Abstract
Background
Human mercaptalbumin and human non-mercaptalbumin have been reported as markers for various pathological conditions, such as kidney and liver diseases. These markers play important roles in redox regulations throughout the body. Despite the recognition of these markers in various pathophysiologic conditions, the measurements of human mercaptalbumin and non-mercaptalbumin have not been popular because of the technical complexity and long measurement time of conventional methods.
Methods
Based on previous reports, we explored the optimal analytical conditions for a high-performance liquid chromatography method using an anion-exchange column packed with a hydrophilic polyvinyl alcohol gel. The method was then validated using performance tests as well as measurements of various patients’ serum samples.
Results
We successfully established a reliable high-performance liquid chromatography method with an analytical time of only 12 min per test. The repeatability (within-day variability) and reproducibility (day-to-day variability) were 0.30% and 0.27% (CV), respectively. A very good correlation was obtained with the results of the conventional method.
Conclusions
A practical method for the clinical measurement of human mercaptalbumin and non-mercaptalbumin was established. This high-performance liquid chromatography method is expected to be a powerful tool enabling the expansion of clinical usefulness and ensuring the elucidation of the roles of albumin in redox reactions throughout the human body.
Albumin is a key multifunctional protein in the regulation of oncotic pressure, the binding and transferring of different ligands and antioxidant functions. Oxidative stress is more or less linked to the pathogenesis of infectious, renal and liver diseases. Recently, much attention has been focused on the role of albumin under these pathologic conditions.
The thiol group of Cys34 in albumin is widely known to exist in either a reduced (-SH) or oxidized (-S-S-) form.1,2 There are 35 cysteine residues in the albumin molecule. Among them, 34 of the cysteine residues together form 17 pairs via disulphide bindings. Only Cys34 does not have a disulphide-binding partner in the albumin molecule. When the thiol radical of Cys34 is in a reduced (-SH) state, the molecule is called reduced albumin or human mercaptalbumin (HMA). When Cys34 instead forms a disulphide bond with another -SH compound in the blood, the molecule is called oxidized albumin or human non-mercaptalbumin (HNA). Cys34 mainly binds with either cysteine (HNA [Cys]) or glutathione (HNA [Glut]). However, more highly oxidized forms of HNA also exist (HNA [Oxi]), such as sulfenic (-SOH), sulfinic (-SO2H), and sulphonic (-SO3H) forms. 3
The amino acid sequences of albumin differ from species to species, as do their molecular weights and isoelectric points. However, Cys34 is shared among all mammals, and reduced and oxidized forms of albumin are found across species. 4 Sogami et al. 5 first described the measurement of HMA and HNA using high-performance liquid chromatography (HPLC); since then, correlations with various clinical conditions have been reported.6–18 The method described by Sogami et al. requires 2-m columns to separate HMA and HNA and takes about 2 h to perform. An improved method was subsequently developed by Era et al., 3 shortening the measurement time. However, the method still requires about 1 h to measure one sample and thus is not applicable in clinical settings requiring the measurement of large numbers of samples simultaneously. A measurement method using anion-exchange chromatography has also been developed.3,8 However, this method uses a three-step linear gradient program of 1%, 3% and 10% ethanol concentrations for eluents, and the analysis conditions are complicated; the method also takes about 1 h to perform, including time for column equilibration. An HPLC method using post-column detection with a BCG reagent was developed for the specific detection of HMA and HNA, but the measurement time was relatively long and the system and measurement conditions were also complicated. 12
Given this background, the present study was undertaken to develop a simple, rapid and precise system for the clinical measurement of HMA and HNA.
Methods
Separation and measurement of HMA and HNA on HPLC
The component of a basic HPLC system (LabSolutions system; Shimazu Co., Ltd, Kyoto, Japan) consists of a degasser (DGU20A3R), two pumps (LC-20AT), an auto-sampler (SIL30AC), a thermostatic oven (CTO-20AC), a fluorescence detector (RF-20Axs) and a system controller (CBM-20 A).
To obtain a high resolution and to produce an anion-exchange gel, a polyvinyl alcohol cross-linked gel (9 μm in diameter) (Asahipak GS-520; Asahi Kasei Co., Ltd, Tokyo, Japan) was dried in a vacuum for more than 16 h and then suspended in 10 mL of dimethyl sulfoxide (DMSO; Tokyo Chemical Industry Co., Ltd, Tokyo, Japan) per 1 g of dried gel. Next, 20 mmol of epichlorohydrin for each 1 g of dried gel was added to suspend the gel; the reaction was then allowed to proceed for 20 h at 30℃.
The activated gel was filtrated and reacted with a 10% aqueous solution of diethyl amine (Wako Pure Chemical Industries, Ltd, Osaka, Japan) for 20 h. The synthesized anion-exchange gel was packed in a stainless column (50 × 7.6 mm I.D.).
Using the above components, the optimal separation conditions were explored, including the choice and combination of separation buffer, pH, flow rate, gradient condition of the eluents (ethanol concentration, magnesium concentration), analysis temperature and sample amount. All the reagents were either HPLC grade or a special grade (Wako Pure Chemical Industries, Ltd, Osaka, Japan). The conditions for fluorescence detection were the same as those previously reported by Hayashi et al., 9 and the excitation and emission wavelengths were 280 nm and 340 nm, respectively.
Using serum samples from 20 patients, the results were comparatively evaluated using a conventional system as follows 3 : two columns of a Shodex Asahipak GS-520 HQ (300 × 7.5 mm I.D.; Showa Denko K.K., Tokyo, Japan) were used for the HPLC system. Ten microlitres of the serum and plasma samples were directly injected into the system. The flow rate was 0.8 mL/min, and the analytical temperature was 30℃. The eluent buffer solutions contained 0.03 M sodium phosphate buffer and 0.3 M trisodium citrate (pH 6.80). All other conditions were the same as those used for the presently reported method.
According to the results obtained using the conventional method, each of the resulting peaks was identified as corresponding to one of the markers, an above 95% purity of gamma globulin, transferrin and albumin from Sigma-Aldrich, Inc. (St. Louis, MO). The peaks for HMA and HNA were identified based on the fractions obtained using the conventional method.
Based on the peak area, the results were expressed as HMA (%) = HMA/(HMA+HNA) × 100, similar to the method used in previous reports.3,5
Evaluation of method performance
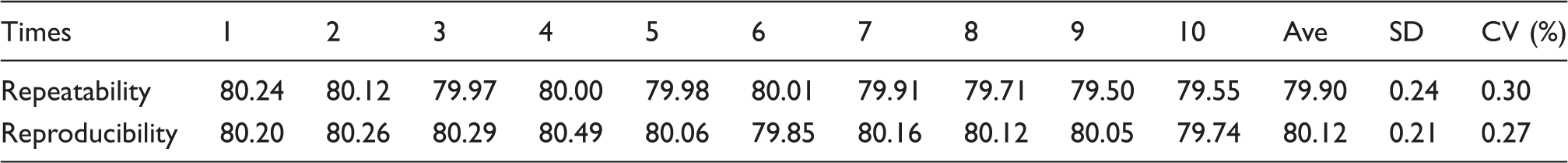
The repeatability was examined 10 consecutive times, and the reproducibility on 10 consecutive days was examined in duplicate.
The effects of interfering substances on the assay system were tested using a commercially available kit (Check A Plus; Sysmex Corporation, Kobe, Japan). Using serum samples from 20 patients, the results of the presently reported method were compared with those of the conventional method. 3
Validation study in clinical settings
The clinical usefulness of the presently reported system was validated using samples from 144 patients with chronic kidney diseases and 93 patients with hepatic diseases for which the serum creatinine or Child-Pugh score can be used as a representative comparative marker. The relationship between HMA and aging was also studied using samples from 69 apparently healthy patients between the ages of 29 and 93 years old. All the serum samples were collected during medical checkups performed at Tokyo University Hospital in July 2015; the samples had been kept at −80℃ after sampling until their use in the clinical validation study. The above studies were performed with the approval of the ethics committee of The University of Tokyo Hospital Graduate School of Medicine (Approval number 10825).
Statistical analysis
A non-repeated measures analysis of variance (ANOVA) and the Pearson product-moment correlation coefficient were used for the statistical analysis.
Results
Separation and measurement of HMA and HNA on HPLC
After evaluating several HPLC eluent conditions, a solution of 25 mM phosphoric acid buffer containing 60 mM sulphuric acid sodium (pH 6.0) was found to be the most suitable for the first eluent (eluent A) to absorb and separate the albumin fraction. To separate HMA and HNA in the anion-exchange column, a high-concentration magnesium chloride solution was the most suitable for eluting HMA and HNA from the column as an eluate (eluent B). The flow rate was set at 1 mL/min after equilibrating the column for 4.5 min with eluent A. The linear gradient time program from eluent A (100%) to eluent B (100%) lasted for 7.5 min and started at the time of sample injection. The total measurement time was 12 min per sample, including both column equilibration and the analysis time. By balancing other analysis conditions, 3 µL of either serum or plasma and an analysis temperature of 40℃ were found to be the ideal conditions.
A representative chromatogram for normal human serum obtained using these HPLC conditions is shown in Figure 1(a). Figure 1(b) shows a representative chromatogram for a patient with liver disease who had a relatively high concentration of HNA (Oxi). Even without a special program for the quantitative determination of the peak area, such as a graphical method for symmetrical resolution,
9
the separation of the HMA and HNA components was sufficient to enable a quantitative analysis under these conditions.
Chromatograms of normal human serum (a) and serum from a patient with liver disease (b). HNA I corresponds to HNA (Cys) and HNA (Glut), and HNA II corresponds to HNA (Oxi).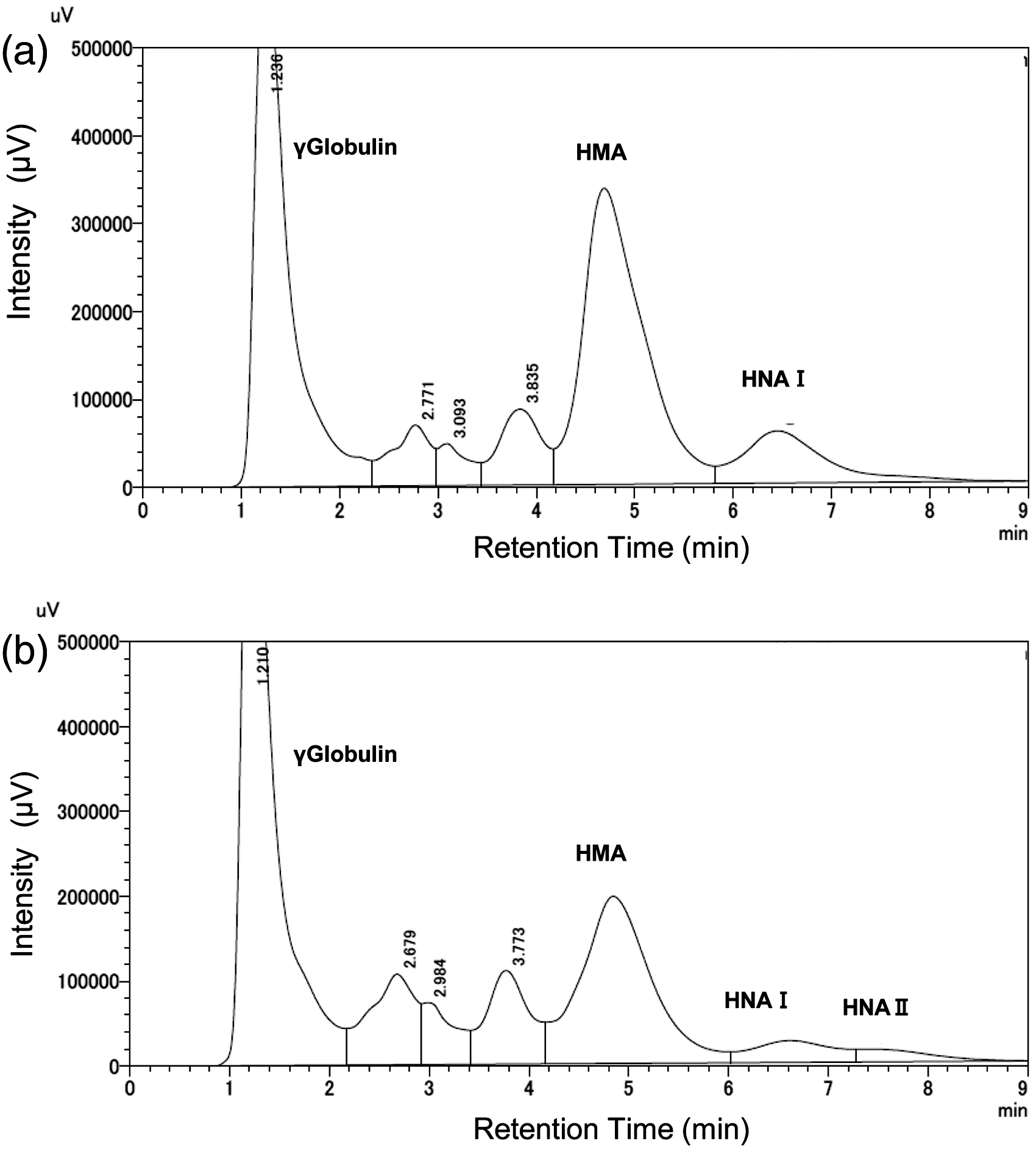
Evaluation of method performance
Repeatability and reproducibility of the HMA (%) assay.
Interference study
Using a kit, the interference effects of four major substances were studied by adding them to serum. Chyle, free bilirubin and conjugated bilirubin did not produce any interference (Figure 2(a) to (c)). However, an increase in haemoglobin produced a false increase in the HMA level, reaching 103% at a concentration of 5 g/dL (Figure 2(d)).
Interference from blood substances. (a) Free bilirubin, (b) conjugated bilirubin, (c) chyle, and (d) haemolytic haemoglobin.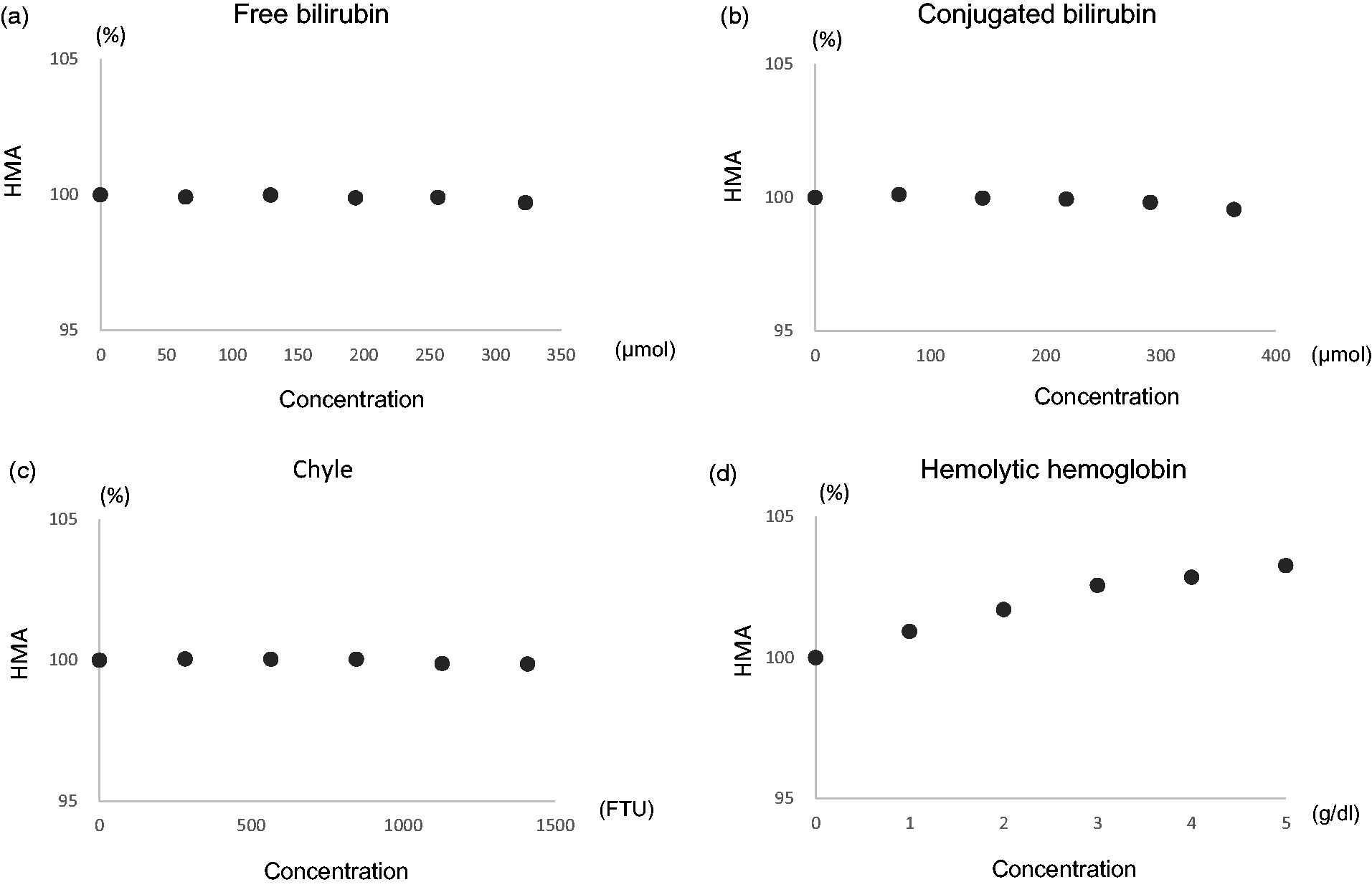
Correlation with conventional method
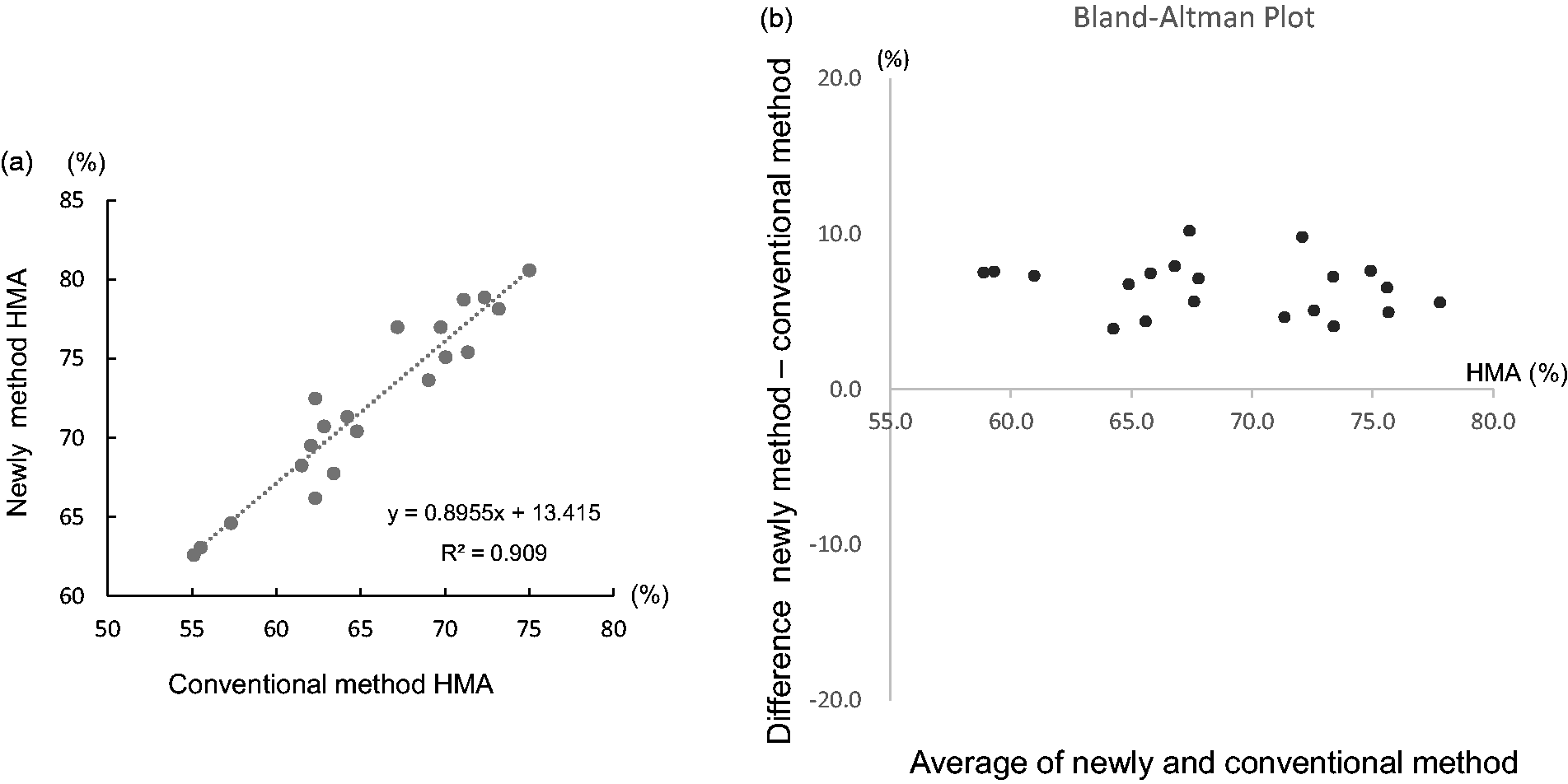
The correlation with the findings obtained using the conventional method is shown in Figure 3. The coefficient of correlation for the HMA% values (n = 20) obtained using the presently reported method and the conventional method was R2 = 0.909. The absolute values obtained using the presently reported method were about 5% higher than those obtained using the conventional method.
Correlation with conventional method. The conditions used for the conventional method were as follows: column, two molecular-sieve columns (Asahipak GS-520H; 7.6 mm I.D. × 500; Showa Denko K.K., Tokyo, Japan); flow rate, 0.8 mL/min; eluent, 0.03 M sodium phosphate buffer containing 0.30 M trisodium citrate (pH 6.8).
15
Validation study of HMA and HNA using various patient serum samples
The relation between HMA% and the creatinine concentration in patients with kidney disease is shown in Figure 4(a) and (b). HMA% was negatively correlated with the creatinine concentration (Figure 4(a)). The HMA% in patients with a creatinine concentration of ≥176.8 µmol (2.0 mg/dL) was significantly lower than that in patients with a creatinine concentration <176.8 µmol (2.0 mg/dL) (P < 0.0001) (Figure 4(b)).
Correlation between HMA% and creatinine concentration in patients with kidney disease and with Child-Pugh score in patients with liver disease. (a) All kidney disease patients (n = 144). (b) Patients with a creatinine concentration ≥176.8 µmol (2.0 mg/dL) (n = 58) and patients with a creatinine concentration <176.8 µmol (2.0 mg/dL) (n = 86). (c) All liver disease patients (n = 93). (d) Patients with a Child-Pugh score of 3 or 4 (n = 53) and patients with a Child-Pugh score of ≥5 (n = 40). Values are the mean ± SD.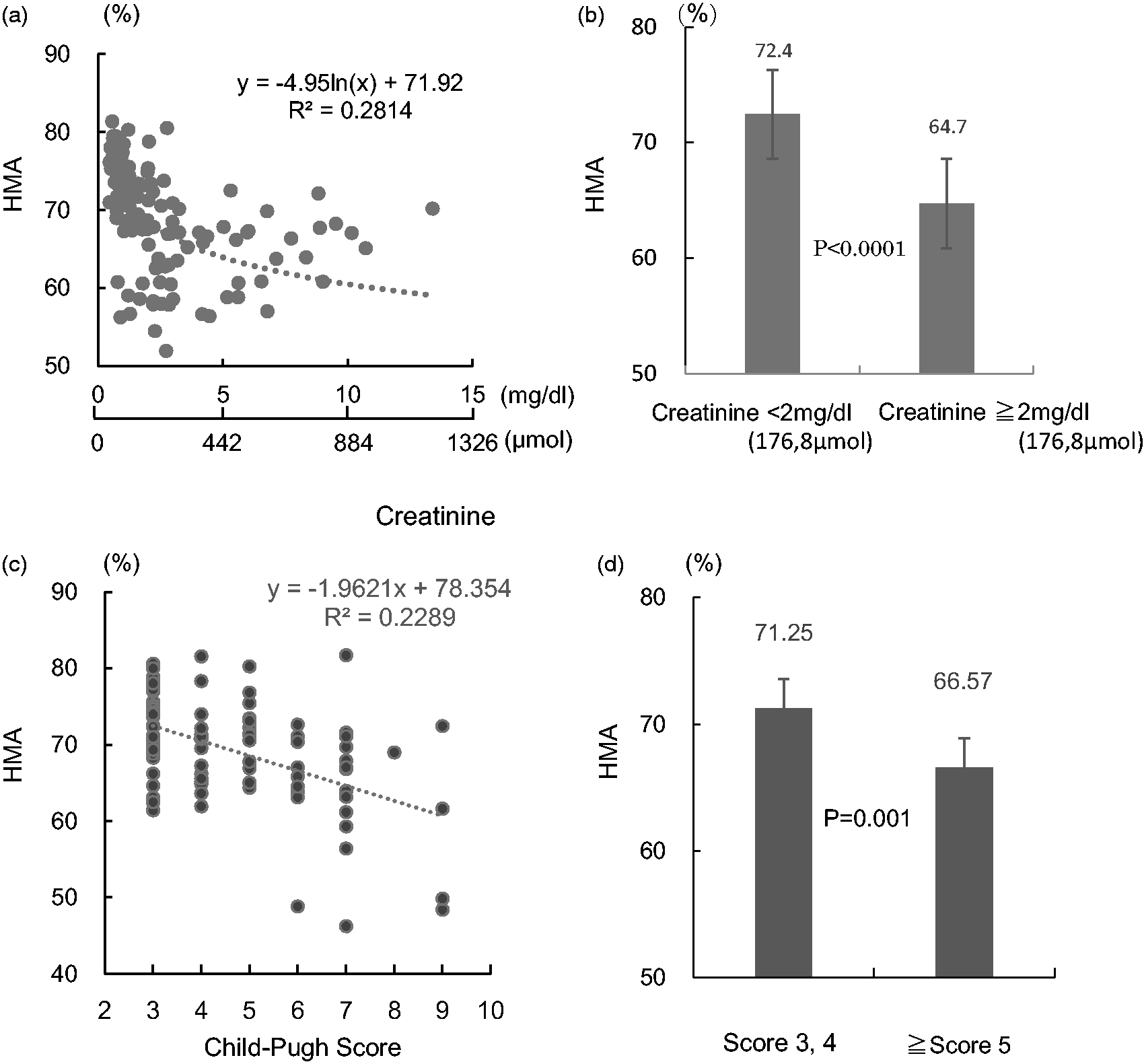
Figure 4(c) and (d) shows the relation between HMA% and the Child-Pugh scores in patients with liver diseases. HMA% was negatively correlated with the Child-Pugh score (Figure 4(c)). The HMA% in patients with a Child-Pugh score ≥5 was significantly lower than that in patients with a Child-Pugh score <4 (P = 0.001) (Figure 4(d)).
All of the above results were mostly comparable to those reported using conventional systems (or methods).7,8,13,14
Correlation between HMA and aging
The correlation between HMA% and age in 69 patients (≥35 g/L albumin, <176.8 µmol [2.0 mg/dL] creatinine) is shown in Figure 5. The HMA% decreased as the age of the patients increased among the study group.
Relation between age and HMA%. The results for 69 patients (albumin ≥35 g/L and creatinine <176.8 µmoL [2.0 mg/dL]) are shown.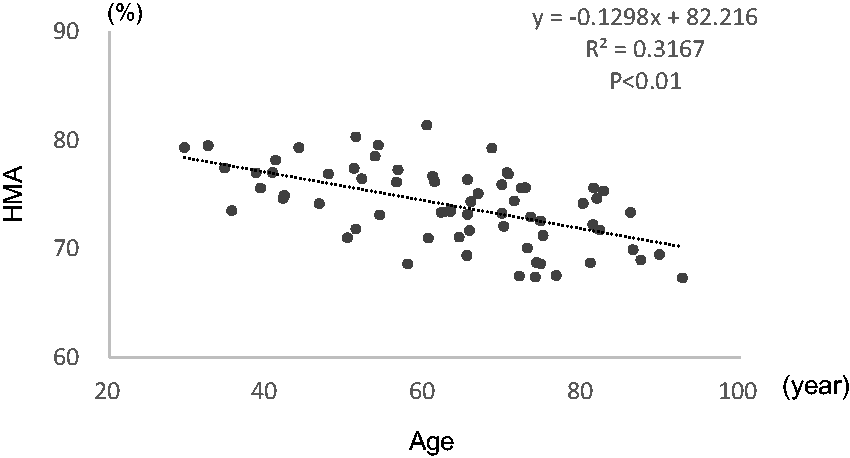
Discussion
Based on previous reports,3,5,12 we developed an easy, precise and practical HPLC method for measuring serum HMA and HNA concentrations simultaneously. The actual test time is now less than 10 min. In total, 100 samples can be measured within 20 h. The test time was reduced by optimizing the conditions, especially the pH of eluent A and applying magnesium chloride to eluent B. It will be possible to reduce the measurement time further by making the column size smaller. As judged by the within-day and day-to-day variations, the performance of the method is equivalent to that of routine HPLC systems for HbA1c and glycated albumin. Thus, the method is expected to be highly applicable to daily clinical practice.
For successful separation, establishing the optimal eluent conditions is key. A high concentration of magnesium chloride was shown to fractionate the targeted fractions more efficiently than the ethanol and thus was used as eluent B in a simple linear gradient. The conventional method uses an eluent with a pH of 4.8, which is almost identical to the isoelectric point of albumin. This parameter influences the yield and the resolution of HMA and HNA from other proteins. 19 We achieved higher yields using an eluent pH of 6.0.
We think that both the ion-exchange action and the hydrophobic interaction of resin with albumin contributed to the resolution of HMA and HNA in the column. Successful separation was possible by controlling the ionic and antichaotropic strength to the resin by adjusting the magnesium concentration and the pH of eluent A.
This simple methodology was also the reason for the high reproducibility of this measure (CV value of 0.30%).
At higher serum haemoglobin concentrations, HMA (%) concentrations increase, and a 3.3% increase was observed at a haemoglobin concentration of 5 g/L of haemolytic haemoglobin. When interpreting clinical data, clinicians should be aware of the possibility of false elevations in HMA concentrations in samples with haemolysis.
The correlation between the measurement values obtained using our method and those obtained using the conventional method was good. The measurement values obtained using our method were about 5% higher than those obtained using the conventional method. In a previous report, the measurement values obtained using LC/MS were also about 5% higher than those obtained using the conventional method, suggesting that the measurement values obtained using LC/MS and our method are likely to be similar. 7
In a validation study using patient serum samples, the HMA% was lower in patients with kidney or liver disease than in healthy controls, and the HMA% also decreased with aging. These results are comparable with those reported in studies using conventional measurement methods.7,8,13,14
HMA and HNA measurements and their correlations with various diseases have been reported.6–18 In addition to kidney and liver diseases, elevated concentrations of albumin oxidation have been reported in the aqueous humor and joint synovia of patients with senile cataracts and arthrosis of the temporomandibular joint, respectively.11,16 In vitro studies have shown that oxidized albumin is cytotoxic, and the HNA% of commercially available albumin preparations is very high; thus, the therapeutic use of albumin preparations with a high HNA content might be problematic. 18
In conclusion, we attempted to establish a simple and rapid HPLC method for measuring human mercaptalbumin and non-mercaptalbumin concentrations suitable for use in actual clinical settings. The column was sufficiently durable and was used to measure about 2000 samples under the various conditions examined in the present study, with no change in column performance. The present HPLC method is expected to provide a powerful tool for expanding the clinical usefulness of HMA and HNA measurements and for elucidating the role of albumin in redox reactions throughout the human body.
Footnotes
Acknowledgement
The authors thank Ms N Kanno for her help with the experiments.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by JSPS KAKENHI (25253040) and The Nakatani Foundation for Advancement of Measuring Technologies in Biomedical Engineering.
Ethical approval
This study was performed with the approval of the ethics committee of The University of Tokyo Hospital Graduate School of Medicine (Approval number 10825).
Guarantor
YY.
Contributorship
KY and YY searched for relevant literature and conceived the study. TS and SO were involved in the protocol development, obtaining ethical approval, patient recruitment, and data analysis. KY wrote the first draft of the manuscript. All the authors reviewed and edited the manuscript and approved the final version of the manuscript.
