourguiba ethics Committee (Ref: centre 0707) approved the study.
Guarantor
MK.
Contributorship
IK participated in the quantification of pentosidine and CML, the recruitment of RA patients and control subjects and the interpretation of data and in writing the manuscript; HB participated in biochemical Laboratory measurements; SZ and NB were responsible of the recruitment of RA patients; MH supervised the study and MK participated in the design of the study, the interpretations of results, the statistical analyses and in the preparation of the manuscript.
