Abstract
Objective
The correlation of adiponectin with cholesterol concentration of fractionated lipoproteins has not been well investigated.
Methods
This study included 174 subjects (79 men and 95 women) without diabetes. The medical record data were investigated retrospectively. The study subjects with adiponectin <8.3, > 8.3 but less 13.9, and ≥ 13.9 were classified into tertile groups: Groups A (n = 59), B (n = 58) and C (n = 57), respectively.
Results
In women, age and HDL-C were higher in Group C than in Groups A and B, but BMI, TG, IDL-C and VLDL-C were lower in Group C than in Groups A and B. In men, BMI was lower in Group C than in Groups A and B, and HDL-C was higher in Group C than in Groups A and B. In multiple stepwise regression analysis, BMI and HDL-C were significantly correlated with adiponectin in whole, male and female subjects, but TG-rich lipoprotein cholesterol concentrations were not independently correlated.
Conclusions
HDL-C and BMI were independently correlated with adiponectin in non-diabetic men and women. These results suggest that high adiponectin may play a role in the increased HDL-C concentrations, implicated in the reduction of cardiovascular disease risk, in non-diabetic subjects.
Introduction
Metabolic syndrome relevant to visceral fat accumulation is a susceptible pathophysiologic state to atherosclerosis resulting from impaired metabolism of lipid and glucose, hypertension, obesity and these complex risk factors.1,2 Visceral fat accumulation is considered to be present upstream for a variety of metabolic disorders, which are dyslipidemia, impaired glucose metabolism, hypertension, and these complex risk factors. 1
Adiponectin, one of the adipocytokines secreted from adipose tissue, and its concentrations in serum are inversely correlated with body mass index (BMI) and visceral fat accumulation.1,3 Cell biology studies have demonstrated that adiponectin has diverse antiatherosclerotic functions including anti-inflammatory actions and inhibitory effects of macrophage foam cell formation.1,4,5 In addition, adiponectin has been shown to increase insulin sensitivity,4,6 and low adiponectin concentrations are found in subjects with obesity, type 2 diabetes mellitus and dyslipidaemia.4,7
In non-diabetic subjects, low serum adiponectin was associated with high serum triglyceride (TG) and low high-density lipoprotein (HDL)-cholesterol, but not with low-density lipoprotein (LDL)-cholesterol concentrations. 8 Serum adiponectin was also reported to correlate positively with HDL-C and inversely with TG in patients with type 2 diabetes.9,10
Non-HDL cholesterol is able to account for total atherogenic burden by measuring the aggregate amount of cholesterol in apolipoprotein B-containing lipoproteins which are LDL, intermediate-density lipoprotein (IDL), very low-density lipoprotein (VLDL), chylomicron (CM) and CM remnant. 11 Previous reports showed that cholesterol concentrations of both IDL and VLDL were associated with atherosclerotic cardiovascular disease risk.12–14 However, the relevance of adiponectin to dyslipidaemia determined by means of measuring lipoproteins separated by ultracentrifugation or high-performance liquid chromatography (HPLC) remains understudied. Recently, we have developed a novel HPLC method with anion exchange column containing a non-porous polymer-based gel to facilitate measurements of cholesterol concentrations in the fractionated serum lipoproteins, which are HDL, LDL, IDL, VLDL and other factions, containing chylomicron, chylomicron remnant and lipoprotein(a) [Lp(a)].15,16 We have reported that low serum adiponectin was found in patients with type 2 diabetes and type IIb hyperlipidaemia, and adiponectin correlated inversely with VLDL-cholesterol determined by our developed HPLC method. 17 However, the association of serum adiponectin with detailed lipoprotein fractions (HDL, LDL, IDL, VLDL, Others fraction) with the HPLC method in non-diabetic subjects has never been reported.
Therefore, the present study was for the first time performed to investigate the association of adiponectin with cholesterol concentrations of lipoprotein fractions, determined by the HPLC method, in non-diabetic subjects.
Patients and methods
Subjects, study protocol and measurements of parameters
This study included 174 subjects (79 men and 95 women, mean age: 67.9 ± 11.3 years, mean body mass index [BMI]: 22.8 ± 3.0 kg/m2) who underwent a medical checkup of the brain, including serum adiponectin, at the Jikei University Kashiwa hospital from April 2008 to March 2010. The medical record data were retrospectively investigated. The patients, who took medications for dyslipidaemia, hypertension and diabetes, were excluded from the study. In addition, smokers and premenopausal women were not included. This study was approved by the Ethics Committee of the Jikei University School of Medicine.
Anthropometric measurements (height and weight) were performed in a standing position. BMI was calculated as the weight divided by the square of height in metres. Fasting blood samples were collected from all participants. Serum lipids, total cholesterol (TC) and TG, were measured by conventional enzymatic methods. Cholesterol concentrations of very low-density lipoprotein (VLDL), intermediate-density lipoprotein (IDL), low-density lipoprotein (LDL) and high-density lipoprotein (HDL), other fraction (chylomicron, chylomicron remnant, Lp(a)) were measured by the HPLC method as we reported previously.15,16 Adiponectin was measured as total adiponectin by a latex particle-enhanced turbidimetric assay (Otsuka Pharmaceutical, Tokyo, Japan). 18 Glycohaemoglobin A1c (HbA1c), plasma glucose and serum insulin were measured by routine methods. The homeostasis model assessment of insulin resistance (HOMA-R), a surrogate marker for insulin resistance, was calculated as fasting insulin (mU/l) × plasma glucose (mg/dl)/405.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation (SD). One-way ANOVA with Fisher PLSD for multiple post hoc comparisons was used for the analysis of differences between groups. Simple correlations were estimated by Spearman's rank test. Multiple stepwise regression analysis was performed to identify any independent explanatory variables, including age and BMI, for serum adiponectin. A value of P < 0.05 was considered as statistically significant.
Results
The characteristics of the study subjects (average ± SD).
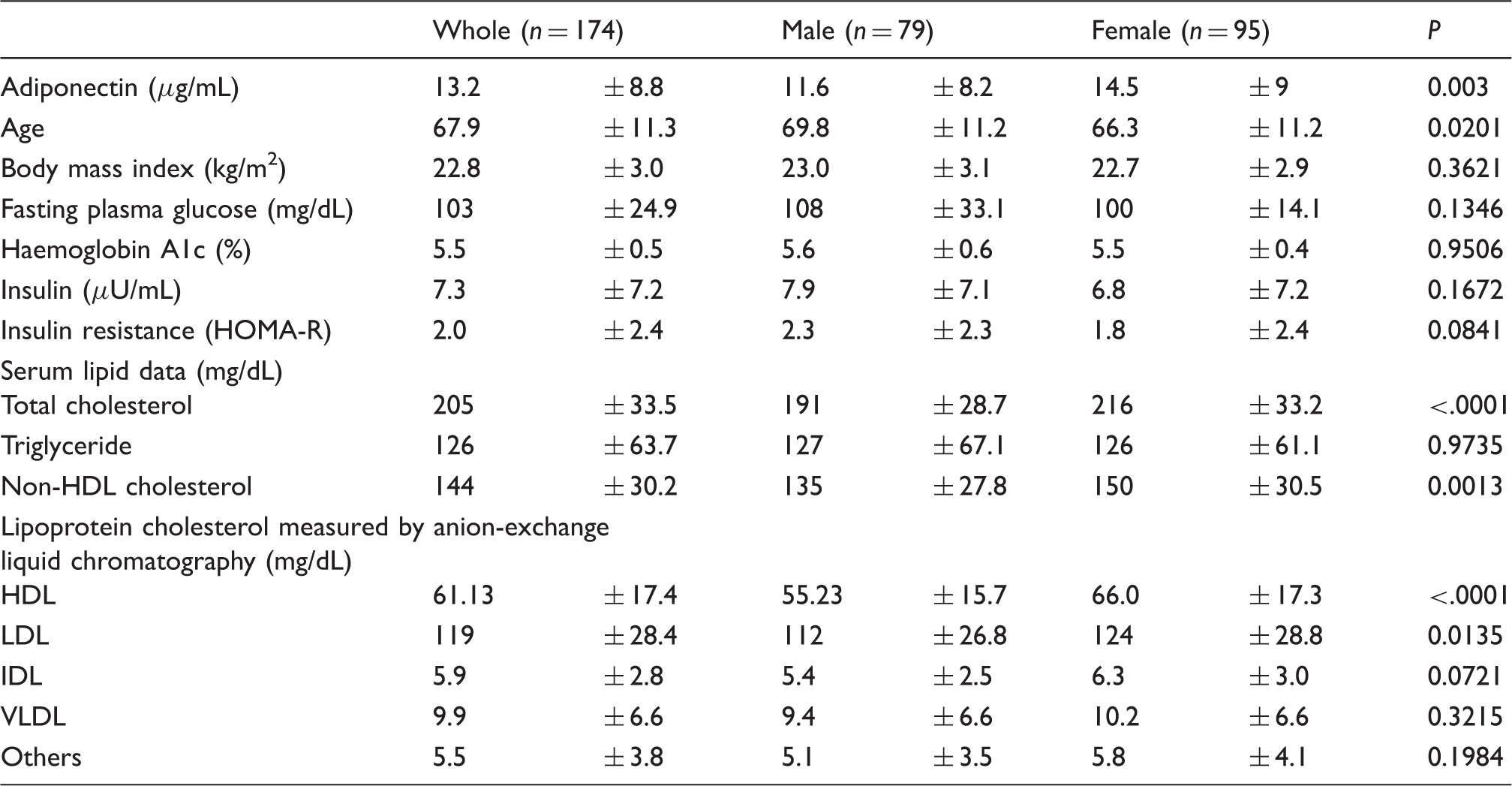
HOMA-R: homeostasis model assessment of insulin resistance; VLDL: very low-density lipoprotein; IDL: intermediate-density lipoprotein; LDL: low-density lipoprotein; HDL: high-density lipoprotein; Others: means cholesterol of chylomicron, chylomicron remnant and lipoprotein (a).
Comparison among tertile groups classified by adiponectin concentration in whole subjects.
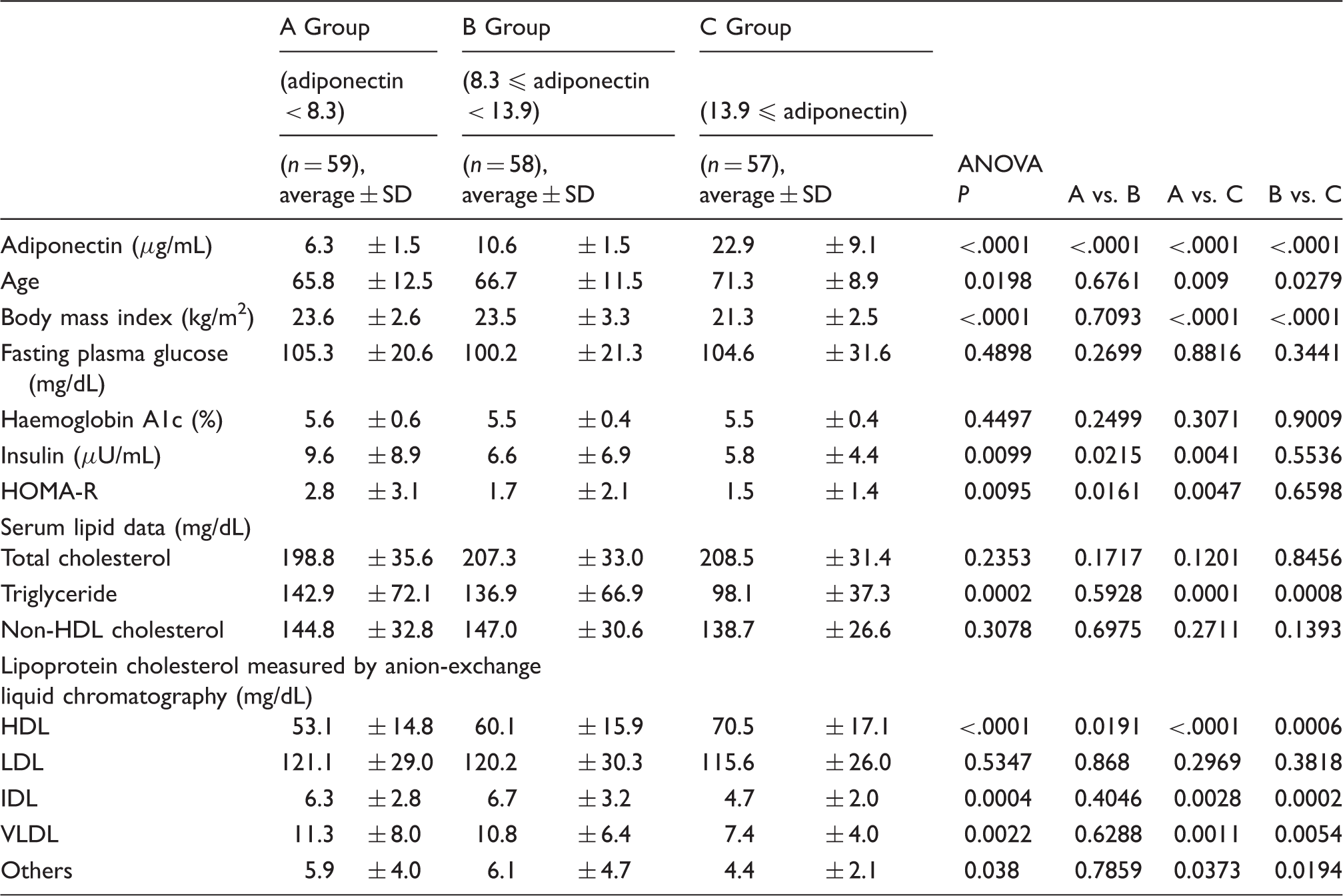
VLDL: very low-density lipoprotein; IDL: intermediate-density lipoprotein; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Comparison among tertile groups classified by adiponectin concentration in men.
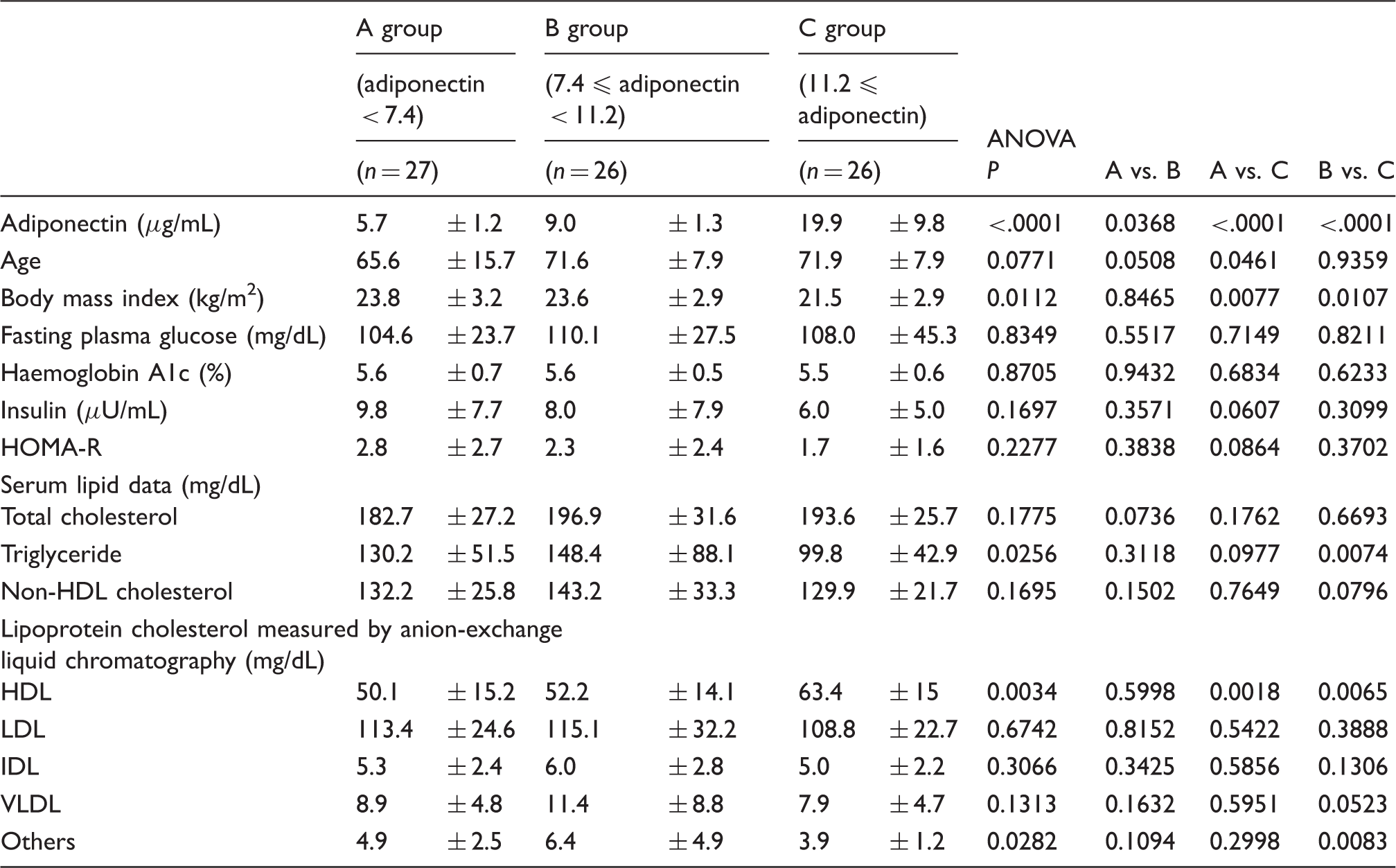
VLDL: very low-density lipoprotein; IDL: intermediate-density lipoprotein; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Comparison among tertile groups classified by adiponectin concentration in women.
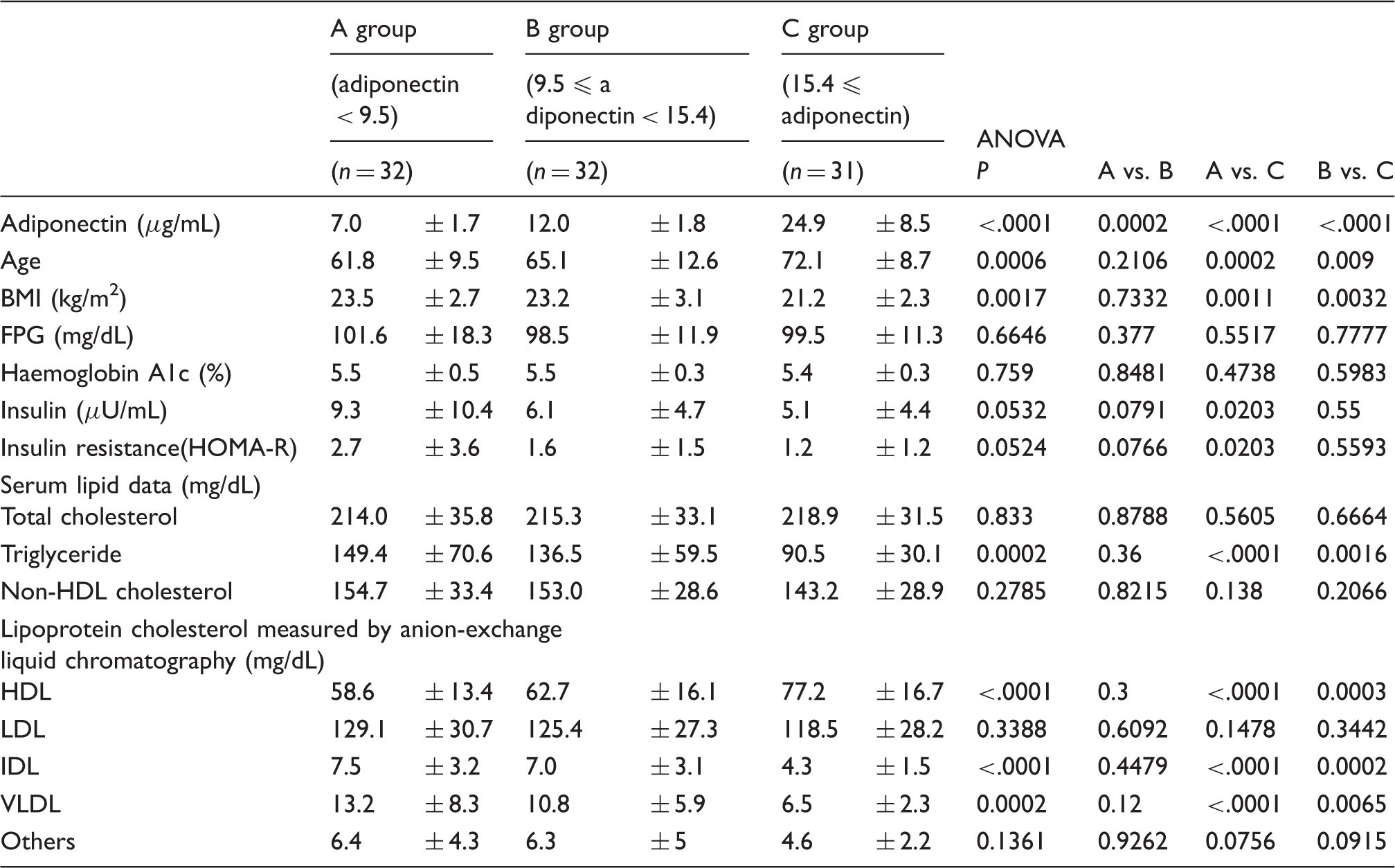
FPG: fasting blood glucose; BMI: body mass index; VLDL: very low-density lipoprotein; IDL: intermediate-density lipoprotein; LDL: low-density lipoprotein; HDL: high-density lipoprotein; Others: means cholesterol of chylomicron, chylomicron remnant and lipoprotein (a)..
Results of multiple regression analysis using adiponectin as a confounding factor.
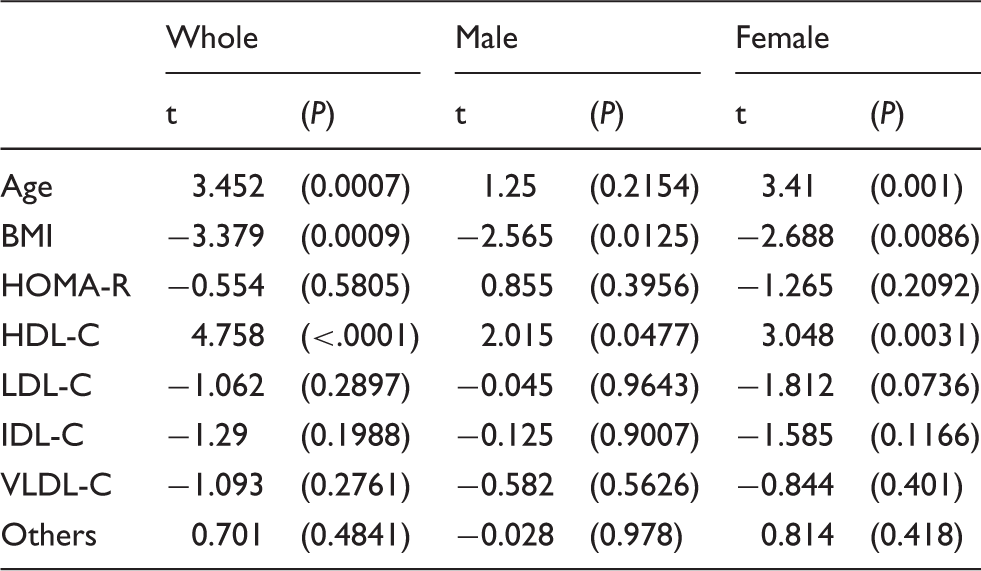
Note: Cholesterol concentrations of HDL, LDL, IDL, VLDL and others were measured by anion-exchange liquid chromatography.
HOMA-R: homeostasis model assessment of insulin resistance; BMI: body mass index; VLDL-C: very low-density lipoprotein-cholesterol; IDL-C: intermediate-density lipoprotein-cholesterol; LDL-C: low-density lipoprotein-cholesterol; HDL-C: high-density lipoprotein-cholesterol; Others: means cholesterol of chylomicron, chylomicron remnant and lipoprotein (a).
Discussion
This study shows that HDL-C and BMI were independently and significantly correlated with adiponectin in both male and female non-diabetic subjects. As for the sex difference in this study subjects, the higher concentrations of TC, non HDL-C, LDL-C, and HDL-C and lower concentrations of age were found in women than in men. Previous studies have reported that serum adiponectin concentrations tended to rise with age.19,21 In the present investigations by sex, age was independently correlated with adiponectin only in women. The subjects with high concentrations of adiponectin live long,1,19,21 and consequently elderly people could have high concentrations of adiponectin, although the reasons why serum adiponectin may rise with age are not well defined. However, the independent correlation of age with adiponectin in men was not found in the present study. This sex difference might be associated with the difference in average life expectancy between men and women, but it remains to be cleared. However, the independent correlations of BMI with adiponectin were found in whole, male and female subjects as reported previously.1,3
The sex difference in serum adiponectin concentrations has been reported.19,20 The distribution of serum adiponectin concentrations was lower in men than in women, but the differences in adiponectin concentrations between pre- and post-menopausal women were not found. In addition, testosterone treatment reduced adiponectin secretion into the culture media from 3T3-L1 adipocytes. Therefore, the sex difference in serum adiponectin concentrations may be attributable in part to androgens-mediated inhibition of adiponectin secretion from adipocytes.
We have previously reported that VLDL-C independently and inversely correlated with adiponectin in patients with type 2 diabetes. 17 Adiponectin might decrease serum TG and VLDL-C, in part by inhibiting VLDL and TG production or by stimulating lipolysis of TG by lipoprotein lipase.1,17,23 It would be considered that the increased concentrations of serum TG and VLDL-C are found in patients with type 2 diabetes partially because of hypoadiponectinaemia. Serum TG concentrations in the highest tertile of adiponectin (Group C) were lower than other Groups in whole, male and female subjects, but TG concentrations were not so high probably because of non-diabetic subjects. Consequently, the independent correlations between adiponectin and TG-rich lipoprotein, including VLDL, might not be found. By contrast, low HDL-C usually is found in hypoadiponectinaemia in part resulting from the effects of adiponectin on cholesterol efflux from macrophages and reverse cholesterol transport.24,25 Namely, adiponectin could increase HDL-C irrespective of TG concentrations, although the positive and inverse correlations of adiponectin with HDL-C and TG, respectively, have been reported.8,26 Therefore, the independent correlations of adiponectin with HDL-C were found in whole, male and female subjects in this study.
This study has several limitations. First, the present study was a retrospective study, and BMI data were found, but waist circumference data were not included. Consequently, the diagnosis of metabolic syndrome could not be conducted by Japanese guideline. 27 However, the mean BMI was 22.8 kg/m2 and defined obese subjects might be excluded. Second, the study subjects were relatively elderly, and the present results may not be extrapolated to the general population.
In conclusion, this study shows that HDL-C and BMI were independently and significantly correlated with adiponectin in non-diabetic men and women, and that simple correlations of TG and TG-rich lipoprotein cholesterol to adiponectin were found but not independently. These results suggest that high adiponectin may have some kind of role in the increased HDL-C concentrations, implicated in the reduction of cardiovascular disease risk, in non-diabetic subjects.
Footnotes
Declaration of conflicting interests
Professor Hiroshi Yoshida received honoraria for speaking activities from Astellas, Amgen, Bayer, Kowa, Mochida, MSD, Shionogi, Takeda.
Funding
Research funds were provided in part by the Jikei University Research Fund and Grant-in-Aid for Scientific Research (26461116) from Japan Ministry of Education, Culture, Sports, Science and Technology (Yoshida H).
Ethical approval
This study was approved by the Ethics Committee of the Jikei University School of Medicine (24-100; 15-166).
Guarantor
HY.
Contributorship
Yoshiharu Tomono is in charge of data collection, data-analysis, data discussion, manuscript writing. Chika Hiraishi is in charge of data collection and data discussion. Hiroshi Yoshida is in charge of data-analysis, data discussion and manuscript writing.
