Abstract
Background
Faecal occult blood testing is an important diagnostic tool for the detection of colorectal cancer. However, it has not been standardized due to the absence of suitable specimens for surveillance.
Methods
We developed a ready-to-use artificial stool made from rice flour. This new artificial stool homogeneously contains not only human haemoglobin A0 (HbA0) but also glycerol as an internal standard material. After the collection of the artificial stool into a buffer, the haemoglobin concentration in dispersed solution was measured using a method based on the peroxidase like activity of haemoglobin. The glycerol concentration was measured using a commercially available triglyceride measurement kit.
Results
With regard to the haemoglobin stability, the decrease in the level of human haemoglobin in the artificial stool was <2% when it was stored at −80℃ for four months, −20℃ for two weeks, and 5℃ for two days. The artificial stool was easily collected with the collecting tubes of a commercially available faecal haemoglobin test kit. The weight of the collected artificial stool could be calculated by measuring the concentration of glycerol in the extracting solution of the collected stool sample. The haemoglobin concentrations could be adjusted based on their collection weights.
Conclusions
The artificial stool has a paste-like consistency and contains both haemoglobin and glycerol homogeneously. Furthermore, the measured haemoglobin concentration could be determined based on the collected stool weight, which was directly related to the glycerol concentration. These features make it a useful material for the surveillance of faecal occult blood testing.
Introduction
Faecal occult blood testing (FOBT) is an important diagnostic tool for the detection of colorectal cancer.1,2 Recently, the methods used for measuring haemoglobin (Hb) have been changing from colorimetric methods to immunochemical methods.3–5 The commercially available immunochemical test kits use various types of antibodies and coagulants, which were individually developed by the kit suppliers; consequently, there are some differences among the different kits. 6 For diagnostic testing to be impartial, Hb measurement must be standardized. At present, however, it has not been standardized due to the very low concentration and unstable structure of Hb in stool and the absence of specimens that are suitable for surveillance.
A meeting of all of the Japanese suppliers of diagnostic kits for faecal Hb testing was organized by the Health Care Technology Foundation (HECTEF) and was held in Tokyo, Japan in 2011. 7 At present, efforts to prepare a ‘standardized Hb solution’ are ongoing, with the aim of standardizing the calibrators of each of the suppliers. However, surveillance studies have not been performed to evaluate these kits on a nationwide scale due to the lack of a suitable surveillance material.
Although there have been several trials to establish surveillance material for FOBT, the following issues still remain to be solved. First, the use of real human stool specimens for surveillance is associated with some problems. For example, the stability of Hb is very poor due to the surviving bacteria and the remaining proteinases. 8 Furthermore, it is almost impossible to define ‘a standard stool’ because real stool has a wide variety of textures and water content. Second, although a small surveillance study was carried out using experimentally made artificial stool, 9 the exact mass of the stool collected with commercially available collecting tubes could not be determined, because the error range of picking weight of specimen with the commercially available collecting tube was more than that of Hb measurement. This makes it impossible to perform a correct evaluation of measuring the exact concentration of the Hb in the stool. Third, a solid–liquid separating type of artificial stool had been commercially launched in Japan. However, the rate of human error in preparing the specimens was significant. In order to overcome these problems, we started to develop an artificial stool for external quality assessment that homogeneously contained both Hb and an appropriate internal control.
Materials and methods
Glycylglycine (Wako special grade) was obtained from Wako Pure Chemical Industries, Ltd, Osaka, Japan. NaOH (JIS special grade), HCl (super special grade), sodium azide (JIS special grade), NaCl (JIS special grade), ethanol (super special grade), methanol (Super special grade), glycerol (ultrapure HPLC grade, Alfa Aesar, UK,), bovine serum albumin (biochemistry grade), DL-homocystein, casein sodium, agarose, catalase (from bovine liver), gelatin (bovine leather and bone, blocking reagents N101 and N102) and hydrolysed collagen peptide enzyme (for biochemistry) were also obtained from Wako Pure Chemical Industries, Ltd.
Trehalose was obtained from Hayashibara Co., Ltd, Okayama, Japan. Rice flour (Joyoko) was obtained from Mamehei Shimayashokai Co. Ltd, Toyama, Japan. Horseradish peroxidase (EC1.11.1.7) was obtained from the Oriental Yeast Co. Ltd, Tokyo, Japan. Human HbA0 (H0267-25MG, HbA0, ferrous stabilized human) was obtained from Sigma-Aldrich Co., US. MAPT (10-Methylaminocarbonyl-3,7-bis(dimethylamino)Phenothiazine) 10 was obtained from HECTEF (Health Care Technology Foundation, Tokyo, Japan). Determiner™ L TGII was obtained from Kyowa Medex Co., Ltd, Tokyo, Japan. Bactogelatin™ was obtained from Muto Pure Chemicals Co., Ltd, Tokyo, Japan. Dextran sulphate (molecular weight 50,000) was obtained from Meito Sangyo Co., Ltd, Nagoya, Japan. Junflon™ PFA (Tetra fluoro ethylene perfluoro alkyl vinyl ether copolymer) TA100 tubes were obtained from Junkosha Co., Ltd, Tokyo Japan (these tubes were used for sealing samples of the artificial stool).
Base buffer solution
Fifteen grams of collagen peptide were added to 2.7 L of distilled water and dissolved by heating to 37℃. After cooling to room temperature, 20.4 g of glycylglycine and 90 g of trehalose was added and dissolved. The solution was adjusted pH 7.5 with 6 N NaOH and 6 N HCl. Six grams of sodium azide was dissolved and adjusted to a final volume of 3.0 L with distilled water.
Pretreated rice flour
One kilogram of rice flour was added to 3.0 L of base buffer solution, and mixed. The mixture was left at room temperature for 1 h, then filtered with a 151G No. 3 glass vacuum filter and wet flour was obtained. The wet flour was dried in a vacuum at a temperature that was lower than room temperature for two days. We finally obtained approximately 0.87 kg of dried and sterilized rice flour.
Hb-stabilizing solution
NaCl (6.8 g) and glycerol (100 g) were added to 1.0 L of the above base solution and mixed well.
The pH was readjusted to 7.5 with 6 N NaOH and 6 N HCl, and 11,000 units of horseradish peroxidase were added to stabilize the Hb. Finally, the solution was mixed and filtered using an MF-Millipore membrane filter HAWP02500 (0.45 μm).
Hb solution
Human HbA0 was dissolved in 100 mL of the above Hb-stabilizing solution and was diluted appropriately with the same solution.
Artificial stool
One hundred grams of pretreated rice flour and 110 mL of Hb solution were mixed together and kneaded well for 10 min in a glass bowl at 3–8℃. We made three types of artificial stool: zero (0 µg/g), middle level (approximately 65 µg/g) and high level (approximately 130 µg/g) of Hb. The mixture was placed in a DK0524 sausage maker (Kaijirusi Co., Ltd, Tokyo) and approximately 1.5 g was pushed out into an 8-mm Junflon PFA tube (TA100). Both ends of the PFA tubes were sealed by heat and stored in glass bottles at a temperature lower than −80℃.
The collection and extraction of artificial stool
Artificial stools were collected with the collection tubes of commercially available faecal Hb test kits according to the manufacturer’s instructions or with mini-spatulas. In the case of mini-spatulas, the artificial stool was collected and smeared in empty 5 mL glass test tubes which had been previously weighed. The total weights of the smeared test tubes were immediately measured using a digital balance. The weights of the collected artificial stool were calculated from the total weights and the weights of the empty tubes. Three millilitres of base buffer solution were added to the test tubes, and the contents were dispersed by stirring. After centrifugation for 5 min at 1500 × g, the supernatants were drawn out and the concentrations of Hb and glycerol were measured. In the case of commercially available collection tubes for faecal Hb test kits, we used the sampling bottles of the OC-SENSOR kit (G-PZ15, Eiken Chemical Co. Ltd, Tokyo Japan). In order to draw out liquid specimens, the two layers of the aluminium bottom seal in the bottle were broken by hand and a fixed quantity of the liquid was drawn out using an Eppendorf micropipette.
The measurement of Hb
The Hb concentration was measured with a highly sensitive method 11 that utilizes the decomposition type chromogen MAPT and cumene hydroperoxide. The absorbance at a wavelength of 666 nm was measured using a glass cell (light path length, 1 cm) with a UV-3600 double beam spectrophotometer (Shimadzu Co., Ltd, Tokyo). The Hb concentration was calculated and compared to Hb-free (Hb 0) artificial stool.
The measurement of glycerol
The glycerol concentration was measured with ‘Determiner L TGII’, a triglyceride measuring kit. The reaction reagent was prepared by mixing 30 mL of R1 reagent and 10 mL of R2 reagent; 30 μL of the sample solution was added to 3 mL of reaction reagent and mixed well. After 10 min of incubation at 37℃, the absorbance at 600 nm was measured using the above-mentioned UV-3600 double beam spectrophotometer. The concentration of glycerol was calculated in comparison to the absorbance of 500 mg/L glycerol standard solution. The coefficient of variation of glycerol measuring was within 1.5% (data not shown).
Results
The recovery of Hb from rice flour matrix
In order to choose the non-specific adsorption preventing agent on the surface of the rice flour, various compounds were added to 50 mmol/L glycylglycine buffer solution (pH = 7.5) and were mixed well. One gram of rice flour was added to 3 mL of the above solution and mixed well in a 10 mL glass tube. After 10 min at room temperature, the mixed solution was centrifuged at 3000 × g for 10 min. The supernatants were completely removed by vacuum drawing and dried in vacuum chamber for 1 day at room temperature. A total of 1.1 mL of 20 μg/mL Hb solution was mixed well with the solid dried residue and left at room temperature for 5 min. In order to extract the Hb from the solid residue, 2 mL of distilled water was added and mixed well at room temperature. After centrifugation at 3000 × g, 100 μL of supernatant was drawn out and the concentration of Hb was measured. The Hb recovered from the rice flour was calculated in comparison to the Hb concentration of the 22.6 μg/mL Hb solution. The Hb concentration in the rice flour was increased by water absorption (3.1/[3.10–0.345] × 20 μg/mL) because 1 g of rice flour absorbed 0.34 mL water; with the exception of large Hb molecules, the water was absorbed by the matrix of the rice flour. The volume of the absorbed water became clear in another experiment (data not shown).
The results of a recovery test are shown in Figure 1. Hydrolysed gelatin and collagen peptide yielded the best recovery. We chose collagen peptide, which has good solvency in water, as an adsorption preventing agent.
Haemoglobin recovery. The recoveries (%) were calculated from concentration of each extracted supernatants compared with Hb standard solution. Gelatin from bovine and its decomposition product (collagen peptide) showed approximately 100% recovery.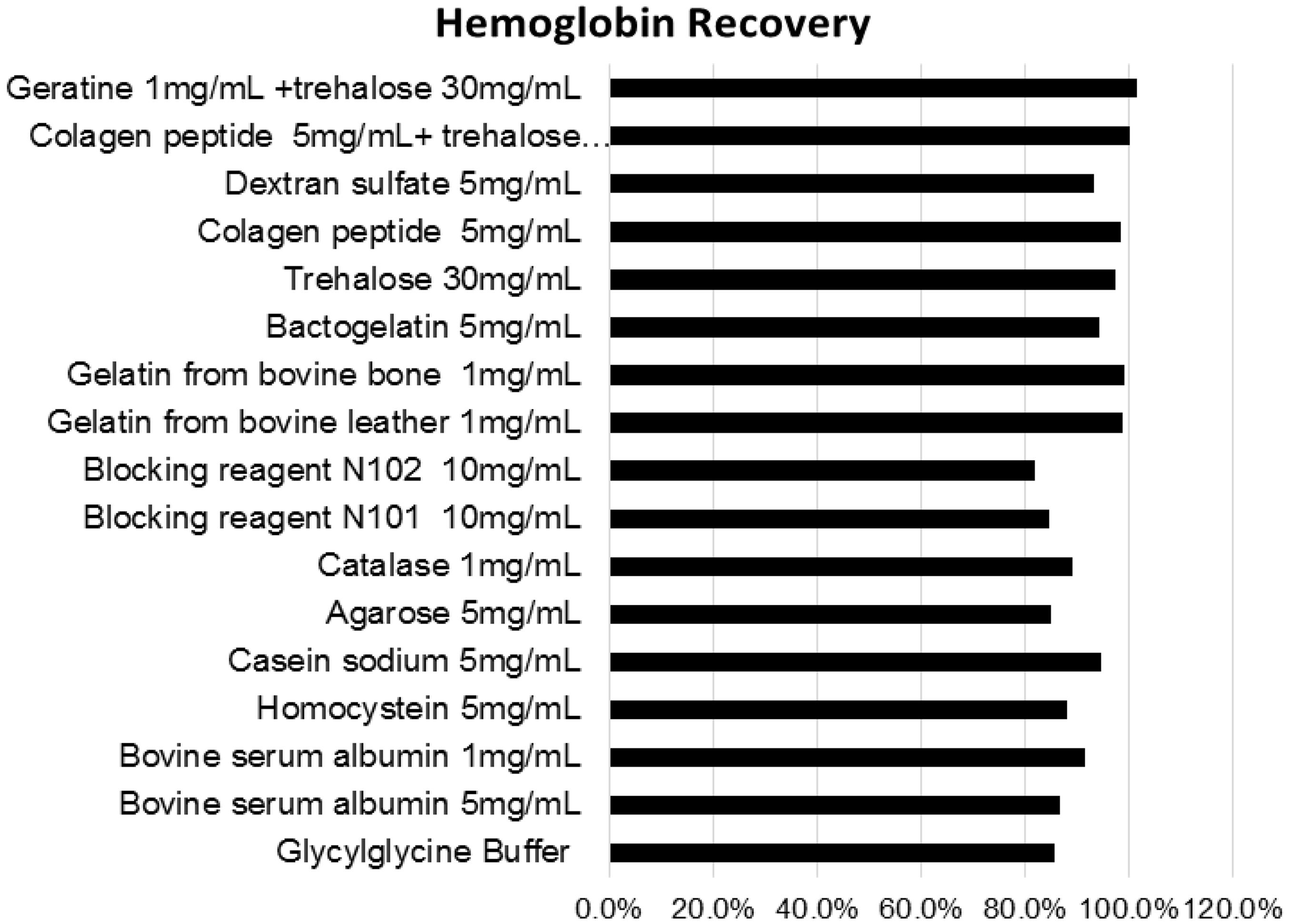
The relationship between the glycerol concentration and collection weight of the stool
Different weights of artificial stool (5–70 mg) were collected with mini-spatulas and smeared in the 5 mL empty glass test tubes. After weighing, 3 mL of base buffer solution was added to the test tubes and the contents were dispersed by stirring. After centrifugation for 5 min at 1500 × g, the supernatants were drawn out and the glycerol concentration of the supernatants was measured. The relationship of measured glycerol concentrations and collected weights of stool are shown in Figure 2.
The relationship between the glycerol concentration and collection weight of the stool. The weights of collected new artificial stool and glycerol concentrations were directly proportional.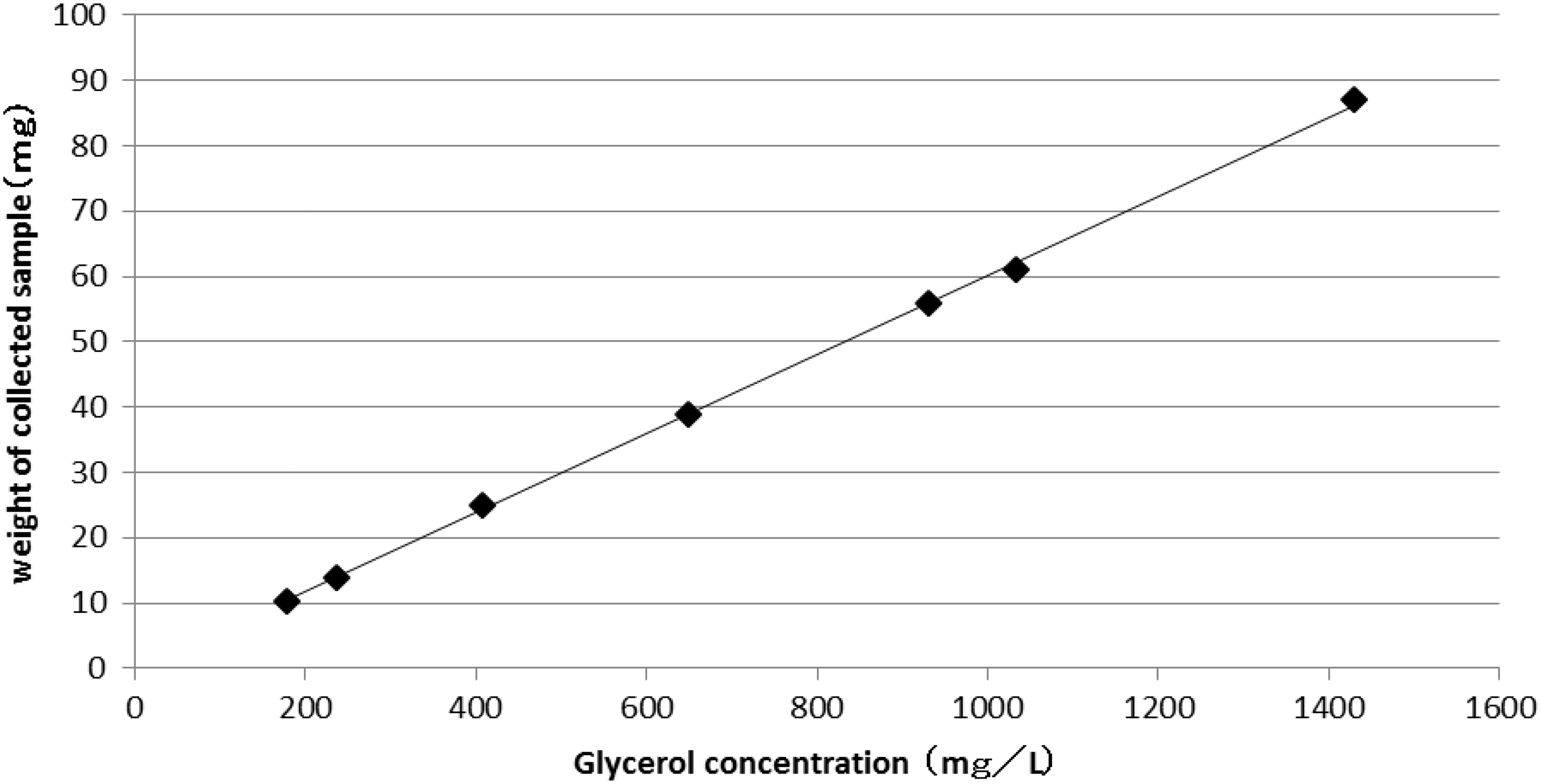
The stability of Hb in artificial stool
High level and low level of artificial stool were placed in Junflon™ PFA tubes, which were packed in a glass bottle and stored at less than −80℃ for one to four months, −20℃ for one to two weeks and 5℃ for one to two days. The stored specimens (approximately 50 mg) were collected three times by mini-spatula and were weighed immediately. The weights of the specimen and the Hb concentration were measured. To make the data easier to understand, all of the Hb concentration data were adjusted based on the weight of the collected specimen (50 mg). The results are shown in Figure 3.
Stability of haemoglobin in artificial stool. The artificial stools in the Junflon TM PFA tube packed in glass bottles were stored at less than −80℃ for one to four months, −20℃ for one to two weeks and 5℃ for one to two days, respectively.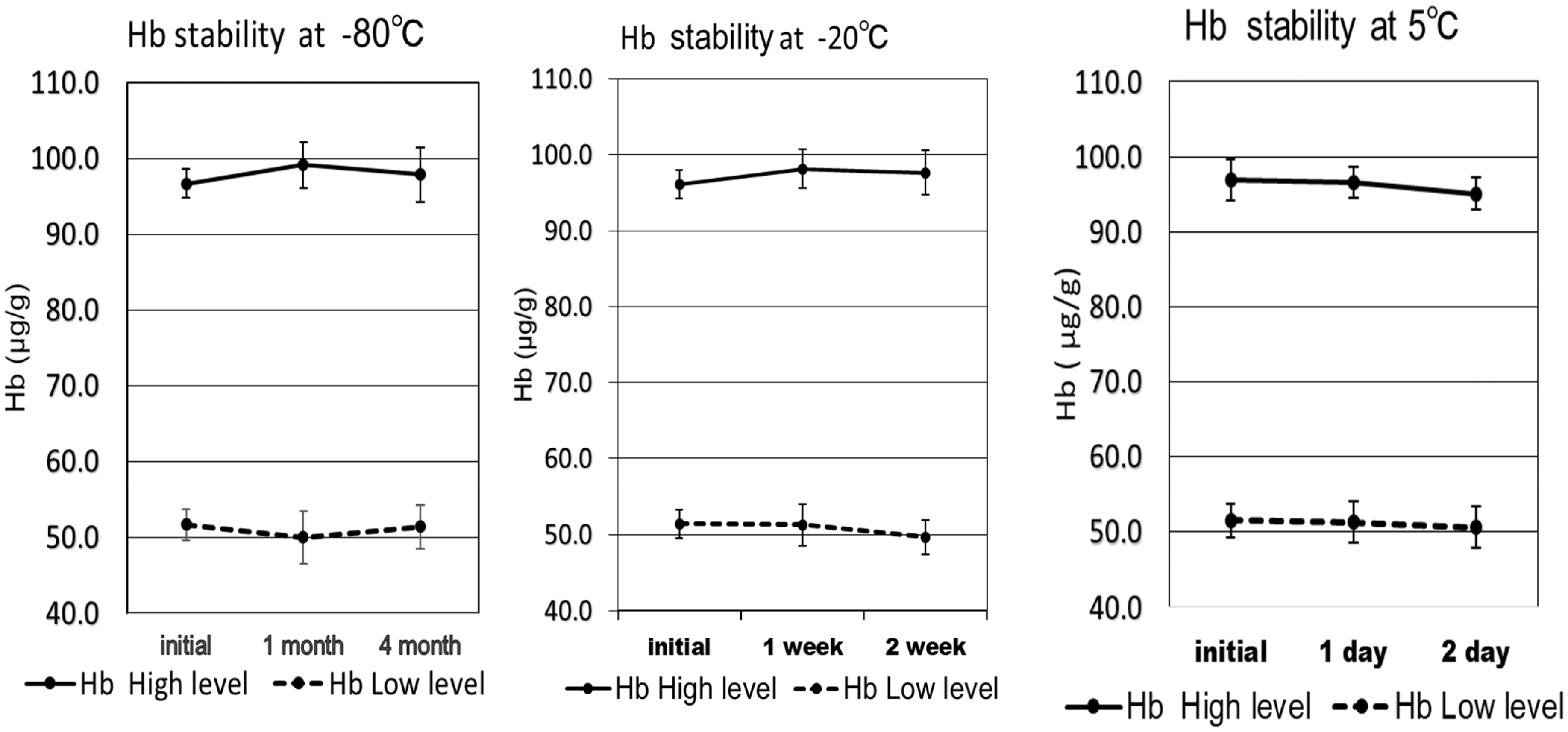
Hb was stable for four months at −80℃, one week at −20℃, and two days at 5℃. A slight decline in the range of 1–2% was observed after two weeks of storage at −20℃; however, Student's t-test revealed that the result was not significant.
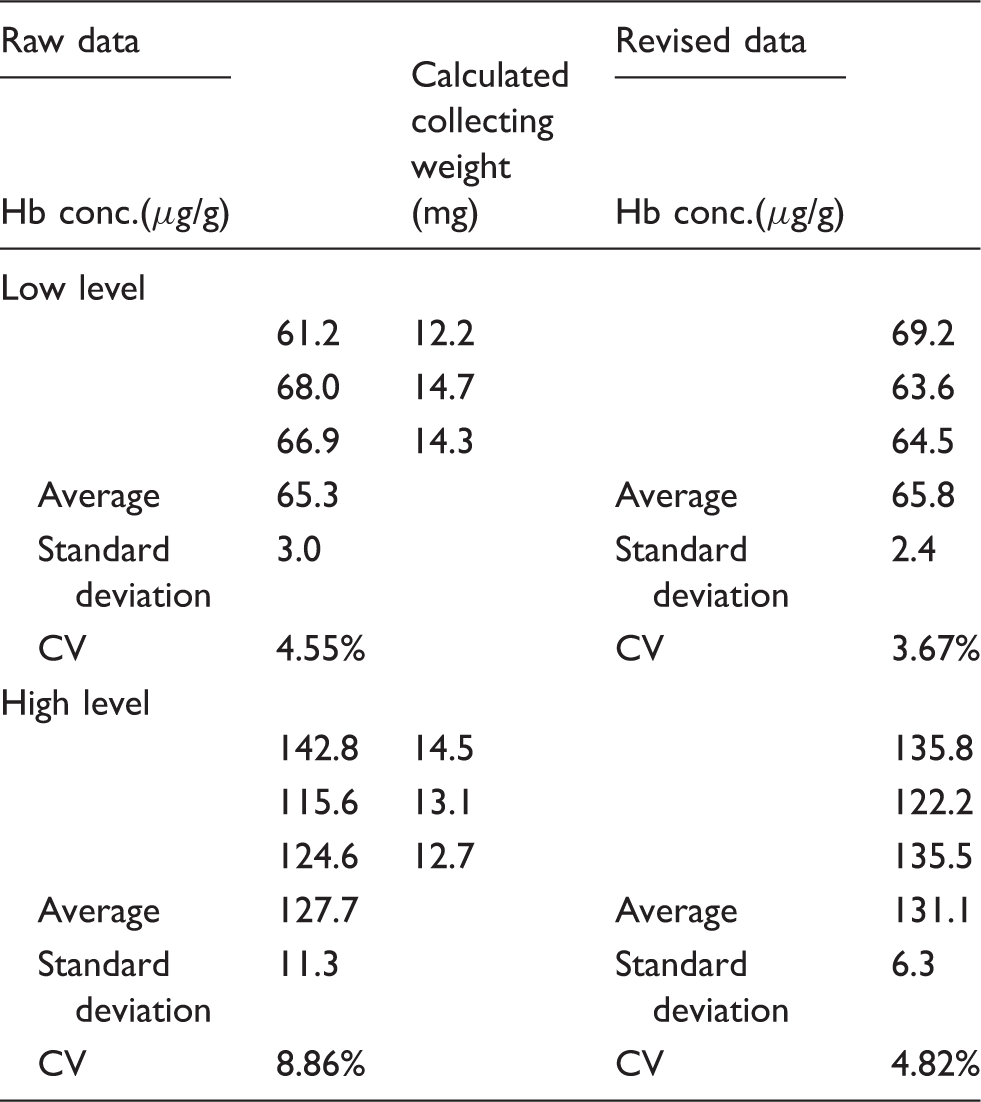
The determination of the Hb concentration using commercially available collecting tubes and adjustment based on the glycerol concentration
Three types of artificial stool (0-level, low-level and high-level) were validated for practical use. The sampling bottles of the G-PZ15 OC-sensor are designed to collect a fixed quantity (approximately 10 mg) of stool. After collecting the artificial stool, the bottom seals of the bottles were broken and filtrated sample solutions were drawn out using an Eppendorf micropipette. We used 30 μL of solution to determine the glycerol concentration and 100 μL to determine the Hb concentration.
Hb determination using commercially available collecting tube and revision with glycerol concentration.
Discussion
We used glycylglycine as a base buffer in the developed artificial stool, expecting that the Schiff base reaction between the aldehyde and amino groups of the Hb molecule would be inhibited. The aldehyde group might have been generated by unsaturated fatty acids contamination, which can be found in natural products, such as rice flour.
Recently, the determination of triglyceride concentrations has been performed without the free glycerol concentration in many clinical laboratories. However, free glycerol became measureable when the reagents of the triglyceride determination kit were rearranged. In the present study, we used ‘Determiner TG’, in which R1 and R2 were premixed into a one reagent system. The reaction that occurred when glycerol-kinase, glycerol-3-phosphate oxidase and peroxidase were added at the same time, allowed free glycerol to directly react to form an equimolar of hydrogen peroxide. Then, free glycerol was measured easily by automated biochemical analyser.
In addition, the added glycerol was very stable and could be precisely measured. The evaluation of commercial collection tubes may be possible by a statistical study of the plural data of the glycerol concentration in drew out solutions of collecting tubes.
We used a biochemical method with a highly sensitive chromogen to measure the peroxidase-like activity of Hb. However, because the new artificial stool contains peroxidase, the peroxidase-like activity is increased in comparison to the activity observed with Hb alone. Thus, the measured Hb concentrations need to be reduced to account for the additional peroxidase activity. This was accomplished by measuring the activity of a zero-Hb sample (blank value). The immunological measurement method that is applied in many clinical laboratories in Japan would not need to reduce the peroxidase activity.
The stability of Hb in the artificial stool suggests that the artificial stool can tolerate transportation between facilities if refrigerated transportation services are used. We selected glycerol as an internal standard material for calculating the collected weight of the artificial stool. Glycerol is freely soluble in aqueous solutions. Moreover, glycerol could be measured with automatic biochemical analysers and triglyceride measurement kits – which can be found in most laboratories – if the reagents are mixed before the assay.
Conclusion
We showed that the artificial stool that we developed may be useful for the surveillance of faecal Hb testing. The artificial stool contains stabilized human Hb as well as glycerol, which could be used to calculate the weight of the collected stool. The new artificial stool has three benefits: (1) it has a paste-like consistency and can therefore be directly used in the laboratory without pretreatment; (2) the Hb concentration can be adjusted according to the collected stool weight, which is directly related to the glycerol concentration and (3) the quality of the collection tubes can be managed by measuring the glycerol concentration of the buffer extracts, which indicates the weight of the collected artificial stool.
Footnotes
Acknowledgements
The authors would like to express their thanks to Eiken chemicals Co. Ltd, for providing the sampling bottles for the OC-sensor. The authors also thank the Department of Clinical Chemistry of Kitasato University for providing the laboratory space and utilities.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AM is a director of General Incorporated HECTEF, IS is the chairperson of the board of General Incorporated HECTEF. ZO has no conflicts of interest in association with this study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
No human subjects or participants were used in this study.
Guarantor
AM.
Contributionship
AM researched literature and conceived the study. AM and ZO carried out the experiments and data analysis of the study. AM wrote the first draft of the manuscript. IS gave the information of laboratory needs and technical advice on developing new artificial stool. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
