Abstract
We report a patient with acquired von Willebrand disease, associated with multiple myeloma. At one stage in his illness, we were unable to analyse a sample sent in a serum separator tube, due to the presence of a gel within the separated serum layer. We suggest this was due to anomalous position of the gel because of the density of the sample caused by its high total protein concentration, exacerbated by fibrin strand formation because of inhibition of appropriate fibrin clot formation secondary to clotting disorder.
Introduction
Monoclonal gammopathies, or paraproteins, are a relatively common finding. In those aged over 50 years, the overall incidence is 3.2%, rising to 5.3% in those over 70 years. 1 Monoclonal gammopathy may be associated with clinical disorders, for example B-cell lymphoproliferative disorders or plasma cell disorders, for example multiple myeloma, or monoclonal gammopathy of undetermined significance (MGUS).
Paraproteins are well recognized as a source of interference in laboratory assays2,3 and the diseases associated with them can be associated with specific clinical complications. We report a patient with myeloma and an IgA paraprotein, with acquired von Willebrand disease, in whom biochemistry analysis was complicated by formation of a gel within the separated serum. We postulate that both these conditions were contributory to the analytical problem.
Case report
A 60-year-old man was transferred to the Royal London Hospital in April 2012 with a painful haematoma on his buttock, following bone marrow biopsy performed at his local hospital. He had been referred there following detection of an IgA paraprotein (26 g/L), initially detected as part of well man screening. Investigations locally showed creatinine, albumin and adjusted calcium concentrations within reference intervals and no lytic lesions on skeletal survey. Urine Bence Jones protein was detected but not quantified and his bone marrow was infiltrated with 25% plasma cells. He had no history of bleeding.
Clotting studies on admission.
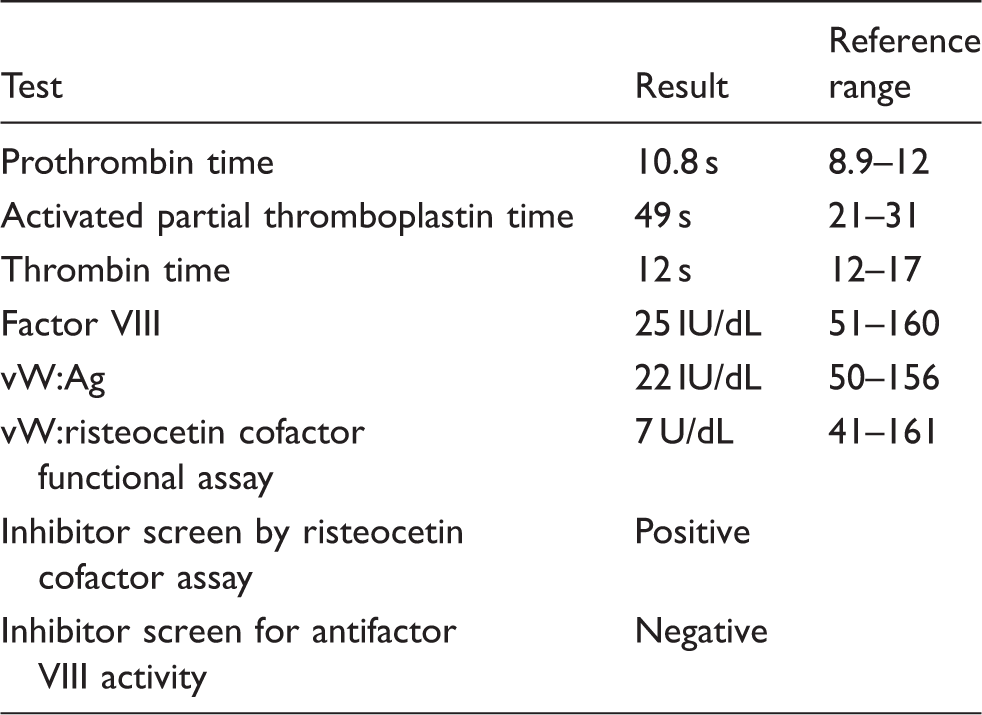
vW: von Willebrand.
A diagnosis of acquired von Willebrand disease was made and this was managed with human von Willebrand Factor/coagulation Factor VIII complex (wilate). He underwent chemotherapy with bortezomib and dexamethasone and, following high dose melphalan, received a stem cell transplant in December 2012.
By July 2013, his IgA paraprotein concentration had reduced to 12 g/L and his von Willebrand activity (vWF:RCo) had improved to 22 U/dL. However, over the following months, his paraprotein increased accompanied by reduction in von Willebrand activity, necessitating intensification of treatment. In May 2015, he experienced a massive splenic haemorrhage of unknown cause, requiring emergency embolization and wilate therapy and was subsequently established on intravenous immunoglobulin prophylaxis to reduce further bleeding episodes. His myeloma continued to progress and his IgA paraprotein concentration rose to 35 g/L, total protein concentration 98 g/L, albumin 42 g/L.
During a further admission to Royal London in June 2015, a sample sent for clinical biochemistry analysis provided unsuitable for analysis due to the formation of a viscous gel within the separated serum. Request for a repeat using a lithium heparin sample circumvented this problem. While gel formation in serum separator tubes in patients with multiple myeloma is well described, we suggest that his associated clotting disorder may have been contributory.
He had a further seven cycles of chemotherapy with good response. In October 2016, his IgA band was 2 g/L and his von Willebrand antigen and vWF:RCO had normalized. The concentrations of IgA paraprotein and von Willebrand activity at various time points in his illness are shown in Figure 1.
Concentration of IgA paraprotein and von Willebrand activity (vWF:RCo) at various time points during the course of his illness, illustrating the relationship between them.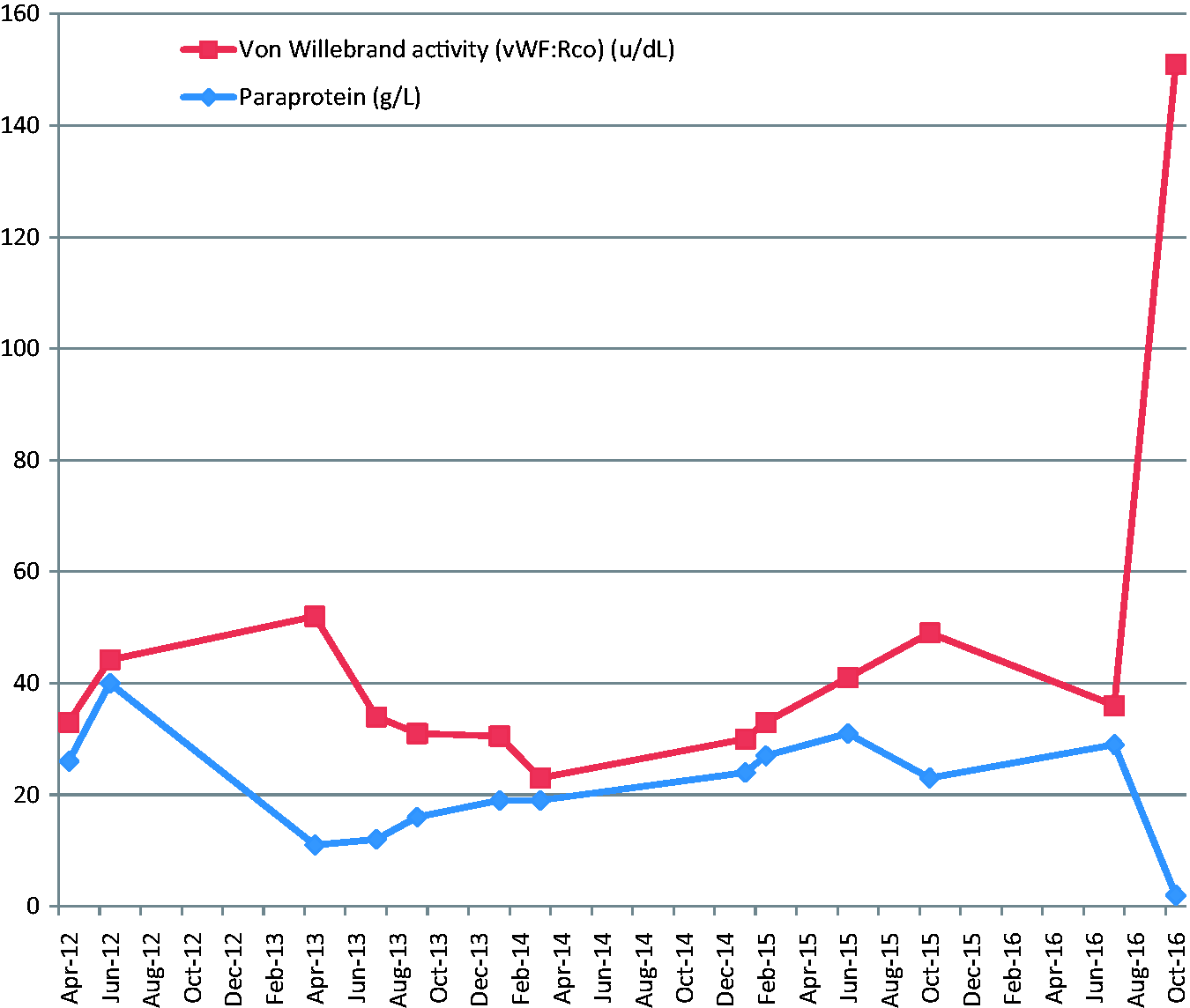
This is characteristic of the acquired condition in which normal haemostasis returns when the underlying condition is treated.
Discussion
von Willebrand disease is the most common autosomally inherited bleeding disorder; it is caused by mutation in the VWF gene, leading to quantitative, structural or functional abnormalities of von Willebrand factor. 4 Acquired von Willebrand disease was first reported in 1968. 5 It is a bleeding disorder which, contrary to the hereditary form, tends to have an onset in adulthood in those with no previous personal or family history of bleeding disorder. Over the next 30 years, 266 cases were reported and in 2000, a retrospective survey identified a further 186 cases. 6 While its exact incidence is unknown, it is acknowledged to be relatively rare. Diagnosis requires the demonstration of abnormalities of von Willebrand factor in the blood and of a causal underlying disease. From reports in the literature, acquired von Willebrand syndrome is most commonly associated with cardiovascular disorders (e.g. aortic stenosis, ventricular assist devices) lymphoproliferative and myeloproliferative disorders and hypothyroidism. Other associations include non-haematological malignancies, autoimmune disorders and drugs, particularly sodium valproate. 7 The aetiology of acquired von Willebrand syndrome is thought to be multifactorial and to differ according to the underlying associated disorder. In patients with associated lymphoproliferative disease, it is thought to arise as a result of autoantibodies against von Willebrand factor which bind to functional epitopes and neutralize its activity, or which form immune complexes which lead to increased clearance from the circulation. 8
Our patient presented with bleeding complications after bone marrow biopsy, performed to confirm a diagnosis of multiple myeloma, and clotting studies were consistent with a diagnosis of acquired von Willebrand syndrome. He was treated with human von Willebrand factor/coagulation Factor VIII complex (wilate). The effects of wilate may be of lesser magnitude in acquired than congenital von Willebrand disease, resulting from mutations in the VWF gene, perhaps because of neutralization by antibodies or increased clearance in the acquired form. Desmopressin increases plasma concentration of factor VIII and von Willebrand factor in deficient patients, enhancing platelet adhesion to vessel walls, so can be used in the treatment of von Willebrand disease. 9 However, its success has also been found to be limited and short lived in acquired disease, particularly associated with underlying cardiovascular and myeloproliferative disorders.
Paraproteins are a common cause of interference in clinical biochemistry analyses.2,3 Our patient is of interest because his sample proved impossible to analyse due to the formation of a viscous gel within the separated serum. We postulate that a combination of his myeloma and the von Willebrand syndrome contributed to this. Serum separator tubes of the type used in our laboratory contain a clot activator and a polymerized gel. Microionized silica particles accelerate clot formation, by the intrinsic pathway, through clot activation. The gel is thixotropic, i.e. it is semi-solid under static conditions but becomes less viscous if force, such as centrifugation, is applied. The specific gravity of the gel is designed so that it is between that of serum and blood cells, acting as a barrier to separate them. 10 The position of the gel in the tube may be influenced by factors which are laboratory controlled, e.g. temperature and centrifugation conditions, or patient specific, e.g. low haematocrit, and increased plasma proteins, 11 administration of iodinated contrast material 12 and contamination of the sample with trisodium citrate (Citra-lock). 13
Different explanations have been proposed for anomalous positioning of the gel with both differences in density and viscosity being suggested. An experiment in which gel position was observed in dextran solutions of differing densities and viscosities would tend to support density as the determinant of gel position. 14 If undetected, abnormal gel formation may lead to occlusion of analyser probes or to falsely low results when insufficient sample is aspirated. 15
In a study of 42,080 patients whose blood was sampled into serum separator containers, only 16 had persistently abnormal separation and of these 14 had multiple myeloma (total protein concentrations (69–141 g/L, median 90 g/L) and two had MGUS (88 and 65 g/L). 16 This suggests that underlying myeloma may be more commonly associated with anomalous gel position than raised total protein alone and a possible additional mechanism is the known inhibition of myeloma paraprotein with fibrin formation. 17 Incomplete or delayed fibrin clot formation may result in the presence of latent fibrin in the form of thin strands or gelatinous masses. Fibrin gel formation in clot activator tubes sufficient to cause interference in biochemical analysis has been reported as an issue in patients on anticoagulant therapy; their samples may require longer to clot prior to centrifugation than those from other patients. 18 Our patient had a low factor VIII level consistent with the diagnosis of acquired VWD and this would lead to prolongation of his clotting time via the intrinsic pathway.
We report a patient with the rare condition of acquired von Willebrand disease associated with multiple myeloma. We were unable to perform biochemical analysis in a sample due to the presence of a gel within the separated serum layer. We postulate two reasons for this – firstly anomalous positioning of the barrier gel due to the density of the sample caused by high total protein and secondly fibrin strand formation because of inhibition of appropriate fibrin clot formation, as a result of not only myeloma paraprotein but also his acquired von Willebrand disease.
He serves to draw the attention of Clinical Biochemists to acquired von Willebrand disease and to illustrate the ways in which myeloma proteins can lead to preanalytical interference.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Patient has given written informed consent.
Guarantor
RMA.
Contributorship
LB was involved in clinical care of the patient. NRR wrote the first draft of the manuscript. All authors reviewed, contributed to and edited the manuscript and approved its final version.
