Abstract
The human precorneal tear film is a special body fluid, since it is a complex mixture of proteins, lipids, small bioactive molecules, and their concentrations and relative distribution represent not only the metabolic state of the ocular surface but also the systemic and local homeostasis of the outer eye and the human body. This suggests that biochemical analysis of the precorneal tear film composition may provide a non-invasive tool for diagnosis and monitoring of disease progression or treatment efficacy in human medicine. However, collecting tears is demanding, and obtaining reproducible and unaltered samples is challenging because of the small sample volumes of tears. Several methods are available for tear collection as a preparatory step of precorneal tear film analysis, and the collection method used has to be assessed since it has a critical impact on the effectiveness of the assays and on the quality of the results. Each sampling method has advantages and disadvantages; therefore, it is not easy to choose the appropriate collecting method for tear collection. To overcome these limitations various methods have been recommended by different authors for special aspects of specific tests. The aim of our review was to evaluate tear sampling methods with regard to our ongoing biochemical analysis.
Introduction
Precorneal tear film (PCTF) as a biological fluid is very easily accessible with non- or very low-invasive methods at a relatively low cost. It not only lubricates the ocular surface carrying secreted molecules from corneal epithelial cells and tissues producing tear components but can also represent the physiological status of the body. Due to the very limited number of samples and the relative instability of the components, sample collection is a critical step in tear research and diagnostics.
In the present review, we summarize the most commonly used tear sampling methods, emphasizing their advantages and disadvantages based particularly on the subsequent analysis.
Tear biology: Functions and pathological relations of the human tear
The tear film covering the ocular surface has several functions including protecting the external surface of the eyeball thus constituting a mechanical and antimicrobial barrier. It lubricates the eye surface and nourishes also the avascular tissues of the cornea. Since the tear film is also an optical refractive medium, its stability is pivotal in achieving appropriate vision.1–3
The human tear film is composed of two layers: there is a lipid layer and an aqueous layer that include soluble proteins and mucins and also membrane-bound mucins. 3 The lipid component, which originates from the Meibomian glands of the tarsus, forms the superficial layer of the tear film. The aqueous component is primarily secreted by the lacrimal gland. This is the thickest layer, which contains water, electrolytes and diverse proteins, peptides and glycoproteins. Mucins, which are glycoproteins expressed by epithelial tissues of mucous surfaces, protect tissues since they are antioxidants, provide lubrication and inhibit bacterial adherence.4–7
Under normal conditions, the PCTF flow in humans is around 0.5–2.2 μL/min with a turnover rate of approximately 16% per minute. 8 PCTF volume present in the human eye is 7–10 μL. 9 A continuous cycle of tear production, evaporation, drainage and absorption results in a dynamic equilibrium of the ocular surface. Osmolarity is the index of tear dynamics and is mainly determined by the electrolytes of the aqueous phase of the PCTF. Under normal conditions the expected range is 302 ± 8 mOsm/L. The dry eye workshop in 2007 identified increased tear osmolarity and tear film instability as ‘core mechanisms’ of DE, regardless of the oetiology.4,6 DE studies using the TearLab Osmolarity System have found that the mean tear osmolarities of the mild-to-moderate and severe DE patients were 315.0 ± 11.4 and 336.4 ± 22.3 mOsm/L, respectively. 5
Furthermore, tears have to be viscous enough to protect and lubricate the surface, but not too viscous so as to avoid ocular surface damages of high shear forces caused by blinking. The normal tear fluid has a viscosity of 1.3–5.9 cP. 7
Properties of precorneal tear film (PCTF).

Irritating stimuli like environmental fluctuations, diurnal patterns and physiological status indicate reflex tear secretion via activation of the corneal nerves.8–12 Open and closed eye PCTF differ in composition and origin, hence eye closure also influences the result of tear analysis. 13
Layers of tears: Origins, components and main roles.

PCTF: precorneal tear film.
Major bioactive components of PCTF
Despite its small volume, tear film is a remarkably complex biological fluid consisting of peptides, electrolytes, lipids, carbohydrates, salts and small bioactive molecules such as amino acids, nucleosides, vitamins, etc.10,19 Normal tears have a total protein concentration of approximately 7 g/L and they contain hundreds of different proteins, though the method of tear collection greatly influences the relative proportion of the proteins present in any individual tear sample, as clarified in the early 1980s.20–22 Many of the tear proteins play an important role in corneal wound healing, inflammatory processes and corneal protection against various pathogens. 23
The most frequent proteins detected in tear samples include lactoferrin, lysozyme, secretory immunoglobulin A (sIgA), lipocalin, superoxide dismutase, cystatins and α1-protease inhibitor. These proteins account for more than 90% of all tear proteins.2,24,25 In the early years, tear film protein profiles were characterized using gel electrophoresis and Edman degradation.26–28 Later, sensitive immunoassay-based methods identified other proteins in mammals' tears, including growth factors, neurotrophic factors, cytokines and cell adhesion molecules, matrix metalloproteinases, immunoglobulins and insulin.29,30
Prospects for the future: Tear diagnostics
Body fluid analysis is a widely accepted, readily repeated, convenient and low cost method in diagnostics. Biomarker screening of various body fluids may have potential benefits not only for the examination of physiological processes but also for the early diagnosis and effective therapy of several diseases. Fluid biomarkers include macromolecules such as lipids, proteins, RNA and DNA, as well as cells such as immune, endothelial or even cancer cells. 31
Tears, a body fluid exposed to both internal and external environment, contain an amazing amount of molecular information, which is useful for the diagnosis, prognosis and treatment of ocular surface diseases. This may promote the development of personalized medicine and the utilization of biomarkers in certain diseases. 10
Tear collection methods and their application in practice
Quantitative determination of tear proteins is of increasing interest in ophthalmology, but still there remains a technical problem due to small sample volumes available and the complexity of sample composition. 32 Tear sampling is definitely a major challenge and has the greatest significant influence on the precision and reproducibility of the analytical results.
Direct sampling methods
Regarding the direct sampling method, microcapillary tubes (MCT) or micropipettes are used for sampling and this manner frequently requires previous stimulation or instillation of different volumes of saline (100–200 μL) into the cul-de-sac and collecting after sufficient mixing. This procedure can cause dilution and may not permit collection of samples from specific sites of the ocular surface. 33 Kalsow et al. investigated tear cytokine response to multipurpose solutions in contact lens wear. Prior to contact lens removal, non-stimulated tears (NST) were collected from each eye from the inferior tear meniscus between the 6 o'clock and lateral canthus positions using a 10 μL flame-polished glass micropipette. Following collection, a 5.5 μL volume of tear was immediately transferred to a sterile 0.2 mL tube containing 49.5 μL of storage solution to produce a 1:10 tear dilution for immediate storage at −80℃. 34
In 2012, Guyette et al. compared low-abundance biomarker concentrations in capillary-collected NST and washout (WO) tears of aqueous-deficient and normal patients. Ten-microliter polished micropipettes were used to collect tears from the inferior marginal strip taking great care in minimizing ocular surface contact. Tear collection rate was continuously monitored. Individual NST samples were collected in 10 min aliquots and then immediately transferred to a sterile polymerase chain reaction (PCR) tube. An equal volume of assay buffer was added and the sample was stored at −86℃. A total of at least 6.5 μL NST was collected from each study participant; each 10 min aliquot was stored without delay in a separate PCR tube. Prior to WO tear sample collection 10 μL sterile physiologic saline solution was added to the lower conjunctiva by a digital pipette. The patient was instructed to gently close their eyes and avoid any eye movements for 1 min. Tears were then collected using the same method as for NST samples, but a shorter collection time of 5 min per aliquot was used to make up the 6.5 μL minimum volume requirement. Tear collection volume and time were continually monitored to measure the tear collection rate. 35
There have been several research projects in dry eye syndrome and today emphasis in dry eye research has shifted towards the role of inflammation in the anterior surface of the eye. 36 Since inflammatory mediators originating from various ocular surface sources and the main lacrimal gland do not constitute a totally homogenous mix the way tears are collected can influence the resulting biomarker profile. NST from the inferior marginal strip covers a broader spectrum of the sources, whereas stimulated tear (ST) samples contain a higher proportion of the lacrimal gland secretion. 1 Explicit protein profile differences between NST and ST demonstrate that these two sample types are not equivalent.37,38 Although NST represents specifically the inflammatory status of the ocular surface, the volume of NST is limited, especially in aqueous deficient dry eye (ADDE). Even though tear sampling frequently makes use of capillaries as they are less irritating and the resulting sample is an exact representative concentration of molecules, the main limitation of the method is the volume of the sample (2–3 μL) to be gained. 8
One way to increase the available tear sample volume is adding fluid (e.g. sterile saline) to the eye prior to sample collection, effectively ‘washing out’ ocular surface molecules.39,40 Validity of the WO method depends on the extent to which it changes the NST biomarker profile. By determining tear sIgA, inducement of reflex tearing is easily detected because tear sIgA concentrations decrease with reflex tear flow rate. Markoulli et al. found equal tear sIgA–total tear ratios in WO and NST, which suggests that WO samples do not significantly induce reflex tearing. Guyette's study evaluated WO tear collection as a replacement for capillary NST and applied this to compare biomarker concentrations between ADDE and non-ADDE patients.35,38,41,42
Indirect methods
Regarding indirect methods, collection of PCTF is carried out using absorbing supports such as Schirmer test strips (STS), filter paper disks, cellulose sponges and polyester rods. The most common method among them is STS collection. 43
Inflammatory markers were analysed in the PCTF of patients with ocular surface disease. Ten microliters of tear was collected by a Weck-Cell Sponge. The concentrations of interleukin (IL)-1β, IL-6 and pro-MMP-9 were measured by enzyme-linked immunosorbent assay (ELISA), and the MMP-9 activity was evaluated with gelatine zymography. 44
Ophthalmic sponges and extraction buffers were compared for quantifying cytokine profiles in tears using Luminex technology. Luminex detection of cytokine/chemokine profiles of tears collected with Merocel sponges was found to be useful in clinical studies, for instance to assess cytokine profiles evaluation in ocular surface diseases. 45
Samples obtained from the Schirmer procedure have a higher mucus, lipid and cellular content than MCT samples. 46 STS also suffers incomplete, non-uniform elution of proteins from the filter matrix. 43 Although micropipette and STS collection provide different biomarker profiles for a given donor, the correctly applied micropipette method is more consistent. 47 STS is widely accepted as the volume of sample collected with this method is larger compared to other methods, but it can cause reflexive tearing due to irritation, which increases the volume of the samples, therefore aggravates the detection of the investigated tear component(s), e.g. drug concentrations.
In comparative studies tears of one and the same patient are collected by several collection methods to determine the same biomarkers from different tear samples.
Green-Church et al. collected tears using small volume (1–5 μL) Drummond glass MCT tubes with 1.6 × slit-lamp magnification. Non-reflex tears were collected from the inferior tear prism without contact with the lower lid until a total of 5 μL were collected. During a separate visit, tear collection was performed by placing a STS over the lower lid. The lid was not anesthetized and the STSs were placed approximately 6 mm nasally from the lateral canthus. The subject was instructed to close their eyes for the 5 min test duration; the wet length was not recorded but was observed to be within reference ranges in all cases. The STS was then placed in a 1.6 mL amber Eppendorf tube at 4℃ until analysis. Their results suggest that the tear film collection method does impact the proteins present in the sample, so care should be exercised in choosing a tear collection method in order to best correlate to the experiment being conducted or the hypothesis being tested. 47
Tear collection methods of some authors including studies from the past few years with a large number of samples. All studies were performed in humans.
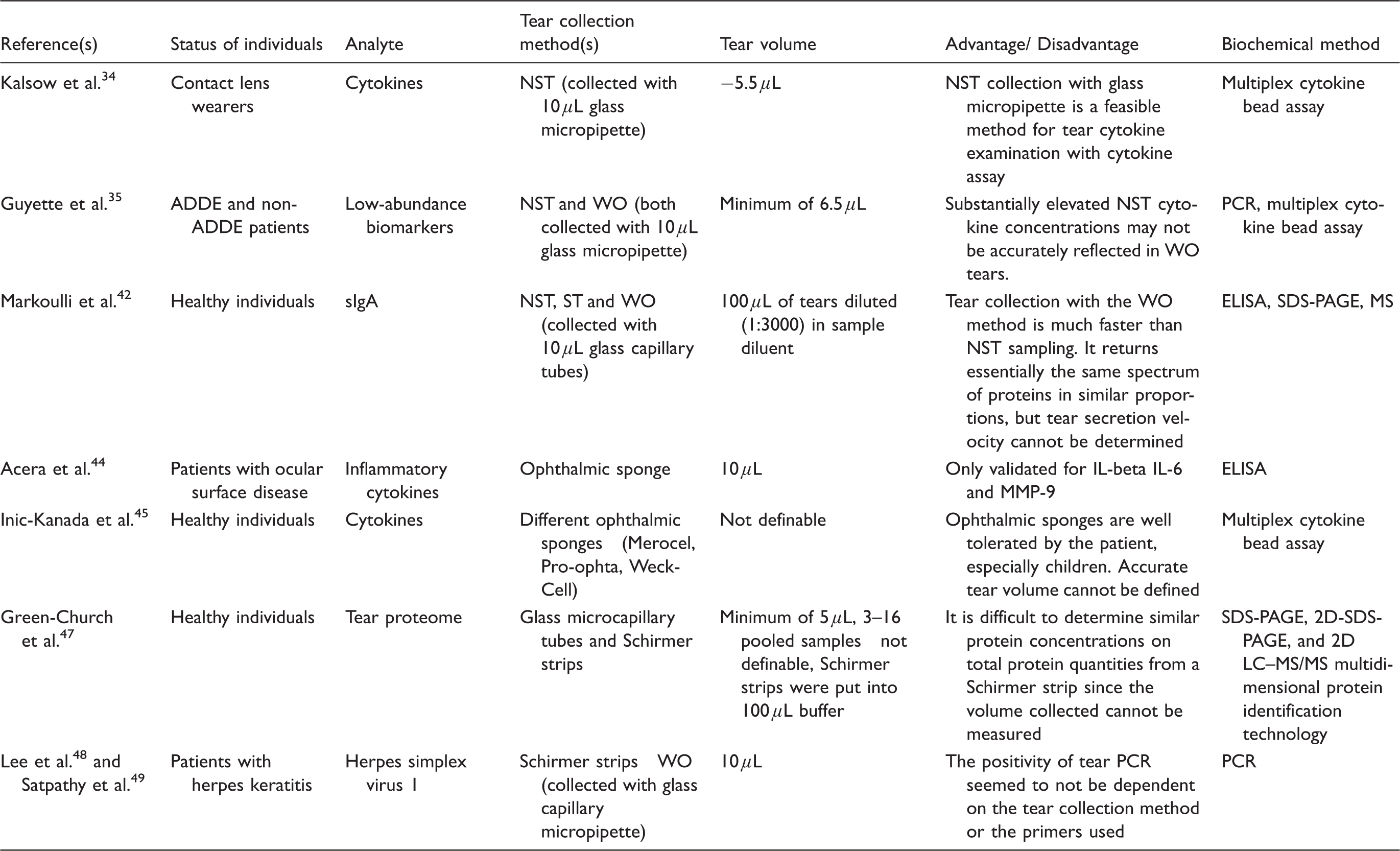
ADDE: aqueous deficient dry eye; ELISA: enzyme-linked immunosorbent assay; MS: mass spectrometry; NST: non-stimulated tear; PCR: polymerase chain reaction; SDS-PGE: sodium dodecyl sulphate polyacrylamide gel electrophoresis; sIgA: secretory immunoglobulin A; ST: stimulated tear; 2D LC: two-dimensional liquid chromatography; WO: washout.
The main advantage of the direct sampling methods is the straight way to collect tears from the ocular surface, and the main disadvantages are the possible need of dilution and the impossibility of sample collection from detailed sites of tears. They may be difficult to perform in practice, but analytically they provide the most proper analyte concentration of tears. The indirect methods, however, are easy to implement, but analytically they do not inevitably represent the biochemical characteristics of tears.
The critical aspects of tear sampling with respect to the subsequent analysis
In the last decade, advances in proteomics/metabolomics/lipidomics technologies have greatly expanded our knowledge of the biochemical composition of the ocular tear fluid. To date, a number of tear proteins and lipids have been identified as possible disease-related biomarkers. 51 The rapid development of various ‘omics’ methods facilitates the identification and examination of tear-based biomarkers. In some cases, these techniques require specific sample collection, handling and storage procedures. Hereunder we would like to summarize the most commonly used methods in proteomics and lipidomics with special regard to their minimum sampling requirements.
Proteomics
Qualitative and quantitative tear protein examination methods include one- and two-dimensional gel electrophoresis, ELISA, high performance liquid chromatography (HPLC), mass spectrometry (MS) related techniques such as MS–MS, matrix-assisted laser desorption/ionization time-of-flight MS, surface-enhanced laser desorption/ionization time-of-flight MS, liquid chromatography–MS, various antibody arrays, multiplex bead analysis, Western blot analysis, etc.52,53 Using highly sensitive techniques – like isobaric tags for relative and absolute quantitation – more than 500 tear proteins have been identified so far.
Previous studies have indicated that sample handling variables such as sample collection conditions and time, storage temperature and time, storage tube, freeze/thaw cycles and protease inhibitors have significant effects on the results of protein analysis. 54 In general, sample collection should be scheduled at the same time of day (e.g. early morning) and samples should be transferred onto dry ice immediately after sampling to prevent protein degradation. Addition of protective or stabilizing compounds (reducing agents, protease and peptidase inhibitors, etc.) would be desirable but inhibitor cocktails may interfere with the subsequent MS analysis. The collected samples should be aliquoted and stored with minimization of thaw/refreeze cycles, preferably at −80℃. 55 Theoretically, the frozen samples (at −20 to −80℃ or in liquid nitrogen) can be stored for years protected from degradation. 56
Another critical aspect of tear analysis is the limited volume of the samples and the relatively low number of proteins of interest. In addition, the stimulus conditions (NST versus ST) and collection technique can strongly affect the protein profile and volume of the tear sample. Fullard and Snyder observed that the concentrations of eight proteins (IgA-SC, IgA1, IgA2, IgM, IgG, IgA, transferrin, serum albumin) from the 12 analysed tear proteins showed significantly higher concentrations in NST, and only four (lactoferrin, tear-specific prealbumin, peroxidase, lysozyme) were in similar concentrations in both types of tears. 38 On the other hand, the total tear protein content of NST samples decreased from 9.1 to 6.0 g/L in the ST ones. 37 These data indicate that the diluting effect of reflexive tearing has a decisive effect on the quantitative composition of the tear sample and highlights the importance of controlling tear flow rate during tear collection. 55 Based on the above observation NST sampling seems to be more beneficial unless the experimental goal is the investigation of ST tears or reflexive tearing.
Lipidomics
While the protein composition of the human tear has been described in great detail, the lipidomic analysis of the tear is noticeably lagging behind due to the low lipid content of the tear fluid. 57 Because of the various difficulties (lipid diversity and complexity, chemical stability or instability of different types of lipids) the qualitative and quantitative analysis of tear lipids is a difficult task.
Regarding the technical limitations (sensitivity and performance of the method; volatility and stability of intact lipids) the commonly used analytical methods – gas chromatography–mass spectroscopy and liquid chromatography–mass spectrometry (LC/MS) – are not sufficiently efficient and accurate for the lipidomic analysis of the human tear. HPLC and its newer and faster ‘relative’, ultra-high performance liquid chromatography (UPLC) mass spectrometry (HPLC/MS and UPLC/MS) and atmospheric pressure ionization MS are more suitable methods for the examination of intact lipids in tear fluid. 58 Nuclear magnetic resonance spectroscopy, Raman and infrared spectroscopy may also be used as alternative approaches, although their sensitivity and selectivity is far below that of MS.4,58
The requirements of sample collection are the same as in the case of protein determination. The samples should be stored frozen at −80℃ or lower, multiple melting of the samples should be avoided and the samples should be handled in deeply frozen conditions (e.g. at −80℃) until assessment. During the examination of lipids that are sensitive to light or the O2 content of the air – such as retinoids – special attention is needed, e.g. usage of amber-coloured Eppendorf tubes. 58 Basal tear collection using capillary tubes is recommended for tear lipid analysis as the concentration of several lipid classes is below the limit of detection in reflex and flush tears. 59
Conclusions
Biomarker-based diagnostics, as well as personalized medicine utilizing its results are becoming more widely used in modern medicine. In addition to the ‘classical’ sampling methods (e.g. biopsy), there is a growing demand for fast, painless and non-invasive sampling procedures such as examination of various easily accessible body fluids. The identification and potential application of biomarkers carried by urine, sweat, amniotic fluid and last but not least precorneal tear are the subjects of intensive interest and research nowadays.
Similarly to other newly developed methods, precorneal tear analysis has no standard methodology. Sampling techniques used by different research groups are not exceptions to this rule. Both the investigation of ‘tear physiology’ and tear biomarker research are based on the biochemical characterization of so-called basal (or NST) tears, which differ from ST tears (also known as reflex tears) both in terms of quantitative and qualitative biochemical characteristics. In practice, two quickly and easily adaptable sampling methods, the STS and the MCT technique are widely used for the collection of NST. Based on the previous statements the authors unequivocally recommend the capillary-collected NST sampling method for protein content determination since it represents the most accurate protein concentration of tears. For lipid content determination, NST is the most commendable sampling method as well, but in this case glass or polished micropipette is also available. There are no special considerations for mucin analysis as they consist of glycocalyx expressed by epithelial tissues of mucous surface and there is no direct simple method to evaluate ocular surface glycocalyx.
Since the STS method triggers more or less intense tearing, this technique is suitable for the isolation of a mixed sample containing NST and ST. Thus, the analysis of samples collected by the STS method does not necessarily represent the biochemical properties of NS tears.
In the last decade, numerous studies have addressed the comparison of STS and MCT methods. It was observed in the late 1960s that the STS method may underestimate the actual protein concentrations of tears due to increased fluid flow. Some studies have since revealed that not only the diluting effect of reflex tearing, but also the protein binding and retention capacity – which is strongly associated with the molecular weight and hydrophobic surface area of the studied proteins – of STS paper is responsible for the lower protein content of STS samples. The STS method modifies both the quantitative and the qualitative characteristics of tear samples. In the late 1970s and 1980s, various research groups reported elevated concentrations of certain proteins – like albumin, IgG, transferrin, urokinase and plasmin as well as various intracellular enzymes involved in metabolism such as aspartate aminotransferase and alanine aminotransferase, lactate dehydrogenase, aldolase, glucose-6-phosphate dehydrogenase – in the STS-collected tears compared to MCT samples. Comparing the proteome of tear samples collected by MCT and STS methods Green-Church et al. found that more than 50 different proteins were detected exclusively in the STS samples (MCT-specific proteins: 13; STS-specific proteins: 54; overlapping proteins: 30; total: 97 proteins; determination: in-gel tryptic digestion followed by liquid chromatography–tandem mass spectrometry and multidimensional protein identification technology). Several studies have confirmed that the STS method – which often triggers irritation in the lower cul-de-sac of the eye – changes the protein composition of tears by injuring the conjunctival surface and microvasculature.
In contrast to STS-based sampling, the MCT technique is believed to be a less invasive procedure. If it is performed by a specialist who has practice and experience in this collection method, the MCT technique does not induce reflex tearing, nor does it involve a potential risk of injury. Therefore, the MCT sampling method is more suitable for the collection of NST than the STS procedure. On the other hand, some researchers have highlighted the following disadvantages of the MCT method: the sampling is interrupted by blinking; STS could be more pleasant for the test subjects than the capillary tube; the investigator has to hold the capillary tube for the duration of the sampling procedure, which entails constant and prolonged work on the open eye.
For the compensation of low sample volume, several research groups use the so-called WO method, in which the tears are ‘flushed out’ through the addition of exogenous fluid (e.g. sterile physiol. saline). The diluting effect of the WO method is reflected by the suppressed concentrations and the decreased variances of the most abundant cytokines (e.g. IL-8, IL-1β and vascular endothelial growth factor). In addition, the concentrations of ‘minor’ tear components drop below the detection limit and the slight differences between the samples become unrecognizable. Therefore, the WO method can be used successfully only in those experiments which target proteins found in high concentration in tears.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Local bioethics committee (DE KK RKEB/IKEB 4242-2014, and HBR/052/01761-2/2014) and Declaration of Helsinki (1996).
Guarantor
AKB.
Contributorship
All authors researched literature to perform the study. AR and KK wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
