Abstract
Background
Plasma proprotein convertase subtilisin/kexin type 9 (PCSK9) has been reported to be related to several risk factors and diseases such as inflammatory markers and coronary artery disease. The aim of present study was to investigate whether plasma PCSK9 concentration was associated with coronary artery calcification.
Methods
A total of 403 consecutive untreated patients with angina-like chest pain, who received electron beam computed tomography, were enrolled and a coronary artery calcification score (CACS) was also measured. The baseline clinical characteristics were collected and blood sample was taken after 12-h fasting. The plasma PCSK9 concentrations were determined by ELISA in all patients, and the relationship between plasma PCSK9 concentrations and CACS was investigated.
Results
Patients with coronary artery calcification (CACS > 0) had significant higher plasma PCSK9 concentrations compared with those (CACS = 0) without coronary artery calcification (258.58 ± 69.53 ng/mL vs. 202.53 ± 52.17 ng/mL, P < 0.001). Patients with highest PCSK9 concentrations had the highest CACS. Multivariable linear regression analysis suggested that PCSK9 was independently associated with coronary artery calcification (P = 0.002) after adjusting for traditional cardiovascular risk factors. Furthermore, the area under the curve for the plasma PCSK9 concentration in predicting coronary artery calcification was 0.736 (95% CI: 0.687–0.785, P < 0.001), with a sensitivity of 66% and specificity of 70%.
Conclusion
A positive association between plasma PCSK9 concentration and coronary artery calcification in untreated patients with angina-like chest pain was observed in our study, suggesting that further investigation may be needed in order to confirm our primary findings and explore the clinical implications.
Keywords
Introduction
Coronary artery calcification (CAC) has long been known as an important pathophysiological phenomenon, which may be associated with atherosclerotic processes. 1 Previous studies have demonstrated that CAC is a specific marker of coronary artery disease (CAD) and is directly related to the atherosclerotic plaque burden. 2 Furthermore, it has been shown that CAC is an efficient risk factor for cardiovascular and all-cause mortality in patients with cardiovascular risk factors and asymptomatic patients.3–6 More recently, there have been many reports that suggest that there is also an association between the presence of CAC and disease (diabetes mellitus [DM], chronic kidney disease), risk factors (age, male gender, smoke, body mass index [BMI]), an inflammatory biomarker (C-reactive protein) and cytokines (endothelia-1 [ET-1]).7–14 Moreover, a number of previous studies have suggested that dyslipidaemia is the earliest and most important risk factor associated with the development of CAC.15,16 Previous data have indicated that there is an association between elevated concentrations plasma low-density lipoprotein cholesterol (LDL-C) and lipoprotein(a) and CAC. 16 However, the mechanism of the development of CAC has not been fully revealed till now. As a consequence, those studies that focus on CAC-related factors may be of clinical interest.
Proprotein convertase subtilisin/kexin type 9 (PCSK9) plays an important role in the regulation of cholesterol metabolism. 17 Recent data have suggested that PCSK9 may be involved in multiple patho-physiological processes, such as inflammation, atherosclerosis and glucose metabolism.18–20 Our previous studies have indicated that the concentrations of plasma PCSK9 show a positive correlation with concentrations of several inflammatory markers.21,22 Based on the results that PCSK9 has been involved in both lipid metabolism and inflammation, we hypothesized that concentrations of plasma PCSK9 might be related to the presence and also to the extent of any CAC. Interestingly, a very recent study has reported that PCSk9 is independently associated with the presence and severity of CAC in statin-treated symptomatic familial hypercholesterolaemia (FH) patients. 16 However, in the Dallas Heart Study, the investigators did not find a positive association between concentrations of plasma PCSK9 and CACS in statin-naive patients. 23 The exact reason for the disparity of results concerning the relationship between PCSK9 concentrations and CAC between the two studies is unknown. Statin treatment may be an explanation because the previous studies have indicated that statin therapy could rapidly enhance PCSK9 expression. 24 Therefore, in this study, we try to investigate the relationship between concentrations of PCSK9 and CAC in patients with angina-like chest pain, who did not receive any lipid-lowering drugs.
Patients and methods
Study design and population
Four hundred and three consecutive patients with angina-like chest pain were included in this cross-sectional study. All subjects attended the Centre for Cardiovascular Diseases, Beijing, between October 2015 and July 2016 and all underwent cardiac CT using a 64-slice multidetector CT scanner. In addition, all subjects had blood samples collected and all underwent a CAC scan.
CAD was defined as a stenosis ≥50% of luminal diameter in at least one of the major epicardial coronary arteries detected by the elective coronary angiography. DM was defined as a fasting plasma glucose (FPG) concentration ≥7 mmol/L on multiple determinations or the current use of insulin or oral hypoglycaemic agents. Hypertension was defined as a blood pressure of ≥140/90 mmHg on at least two occasions in different environments or the current use of antihypertensive drugs.
To avoid the effect of lipid-lowing drugs on plasma PCSK9 concentrations, we enrolled subjects with no previous history of treatment with statins and/or other lipid-lowing drugs. Other exclusion criteria were: a history of coronary revascularization (percutaneous coronary intervention or coronary artery bypass graft surgery), heart failure or cardiomyopathies, hepatic failure, renal dysfunction, haemolytic disorders, thyroid disease, neoplastic diseases and acute infectious or inflammatory conditions.
Laboratory examinations
Fasting blood samples (12 h overnight) were obtained between 7:00 and 9:00 a.m. from patients. All plasma samples were collected using EDTA-containing tubes and were store at −80℃. The concentrations of plasma total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C) and LDL-C were measured using a Hitachi 7150 automated analyser (Hitachi, Japan). Haemoglobin A1c (HbA1c) concentrations were measured using a Tosoh G7 HPLC Analyser (TOSOH Bioscience, Japan). Plasma PCSK9 concentrations were measured using a commercial sandwich enzyme immunoassay (Quantikine ELISA, R&D System Europe Ltd) with a lower limit of detection of 0.096 ng/mL. The measurement range of this assay is 0.16 ng/mL to 10 ng/mL; intra-assay imprecision (CV) is 1.5–2.6% and inter-assay imprecision 2.90–7.09%. All other parameters were measured using standard methods as outlined previously.18–22
Measurement of CACS
A 64-row spiral CT scanner (Light Speed VCT, GE Healthcare, Milwaukee, Wisconsin) was used to obtain CACS. The machine was set up with a rotation time of 0.35-s and a pitch of 0.16 n to 0.22, while the tube current of 200 to 550 mA and tube voltage of 120 kV. Patients undergoing this procedure should have heart rates ≤70 beats/min. If necessary, those individuals in whom treatment with beta-blockers was contraindicated, were therefore given 25 to 50 mg of metoprolol (Selokeen, AstraZeneca, Zoetermeer, the Netherlands) orally 1 h before scanning, as described in our previous report. 13 At least three contiguous pixels present, and a density of 130 HU was defined as calcium. The CACS was the sum of calcium scores of four main coronary arteries according to the Agatston scoring algorithm. 25 The main coronary arteries include the left main, left anterior descending, left circumflex and right coronary artery.
Statistical analysis
The data were conveyed as means ± standard deviation (SD) for the continuous variables and as the number (percentage) for the categorical variables. The correlations between the parameters were assessed by Spearman’s correlation (non-parametric data) and Pearson’s correlation (parametric data).
The patients were grouped according to their CACS. We compared the CAC risk factors between the two groups. The differences of clinical and biochemical parameters between groups were analysed using independent sample t-test, Mann-Whitney U test, χ2-tests, and Fisher’s exact test where appropriate. Multivariable linear regression analysis was applied to identify significant independent association variables with the absence of CAC. Receiver-operator characteristic (ROC) curve analysis was used to assess the value of the PCSK9 as a predictor for CAC. The Youden index (sensitivity + specificity−1) was calculated to determine the locally adequate cut-off point for the PCSK9 and the optimal cut-off value was considered as the corresponding cut-off value for the highest Youden index. The SPSS Version 19.0. Software Program (Chicago, Illinois, USA) was performed for all analysis and a P-value of less than 0.05 was considered to be statistically significant.
Results
Baseline clinical characteristics and laboratory findings
Comparison of baseline clinical characteristics between patients with and without coronary calcification.
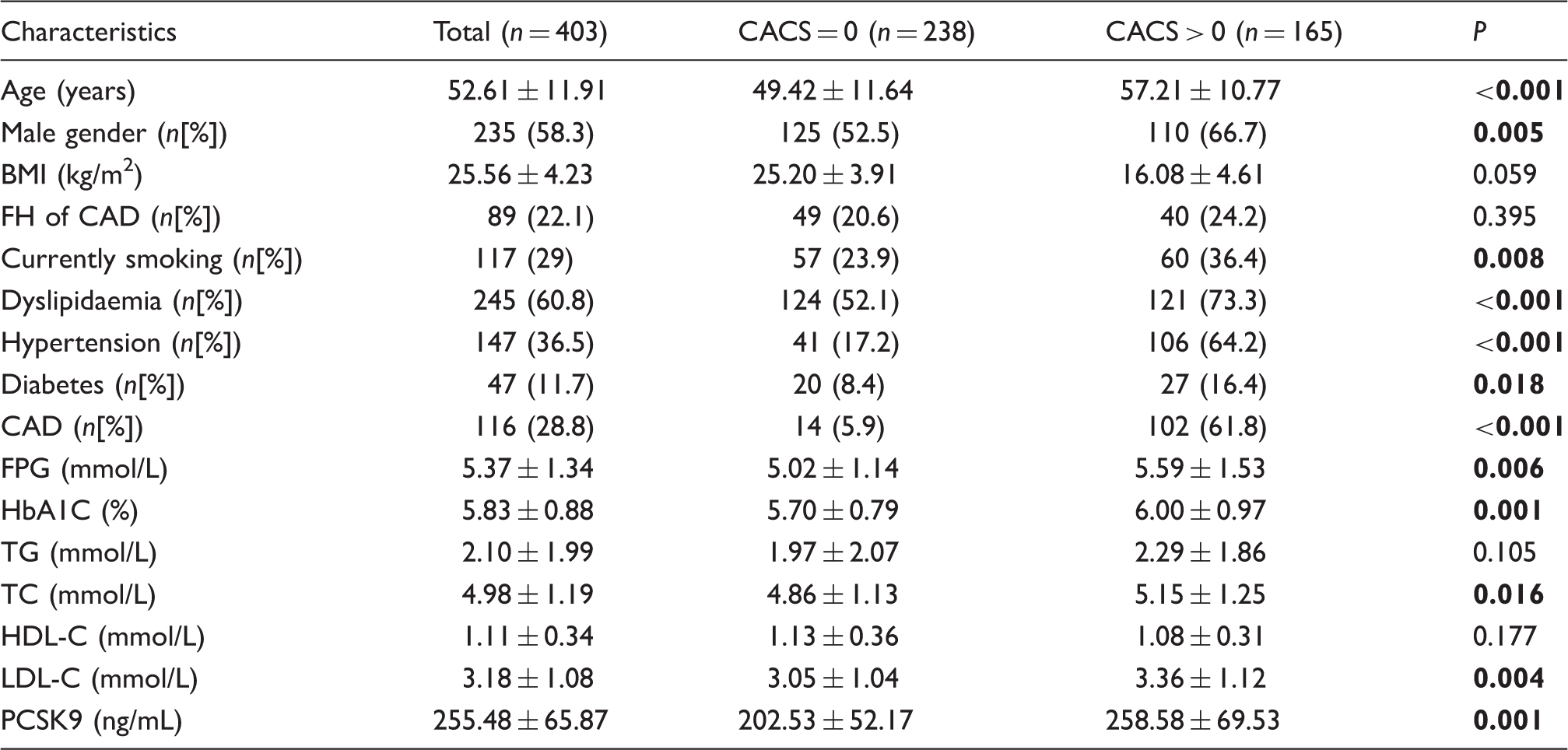
CACS: coronary artery calcification score; BMI: body mass index; CAD: coronary artery disease; FH: family history; FPG: fasting plasma glucose; HbA1c: haemoglobin A1c; PCSK9: proprotein convertase subtilisin-kexin type 9; TG: triglyceride; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol.
Note: The data shown are the mean ± SD or n (%). Bold values indicate statistical significance.
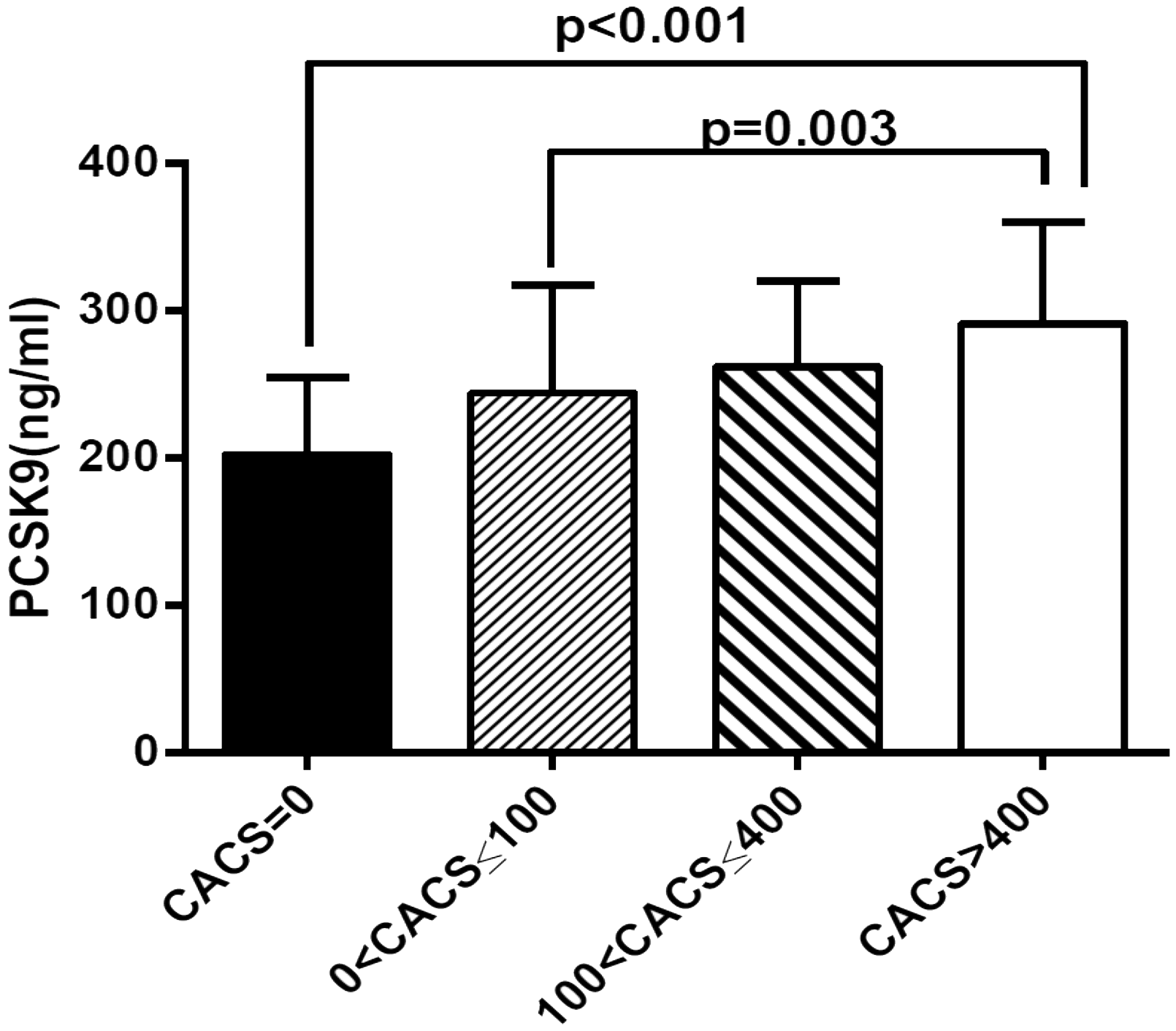
Relationship between concentration of plasma PCSK9 and CACS (202.53 ± 52.17 ng/mL vs. 244.24 ± 73.10 ng/mL vs. 261.95 ± 58.13 ng/mL vs. 291.27 ± 68.73 ng/mL, P < 0.001).
Correlations of CACS with PCSK9 concentrations and other factors
Correlations of CACS with variables.
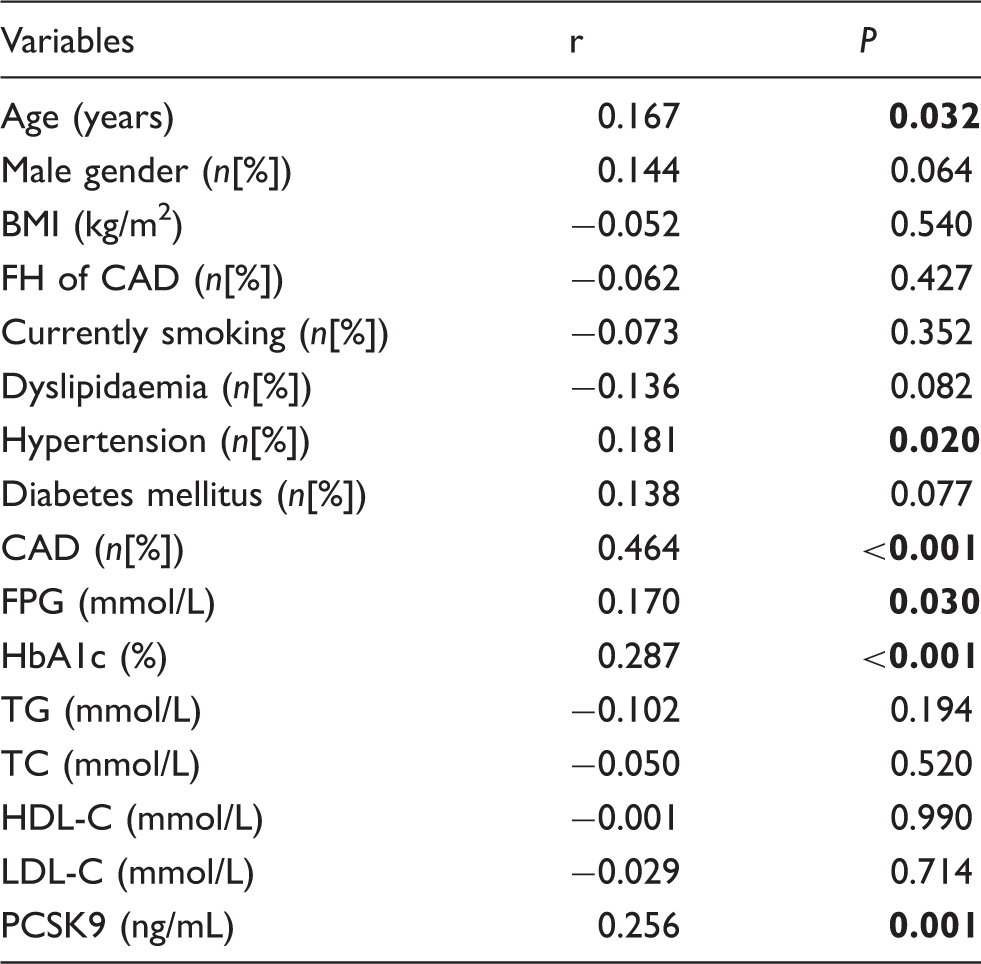
Note: Spearman correlation analysis is shown. Bold values indicate statistical significance.
CACS: coronary artery calcification score; PCSK9: proprotein convertase subtilisin-kexin type 9; CAD: coronary artery disease; FH: family history; BMI: body mass index; TG: triglyceride; TC: total cholesterol; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; FPG: fasting plasma glucose;HbA1c: haemoglobin A1c.
Moreover, the patients with CAC were further stratified into three groups according to the CACS (group 1: CACS = 0–100; group 2: CACS = 100–400; group 3: CACS > 400). The plasma concentrations of PCSK9 increased in each of the three groups as the CACs increased. PCSK9 concentrations were: group 1, 244.24 ± 73.10 ng/mL; group 2, 261.95 ± 58.13 ng/mL; group 3 291.27 ± 68.73 ng/mL, (P = 0.003; Figure 1).
Furthermore, the PCSK9 concentration tertiles were positively related to CACS (Tertile 1: Patients with plasma PCSK9 concentrations were between 120.13 and 222.55 ng/mL, n = 55; Tertile 2: Patients with plasma PCSK9 concentrations between 223.05 and 275.36 ng/mL, n = 55; Tertile 3: Patients with plasma PCSK9 concentrations between 275.40 and 487.96 ng/mL, n = 55; P = 0.009; Figure 2).
Relationship between log(CACS + 1) and PCSK9 concentration tertiles. Tertile 1, Patients with plasma PCSK9 concentrations between 120.13 and 222.55 ng/mL, n = 55; Tertile 2, Patients with plasma PCSK9 concentrations between 223.05 and 275.36 ng/mL, n = 55; Tertile 3, Patients with plasma PCSK9 concentrations between 275.40 and 487.96 ng/mL, n = 55.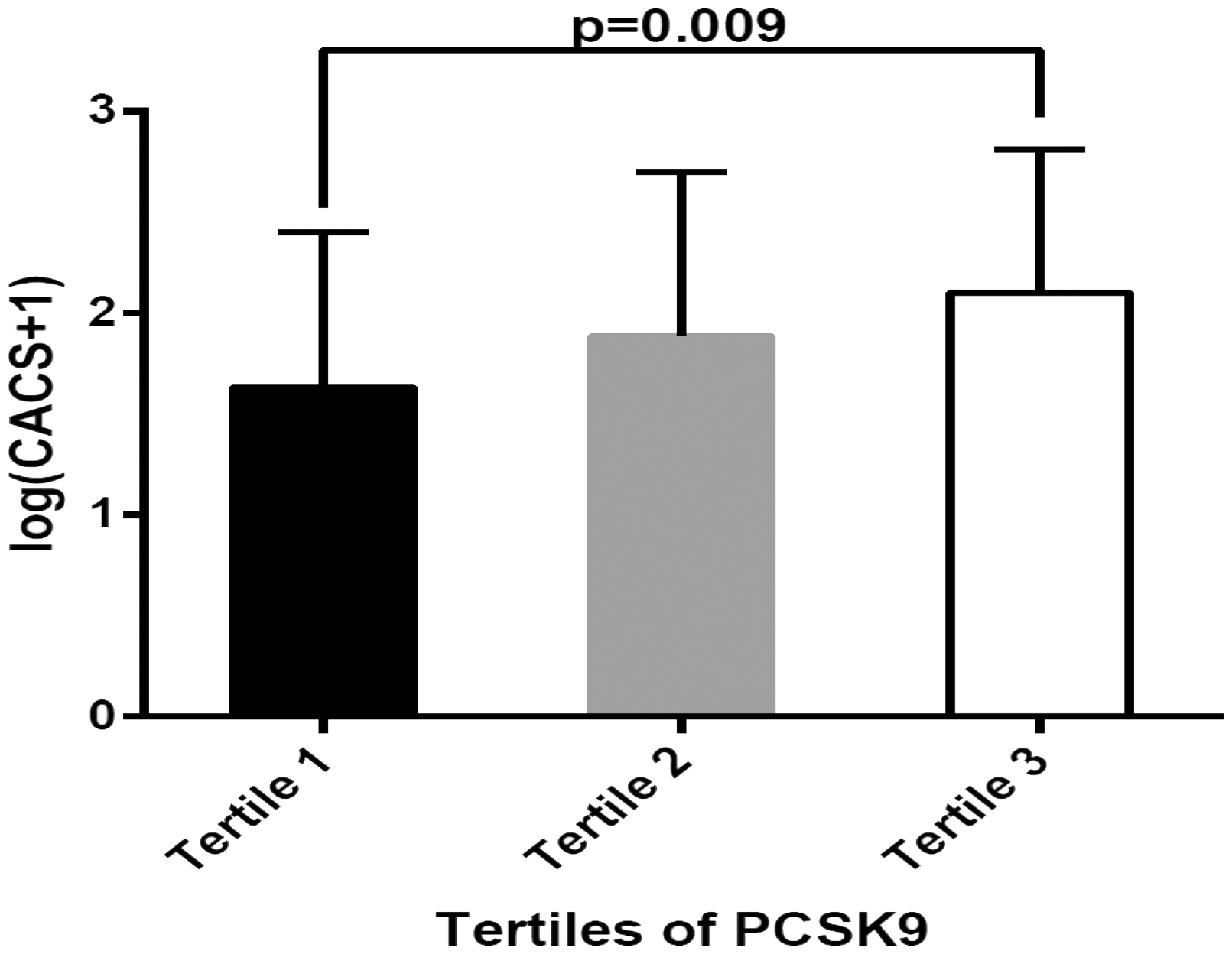
Multivariable linear analysis of CACS and plasma PCSK9 concentrations
Contributors to CACS as assessed by multivariable linear regression analysis.
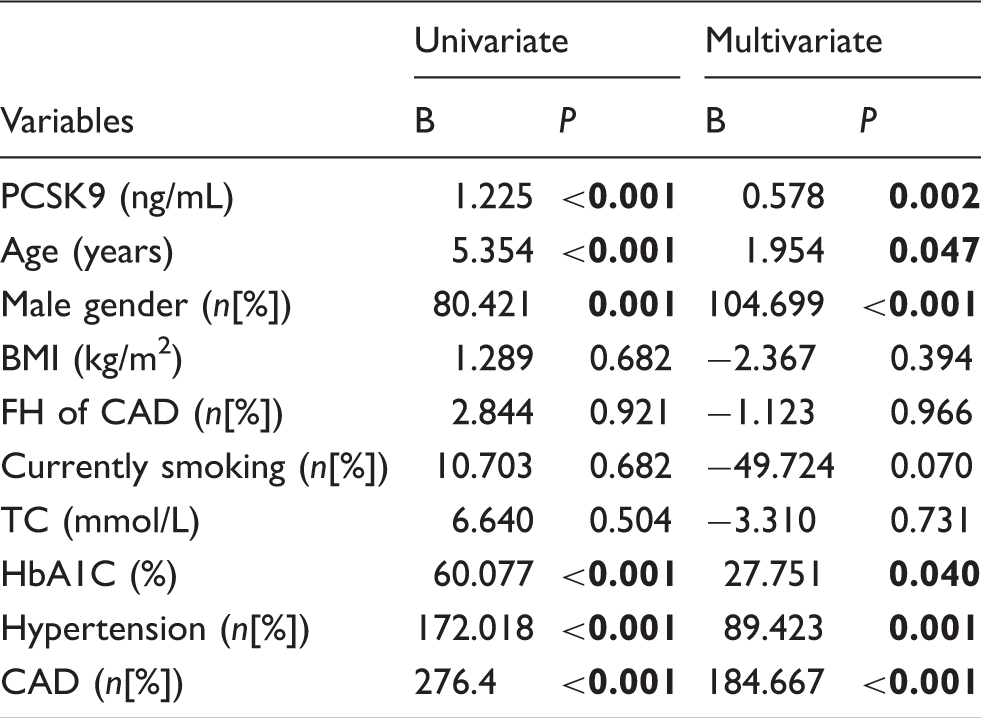
Note: Multivariable linear logistic regression analyses were used. Bold values indicate statistical significance. The adjusting known confounders were including age, gender, BMI, TC, HbA1C, hypertension, smoking, family history of CAD, and CAD.
CACS: coronary artery calcification score; PCSK9: proprotein convertase subtilisin/kexin type 9; BMI: body mass index; FH: family history; TC: total cholesterol; HbA1C: haemoglobin A1c; CAD: coronary artery disease.
Discussion
CAC is the hot topic in the area of cardiovascular research, and the potential mechanisms that give rise to it are not fully understood. In this study, our data from patients who were not receiving lipid-lowering medication showed a positive association between plasma PCSK9 concentration and both the presence and severity of CAC, even after adjusting for age, male gender, hypertension, DM and CAD. This suggests a potential role of PCSK9 in CAC susceptibility.
The clinical presence of CAC is seen in aging, hypertension, chronic kidney disease, DM and is a known risk factor for cardiovascular disease.7–9,26 In recent years, abundant evidence has demonstrated that CAC may also be associated with subsequent coronary events such as all-cause mortality in individuals with or without CAD. 3 Hence, the identification of the potential risk factors for CAC may have important clinical implications. Recent studies have shown that lipoprotein(a) concentration was associated with CAC and that higher concentrations of serum LDL particles indicated an increased risk of CAC progression in patients with CAD using repeated CTA examination.16,27 Moreover, previous studies have provided evidence that increased plasma concentrations of pro-inflammatory interleukin (IL)-6 and reduced concentrations of anti-inflammatory IL-8 and IL-13 were found to be linked to CAC. 28 A prospective study has demonstrated that tumour necrosis factor (TNF)-a, a biomarker of inflammation, was independently associated with CAC progression. 29 Additionally, several studies have suggested that novel inflammatory biomarkers such as serum soluble IL-2 receptor and soluble urokinase plasminogen activator receptor have also been found to be associated with CAC.30,31 Our previous study indicated that plasma big ET-1 was independently associated with CAC in Chinese Han population who had a manifestation of chest pain. 13 Taken together, the findings of these previous studies strongly suggest that CAC is a complex patho-physiological process, which requires further investigation.
Interestingly, a recent study reported that, in symptomatic FH patients, PCSK9 was independently associated with the presence and severity of CAC. 16 However, there is no data currently available with regard to the relationship between plasma PCSK9 concentration and CAC in the general population. Notably, our previous studies have demonstrated an association between plasma PCSK9 concentrations and fibrinogen, resistin and small dense LDL-C in CAD patients.21,22,32 Plasma PCSK9 concentrations were also independently associated with the severity of coronary artery stenosis and even major adverse cardiovascular events in stable CAD patients.18,19 Hence, we hypothesized that there might be an independent association between plasma PCSK9 concentrations and CAC in the general population. In the present study, we chose 403 consecutive patients with angina-like chest pain who had also undergone 64-slice multidetector CT scanning in our lipid clinics. Those patients did not receive lipid-lowering drugs in order to ensure the quality of the study; we had previously shown that lipid-lowering drugs such as statins and ezetimibe could rapidly increase the concentrations of plasma PCSK9. Indeed, we found that those patients with CACS had higher concentrations of PCSK9 than those without CACS. The positive association of plasma PCSK9 concentration and CACS was still observed, even after adjustment for traditional risk factors. This suggests that the role of PCSK9 in the process of coronary calcification merits further investigation.
PCSK9 has also been demonstrated to be a marker the presence and severity of several diseases. For example, recent studies have shown a correlation between circulating PCSK9 concentrations and the severity of CAD, together with poor prognosis.18,19 Moreover, the findings of one study has suggested that increased concentrations of PCSK9 are associated with DM and insulin resistance. 33 At the same time, increased circulating concentrations of PCSK9 have also been shown to be associated with HbA1c concentrations. 20 Furthermore, a post hoc analysis showed that plasma PCSK9 concentrations correlated with both the severity and the extent of peripheral artery disease. 34 These findings might suggest that PCSK9 has a role in different patho-physiological status. That is the reason why our present study focuses on the relationship between plasma PCSK9 concentrations and CAC. In the present study, our results have shown that PCSK9 has a discriminatory role for predicting CACS, while the sensitivity is 66% and specificity is 70% (AUC = 0.736, P < 0.001), although the power is relatively lower. We believe that the findings of the present study have provided novel information regarding the possible role of PCSK9 and coronary calcification, suggesting that further study is needed.
The present study has several limitations. Firstly, and most importantly, there is possible selection bias, in terms of the population examined, for example, a single centre with small sample size. Secondly, because the study was observational, the conclusion of causality between PCSK9 concentration and CAC cannot be made. Thirdly, as only a Chinese population was studied, the data might only be applicable to a single ethnic group.
In conclusion, the findings of the present study have shown a positive correlation between plasma PCSK9 concentration and CAC in untreated Chinese patients with angina-like chest pain. We believe further studies are required to confirm these preliminary findings and explore further clinical implications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was partly supported by National Natural Scientific Foundation (81241121), Capital Special Foundation of Clinical Application Research (Z121107001012015), Capital Health Development Fund (2011400302, 201614035), Beijing Natural Scientific Foundation (7131014), and CAMS Major Collaborative Innovation Project (2016-I2M-1-011) awarded to Dr. Jian-Jun Li, MD, PhD.
Ethical approval
The study complied with the Declaration of Helsinki. The hospital ethics review board (Fu Wai Hospital & National Centre for Cardiovascular Diseases, Beijing, China) approved this study (REC number: 2013-442). Informed written consent of all the patients was obtained in this study.
Guarantor
JJL.
Contributorship
JJL, XZ and HWZ researched literature and conceived the study. RXX, YLG, CGZ, QD, JS, GL, XLL, PQ and NQW were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. XZ, YZ and SL wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
