Abstract
Background
Interference from opiates in the Microgenics CEDIA® Buprenorphine assay is known to produce false-positive buprenorphine screening immunoassay results necessitating confirmatory buprenorphine testing by chromatography/mass spectrometry methods.
Method
We reviewed data on falsely positive buprenorphine immunoassay screen (cut-off ≥ 5
Results
We found that cross-reactivity in the CEDIA® buprenorphine immunoassay by opiates at concentrations <2000
Conclusion
Samples with CEDIA® opiate immunoassay result <2000
Introduction
Buprenorphine is a semi-synthetic and relatively long-acting partial opioid receptor agonist that has gained significant importance as a substitution treatment for opioid dependence. In Australia, a mono-buprenorphine (BPN) tablet (marketed as Subutex®) has been available since 2001. To discourage parenteral abuse of BPN, a co-formulation of BPN and the opioid antagonist naloxone (NX) was developed (BNX tablets, marketed as Suboxone®) and has been available in Australia since 2006. More recently, a buprenorphine–naloxone sublingual formulation (BNX film, marketed as Suboxone® Film) has been introduced. A transdermal buprenorphine patch, which delivers buprenorphine at a constant rate over seven days, is also available for management of chronic, severe disabling pain not responding to non-opioid analgesics. After sublingual dosage, buprenorphine is quickly and extensively metabolized by hepatic N-dealkylation to norbuprenorphine. Both buprenorphine and norbuprenorphine are conjugated with glucuronic acid prior to urinary excretion. 1
Considerable diversion and abuse of both BPN and BNX and several fatal BPN poisonings have been reported.2–8 Many studies have proven that the abuse potential of BNX is lesser than that of BPN but still significant in both opioid-dependent and opioid non-dependent abusers.9–13 The BNX film is advantageous in preventing abuse because the sublingual bioavailability of BPN is relatively high while that of NX is low. When BNX is taken parenterally, the bioavailability of NX is high, which should precipitate withdrawal and attenuation of the pleasurable effects in opioid-dependent subjects. However, the diversion rates for BNX film have been reported to be similar to that of BNX tablets.14,15
In order to minimize undisclosed drug use and potential diversion, physicians generally require patients in substitution programmes to receive medications only from a single prescriber and not to use non-prescribed medications or illicit drugs. 16 Because patient self-reporting of drug use is frequently unreliable, clinicians treating these patients often perform urine drugs of abuse screening for verification of opiate abstinence, and/or compliance with substitution therapy and illicit use of the drug (drug diversion). In Australia, the National Guidelines for Medication-Assisted Treatment of Opioid Dependence 16 identified urine drug screens as an important means to enhance the validity of patients’ self-reported use of substances, identify substances not reported by the patient that may assist diagnosis/management and assist in determining eligibility for takeaway or unsupervised dosing.
The laboratory procedure for the measurement of buprenorphine in urine is performed in two stages. After an initial screening procedure with a class specific-immunoassay, positive screen findings are confirmed by highly specific chromatography-mass spectrometry (GCMS or LCMS) procedures. The advantages of the immunoassay screening tests are that it is less labour-intensive, can be performed quickly with a result available in a relatively short time frame and is relatively cheap. GCMS or LCMS is labour-intensive and time-consuming making it relatively expensive, as many drugs require considerable preparation before they can be analysed. Therefore, it would be very useful to obtain the best information from the immunoassay and reserve the confirmation assay to cases where clinically meaningful extra information could be gained.
The main disadvantage of the immunoassay screen is that it is specific only to the class of drug. Structurally similar compounds of different classes may also react with the antibody, giving a falsely positive result. Patients in buprenorphine substitution programmes are often prescribed potentially interfering medications or may be abusing other opiates which may produce false-positive buprenorphine screening results. The Microgenics CEDIA® burprenorphine immunoassay has been shown to have non-specific cross-reactivity with other opioids including codeine, morphine, methadone 17 tramadol 18 as well as hydroxychloroquine, 19 and the antipsychotic amisulpride. 20
Most clinical laboratories report results as detected or not detected based on the concentration above or below a defined buprenorphine threshold. We are aware that buprenorphine cut-offs used for immunoassays as well as confirmatory assays vary between laboratories (Lisa Jolly RCPA QAP, personal communication). Thus, a negative result does not necessarily mean that the drug is not present, only that it is not present at a concentration above the defined threshold for that laboratory. Buprenorphine as well as opiate immunoassays used currently in clinical laboratories are optimized to measure concentrations correctly around the cut-off. Also manufacturers do not routinely provide a linear range of the assay except to say that it is linear around the cut-off.21,22
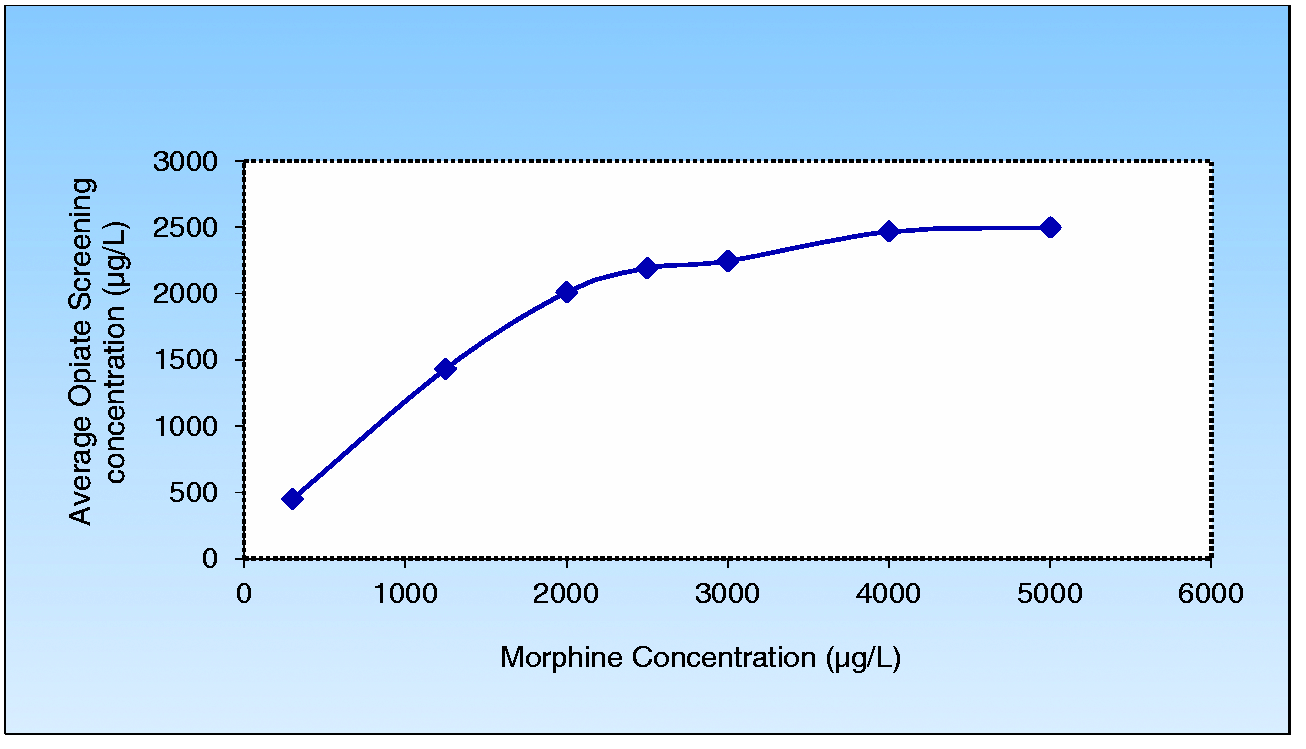
To perform confirmatory testing on all positive samples incur significant labour, cost and cause delay in reporting. It would be advantageous to establish a specific opiate immunoassay screen concentration below which we can be confident that cross-reactivity in the buprenorphine immunoassay would be insufficient to cause a false-positive buprenorphine. Then buprenorphine immunoassay results with an opiate immunoassay result below this threshold can be confidently called truly positive without the need for confirmation. In order to set this opiate threshold, it is essential we are confident that the opiate immunoassay screen is linear at this concentration.
Methods
We reviewed all confirmatory testing by GC/MS for buprenorphine performed in our laboratory over a three-month period. In our laboratory screening, buprenorphine and opiate immunoassays used are Microgenics CEDIA® Buprenorphine assay and Microgenics CEDIA® Opiate assay on a Beckman-Coulter AU680 analyser. The buprenorphine screening assay has coefficients of variation (CV) of 14.9% at 3.75
Results
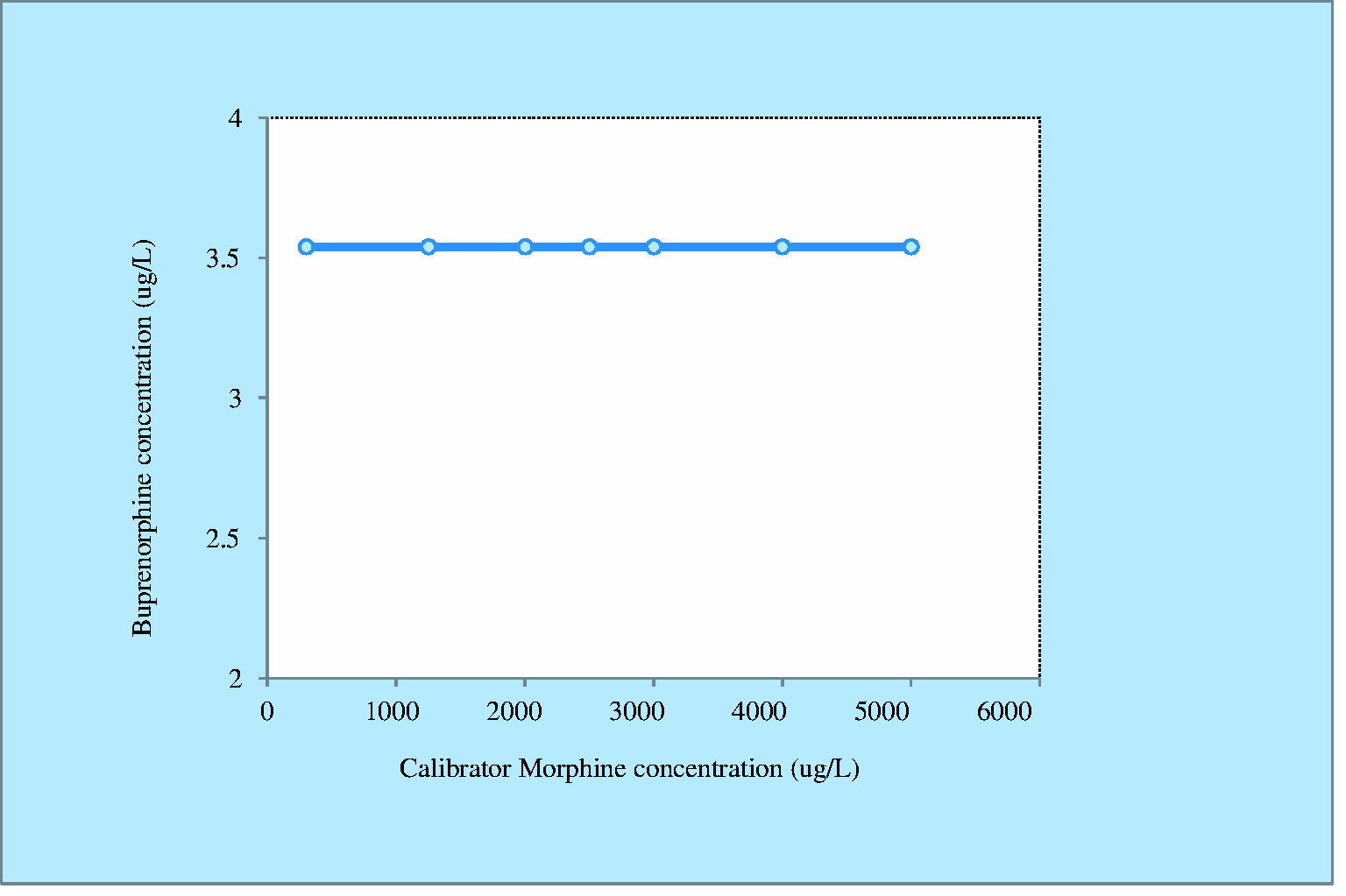
In total, 222 samples positive for buprenorphine by immunoassay were analysed by the GCMS enzyme hydrolysis method for confirmation of buprenorphine over the study period; 133 were confirmed by GCMS to be truly positive (above the cut-off of 5 Flow diagram of the data analysed. Linearity of the CEDIA® Opiate assay assessed using morphine calibrator. Buprenorphine interference testing using morphine calibrator.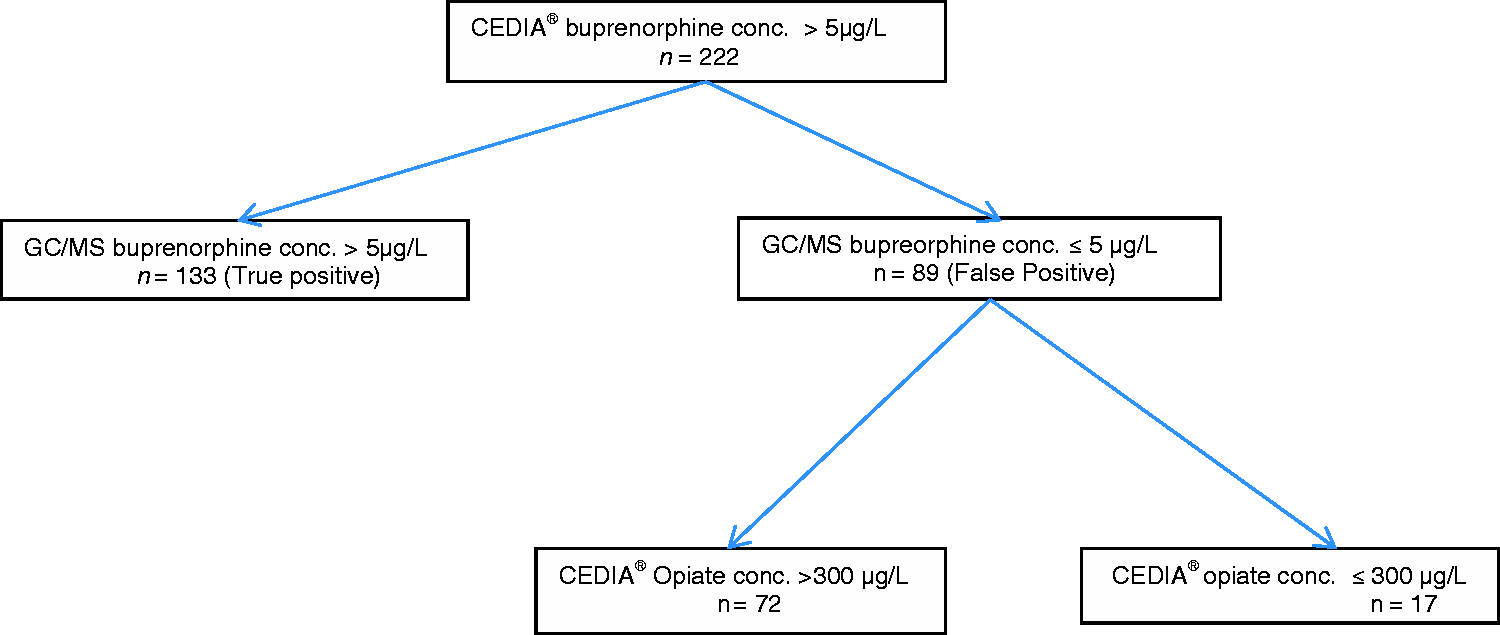
Discussion
While guidelines for urine drug testing in the setting of opioid substitution programmes do not clearly define the testing process, they are commonly performed to identify use of undisclosed substances, uncover possible diversion of prescribed substances and determine compliance with prescribed substances. Patients who are substituted with BNX are often prescribed or use non-prescribed medication that may cause a false-positive urine buprenorphine screening result which may need to be confirmed. If the information obtained from the screening buprenorphine assay can be optimized to obtain the answer to these questions without having to do the confirmation test in some of these samples, it may provide a more cost-effective service. In an attempt to optimize our service, we aimed to determine whether there is a threshold opiate concentration by the screening opiate assay below which cross-reactivity in the buprenorphine immunoassay does not cause a false-positive buprenorphine.
We found that when both buprenorphine and opiate screening assays are positive, at opiate concentrations lesser than 2000
In our study, 17/89 specimens had buprenorphine immunoassay >5
A limitation of our study is that this is a retrospective analysis of laboratory results and full clinical information on these patients was not always available. Although this does not affect the current question being asked, clinical information is very important in the interpretation of urine drug screens. For example, confirmation of prescription of buprenorphine on patient with GCMS buprenorphine detected below the cut-off would have been important both for our purposes as well as in the clinical management of these patients. This is an example, where over-reliance on a defined cut-off and reporting of a qualitative negative result might be assumed categorically to be non-compliance which may adversely affect patient management. We have changed our practice to add a comment to the report about the detection of buprenorphine and metabolites by GCMS below the cut-off and have a conversation with the clinician about the possibilities for this.
Conclusion
Cross-reactivity in the CEDIA® buprenorphine immunoassay by opiates at concentrations <2000
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Not applicable.
Guarantor
MS.
Contributorship
All authors contributed to the design of the experiments. AT performed the linearity studies. MS wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
